Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (1): 27-34.DOI: 10.19852/j.cnki.jtcm.20231024.002
• Original articles • Previous Articles Next Articles
B-cell lymphoma-2 phosphorylation at Ser70 site-related autophagy mediates puerarin-inhibited the apoptosis of MC3T3-E1 cells during osteoblastogenesis
LI Xi1,2, LIN Xiangquan1,2, CHEN Dongdong1,2, LIU Hui1,2( )
)
- 1 the Third Clinical Medical College, Fujian Medical University
2 Department of Orthopedics, Fuzhou Second Hospital, Fuzhou 350007, China
-
Received:2022-09-14Accepted:2022-12-22Online:2024-02-15Published:2023-10-24 -
Contact:LIU Hui, Department of orthopedics, Fuzhou Second Hospital, Fuzhou 350007, China. Liuhuifzse@163.com. Telephone: +86-591-22169038 -
Supported by:Fujian Provincial Natural Science Foundation Project: Exploration of Crisis Warning Values for Intraoperative Electrophysiological Monitoring in High-risk Cervical Spondylosis Patients on the Brink of Paralysis(2019J01548);Fujian Provincial Clinical Medical Research Center for First Aid and Rehabilitation in Orthopaedic Trauma: Study on the Treatment of Osteoporosis(2020Y2014)
Cite this article
LI Xi, LIN Xiangquan, CHEN Dongdong, LIU Hui. B-cell lymphoma-2 phosphorylation at Ser70 site-related autophagy mediates puerarin-inhibited the apoptosis of MC3T3-E1 cells during osteoblastogenesis[J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 27-34.
share this article
| Gene | Forward(5'-3') | Reverse(5'-3') |
|---|---|---|
| OCN | AGCAGCTTGGCCCAGACCTA | TAGCGCCGGAGTCTGTTCACTAC |
| Runx2 | CCCAGCCACCTTTACCTACA | TATGGAGTGCTGCTGGTCTG |
| COL1 Osterix GAPDH | AGAACAGCGTGGCCT ATGGCGTCCTCTCTGCTTG ACCACAGTCCATGCCATCAC | TCCGGTGTGACTCGT TGAAAGGTCAGCGTATGGCTT TCCACCACCCTGTTGCTGTA |
Table 1 Specific primer sequences for qRT-PCR
| Gene | Forward(5'-3') | Reverse(5'-3') |
|---|---|---|
| OCN | AGCAGCTTGGCCCAGACCTA | TAGCGCCGGAGTCTGTTCACTAC |
| Runx2 | CCCAGCCACCTTTACCTACA | TATGGAGTGCTGCTGGTCTG |
| COL1 Osterix GAPDH | AGAACAGCGTGGCCT ATGGCGTCCTCTCTGCTTG ACCACAGTCCATGCCATCAC | TCCGGTGTGACTCGT TGAAAGGTCAGCGTATGGCTT TCCACCACCCTGTTGCTGTA |
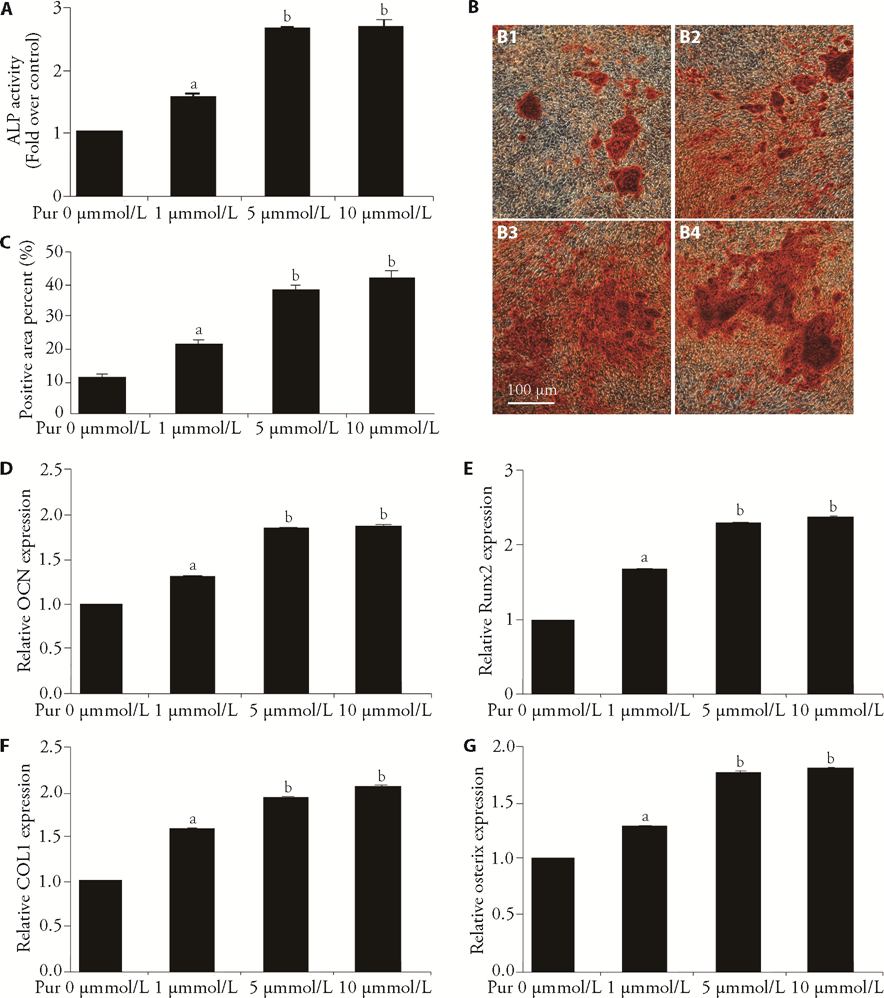
Figure 1 Puerarin promotes osteoblast differentiation A: after incubation with different concentrations of puerarin (0, 1, 5, 10 μM) for 7 d in the presence of osteoblast-inducing reagent, osteoblastic differentiation level of MC3T3-E1 cells was measured by detecting ALP activity. B-C: After treatment as described in A, mineralized nodules of differentiated osteoblasts were assessed using Alizarin red staining and quantified with ImageJ 1.8.0 software (5 visible areas/group). Scale bar, 100 μm. B1: control group; B2: 1 μM of puerarin group; B3: 5 μM of puerarin group; B4: 10 μM of puerarin group; C: quantitative results in B. D-G: after treatment as described in A, mRNA levels of OCN, Runx2, Col1 and Osterix in differentiated osteoblasts were detected using qPCR assays. D: quantitative results of OCN; E: quantitative results of Runx2; F: quantitative results of Col1; G: quantitative results of Osterix. Pur: puerarin; μM: μmol/L; d: days; ALP: alkaline phosphatase; qPCR: quantitative polymerase chain reaction; OCN: osteocalcin; Runx2: runt-related transcription factor 2; COL1: collagen type I. All experiments include a minimum of three samples. The data are represented as the mean ± standard error of mean from three independent experiments. aP < 0.05 between control group and 1 μM of puerarin group in A, C-G; bP < 0.05 between 1 μM of puerarin group and 5 or 10 μM of puerarin group in A, C-G.

Figure 2 Puerarin enhances osteoblast precursor autophagy and inhibits osteoblast precursor apoptosis A: after treatment with different concentrations of puerarin (0, 1, 5, 10 μM) for 8 h, the protein levels of Beclin1 and LC3II in MC3T3-E1 cells were measured via Western Blotting. B: After treatment with puerarin (10 μM) for 8 h in the presence or absence of serum-free DMEM or serum-free DMEM plus E64D / Pepstain A, LC3II protein expression in MC3T3-E1 cells was measured using Western Blotting. C: After treatment with puerarin (10 μM) for 24 h, the autophagosomes (red arrows) were imaged using TEM. Scale bar, 5 μm or 2 μm. C1: control group-5 μm; C2: puerarin group-5 μm; C3: control group-2 μm; C4: puerarin group-2 μm. D: after treatment with different concentrations of puerarin for 12 h, the protein level of PARP in MC3T3-E1 cells was measured via Western Blotting. E-F: after treatment with different concentrations of puerarin for 24 h, the apoptosis level of MC3T3-E1 cells was detected via AV/PI staining. E1: control group; E2: 1 μM of puerarin group; E3: 5 μM of puerarin group; E4: 10 μM of puerarin group; F: The histogram is related to the quantitative results of apoptotic cells from each group (Annexin-A+ cells). Pur: puerarin; E: E64D; P: Pepstain A; Starv: serum-free DMEM; h: hour; DMEM: Dulbecco modified Eagle medium; TEM: transmission electron microscope; AV/PI: Annexin V-FITC/PI; PARP: poly (ADP-ribose) polymerase family, member 1. All experiments include a minimum of three samples. The data are represented as the mean ± standard error of mean from three independent experiments. aP < 0.05 between control group and puerarin group in D; aP < 0.05 between control group and 1 or 5 μM of puerarin group in G; bP < 0.05 between 5 μM of puerarin group and 10 μM of puerarin group in G.
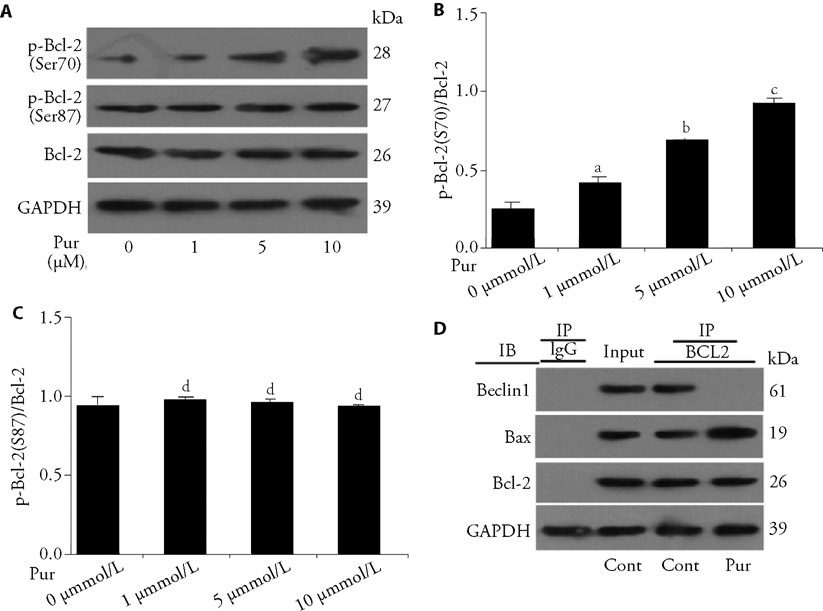
Figure 3 Puerarin alters the interaction of Bcl-2 and Beclin1/Bax in osteoblast precursors C: After treatment with different concentrations of puerarin (0, 1, 5, 10 μM) for 2 h, p-Bcl-2 (Ser70 and Ser87) expression of MC3T3-E1 cells was detected via Western Blotting. B: quantitative results of p-Bcl-2-Ser70 in A. C: quantitative results of p-Bcl-2-Ser87 in A. D: MC3T3-E1 cells were treated with puerarin (10 μM) for 2 h, and cell lysates were extracted for Co-IP with anti-Bcl-2 antibody. Next, precipitates were detected using anti-Beclin1, anti-Bax or anti-Bcl-2 antibody via Western Blotting. Pur: puerarin; Cont: control group; IP: the antibody for immunoprecipitation; IB: the antibody for immunoblot; h: hour; Ser: serine; Bcl-2: B-cell lymphoma-2; Bax: Bcl-2-associated X protein; Co-IP: Co-immunoprecipitation. All experiments include a minimum of three samples. The data are represented as the mean ± standard error of mean from three independent experiments. aP < 0.05 between control group and 1 μM of puerarin group in B; bP < 0.05 between 1 μM of puerarin group and 5 μM of puerarin group in B; cP < 0.05 between 5 μM of puerarin group and 10 μM of puerarin group in B; dP > 0.05 between control group and 1, 5 or 10 μM of puerarin group in C.

Figure 4 Puerarin increases mitochondrial membrane potential of osteoblast precursors A: MC3T3-E1 cells were treated with puerarin (0, 5, 10 μM) for 24 h, and mitochondrial membrane potential was detected via measuring the ratio of red/green fluorescence intensity using JC-10 kit. Flow cytometry was used for quantitative analysis of mitochondrial membrane potential. A1: control group; A2: 5 μM of puerarin group; A3: 10 μM of puerarin group. B-C: percentages of cells in each quadrant are displayed in the histograms according to A. B: The higher proportion of cells in H2 quadrant indicates higher mitochondrial membrane potential; C: The higher proportion of cells in the H4 quadrant indicates lower mitochondrial membrane potential. Pur: puerarin; Cont: control group; h: hour. All experiments include a minimum of three samples. The data are represented as the mean ± standard error of mean from three independent experiments. aP < 0.05 between control group and 5 μM of puerarin group in B and C; bP < 0.05 between 5 μM of puerarin group and 10 μM of puerarin group in B and C.
| 1. |
Fu R, Zhang Y, Guo Y, Zhang Y, Xu Y, Chen F. Digital gene expression analysis of the pathogenesis and therapeutic mechanisms of ligustrazine and puerarin in rat atherosclerosis. Gene 2014; 552: 75-80.
DOI PMID |
| 2. |
Cho HJ, Jun HJ, Lee JH, et al. Acute effect of high-dose isoflavones from pueraria lobata (Willd.) ohwi on lipid and bone metabolism in ovariectomized mice. Phytother Res 2012; 26: 1864-71.
DOI PMID |
| 3. |
Guo CJ, Xie JJ, Hong RH, Pan HS, Zhang FG, Liang YM. Puerarin alleviates streptozotocin (STZ)-induced osteoporosis in rats through suppressing inflammation and apoptosis via HDAC1/HDAC3 signaling. Biomed Pharmacother 2019; 115: 108570.
DOI URL |
| 4. |
Urasopon N, Hamada Y, Cherdshewasart W, Malaivijitnond S. Preventive effects of pueraria mirifica on bone loss in ovariectomized rats. Maturitas 2008; 59: 137-48.
DOI PMID |
| 5. |
Cao J, Qiu X, Gao Y, Cai L. Puerarin promotes the osteogenic differentiation of rat dental follicle cells by promoting the activation of the nitric oxide pathway. Tissue Cell 2021; 73: 101601.
DOI URL |
| 6. |
Jiang X, Chen W, Su H, Shen F, Xiao W, Sun W. Puerarin facilitates osteogenesis in steroid-induced necrosis of rabbit femoral head and osteogenesis of steroid-induced osteocytes via miR-34a upregulation. Cytokine 2021; 143: 155512.
DOI URL |
| 7. |
Zeng X, Feng Q, Zhao F, et al. Puerarin inhibits TRPM3/miR-204 to promote MC3T3-E1 cells proliferation, differentiation and mineralization. Phytother Res 2018; 32: 996-1003.
DOI PMID |
| 8. |
Vidoni C, Ferraresi A, Secomandi E, et al. Autophagy drives osteogenic differentiation of human gingival mesenchymal stem cells. Cell Commun Signal 2019; 17: 98.
DOI PMID |
| 9. |
Kang C, Wei L, Song B, et al. Involvement of autophagy in tantalum nanoparticle-induced osteoblast proliferation. Int J Nanomedicine 2017; 12: 4323-33.
DOI URL |
| 10. | Weng YM, Ke CR, Kong JZ, Chen H, Hong JJ, Zhou DS. The significant role of ATG5 in the maintenance of normal functions of Mc3T3-E1 osteoblast. Eur Rev Med Pharmacol Sci 2018; 22: 1224-32. |
| 11. | Wang L, Jiang W, Wang J, Xie Y, Wang W. Puerarin inhibits FUNDC1-mediated mitochondrial autophagy and CSE-induced apoptosis of human bronchial epithelial cells by activating PI3K/AKT/mTOR signaling pathway. Aging (Albany NY) 2022; 14: 1253-64. |
| 12. |
Han Y, Wang H, Wang Y, Dong P, Jia J, Yang S. Puerarin protects cardiomyocytes from ischemia-reperfusion injury by upregulating LncRNA ANRIL and inhibiting autophagy. Cell Tissue Res 2021; 385: 739-51.
DOI PMID |
| 13. |
Zhang G, Wang Y, Tang G, Ma Y. Puerarin inhibits the osteoclastogenesis by inhibiting RANKL-dependent and -independent autophagic responses. BMC Complement Altern Med 2019; 19: 269.
DOI |
| 14. |
Wang T, Wang L, Zhang Y, et al. Puerarin restores autophagosome-lysosome fusion to alleviate cadmium-induced autophagy blockade via restoring the expression of Rab 7 in hepatocytes. Front Pharmacol 2021; 12: 632825.
DOI URL |
| 15. | Chang X, Zhang T, Liu D, et al. Puerarin attenuates LPS-induced inflammatory responses and oxidative stress injury in human umbilical vein endothelial cells through mitochondrial quality control. Oxid Med Cell Longev 2021; 2021: 6659240. |
| 16. |
Li G, Rao H, Xu W. Puerarin plays a protective role in chondrocytes by activating Beclin1-dependent autophagy. Biosci Biotechnol Biochem 2021; 85: 621-5.
DOI URL |
| 17. |
Fitzwalter BE, Thorburn A. Recent insights into cell death and autophagy. FEBS J 2015; 282: 4279-88.
DOI PMID |
| 18. |
Fitzwalter BE, Towers CG, Sullivan KD, et al. Autophagy inhibition mediates apoptosis sensitization in cancer therapy by relieving FOXO3a turnover. Dev Cell 2018; 44: 555-65.
DOI PMID |
| 19. |
Young TM, Reyes C, Pasnikowski E, et al. Autophagy protects tumors from T cell-mediated cytotoxicity via inhibition of TNF-α-induced apoptosis. Sci Immunol 2020; 5: eabb9561.
DOI URL |
| 20. |
Lian WS, Ko JY, Chen YS, et al. Chaperonin 60 sustains osteoblast autophagy and counteracts glucocorticoid aggravation of osteoporosis by chaperoning RPTOR. Cell Death Dis 2018; 9: 938.
DOI |
| 21. |
Yue C, Jin H, Zhang X, et al. Aucubin prevents steroid-induced osteoblast apoptosis by enhancing autophagy via AMPK activation. J Cell Mol Med 2021; 25: 10175-84.
DOI URL |
| 22. |
Guo X, Liang M. Metformin alleviates dexamethasone-induced apoptosis by regulating autophagy via AMPK/mTOR/p70S6K in osteoblasts. Exp Cell Res 2022; 415: 113120.
DOI URL |
| 23. |
Ke D, Yu Y, Li C, et al. Phosphorylation of Bcl-2 at the Ser 70 site mediates RANKL-induced osteoclast precursor autophagy and osteoclastogenesis. Mol Med 2022; 28: 22.
DOI |
| 24. |
Levine B, Sinha SC, Kroemer G. Bcl-2 family members: dual regulators of apoptosis and autophagy. Autophagy 2008; 4: 600-6.
PMID |
| 25. |
Wu H, Xue Y, Zhang Y, Wang Y, Hou J. PTH1-34 promotes osteoblast formation through Beclin1-dependent autophagic activation. J Bone Miner Metab 2021; 39: 572-82.
DOI PMID |
| 26. |
Ke D, Wang X, Lin Y, et al. Lactoferrin promotes the autophagy activity during osteoblast formation via Bcl-2-Beclin1 signaling. Mol Biol Rep 2022; 49: 259-66.
DOI |
| 27. |
Wei Y, Sinha S, Levine B. Dual role of JNK1-mediated phosphorylation of Bcl-2 in autophagy and apoptosis regulation. Autophagy 2008; 4: 949-51.
DOI PMID |
| 28. |
Bassik M, Scorrano L, Oakes SA, Pozzan T, Korsmeyer SJ. Phosphorylation of BCL-2 regulates ER Ca2+ homeostasis and apoptosis. EMBO J 2004; 23: 1207-16.
DOI URL |
| 29. |
Liu J, Liu W, Lu Y, et al. Piperlongumine restores the balance of autophagy and apoptosis by increasing Bcl-2 phosphorylation in rotenone-induced parkinson disease models. Autophagy 2018; 14: 845-61.
DOI URL |
| 30. |
Saatci Ö, Borgoni S, Akbulut Ö, et al. Targeting PLK1 overcomes T-DM1 resistance via CDK1-dependent phosphorylation and inactivation of Bcl-2/xL in HER2-positive breast cancer. Oncogene 2018; 37: 2251-69.
DOI PMID |
| 31. |
Liu XA, Liao K, Liu R, et al. Tau dephosphorylation potentiates apoptosis by mechanisms involving a failed dephosphorylation/ activation of Bcl-2. J Alzheimers Dis 2010; 19: 953-62.
DOI URL |
| [1] | ZHUANG Xingxing, LIU Tao, WEI Liangbing, SONG Junmei, GAO Jiarong. Qiteng Xiaozhuo granules (芪藤消浊颗粒) medicated serum inhibits excessive proliferation and promotes apoptosis of human glomerular mesangial cells by targeting fat mass and obesity associated proteins [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1072-1080. |
| [2] | SU Chengguo, ZHAO Xiaoyan, YE Jiangnan, ZHANG Xin, JIANG Yuqing, GUO Junjie, ZHANG Xiyuan, QI Wenchuan, ZHU Jun. Effect of Tuina along “bladder meridian” alleviating intervertebral disc degeneration by regulating the transforming growth factor-β1/Smad signaling pathway in a rabbit model [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 991-1000. |
| [3] | ZHU Bin, YU Ning, WANG Lei, TIAN Yue, WU Mingfen, ZHAO Zhigang. Synergistic effect of schizandrin A and DNase I knockdown on high glucose induced beta cell apoptosis by decreasing intracellular calcium concentration [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 661-666. |
| [4] | GUO Zhaoan, SUN Lina, LIU Yingying, LI Ruifeng, LIU Chong, DIAO Ke, SHI Jing, SUN Jun. Qizhi Jiangtang capsule (芪蛭降糖胶囊) activates podocyte autophagy in diabetic kidney disease by inhibiting phosphatidylinositol 3-kinase/protein kinase B/mammalian target of rapamycin pathways [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 667-675. |
| [5] | JIANG Yiqian, ZHOU Xibin, PU Wenyuan, ZHOU Chunxiang. Sanwu Baisan decoction (三物白散) inhibits colorectal cancer progression in mice by remodeling gut microbiota and tumorigenesis [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 466-473. |
| [6] | WANG Tianqi, LI Zining, CHEN Ting, CHEN Rui, JIN Ya, Oduro Patrick kwabena, ZHANG Han, WANG Yi. Zuogui (左归丸) and Yougui pills (右归丸) improve perimenopausal syndrome via regulation of apoptosis in mice [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 474-483. |
| [7] | WU Haiyang, WANG Ying, HAN Wei, LI Huihui, JI Haisheng, LIU Xiuxiu. Protective effect of Tongdu Tiaoshen acupuncture combined with Xiaoxuming decoction (小续命汤) on dopaminergic neurons in Parkinson’s disease model [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 484-493. |
| [8] | XIE Jing, BI Zheng, WANG Sihai, SHEN Guoming, FANG Zhaohui. Danzhi Jiangtang capsule (丹蛭降糖胶囊) reduces renal injury in rats with diabetes induced by high fat diet and streptozotocin via downregulating toll-like receptor 4-nuclear factor-κB pathway and apoptosis [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 312-321. |
| [9] | DING Yajie, LIU Feng, LI Zhaoyan, Xu Yan, CAO Nida, ZHANG Guangao, WANG Rui, ZHAO Aiguang. Efficacy of luteolin on the human gastric cancer cell line MKN45 and underlying mechanism [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 34-41. |
| [10] | JIANG Wen, ZHANG Wei, ZHANG Yuxiang, YANG Hao, PAN Xiaomei, CHEN Qiang, CHEN Junhui. Tilianin extracted from Xiangqinglan (Herba Dracocephali Moldovicae) inhibits apoptosis induced by mitochondrial pathway and endoplasmic reticulum stress in H9c2 cells after oxygen-glucose deprivation/reoxygenation [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 42-50. |
| [11] | YANG Ye, ZHANG Aihui, LI Anmao. Celastrol promotes apoptotic cell death in children neuroblastoma cells through caspases dependent pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 877-884. |
| [12] | ZHENG Wei, WANG Mingxing, LIU Shanxue, LUAN Chao, ZHANG Yanqiu, XU Duoduo, WANG Jian. Buyang Huanwu Tang (补阳还五汤) protects H2O2-induced RGC-5 cell against oxidative stress and apoptosis via reactive oxygen species-mitogen-activated protein kinase signaling pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 885-891. |
| [13] | YANG Mengzhe, ZHANG Beibei, LIANG Zhenqiang, CHENG Nannan, Lü Anqiao, YANG Jianyu, GUO Xingzhe, BAI Xianyu, HUANG Yuanjiao, JIAO Aijun, XU Ning. Sanguinarine suppresses cell proliferation, migration and invasion in nasopharyngeal carcinoma via inhibiting mTOR signaling [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 687-692. |
| [14] | DING Jiamin, XING Yifeng, CHEN Zuoliang, CHEN Wanlu, MA Zhongxiong, XIE Yunde, ZHOU Lin. Qilan preparation (芪蓝颗粒) inhibits proliferation and induces apoptosis by down-regulating microRNA-21 in human Tca8113 tongue squamous cell carcinoma cells [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 693-700. |
| [15] | LI Miao, ZHENG Jialu, WANG Shuangshuang, CHEN Lei, PENG Xiao, CHEN Jinfang, AN Hongmei, HU Bing. Tenglong Buzhong granules (藤龙补中颗粒) inhibits the growth of SW620 human colon cancer in vivo [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 701-706. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

