Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (2): 176-186.DOI: 10.19852/j.cnki.jtcm.20220311.002
• Research Articles • Previous Articles Next Articles
Protective effect of resveratrol on rat cardiomyocyte H9C2 cells injured by hypoxia/reoxygenation by regulating mitochondrial autophagy via PTEN-induced putative kinase protein 1/Parkinson disease protein 2 signaling pathway
ZHAO Lixia1,2, SUN Wei3, BAI Decheng1( )
)
- 1 Institute of Integrated Traditional Chinese and Western Medicine, School of Basic Medical Sciences, Lanzhou University, Lanzhou 730000, China
2 School of Nursing, Gansu University of Chinese Medicine, Lanzhou 730000, China; Key Laboratory of Dunhuang Medicine, Ministry of Education, Gansu University of Chinese Medicine, Lanzhou 730000, China
3 Cardiac Surgery, the First Hospital of Lanzhou University, Lanzhou, Gansu 730000, China
-
Received:2021-09-12Accepted:2021-12-29Online:2022-04-15Published:2022-03-11 -
Contact:BAI Decheng -
About author:Prof. BAI Decheng, Lanzhou university, Lanzhou 730000, Gansu, China. bdc8637@163.com, Telephone: +86-13088758222
-
Supported by:Open Fund of Key Laboratory of Dunhuang Medicine, Ministry of Education(DHYX20-09);Youth Research Foundation of the Gansu University of Chinese Medicine(ZQ2017-14)
Cite this article
ZHAO Lixia, SUN Wei, BAI Decheng. Protective effect of resveratrol on rat cardiomyocyte H9C2 cells injured by hypoxia/reoxygenation by regulating mitochondrial autophagy via PTEN-induced putative kinase protein 1/Parkinson disease protein 2 signaling pathway[J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 176-186.
share this article
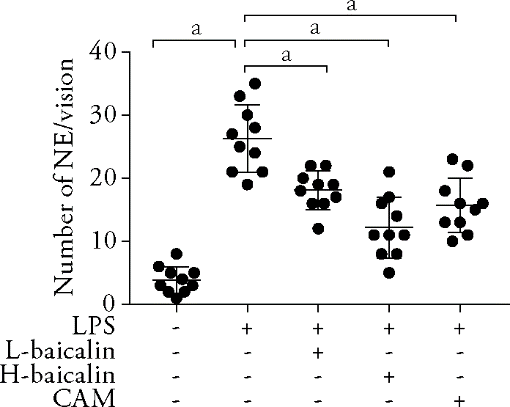
Figure 1 Resveratrol inhibited the proliferation of H9C2 cells in a time and concentration dependent manner, which was determined using an 3-(4, 5-Dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide cell survival assay H9c2 cells were treated with resveratrol at the concentrations of 0, 6.25, 12.5, 25, 50, 100 and 200 g/L for 24, 48 and 72 h, respectively.
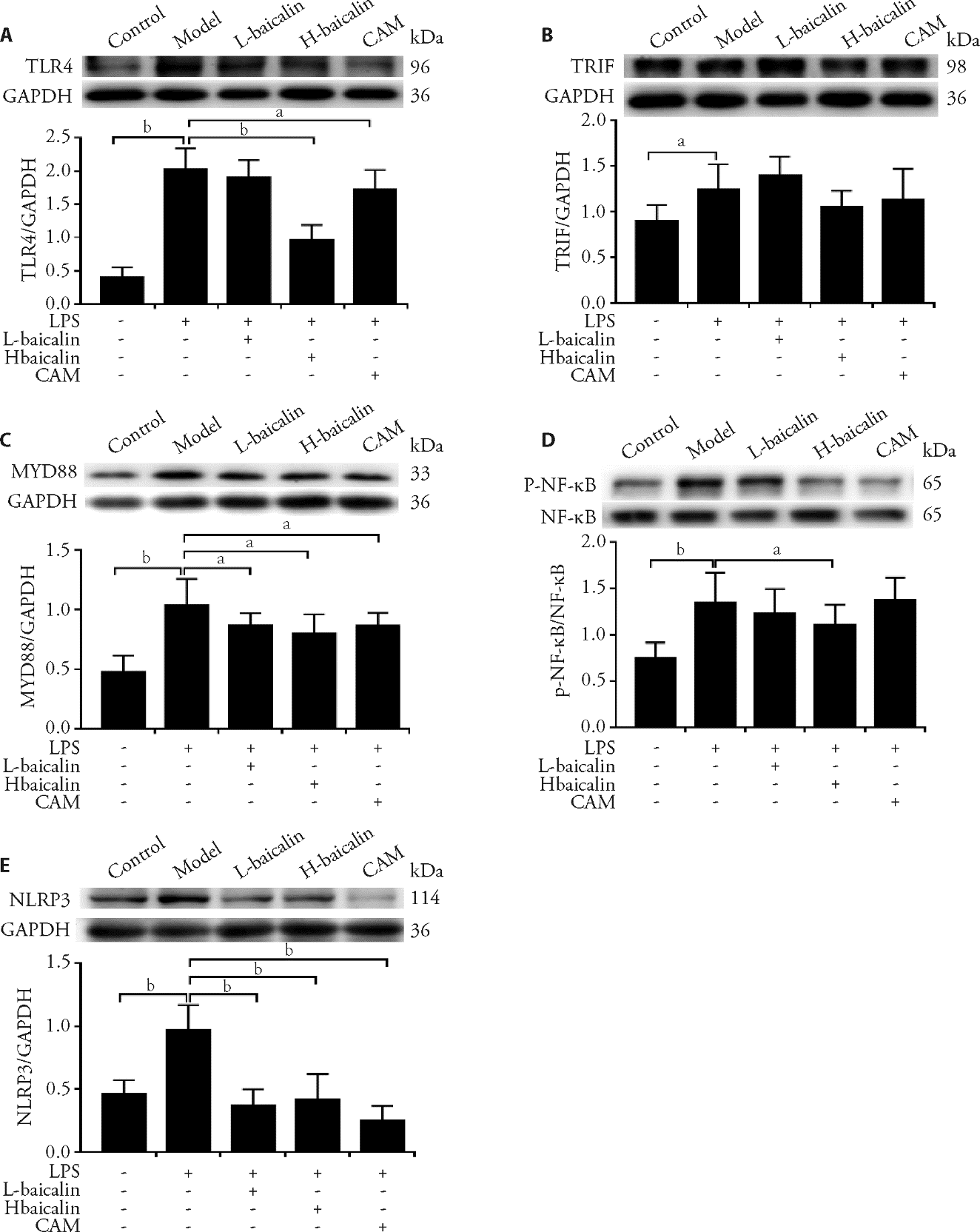
Figure 2 Resveratrol can increase decrease of the mitochondrial membrane potential induced by H/R in H9C2 cells (× 200) The mitochondrial membrane potential was detected by JC-1 staining (A, F, K: control, no treatment; B, G, L: H/R, hypoxia/reoxygenation injury; C, H, M: L-dose Res, 6.25 g/L for 24 h, D, I, N: H-dose Res, 12.5 g/L for 24 h; E, J, O: BHH, 100 μM for 24 h) and the fluorescence intensity was measured with ImageProPlus software and the Red/Green ratio was analyzed by SPSS25.0 Software (P). H/R: hypoxia/reoxygenation; JC-1: 5,5',6,6'-Tetrachloro-1,1',3,3'-tetraethyl-imidacarbocyanine iodide; L-dose Res: low-dose resveratrol; H-dose Res: high-dose resveratrol; BHH: bromhexine hydrochloride. aP < 0.01 vs control group, bP < 0.01 vs H/R group.

Figure 3 Resveratrol can activate mitochondrial autophagy (× 15 000) The ultrastructure of H9C2 cells was detected by transmission electron microscope. The red arrow indicates the mitochondrial cristae and the white arrow indicates the mitochondrial membrane. A: control, no treatment; B: H/R, hypoxia/reoxygenation injury; C: L-dose Res, 6.25 g/L for 24 h; D: H-dose Res, 12.5 g/L for 24 h; E: BHH, 100 μM for 24 h. H/R: hypoxia/reoxygenation; BHH: bromhexine hydrochloride.
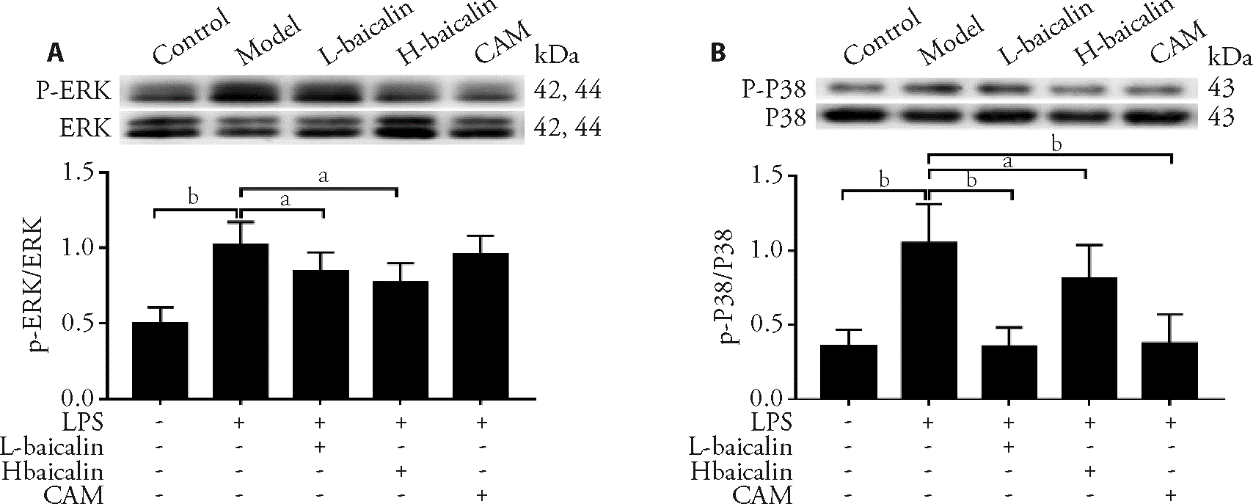
Figure 4 Resveratrol promotes the expression of PINK1 and PARKIN protein and mRNA and inhibits the expression of TOM20, TIM23 and PARL protein in H9C2 cells induced by H/R, which were detected by Western blot (A, B) and qPCR (C), respectively PINK1: PTEN-induced putative kinase protein 1; PARKIN: Parkinson disease protein 2; TOM20: mitochondrial 20 kDa outer membrane protein; TIM23: translocase of inner mitochondrial membrane 23; PARL: presenilins associated rhomboid-like protein; H/R: hypoxia/reoxygenation; qPCR: quantitative polymerase chain reaction. aP < 0.01 vs control group, bP < 0.01 vs H/R group.

Figure 5 Resveratrol promotes interaction between PARKIN and ubiquitin in the H/R-induced H9C2 cells A: Co-IP assay; B: the semi-quantitative analysis of the interaction between PARKIN and ubiquitin was conducted by the ImageProPlus software. PARKIN: Parkinson disease protein 2; H/R: hypoxia/reoxygenation. aP < 0.01 vs Control group, bP < 0.01 vs H/R group.
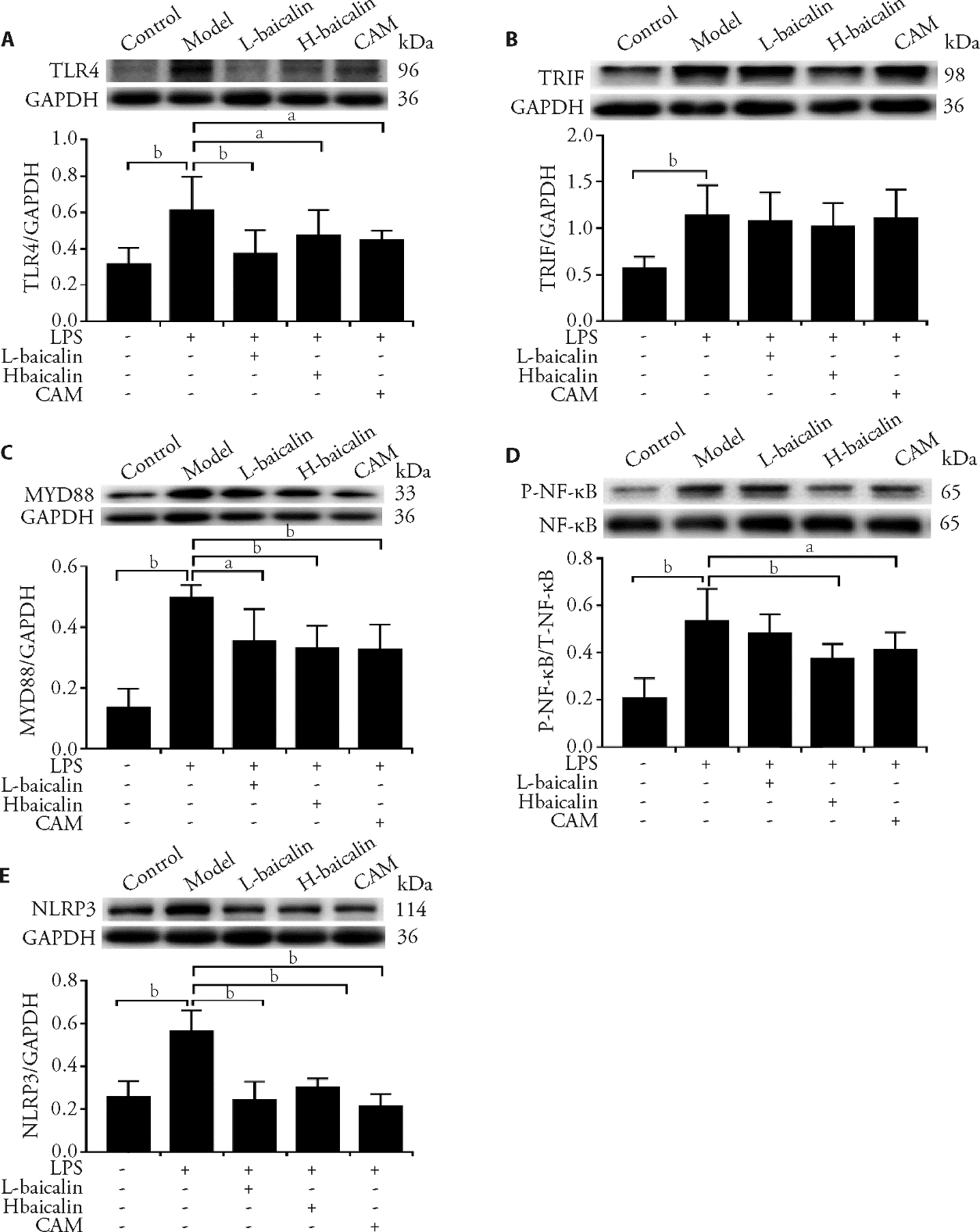
Figure 6 Resveratrol promotes the expression of Beclin 1, p62 and LC3 in H9C2 cells induced by H/R, which were detected by Western blot (A, B) LC3: microtubule associated protein light chain 3; H/R: hypoxia/reoxygenation. aaP<0.01 vs Control group, bbP<0.01 vs H/R group.
| 1 |
Boateng S, Sanborn T. Acute myocardial infarction. Dis Mon 2013; 59: 83-96
DOI URL |
| 2 |
Reed GW, Rossi JE, Cannon CP. Acute myocardial infarction. Lancet 2017; 389: 197-210.
DOI URL |
| 3 |
Frank A, Bonney M, Bonney S, et al. Myocardial ischemia reperfusion injury: from basic science to clinical bedside. Semin Cardiothorac Vasc Anesth 2012; 16: 123-32.
DOI PMID |
| 4 |
Wu MY, Yiang GT, Liao WT, et al. Current mechanistic concepts in ischemia and reperfusion injury. Cell Physiol Biochem 2018; 46: 1650-67.
DOI URL |
| 5 |
Toldo S, Mauro AG, Cutter Z, et al. Inflammasome, pyroptosis, and cytokines in myocardial ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol 2018; 315: H1553-68.
DOI URL |
| 6 |
Shi B, Ma M, Zheng Y, et al. mTOR and Beclin1: two key autophagy-related molecules and their roles in myocardial ischemia/reperfusion injury. J Cell Physiol 2019; 234: 12562-8.
DOI URL |
| 7 |
Boag SE, Andreano E, Spyridopoulos I. Lymphocyte communication in myocardial ischemia/reperfusion injury. Antioxid Redox Signal 2017; 26: 660-75.
DOI URL |
| 8 | Yang CF. Clinical manifestations and basic mechanisms of myocardial ischemia/reperfusion injury. Ci Ji Yi Xue Za Zhi 2018; 30: 209-15. |
| 9 | Morciano G, Bonora M, Campo G, et al. Mechanistic role of mPTP in ischemia-reperfusion injury. Adv Exp Med Biol 2017; 982: 169-89. |
| 10 | Minutoli L, Puzzolo D, Rinaldi M, et al. ROS-mediated NLRP3 inflammasome activation in brain, heart, kidney, and testis ischemia/reperfusion injury. Oxid Med Cell Longev 2016; 2016: 2183026. |
| 11 |
Lesnefsky EJ, Chen Q, Tandler B, et al. Mitochondrial dysfunction and myocardial ischemia-reperfusion: implications for novel therapies. Annu Rev Pharmacol Toxicol 2017; 57: 535-65.
DOI PMID |
| 12 |
Bugger H, Pfeil K. Mitochondrial ROS in myocardial ischemia reperfusion and remodeling. Biochim Biophys Acta Mol Basis Dis 2020; 1866: 165768.
DOI URL |
| 13 |
Melser S, Lavie J, Bénard G. Mitochondrial degradation and energy metabolism. Biochim Biophys Acta 2015; 1853: 2812-21.
DOI PMID |
| 14 |
Vásquez-Trincado C, García-Carvajal I, Pennanen C, et al. Mitochondrial dynamics, mitophagy and cardiovascular disease. J Physiol 2016; 594: 509-25.
DOI URL |
| 15 |
Murata D, Arai K, Iijima M, et al. Mitochondrial division, fusion and degradation. J Biochem 2020; 167: 233-41.
DOI URL |
| 16 |
Tong M, Sadoshima J. Mitochondrial autophagy in cardiomyopathy. Curr Opin Genet Dev 2016; 38: 8-15.
DOI URL |
| 17 |
Zhang P, Liu X, Huang G, et al. WITHDRAWN: Schisandrin B suppresses NLRP3 inflammasome activation to alleviate myocardial ischemia reperfusion injury via maintaining mitochondrial autophagy. Biochem Biophys Res Commun 2017; 494: 425.
DOI URL |
| 18 |
Gustafsson AB, Gottlieb RA. Eat your heart out: role of autophagy in myocardial ischemia/reperfusion. Autophagy 2008; 4: 416-21.
DOI PMID |
| 19 |
López De Figueroa P, Lotz MK, Blanco FJ, et al. Autophagy activation and protection from mitochondrial dysfunction in human chondrocytes. Arthritis Rheumatol 2015; 67: 966-76.
DOI URL |
| 20 |
Sekine S, Youle RJ. PINK1 import regulation; a fine system to convey mitochondrial stress to the cytosol. BMC Biol 2018; 16: 2.
DOI URL |
| 21 |
Eiyama A, Okamoto K. PINK1/Parkin-mediated mitophagy in mammalian cells. Curr Opin Cell Biol 2015; 33: 95-101.
DOI URL |
| 22 |
Zhao Y, Zhu Q, Song W, et al. Exercise training and dietary restriction affect PINK1/Parkin and Bnip3/Nix-mediated cardiac mitophagy in mice. Gen Physiol Biophys 2018; 37: 657-66.
DOI URL |
| 23 | Ney PA. Mitochondrial autophagy: origins, significance, and role of BNIP3 and NIX. Biochim Biophys Acta 2015; 1853: 2775-83. |
| 24 |
Ashrafi G, Schwarz TL. The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ 2013; 20: 31-42.
DOI PMID |
| 25 |
Springer MZ, Macleod KF. In Brief: Mitophagy: mechanisms and role in human disease. J Pathol 2016; 240: 253-55.
DOI URL |
| 26 |
Zhu W, Liu F, Wang L, et al. pPolyHb protects myocardial H9C2 cells against ischemia-reperfusion injury by regulating the Pink1-Parkin-mediated mitochondrial autophagy pathway. Artif Cells Nanomed Biotechnol 2019; 47: 1248-55.
DOI URL |
| 27 |
Dhanabalan K, Huisamen B, Lochner A. Mitochondrial oxidative phosphorylation and mitophagy in myocardial ischaemia/ reper-fusion: effects of chloroquine. Cardiovasc J Afr 2020; 31: 169-79.
DOI PMID |
| 28 |
Nunes S, Danesi F, Del Rio D, et al. Resveratrol and inflammatory bowel disease: the evidence so far. Nutr Res Rev 2018; 31: 85-97.
DOI URL |
| 29 |
Vestergaard M, Ingmer H. Antibacterial and antifungal properties of resveratrol. Int J Antimicrob Agents 2019; 53: 716-23.
DOI URL |
| 30 |
Vervandier-Fasseur D, Latruffe N. The potential use of resveratrol for cancer prevention. Molecules 2019; 24: 4506.
DOI URL |
| 31 |
Valentovic MA. Evaluation of resveratrol in cancer patients and experimental models. Adv Cancer Res 2018; 137: 171-88.
DOI PMID |
| 32 |
Dyck GJB, Raj P, Zieroth S, et al. The effects of resveratrol in patients with cardiovascular disease and heart failure: a narrative review. Int J Mol Sci 2019; 20: 904-32.
DOI URL |
| 33 |
Xu H, Cheng J, Wang X, et al. Resveratrol pretreatment alleviates myocardial ischemia/reperfusion injury by inhibiting STIM1-mediated intracellular calcium accumulation. J Physiol Biochem 2019; 75: 607-18.
DOI URL |
| 34 |
Liu MH, Shan J, Li J, et al. Resveratrol inhibits doxorubicin-induced cardiotoxicity via sirtuin 1 activation in H9c2 cardiomyocytes. Exp Ther Med 2016; 12: 1113-18.
DOI URL |
| 35 |
Zhao XY, Li GY, Liu Y, et al. Resveratrol protects against arsenic trioxide-induced cardiotoxicity in vitro and in vivo. Br J Pharmacol 2008; 154: 105-13.
DOI URL |
| 36 | Feng H, Mou SQ, Li WJ, et al. Resveratrol inhibits ischemia-induced myocardial senescence signals and NLRP3 inflammasome activation. Oxid Med Cell Longev 2020; 2020: 2647807. |
| 37 |
Sun ZM, Guan P, Luo LF, et al. Resveratrol protects against CIH-induced myocardial injury by targeting Nrf2 and blocking NLRP3 inflammasome activation. Life Sci 2020; 245: 117362.
DOI URL |
| 38 |
Tian M, Xie Y, Meng Y, et al. Resveratrol protects cardiomyocytes against anoxia/reoxygenation via dephos-phorylation of VDAC1 by Akt-GSK3 β pathway. Eur J Pharmacol 2019; 843: 80-7.
DOI URL |
| 39 | Zhang X, Huang L F, Hua L, et al. Resveratrol protects myocardial apoptosis induced by ischemia-reperfusion in rats with acute myocardial infarction via blocking P13K/Akt/e-NOS pathway. Eur Rev Med Pharmacol Sci 2019; 23: 1789-96. |
| 40 |
Yu D, Xiong J, Gao Y, et al. Resveratrol activates PI3K/AKT to reduce myocardial cell apoptosis and mitochondrial oxidative damage caused by myocardial ischemia/reperfusion injury. Acta Histochem 2021; 123: 151739.
DOI URL |
| 41 | Sebori R, Kuno A, Hosoda R, et al. Resveratrol decreases oxidative stress by restoring mitophagy and improves the pathophysiology of dystrophin-deficient MDX mice. Oxid Med Cell Longev 2018; 2018: 9179270. |
| 42 |
Lin KL, Lin KJ, Wang PW, et al. Resveratrol provides neuroprotective effects through modulation of mitochondrial dynamics and ERK1/2 regulated autophagy. Free Radic Res 2018; 52: 1371-86.
DOI URL |
| 43 | Qu X, Chen X, Shi Q, et al. Resveratrol alleviates ischemia/reperfusion injury of diabetic myocardium via inducing autophagy. Exp Ther Med 2019; 18: 2719-25. |
| 44 |
Zhang B, Xu L, Zhuo N, et al. Resveratrol protects against mitochondrial dysfunction through autophagy activation in human nucleus pulposus cells. Biochem Biophys Res Commun 2017; 493: 373-81.
DOI URL |
| 45 |
Liu MH, Lin XL, Guo DM, et al. Resveratrol protects cardiomyocytes from doxorubicin-induced apoptosis through the AMPK/P53 pathway. Mol Med Rep 2016; 13: 1281-6.
DOI URL |
| 46 |
Wu X, Zhou S, Zhu N, et al. Resveratrol attenuates hypoxia/reoxygenation-induced Ca2+ overload by inhibiting the Wnt5a/Frizzled-2 pathway in rat H9c2 cells. Mol Med Rep 2014; 10: 2542-8.
DOI URL |
| 47 |
Xu RY, Xu XW, Deng YZ, et al. Resveratrol attenuates myocardial hypoxia/reoxygenation-induced cell apoptosis through DJ-1-mediated SIRT1-p53 pathway. Biochem Biophys Res Commun 2019; 514: 401-6.
DOI URL |
| 48 |
Hong W, Tatsuo S, Shou-Dong W, et al. Resveratrol upregulates cardiac SDF-1 in mice with acute myocardial infarction through the deacetylation of cardiac p53. PLoS One 2015; 10: e0128978.
DOI URL |
| 49 |
Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol 2015; 6: 524-51.
DOI PMID |
| 50 |
Cadenas S. ROS and redox signaling in myocardial ischemia-reperfusion injury and cardioprotection. Free Radic Biol Med 2018; 117: 76-89.
DOI URL |
| 51 |
Jin X, Xue B, Zhou Q, et al. Mitochondrial damage mediated by ROS incurs bronchial epithelial cell apoptosis upon ambient PM (2.5) exposure. J Toxicol Sci 2018; 43: 101-111.
DOI URL |
| 52 |
Sinha K, Das J, Pal PB, et al. Oxidative stress: the mitochondria-dependent and mitochondria-independent pathways of apoptosis. Arch Toxicol 2013; 87: 1157-80.
DOI URL |
| 53 |
Kaminskyy VO, Zhivotovsky B. Free radicals in cross talk between autophagy and apoptosis. Antioxid Redox Signal 2014; 21: 86-102.
DOI URL |
| 54 |
Yamashita SI, Kanki T. How autophagy eats large mitochondria: autophagosome formation coupled with mitochondrial fragmentation. Autophagy 2017; 13: 980-1.
DOI URL |
| 55 |
Sun L, Zhao M, Yang Y, et al. Acetylcholine attenuates hypoxia/reoxygenation injury by inducing mitophagy through PINK1/parkin signal pathway in H9c2 cells. J Cell Physiol 2016; 231: 1171-81.
DOI URL |
| 56 | Mcwilliams TG, Prescott AR, Montava-Garriga L, et al. Basal mitophagy occurs independently of PINK1 in mouse tissues of high metabolic demand. Cell Metab 2018; 27: 439-49.e435. |
| 57 |
Okatsu K, Iemura S, Koyano F, et al. Mitochondrial hexokinase HKI is a novel substrate of the Parkin ubiquitin ligase. Biochem Biophys Res Commun 2012; 428: 197-202.
DOI URL |
| 58 |
Ham SJ, Lee D, Yoo H, et al. Decision between mitophagy and apoptosis by Parkin via VDAC1 ubiquitination. Proc Natl Acad Sci USA 2020; 117: 4281-91.
DOI URL |
| 59 |
Mclelland GL, Goiran T, Yi W, et al. Mfn2 ubiquitination by PINK1/parkin gates the p97-dependent release of ER from mitochondria to drive mitophagy. Elife 2018; 7: e32866.
DOI URL |
| 60 |
Yamada T, Dawson TM, Yanagawa T, et al. SQSTM1/p62 promotes mitochondrial ubiquitination independently of PINK1 and PRKN/parkin in mitophagy. Autophagy 2019; 15: 2012-8.
DOI URL |
| 61 |
Lazarou M, Sliter DA, Kane LA, et al. The ubiquitin kinase PINK1 recruits autophagy receptors to induce mitophagy. Nature 2015; 524: 309-14.
DOI URL |
| 62 |
Marques-Aleixo I, Santos-Alves E, Torrella JR, et al. Exercise and doxorubicin treatment modulate cardiac mitochondrial quality control signaling. Cardiovasc Toxicol 2018; 18: 43-55.
DOI PMID |
| 63 |
Liu H, Dai C, Fan Y, et al. From autophagy to mitophagy: the roles of P62 in neurodegenerative diseases. J Bioenerg Biomembr 2017; 49: 413-22.
DOI URL |
| 64 |
Park S, Choi SG, Yoo SM, et al. Choline dehydrogenase interacts with SQSTM1/p62 to recruit LC3 and stimulate mitophagy. Autophagy 2014; 10: 1906-20.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

