Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (1): 85-94.DOI: 10.19852/j.cnki.jtcm.2026.01.008
• Original Articles • Previous Articles Next Articles
Alleviation of nitroglycerin-induced migraine in rats: inhibition of central sensitization by optimizing Qinggan Jieyu decoction (清肝解郁方) via regulation of purinergic receptor P2X ligand-gated ion channel 7 and autophagy
- Department of Neurology, Xiyuan Hospital of China Academy of Chinese Medical Sciences, Beijing 100091, China
-
Received:2024-11-13Accepted:2025-05-19Online:2026-02-15Published:2026-01-28 -
Contact:ZHOU Bo, Department of Neurology, Xiyuan Hospital of China Academy of Chinese Medical Sciences, Beijing 100091, China.zhoubokingkong@163.com ;Telephone: + 86-10-62835332 -
Supported by:China Academy of Chinese Medical Sciences Innovation Fund:Multicenter Randomized Controlled Study on the Intervention of Yiqi Huoxue Huatan Tongluo decoction in Post-Stent Restenosis of Vertebral Arteries(CI2021A01308);In-Hospital Mentorship Program of Xiyuan Hospital, China Academy of Chinese Medical Sciences-Zhou Shaohua(0203055)
Cite this article
LI Jiazheng, ZHOU Bo. Alleviation of nitroglycerin-induced migraine in rats: inhibition of central sensitization by optimizing Qinggan Jieyu decoction (清肝解郁方) via regulation of purinergic receptor P2X ligand-gated ion channel 7 and autophagy[J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 85-94.
share this article

Figure 1 QGJY intervention alleviates NTG-induced migraines A: cage-climbing frequency; B: head-scratching frequency; C: western blot of central sensitization-related indicators; D: SP protein expression; E: CGRP protein expression; F: c-fos protein expression. Sham group: received daily gavage of 0.9% saline (10 mL/kg) for 7 consecutive days, followed by a subcutaneous injection of saline at the posterior neck on day 7; NTG group: received daily gavage of 0.9% saline for 7 d, followed by a subcutaneous injection of 10 mg/kg NTG at the posterior neck on day 7; NTG + QGJY Group: received daily gavage of QGJY (at the same volume as saline) for 7 d, followed by a subcutaneous injection of 10 mg/kg NTG on day 7; NTG + QGJY + BzATP Group: received daily gavage of QGJY for 7 d, followed by a co-injection of 10 mg/kg NTG and 3.5 mg/kg BzATP subcutaneously on day 7. NTG: nitroglycerin; QGJY: optimizing Qinggan Jieyu decoction; BzATP: BzATP triethylammonium salt; SP: substance P; CGRP: calcitonin gene-related peptide; c-fos: c-fos induced growth factor. One-way analysis of variance was used to compare more than two groups, followed by Tukey’s post hoc test for pairwise comparisons. Values are expressed as mean ± standard deviation (n = 6). aP < 0.05, sham group compared to NTG group; bP < 0.05, NTG + QGJY group compared to NTG + QGJY + BzATP group; cP < 0.05, NTG group compared to NTG + QGJY group.

Figure 2 QGJY inhibits the levels of inflammatory cytokines IL-1β, IL-6, and TNF-α A: IL-1β levels in serum; B: IL-6 levels in serum; C: TNF-α levels in serum; D: IL-1β levels in TNC tissues; E: IL-6 levels in TNC tissues; F: TNF-α levels in TNC tissues. Sham group: received daily gavage of 0.9% saline (10 mL/kg) for 7 consecutive days, followed by a subcutaneous injection of saline at the posterior neck on day 7; NTG group: received daily gavage of 0.9% saline for 7 d, followed by a subcutaneous injection of 10 mg/kg NTG at the posterior neck on day 7; NTG + QGJY Group: received daily gavage of QGJY (at the same volume as saline) for 7 d, followed by a subcutaneous injection of 10 mg/kg NTG on day 7; NTG + QGJY + BzATP Group: received daily gavage of QGJY for 7 d, followed by a co-injection of 10 mg/kg NTG and 3.5 mg/kg BzATP subcutaneously on day 7. NTG: nitroglycerin; QGJY: optimizing Qinggan Jieyu decoction; BzATP: BzATP triethylammonium salt. One-way analysis of variance was used to compare more than two groups, followed by Tukey’s post hoc test for pairwise comparisons. Values are expressed as mean ± standard deviation (n = 6). aP < 0.05, sham group compared to NTG group; bP < 0.05, NTG + QGJY group compared to NTG group.

Figure 3 QGJY inhibited microglia activation and P2X7R expression in the TNC tissues A: P2X7R/Iba1 co-staining in TNC; B: group comparisons of P2X7R and Iba1 expression; A1: nuclear staining with DAPI; A2: immunostaining for the microglial marker Iba1; A3: immunostaining for the P2X7 receptor; A4: merged image of A1-A3; B1: sham (DAPI); B2: NTG (DAPI); B3: NTG + QGJY (DAPI); B4: NTG + QGJY + BzATP (DAPI); B5: sham (Iba1); B6: NTG (Iba1); B7: NTG + QGJY (Iba1); B8: NTG + QGJY + BzATP (Iba1); B9: sham (merge); B10: NTG (merge); B11: NTG + QGJY (merge); B12: NTG + QGJY + BzATP (merge); C: total length of process processes per microglia; D: quantitative analysis of the Iba1 positive cells for FOV; E: mean length of processes per microglia; F: Western blot of P2X7R; G: relative expression levels of P2X7R proteins; H: QRT-PCR analysis of the relative P2X7R mRNA level. Sham group: received daily gavage of 0.9% saline (10 mL/kg) for 7 consecutive days, followed by a subcutaneous injection of saline at the posterior neck on day 7; NTG group: received daily gavage of 0.9% saline for 7 d, followed by a subcutaneous injection of 10 mg/kg NTG at the posterior neck on day 7; NTG + QGJY Group: received daily gavage of QGJY (at the same volume as saline) for 7 d, followed by a subcutaneous injection of 10 mg/kg NTG on day 7; NTG + QGJY + BzATP Group: received daily gavage of QGJY for 7 d, followed by a co-injection of 10 mg/kg NTG and 3.5 mg/kg BzATP subcutaneously on day 7. TNC: trigeminal nucleus caudalis; QRT-PCR: quantitative reverse transcription polymerase chain reaction; QGJY: optimizing Qinggan Jieyu decoction; P2X7R: P2X7 receptor; Iba1: ?ionized calcium-binding adapter molecule 1?; DAPI: 4',6-diamidino-2-phenylindole; NTG: nitroglycerin; BzATP: BzATP triethylammonium salt; FOV: field of view. One-way analysis of variance was used to compare more than two groups, followed by Tukey’s post hoc test for pairwise comparisons. Values are expressed as mean ± standard deviation (n = 6). aP < 0.05, sham group compared to NTG group; bP < 0.05, NTG + QGJY group compared to NTG group; cP < 0.05, NTG + QGJY group compared to NTG + QGJY + BzATP group.
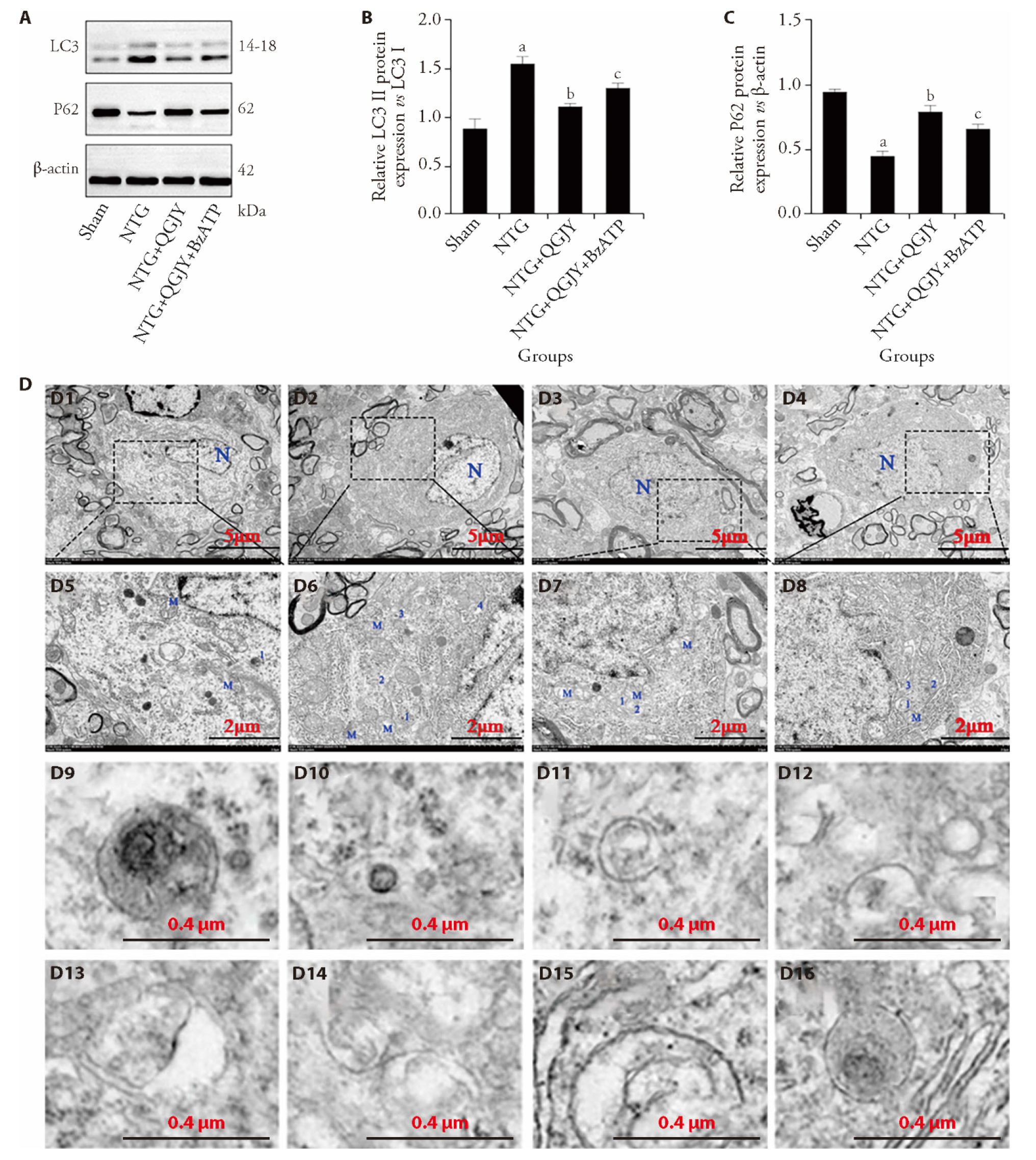
Figure 4 QGJY inhibits autophagy in TNC tissues of migraine rats A: Western blot of LC3 and P62; B: relative LC3 Ⅱ protein expression vs LC3 Ⅰ; C: relative P62 protein expression vs β-actin; D: TEM images of the TNC tissues in different groups; D1: sham (TEM); D2: NTG (TEM); D3 NTG + QGJY (TEM); D4: NTG + QGJY + BzATP (TEM); D5: sham (enlarge); D6: NTG (enlarge); D7 NTG + QGJY (enlarge); D8: NTG + QGJY + BzATP (enlarge); D9: autophagosome (sham); D10, D11, D12, D13: autophagosomes (NTG); D14: autophagosome (NTG + QGJY); D15, D16: autophagosome (NTG + QGJY + BzATP). Scale bar: 5 μm (2 μm, enlarged); Sham group: received daily gavage of 0.9% saline (10 mL/kg) for 7 consecutive days, followed by a subcutaneous injection of saline at the posterior neck on day 7; NTG group: received daily gavage of 0.9% saline for 7 d, followed by a subcutaneous injection of 10 mg/kg NTG at the posterior neck on day 7; NTG + QGJY Group: received daily gavage of QGJY (at the same volume as saline) for 7 d, followed by a subcutaneous injection of 10 mg/kg NTG on day 7; NTG + QGJY + BzATP Group: received daily gavage of QGJY for 7 d, followed by a co-injection of 10 mg/kg NTG and 3.5 mg/kg BzATP subcutaneously on day 7. TNC: trigeminal nucleus caudalis; QGJY: optimizing Qinggan Jieyu decoction; LC3: light chain 3; P62: elevated ubiquitin-binding protein p62; TEM: transmission electron microscopy; NTG: nitroglycerin; BzATP: BzATP triethylammonium salt. One-way analysis of variance was used to compare more than two groups, followed by Tukey’s post hoc test for pairwise comparisons. Values are expressed as mean ± standard deviation (n = 6). aP < 0.05, sham group compared to NTG group; bP < 0.05, NTG + QGJY group compared to NTG group; cP < 0.05, NTG + QGJY group compared to NTG + QGJY + BzATP group.
| 1. |
Qubty W, Patniyot I. Migraine pathophysiology. Pediatr Neurol 2020; 107: 1-6.
DOI PMID |
| 2. |
Khan J, Asoom LIA, Sunni AA, et al. Genetics, pathophysiology, diagnosis, treatment, management, and prevention of migraine. Biomed Pharmacother 2021; 139: 111557.
DOI PMID |
| 3. |
Su M, Yu SY. Chronic migraine: a process of dysmodulation and sensitization. Mol Pain 2018; 14: 1744806918767697.
DOI URL |
| 4. |
Boyer N, Dallel R, Artola A, Monconduit L. General trigeminospinal central sensitization and impaired descending pain inhibitory controls contribute to migraine progression. Pain 2014; 155: 1196-205.
DOI PMID |
| 5. |
Illes P, Müller CE, Jacobson KA, et al. Update of P2X receptor properties and their pharmacology: IUPHAR Review 30. Br J Pharmacol 2021; 178: 489-514.
DOI URL |
| 6. |
Jiang L, Zhang YX, Jing F, et al. P2X7R-mediated autophagic impairment contributes to central sensitization in a chronic migraine model with recurrent nitroglycerin stimulation in mice. J Neuroinflammation 2021; 18: 5.
DOI |
| 7. |
Mizushima N. A brief history of autophagy from cell biology to physiology and disease. Nat Cell Biol 2018; 20: 521-7.
DOI PMID |
| 8. |
Clarke AJ, Simon AK. Autophagy in the renewal, differentiation and homeostasis of immune cells. Nat Rev Immunol 2019; 19: 170-83.
DOI PMID |
| 9. |
Takenouchi T, Fujita M, Sugama S, Kitani H, Hashimoto M. The role of the P2X7 receptor signaling pathway for the release of autolysosomes in microglial cells. Autophagy 2009; 5: 723-4.
PMID |
| 10. |
Kim JE, Ko AR, Hyun HW, Min SJ, Kang TC. P2RX7-MAPK1/2-SP 1 axis inhibits MTOR independent HSPB1-mediated astroglial autophagy. Cell Death Dis 2018; 9: 546.
DOI |
| 11. |
Fabbrizio P, Amadio S, Apolloni S, Volonté C. P2X7 receptor activation modulates autophagy in SOD1-G93A mouse microglia. Front Cell Neurosci 2017; 11: 249.
DOI PMID |
| 12. |
Young CNJ, Sinadinos A, Lefebvre A, et al. A novel mechanism of autophagic cell death in dystrophic muscle regulated by P2RX7 receptor large-pore formation and HSP90. Autophagy 2015; 11: 113-30.
DOI PMID |
| 13. | Zhou B. Biological basis of the association between liver depression and heat syndrome and optimizing Qinggan Jieyu decoction. Beijing: Beijing University of Chinese Medicine, 2020: 1-134. |
| 14. | Huang JH, Huang XH, Chen ZY, Zheng QS, Sun R. Dose conversion among different animals and healthy volunteers in pharmacological study. Chin J Clin Pharmacol Ther 2004; 9: 1069-72. |
| 15. |
Targowska-Duda KM, Ozawa A, Bertels Z, et al. NOP receptor agonist attenuates nitroglycerin-induced migraine-like symptoms in mice. Neuropharmacology 2020; 170: 108029.
DOI URL |
| 16. |
Pradhan AA, Smith ML, McGuire B, Tarash I, Evans CJ, Charles A. Characterization of a novel model of chronic migraine. Pain 2014; 155: 269-74.
DOI PMID |
| 17. |
Wang YJ, Shan ZM, Zhang LL, et al. P2X7R/NLRP3 signaling pathway-mediated pyroptosis and neuroinflammation contributed to cognitive impairment in a mouse model of migraine. J Headache Pain 2022; 23: 75.
DOI PMID |
| 18. |
Huang YL, Ni N, Hong YL, Lin X, Feng Y, Shen L. Progress in Traditional Chinese Medicine for the treatment of migraine. Am J Chin Med 2020; 48: 1731-48.
DOI URL |
| 19. |
Gribbin CL, Dani KA, Tyagi A. Chronic migraine: an update on diagnosis and management. Neurol India 2021; 69: S67-75.
DOI PMID |
| 20. | Pan X, Gao L, Xu W, Wen L. Distinguishing “Huo” and “Yan”: a comparative analysis of Traditional Chinese Medicine and Western Medicine. MJITCM 2009; 18: 2522-3. |
| 21. |
Zhang GL, Yu L, Chen ZY, et al. Activation of corticotropin-releasing factor neurons and microglia in paraventricular nucleus precipitates visceral hypersensitivity induced by colorectal distension in rats. Brain Behav Immun 2016; 55: 93-104.
DOI PMID |
| 22. | Wei ZH, Mo YX. Application of SF-36 quality of life measurement scale. Chin J Soc Med 1997; 4: 145-7. |
| 23. |
Edvinsson L, Haanes KA, Warfvinge K. Does inflammation have a role in migraine? Nat Rev Neurol 2019; 15: 483-90.
DOI PMID |
| 24. |
Sennholz A, Szikszay TM, Marusich T, Luedtke K, Carvalho GF. Association between central sensitization, pain sensitivity and balance control in patients with migraine. Eur J Pain 2024; 28: 786-96.
DOI URL |
| 25. |
Latremoliere A, Woolf CJ. Central sensitization: a generator of pain hypersensitivity by central neural plasticity. J Pain 2009; 10: 895-926.
DOI PMID |
| 26. |
Iyengar S, Johnson KW, Ossipov MH, Aurora SK. CGRP and the trigeminal system in migraine. Headache 2019; 59: 659-81.
DOI PMID |
| 27. |
Afrah AW, Fiskå A, Gjerstad J, et al. Spinal substance P release in vivo during the induction of long-term potentiation in dorsal horn neurons. Pain 2002; 96: 49-55.
DOI URL |
| 28. |
Khasabov SG, Rogers SD, Ghilardi JR, Peters CM, Mantyh PW, Simone DA. Spinal neurons that possess the substance P receptor are required for the development of central sensitization. J Neurosci 2002; 22: 9086-98.
PMID |
| 29. | Chen G, Zhang YQ, Qadri YJ, Serhan CN, Ji RR. Microglia in pain: detrimental and protective roles in pathogenesis and resolution of pain. Neuron 2018; 100: 1292-311. |
| 30. |
Inoue K, Tsuda M. Microglia in neuropathic pain: cellular and molecular mechanisms and therapeutic potential. Nat Rev Neurosci 2018; 19: 138-12.
DOI PMID |
| 31. |
Wieseler J, Ellis A, McFadden A, et al. Supradural inflammatory soup in awake and freely moving rats induces facial allodynia that is blocked by putative immune modulators. Brain Res 2017; 1664: 87-94.
DOI PMID |
| 32. |
McWhorter FY, Wang T, Nguyen P, Chung T, Liu WF. Modulation of macrophage phenotype by cell shape. Proc Natl Acad Sci U S A 2013; 110: 17253-8.
DOI URL |
| 33. |
Kobayashi K, Imagama S, Ohgomori T, et al. Minocycline selectively inhibits M1 polarization of microglia. Cell Death Dis 2013; 4: e525.
DOI |
| 34. |
Hu XM, Leak RK, Shi YJ, et al. Microglial and macrophage polarization—new prospects for brain repair. Nat Rev Neurol 2015; 11: 56-64.
DOI PMID |
| 35. |
Xia CY, Zhang S, Chu SF, et al. Autophagic flux regulates microglial phenotype according to the time of oxygen-glucose deprivation/reperfusion. Int Immunopharmacol 2016; 39: 140-8.
DOI URL |
| 36. |
Ma K, Guo JJ, Wang G, Ni QY, Liu XJ. Toll-like receptor 2-mediated autophagy promotes microglial cell death by modulating the microglial M1/M2 phenotype. Inflammation 2020; 43: 701-11.
DOI PMID |
| 37. |
Debnath J, Gammoh N, Ryan KM. Autophagy and autophagy-related pathways in cancer. Nat Rev Mol Cell Biol 2023; 24: 560-75.
DOI |
| 38. |
Klionsky DJ, Abdalla FC, Abeliovich H, et al. Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 2012; 8: 445-544.
PMID |
| 39. |
Huang R, Xu YF, Wan W, et al. Deacetylation of nuclear LC3 drives autophagy initiation under starvation. Mol Cell 2015; 57: 456-66.
DOI PMID |
| 40. |
You ZY, Xu YF, Wan W, et al. TP53INP2 contributes to autophagosome formation by promoting LC3-ATG7 interaction. Autophagy 2019; 15: 1309-21.
DOI PMID |
| 41. |
Kellermann M, Scharte F, Hensel M. Manipulation of host cell organelles by intracellular pathogens. Int J Mol Sci 2021; 22: 6484.
DOI URL |
| 42. |
Cao WY, Li JH, Yang KP, Cao DL. An overview of autophagy: mechanism, regulation and research progress. Bull Cancer 2021; 108: 304-22.
DOI PMID |
| 43. |
Chessell IP, Hatcher JP, Bountra C, et al. Disruption of the P2X7 purinoceptor gene abolishes chronic inflammatory and neuropathic pain. Pain 2005; 114: 386-96.
DOI PMID |
| 44. |
Broom DC, Matson DJ, Bradshaw E, et al. Characterization of N-(adamantan-1-ylmethyl)-5-[(3R-amino-pyrrolidin-1-yl)methyl]-2-chloro-benzamide, a P2X7 antagonist in animal models of pain and inflammation. J Pharmacol Exp Ther 2008; 327: 620-33.
DOI PMID |
| 45. |
Perez-Medrano A, Donnelly-Roberts DL, Honore P, et al. Discovery and biological evaluation of novel cyanoguanidine P2X(7) antagonists with analgesic activity in a rat model of neuropathic pain. J Med Chem 2009; 52: 3366-76.
DOI PMID |
| 46. |
Zhang Y, Huang LM, Kozlov SA, Rubini P, Tang Y, Illes P. Acupuncture alleviates acid- and purine-induced pain in rodents. Br J Pharmacol 2020; 177: 77-92.
DOI URL |
| 47. |
Tewari M, Seth P. Emerging role of P2X7 receptors in CNS health and disease. Ageing Res Rev 2015; 24: 328-42.
DOI PMID |
| 48. |
Jacobson KA, Gao ZG. Adenosine receptors as therapeutic targets. Nat Rev Drug Discov 2006; 5: 247-64.
DOI PMID |
| 49. |
Jarvis MF, Khakh BS. ATP-gated P2X cation-channels. Neuropharmacology 2009; 56: 208-15.
DOI PMID |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||


