Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (1): 49-57.DOI: 10.19852/j.cnki.jtcm.20210707.002
• Research Articles • Previous Articles Next Articles
Therapeutic effects of salidroside vs pyrrolidine dithiocarbamate against severe acute pancreatitis in rats
Jing QIAN1, Xiaohong WANG2( ), Benzhong WEI3, Guoxiong ZHOU4, Shunxing ZHU5, Chun LIU5
), Benzhong WEI3, Guoxiong ZHOU4, Shunxing ZHU5, Chun LIU5
- 1 Department of General Surgery, Yizheng Hospital of Nanjing Drum Tower Hospital Group, Yizheng 211900, China
2 Department of Gastroenterology, Yizheng Hospital of Nanjing Drum Tower Hospital Group, Yizheng 211900, China
3 Department of Anesthesiology, Yizheng Hospital of Nanjing Drum Tower Hospital Group, Yizheng 211900, China
4 Department of Gastroenterology, Affiliated Hospital of Nantong University, Nantong 226001, China
5 Department of Laboratory Animal Center, Nantong University, Nantong 226001, China
-
Received:2021-01-09Accepted:2021-04-03Online:2022-02-15Published:2021-07-07 -
Contact:Xiaohong WANG -
About author:Dr. WANG Xiaohong, Department of Gastroenterology, Yizheng Hospital of Nanjing Drum Tower Hospital Group, Yizheng 211900, China. wx102474wx@163.com
-
Supported by:Traditional Chinese Medicine Technology Development Plan Project of Jiangsu Province in 2020: Basic and Clinical Study on Salidroside in the Adjuvant Treatment of Severe Acute Pancreatitis(YB2020088);Health Innovation Project of Lvyang Jinfeng Plan of Yangzhou City in 2020: Basic and Clinical Study on Salidroside in the Adjuvant Treatment of Severe Acute Pancreatitis(LJ202037)
Cite this article
Jing QIAN, Xiaohong WANG, Benzhong WEI, Guoxiong ZHOU, Shunxing ZHU, Chun LIU. Therapeutic effects of salidroside vs pyrrolidine dithiocarbamate against severe acute pancreatitis in rats[J]. Journal of Traditional Chinese Medicine, 2022, 42(1): 49-57.
share this article
| Gene name | Forward primer (5’-3’) | Reverse primer (5’-3’) | |
|---|---|---|---|
| Beclin-1 | CCTCTTCCCTGGAAACAACTAA | TAGTTAGCACAGTAAGCGTTCA | |
| LC3II | ACCAAGTTCCTTGTACCTGA | CTCTCTCGCTCTCGTACACT | |
| LAMP2 | ACAACATCACGCTTTCTTAC | CTCACTGTACCATTTTGGAC | |
| IRAK1 | CCAGCCAGTGGAGAGTGATG | CTGGGCCACTCCTCAAACTT | |
| IκBα | CTGTTGAAGTGTGGGGCTGA | ACACACAGTCATCGTAGGGC | |
| p65 | ACGATCTGTTTCCCCTCATCT | TGCTTCTCTCCCCAGGAATA | |
| GAPDH | CATGGTCTACATGTTCCAGT | GGCTAAGCAGTTGGTGGTGC | |
Table 1 Primers for quantitative real-time polymerase chain reaction
| Gene name | Forward primer (5’-3’) | Reverse primer (5’-3’) | |
|---|---|---|---|
| Beclin-1 | CCTCTTCCCTGGAAACAACTAA | TAGTTAGCACAGTAAGCGTTCA | |
| LC3II | ACCAAGTTCCTTGTACCTGA | CTCTCTCGCTCTCGTACACT | |
| LAMP2 | ACAACATCACGCTTTCTTAC | CTCACTGTACCATTTTGGAC | |
| IRAK1 | CCAGCCAGTGGAGAGTGATG | CTGGGCCACTCCTCAAACTT | |
| IκBα | CTGTTGAAGTGTGGGGCTGA | ACACACAGTCATCGTAGGGC | |
| p65 | ACGATCTGTTTCCCCTCATCT | TGCTTCTCTCCCCAGGAATA | |
| GAPDH | CATGGTCTACATGTTCCAGT | GGCTAAGCAGTTGGTGGTGC | |

Figure 1 Pathological changes of the pancreas detected by HE staining (×200) A: SO group at 3 h; B: SO group at 24 h; C: SAP group at 3 h; D: SAP group at 24 h; E: Sal L + S group at 24 h; F: Sal M+S group at 24 h; G: Sal H + S group at 24 h; H: PDTC + S group at 24 h. HE: hematoxylin-eosin staining; SO: sham operation; Sal: salidroside; PDTC: pyrrolidine dithiocarbamate; SAP: severe acute pancreatitis. SAP group: the rats were treated with 0.1 mL/min speed uniformly retrograde infusion of a freshly prepared 3.5% sodium taurocholate solution (0.1 mL/100 g) into the biliopancreatic duct after laparotomy. SO group: the rats were treated with an equivalent volume of normal saline solution as the 3.5% sodium taurocholate solution in the SAP group. Sal L + S group: SAP rats were treated with low-dose (5 mg/kg) Sal; Sal M + S group: SAP rats were treated with middle-dose (10 mg/kg) Sal; Sal H + S group: SAP rats were treated with high-dose (20 mg/kg) Sal; PDTC + S group: SAP rats were treated with PDTC (100 mg/kg).
| Group | n | Pancreas pathological scores (points) | Serum amylase level (U/L) |
|---|---|---|---|
| SO 3 h | 10 | 0.9±0.6a | 471.2±37.9a |
| SO 24 h | 10 | 1.1±0.3a | 482.7±24.5a |
| SAP 3 h | 10 | 4.3±0.8b | 1845±194.8b |
| SAP 24 h | 6 | 10.3±1.6b,c | 4208±177.3b,c |
| Sal L+S 24 h | 6 | 9.8±0.9b | 4162±209.2b |
| Sal M+S 24 h | 7 | 8.4±0.5a,b,d | 3933±198.8a,b,f |
| Sal H+S 24 h | 8 | 7.0±0.9a,b,e | 3557±188.5a,b,e |
| PDTC+S 24 h | 8 | 7.3±0.9a,b | 3591±192.8a,b |
Table 2 Comparison of pancreas pathological scores and serum amylase level in 8 groups ($\bar{x}$± s)
| Group | n | Pancreas pathological scores (points) | Serum amylase level (U/L) |
|---|---|---|---|
| SO 3 h | 10 | 0.9±0.6a | 471.2±37.9a |
| SO 24 h | 10 | 1.1±0.3a | 482.7±24.5a |
| SAP 3 h | 10 | 4.3±0.8b | 1845±194.8b |
| SAP 24 h | 6 | 10.3±1.6b,c | 4208±177.3b,c |
| Sal L+S 24 h | 6 | 9.8±0.9b | 4162±209.2b |
| Sal M+S 24 h | 7 | 8.4±0.5a,b,d | 3933±198.8a,b,f |
| Sal H+S 24 h | 8 | 7.0±0.9a,b,e | 3557±188.5a,b,e |
| PDTC+S 24 h | 8 | 7.3±0.9a,b | 3591±192.8a,b |
| Groups | n | Serum TNF-α contents (pg/mL) | Serum IL-1β contents (pg/ml) | Serum IL-10 contents (pg/ml) |
|---|---|---|---|---|
| SO 3 h | 10 | 14.5±4.9a | 16.0±3.1a | 20.9±3.8a |
| SO 24 h | 10 | 15.1±4.7a | 16.5±3.6a | 19.7±2.9a |
| SAP 3 h | 10 | 67.8±9.9b | 32.5±6.1b | 16.3±3.4b |
| SAP 24 h | 6 | 167.1±13.3b,c | 52.5±7.4b,c | 6.0±1.9b,c |
| Sal L+S 24 h | 6 | 165.6±10.0b | 53.4±8.8b | 6.1±2.1b |
| Sal M+S 24 h | 7 | 121.9±11.9a,b,d | 45.7±4.0b,a1,d1 | 9.8±2.1b,a1,d1 |
| Sal H+S 24 h | 8 | 92.7±10.4a,b,e | 38.6±4.8a,b,e1 | 13.0±2.6a,b,e1 |
| PDTC+S 24 h | 8 | 90.1±15.2a,b | 39.2±5.3a,b | 13.1±2.4a,b |
Table 3 Comparison of serum TNF-α, IL-1 β and IL-10 contents in 8 groups ($\bar{x}$± s)
| Groups | n | Serum TNF-α contents (pg/mL) | Serum IL-1β contents (pg/ml) | Serum IL-10 contents (pg/ml) |
|---|---|---|---|---|
| SO 3 h | 10 | 14.5±4.9a | 16.0±3.1a | 20.9±3.8a |
| SO 24 h | 10 | 15.1±4.7a | 16.5±3.6a | 19.7±2.9a |
| SAP 3 h | 10 | 67.8±9.9b | 32.5±6.1b | 16.3±3.4b |
| SAP 24 h | 6 | 167.1±13.3b,c | 52.5±7.4b,c | 6.0±1.9b,c |
| Sal L+S 24 h | 6 | 165.6±10.0b | 53.4±8.8b | 6.1±2.1b |
| Sal M+S 24 h | 7 | 121.9±11.9a,b,d | 45.7±4.0b,a1,d1 | 9.8±2.1b,a1,d1 |
| Sal H+S 24 h | 8 | 92.7±10.4a,b,e | 38.6±4.8a,b,e1 | 13.0±2.6a,b,e1 |
| PDTC+S 24 h | 8 | 90.1±15.2a,b | 39.2±5.3a,b | 13.1±2.4a,b |

Figure 2 Performance of the pancreas by TEM (×2000) A: SAP group at 3 h; B: SAP group at 24 h; C: SO group at 3 h; D: SO group at 24 h; E: Sal L + S group at 24 h; F: Sal M+S group at 24 h; G: Sal H + S group at 24 h; H: PDTC + S group at 24 h. SO: sham operation; Sal: salidroside; PDTC: pyrrolidine dithiocarbamate; SAP: severe acute pancreatitis; SAP group: the rats were treated with 0.1 mL/min speed uniformly retrograde infusion of a freshly prepared 3.5% sodium taurocholate solution (0.1 mL/100 g) into the biliopancreatic duct after laparotomy. SO group: the rats were treated with an equivalent volume of normal saline solution as the 3.5% sodium taurocholate solution in the SAP group. Sal L + S group: SAP rats were treated with low-dose (5 mg/kg) Sal; Sal M + S group: SAP rats were treated with middle-dose (10 mg/kg) Sal; Sal H + S group: SAP rats were treated with high-dose (20 mg/kg) Sal; PDTC + S group: SAP rats were treated with PDTC (100 mg/kg).
| Group | n | Beclin-1/GAPDH | LC3II/GAPDH | LAMP2/GAPDH |
|---|---|---|---|---|
| SO 3 h | 10 | 1.06±0.07a | 0.97±0.14a | 0.99±0.10a |
| SO 24 h | 10 | 1.08±0.13a | 1.02±0.14a | 1.02±0.16a |
| SAP 3 h | 10 | 1.61±0.10b | 1.55±0.14b | 0.69±0.11b |
| SAP 24 h | 6 | 2.43±0.07bc | 2.52±0.29bc | 0.24±0.10bc |
| Sal L+S 24 h | 6 | 2.38±0.15b | 2.48±0.19b | 0.26±0.09b |
| Sal M+S 24 h | 7 | 2.06±0.08abd | 2.19±0.20abd1 | 0.42±0.09abd1 |
| Sal H+S 24 h | 8 | 1.78±0.11abe | 1.88±0.20abe | 0.54±0.10abe1 |
| PDTC+S 24 h | 8 | 1.80±0.15ab | 1.93±0.26ab | 0.58±0.12ab |
Table 4 Comparison of Beclin-1 mRNA-GAPDH ratio, LC3II mRNA-GAPDH ratio and LAMP2 mRNA-GAPDH ratio in the pancreas tissue in 8 groups ($\bar{x}$± s)
| Group | n | Beclin-1/GAPDH | LC3II/GAPDH | LAMP2/GAPDH |
|---|---|---|---|---|
| SO 3 h | 10 | 1.06±0.07a | 0.97±0.14a | 0.99±0.10a |
| SO 24 h | 10 | 1.08±0.13a | 1.02±0.14a | 1.02±0.16a |
| SAP 3 h | 10 | 1.61±0.10b | 1.55±0.14b | 0.69±0.11b |
| SAP 24 h | 6 | 2.43±0.07bc | 2.52±0.29bc | 0.24±0.10bc |
| Sal L+S 24 h | 6 | 2.38±0.15b | 2.48±0.19b | 0.26±0.09b |
| Sal M+S 24 h | 7 | 2.06±0.08abd | 2.19±0.20abd1 | 0.42±0.09abd1 |
| Sal H+S 24 h | 8 | 1.78±0.11abe | 1.88±0.20abe | 0.54±0.10abe1 |
| PDTC+S 24 h | 8 | 1.80±0.15ab | 1.93±0.26ab | 0.58±0.12ab |
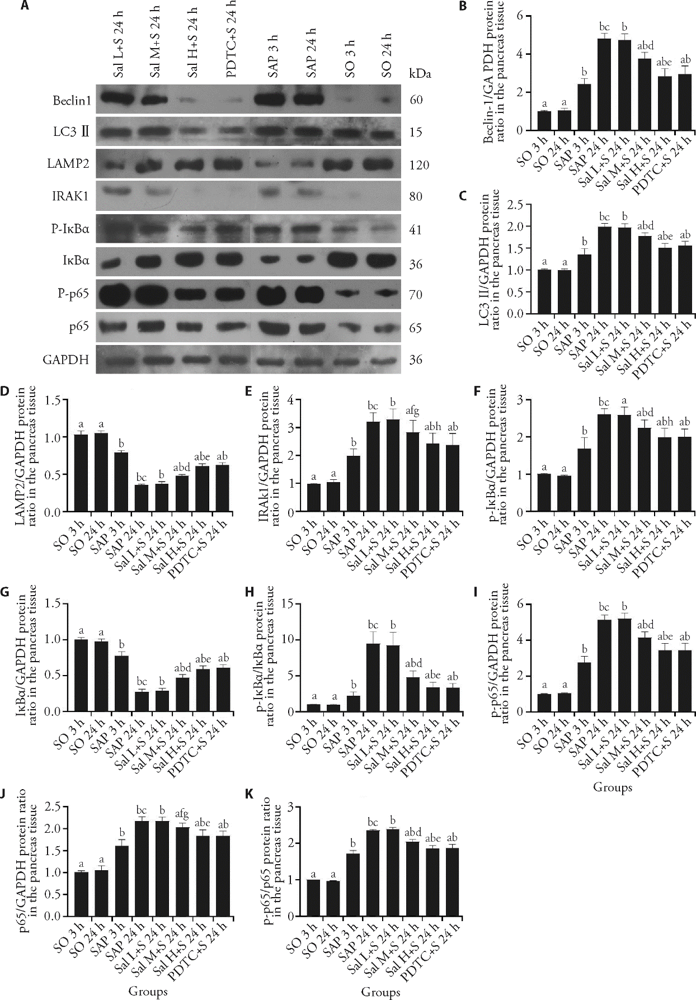
Figure 3 Protein levels of Beclin-1, LC3Ⅱ, LAMP2, IRAK1, IκBα, p-IκBα, p65 and p-p65 in each group A: example blots of Beclin-1, LC3Ⅱ, LAMP2, IRAK1, p-IκBα, IκBα, p-p65, p65 and GAPDH; B: relative protein level of Beclin-1; C: relative protein level of LC3Ⅱ; D: relative protein level of LAMP2; E: relative protein level of IRAK1; F: relative protein level of p-IκBα; G: relative protein level of IκBα; H: relative protein level of p-IκBα/IκBα; I: relative protein level of p-p65; J: relative protein level of p65; K: relative protein level of p-p65/p65. SAP group: the rats were treated with 0.1 mL/min speed uniformly retrograde infusion of a freshly prepared 3.5% sodium taurocholate solution (0.1 mL/100 g) into the biliopancreatic duct after laparotomy. SO group: the rats were treated with an equivalent volume of normal saline solution as the 3.5% sodium taurocholate solution in the SAP group. Sal L + S group: SAP rats were treated with low-dose (5 mg/kg) Sal; Sal M + S group: SAP rats were treated with middle-dose (10 mg/kg) Sal; Sal H + S group: SAP rats were treated with high-dose (20 mg/kg) Sal; PDTC + S group: SAP rats were treated with PDTC (100 mg/kg). LC3Ⅱ: microtubule-associated protein light chain 3II; LAMP2: lysosome associated membrane protein 2; IRAK1: interleukin-1 receptor associated kinase 1; IκBα: inhibitor α of nuclear transcription factor-κB; p-IκBα: phosphorylation of inhibitor α of nuclear transcription factor-κB; p65: nuclear transcription factor-κB 65; p-p65: phosphorylation of nuclear transcription factor-κB 65; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; SO: sham operation; Sal: salidroside; PDTC: pyrrolidine dithiocarbamate; SAP: severe acute pancreatitis. bP < 0.01 vs SO group at the same time point; aP < 0.01, fP < 0.05 vs SAP group at the same time point; cP < 0.01 24 h vs 3 h in SAP group; dP < 0.01, gP < 0.05 Sal L + S 24 h group vs Sal M + S 24 h group; eP < 0.01, hP < 0.05 Sal M+S 24 h group vs Sal H + S 24 h group.
| Group | n | IRAK1/GAPDH | IκBα/GAPDH | p65/GAPDH |
|---|---|---|---|---|
| SO 3 h group | 10 | 1.02±0.04b | 0.98±0.12b | 0.95±0.13b |
| SO 24 h group | 10 | 1.05±0.11b | 1.01±0.15b | 1.02±0.17b |
| SAP 3 h group | 10 | 1.51±0.12a | 0.79±0.13a | 1.93±0.15a |
| SAP 24 h group | 6 | 2.47±0.13a,c | 0.33±0.10a,c | 3.11±0.34a,c |
| Sal L+S 24 h group | 6 | 2.44±0.13a | 0.32±0.10a | 3.05±0.29a |
| Sal M+S 24 h group | 7 | 2.03±0.11a,b,d | 0.49±0.12a,b1,d1 | 2.69±0.27a,b,d |
| Sal H+S 24 h group | 8 | 1.75±0.13a,b,e | 0.63±0.16a,b,e1 | 2.24±0.14a,b,e |
| PDTC+S 24 h group | 8 | 1.69±0.19a,b | 0.63±0.16a,b | 2.31±0.15a,b |
Table 5 Comparison of IRAK1 mRNA-GAPDH ratio, IκBα mRNA-GAPDH ratio, and p65 mRNA-GAPDH ratio in the pancreas tissue in 8 groups ($\bar{x}$s± s)
| Group | n | IRAK1/GAPDH | IκBα/GAPDH | p65/GAPDH |
|---|---|---|---|---|
| SO 3 h group | 10 | 1.02±0.04b | 0.98±0.12b | 0.95±0.13b |
| SO 24 h group | 10 | 1.05±0.11b | 1.01±0.15b | 1.02±0.17b |
| SAP 3 h group | 10 | 1.51±0.12a | 0.79±0.13a | 1.93±0.15a |
| SAP 24 h group | 6 | 2.47±0.13a,c | 0.33±0.10a,c | 3.11±0.34a,c |
| Sal L+S 24 h group | 6 | 2.44±0.13a | 0.32±0.10a | 3.05±0.29a |
| Sal M+S 24 h group | 7 | 2.03±0.11a,b,d | 0.49±0.12a,b1,d1 | 2.69±0.27a,b,d |
| Sal H+S 24 h group | 8 | 1.75±0.13a,b,e | 0.63±0.16a,b,e1 | 2.24±0.14a,b,e |
| PDTC+S 24 h group | 8 | 1.69±0.19a,b | 0.63±0.16a,b | 2.31±0.15a,b |
| [1] | Yang ZW, Meng XX, Xu P. Central role of neutrophil in the pathogenesis of severe acute pancreatitis. J Cell Mol Med 2015;19:2513-20. |
| [2] | Gukovskaya AS, Gukovsky I, Algul H, Habtezion A. Autophagy, inflammation, and immune dysfunction in the pathogenesis of pancreatitis. Gastroenterology 2017;153:1212-26. |
| [3] | Qie T, Xu P, Zhang BX, Cao XB. The effects of salidroside on the apoptosis pathway of myocardial cells in acute exhausted rats. Zhong Guo Ying Yong Sheng Li Xue Za Zhi 2019;35:376-80. |
| [4] | Dong X, Zhang X, Li D, et al. Protective effect of salidroside against high altitude hypoxia-induced brain injury in rats. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 2015;31:1327-31. |
| [5] | Chen D, Lu D, Liu H, et al. Pharmacological blockade of PCAF ameliorates osteoarthritis development via dual inhibition of TNF-alpha-driven inflammation and ER stress. EBioMedicine 2019;50:395-407. |
| [6] | You B, Dun Y, Zhang W, et al. Anti-insulin resistance effects of salidroside through mitochondrial quality control. J Endocrinol 2020;244:383-93. |
| [7] | Zhang Y, Yue Q, Wen Z, et al. Salidroside induces anti-tumor effect in dendritic cells via ERK pathway. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 2019;35:979-85. |
| [8] | Liao ZL, Su H, Tan YF, et al. Salidroside protects PC-12 cells against amyloid beta-induced apoptosis by activation of the ERK1/2 and AKT signaling pathways. Int J Mol Med 2019;43:1769-77. |
| [9] | Hu R, Wang MQ, Ni SH, et al. Salidroside ameliorates endothelial inflammation and oxidative stress by regulating the AMPK/NF-kappaB/NLRP3 signaling pathway in AGEs-induced HUVECs. Eur J Pharmacol 2020;867:172797. |
| [10] | Pu WL, Zhang MY, Bai RY, et al. Anti-inflammatory effects of Rhodiola rosea L.: a review. Biomed Pharmacother 2020;121:109552. |
| [11] | Chen P, Liu J, Ruan H, et al. Protective effects of Salidroside on cardiac function in mice with myocardial infarction. Sci Rep 2019;9:18127. |
| [12] | Lin SY, Dan X, Du XX, et al. Protective effects of salidroside against carbon tetrachloride (CCl4)-induced liver injury by initiating mitochondria to resist oxidative stress in mice. Int J Mol Sci 2019;20(13):E3187. |
| [13] | Huang X, Xue H, Ma J, et al. Salidroside ameliorates Adriamycin nephropathy in mice by inhibiting beta-catenin activity. J Cell Mol Med 2019;23:4443-53. |
| [14] | Deheng C, Kailiang Z, Weidong W, et al. Salidroside promotes random skin flap survival in rats by enhancing angiogenesis and inhibiting apoptosis. J Reconstr Microsurg 2016;32:580-6. |
| [15] | Zhang D, Cao L, Wang Z, et al. Salidroside mitigates skeletal muscle atrophy in rats with cigarette smoke-induced COPD by up-regulating myogenin and down-regulating myostatin expression. Biosci Rep 2019; 39: BSR20190440. |
| [16] | Zhang X, Lai W, Ying X, et al. Salidroside reduces inflammation and brain injury after permanent middle cerebral artery occlusion in rats by regulating PI3K/PKB/Nrf2/NFkappa B signaling rather than complement C3 activity. Inflammation 2019;42:1830-42. |
| [17] | Zhu YP, Zhu JB, Ma XL, Tian YJ, Wan XY, Zhang TB. Evaluation for developmental toxicity of Salidroside injection in rats. Zhong Guo Xin Yao Za Zhi 2009;18:2069-71. |
| [18] | Zhu YP, Zhang TB, Wan XY, et al. Genetic toxicity of Salidroside injection. Pharm Care Res 2009;9:279-82. |
| [19] | Chen S, Cai F, Wang J, et al. Salidroside protects SHSY5Y from pathogenic alpha synuclein by promoting cell autophagy via mediation of mTOR/p70S6K signaling. Mol Med Rep 2019;20:529-38. |
| [20] | Feng J, Zhang Q, Mo W, et al. Salidroside pretreatment attenuates apoptosis and autophagy during hepatic ischemia-reperfusion injury by inhibiting the mitogen-activated protein kinase pathway in mice. Drug Des Devel Ther 2017;11:1989-2006. |
| [21] | Feng J, Niu P, Chen K, et al. Salidroside mediates apoptosis and autophagy inhibition in concanavalin A-induced liver injury. Exp Ther Med 2018;15:4599-614. |
| [22] | Feng J, Chen K, Xia Y, et al. Salidroside ameliorates autophagy and activation of hepatic stellate cells in mice via NF-kappa B and TGF-beta1/Smad3 pathways. Drug Des Devel Ther 2018;12:1837-53. |
| [23] | Lan KC, Chao SC, Wu HY, et al. Salidroside ameliorates sepsis-induced acute lung injury and mortality via downregulating NF-kappa B and HMGB1 pathways through the upregulation of SIRT1. Sci Rep 2017;7:12026. |
| [24] | Chang X, Luo F, Jiang W, et al. Protective activity of salidroside against ethanol-induced gastric ulcer via the MAPK/NF-kappa B pathway in vivo and in vitro. Int Immunopharmacol 2015;28:604-615. |
| [25] | Wang X, Zhou G, Liu C, et al. Acanthopanax versus 3-methyladenine ameliorates sodium taurocholate-induced severe acute pancreatitis by inhibiting the autophagic pathway in rats. Mediators Inflamm 2016;2016(12):8369704. |
| [26] | Wang X, Chu L, Liu C, et al. Therapeutic effects of Saussurea involucrata injection against severe acute pancreatitis- induced brain injury in rats. Biomed Pharmacother 2018;100:564-74. |
| [27] | Schmidt J, Rattner DW, Lewandrowski K, et al. A better model of acute pancreatitis for evaluating therapy. Ann Surg 1992;215:44-56. |
| [28] | Wang P, Zhu Q, Wu N, et al. Tyrosol attenuates ischemia-reperfusion-induced kidney injury via inhibition of inducible nitric oxide synthase. J Agric Food Chem 2013;61(15):3669-75. |
| [29] | Netea-Maier RT, Plantinga TS, van de Veerdonk FL, Smit JW, Netea MG. Modulation of inflammation by autophagy: Consequences for human disease. Autophagy 2016;12:245-60. |
| [30] | Pérez S, Pereda J, Sabater L, Sastre J. Redox signaling in acute pancreatitis. Redox Biol 2015;5:1-14. |
| [31] | Antonucci L, Fagman JB, Kim JY, et al. Basal autophagy maintains pancreatic acinar cell homeostasis and protein synthesis and prevents ER stress. Proc Natl Acad Sci USA 2015;112:E6166-74. |
| [32] | Mareninova OA, Sendler M, Malla SR, et al. Lysosome associated membrane proteins maintain pancreatic acinar cell homeostasis: LAMP-2 deficient mice develop pancreatitis. Cell Mol Gastroenterol Hepatol 2015;1:678-94. |
| [33] | Wan J, Chen J, Wu D, et al. Regulation of autophagy affects the prognosis of mice with severe acute pancreatitis. Dig Dis Sci 2018;63:2639-50. |
| [34] | Yin WY, Ye Q, Huang HJ, et al. Salidroside protects cortical neurons against glutamate-induced cytotoxicity by inhibiting autophagy. Mol Cell Biochem 2016;419:53-64. |
| [35] | Deng AQ, Ma LM, Wang X, Wang SY. Effects of salidroside on expression levels of LC3 and Beclin-1 in HT22 cells injured by sodium nitroprusside. Nan Tong Da Xue Xue Bao (Yi Xue Ban) 2016;36:403-7. |
| [36] | Li D, Mao XN, Xu YB, Yao J, Zhang PJ. Salidroside in hepatic ischemia-reperfusion injury in rats and its induced autophagy reaction. Guo Ji Zhong Yi Zhong Yao Za Zhi 2018;40:432-6. |
| [37] | Kondylis V, Kumari S, Vlantis K, Pasparakis M. The interplay of IKK, NF-kappaB and RIPK1 signaling in the regulation of cell death, tissue homeostasis and inflammation. Immunol Rev 2017;277:113-27. |
| [38] | Wang X, Zhuang X, Wei R, et al. Protective effects of Acanthopanax vs Ulinastatin against severe acute pancreatitis-induced brain injury in rats. Int Immunopharmacol 2015;24:285-98. |
| [39] | Yang S, Bing M, Chen F, et al. Autophagy regulation by the nuclear factor kappa B signal axis in acute pancreatitis. Pancreas 2012;41:367-73. |
| [40] | Yan GH, Choi YH. Salidroside attenuates allergic airway inflammation through negative regulation of nuclear factor-kappa B and p38 mitogen-activated protein kinase. J Pharmacol Sci 2014;126:126-35. |
| [41] | Lu R, Wu Y, Guo H, Huang X. Salidroside protects lipopolysaccharide-induced acute lung injury in mice. Dose Response 2016;14:1559325816678492. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

