Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (2): 326-338.DOI: 10.19852/j.cnki.jtcm.2026.02.006
• Original Articles • Previous Articles Next Articles
Transcutaneous auricular vagus nerve stimulation improves emotional and cognitive functions in post-traumatic stress disorder rats through anti-inflammation, neuroprotection, and modulation of the hippocampal nuclear factor erythroid 2-related factor 2-heme oxygenase-1-glutathione peroxidase 4 pathway
ZHENG Yanfeng1,2, ZHANG Xinjiang2, ZHANG Xiaomeng1, LI Xiangji3, XIN Chen1, KONG Jingwei1, WANG Xin4, SUN Lan1( ), RONG Peijing1(
), RONG Peijing1( )
)
- 1
Institute of Basic Research in Clinical Medicine ,China Academy of Chinese Medical Sciences Beijing 100700, China
2Institute of Acupuncture and Moxibustion ,China Academy of Chinese Medical Sciences Beijing 100700, China
3Department of Gastroenterology ,State Key Laboratory of Digestive Health, National Clinical Research Center for Digestive Diseases, Beijing Friendship Hospital, Capital Medical University Beijing 100050, China
4Beijing Hospital of Traditional Chinese Medicine ,Capital Medical University Beijing 100010, China
-
Received:2025-10-16Accepted:2026-02-05Online:2026-04-15Published:2026-04-04 -
Contact:SUN Lan, Institute of Basic Research in Clinical Medicine, China Academy of Chinese Medical Sciences, Beijing 100700, China. sunlan462@163.com; RONG Peijing, Institute of Basic Research in Clinical Medicine, China Academy of Chinese Medical Sciences, Beijing 100700, China. drrongpj@163.com; Telephone: +86-18022386176 -
Supported by:Scientific and Technological Innovation Project of China Academy of Chinese Medical Sciences Neurobiophysical Investigation on the Amelioration of Post-traumatic Stress Disorder through Transcutaneous Auricular Electromagnetic Stimulation in the THz Frequency Range(CI2023C017YL);National Natural Science Foundation of China Explore the Mechanism by which Transcutaneous Auricular Vagus Nerve Stimulation Improves Depressive Behaviors Based on the Sirtuin 1/Nuclear Factor Erythroid 2-related Factor 2/Glutathione Peroxidase 4 Signaling Pathway(82304914)
Cite this article
ZHENG Yanfeng, ZHANG Xinjiang, ZHANG Xiaomeng, LI Xiangji, XIN Chen, KONG Jingwei, WANG Xin, SUN Lan, RONG Peijing. Transcutaneous auricular vagus nerve stimulation improves emotional and cognitive functions in post-traumatic stress disorder rats through anti-inflammation, neuroprotection, and modulation of the hippocampal nuclear factor erythroid 2-related factor 2-heme oxygenase-1-glutathione peroxidase 4 pathway[J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 326-338.
share this article

Figure 1 Effect of taVNS on emotional and cognitive functions A: travel distance of OFT; B: time in centre zone of OFT; C: time in open arms of EPM; D: discrimination index of novel object; E: mistake times of passive avoidance; F: time spent in dark box; H: travel distance of OFT; H: time in centre zone of OFT; I: time in open arms of EPM; J: discrimination index of novel object; K: mistake times of passive avoidance. L: time spent in dark box. Con: drink freely and eat freely; Mod: Con+ SPS model; Iso: Con + SPS model + 14-d of anesthesia by isoflurane (30 min/d); taVNS: Con + SPS model + 14-d of taVNS anesthesia by isoflurane (30 min/d). taVNS: transcutaneous auricular vagus nerve stimulation; SPS: single prolonged stress. Iso: isoflurane; OFT: open field test; EPM: elevated plus maze; NOR: novel object recognition; SEM: standard error of the mean. Data are presented as mean ± SEM (n = 10 per group). Data were analyzed using one-way repeated-measures analysis of variance, followed by Tukey’s post hoc test when significant F ratios were obtained. aP < 0.01, versus the Con group; bP < 0.01 versus the Mod group.
| Biomarker | Con (n = 10) | Mod (n = 10) | Iso (n = 10) | taVNS (n = 10) |
|---|---|---|---|---|
| IL-1β (pg/mL) | 88.16±11.33 | 319.00±11.73a | 247.30±6.94b | 188.80±15.66b |
| IL-6 (pg/mL) | 68.23±11.04 | 215.80±5.67a | 158.20±7.52b | 137.00±6.19b |
| IL-12 (pg/mL) | 105.00±7.37 | 296.4±8.99a | 231.40±10.42b | 184.20±11.45b |
| TNF-α (pg/mL) | 340.80±40.75 | 1035.00±19.21a | 765.40±30.64b | 623.50±40.98b |
| IL-4 (pg/mL) | 561.50±15.68 | 165.50±21.38a | 310.60±14.07b | 400.20±26.43b |
| IL-10 (pg/mL) | 54.73±1.75 | 15.93±2.43a | 32.76±1.85b | 39.68±1.89b |
| CAT (pg/mL) | 780.30±40.90 | 462.10±52.86a | 594.00±50.12b | 604.40±33.01b |
| GSH (pg/mL) | 6.14±0.50 | 3.58±0.28a | 4.489±0.59b | 5.01±0.32b |
| SOD (pg/mL) | 72.35±5.90 | 42.60±3.84a | 54.45±3.18b | 60.72±3.89b |
| ROS (U/mL) | 36.72±4.78 | 60.62±2.95a | 49.91±5.03b | 48.87±3.62b |
| MDA(nmol/mL) | 4.08±0.22 | 5.993±0.45a | 5.01±0.23b | 4.55±0.47b |
Table 1 The concentration of the immune-related and oxidative stress-related cytokines in serum, including IL-1β, IL-6, IL-12, TNF-α, IL-4, IL-10, CAT, GSH, SOD, ROS and MDA
| Biomarker | Con (n = 10) | Mod (n = 10) | Iso (n = 10) | taVNS (n = 10) |
|---|---|---|---|---|
| IL-1β (pg/mL) | 88.16±11.33 | 319.00±11.73a | 247.30±6.94b | 188.80±15.66b |
| IL-6 (pg/mL) | 68.23±11.04 | 215.80±5.67a | 158.20±7.52b | 137.00±6.19b |
| IL-12 (pg/mL) | 105.00±7.37 | 296.4±8.99a | 231.40±10.42b | 184.20±11.45b |
| TNF-α (pg/mL) | 340.80±40.75 | 1035.00±19.21a | 765.40±30.64b | 623.50±40.98b |
| IL-4 (pg/mL) | 561.50±15.68 | 165.50±21.38a | 310.60±14.07b | 400.20±26.43b |
| IL-10 (pg/mL) | 54.73±1.75 | 15.93±2.43a | 32.76±1.85b | 39.68±1.89b |
| CAT (pg/mL) | 780.30±40.90 | 462.10±52.86a | 594.00±50.12b | 604.40±33.01b |
| GSH (pg/mL) | 6.14±0.50 | 3.58±0.28a | 4.489±0.59b | 5.01±0.32b |
| SOD (pg/mL) | 72.35±5.90 | 42.60±3.84a | 54.45±3.18b | 60.72±3.89b |
| ROS (U/mL) | 36.72±4.78 | 60.62±2.95a | 49.91±5.03b | 48.87±3.62b |
| MDA(nmol/mL) | 4.08±0.22 | 5.993±0.45a | 5.01±0.23b | 4.55±0.47b |
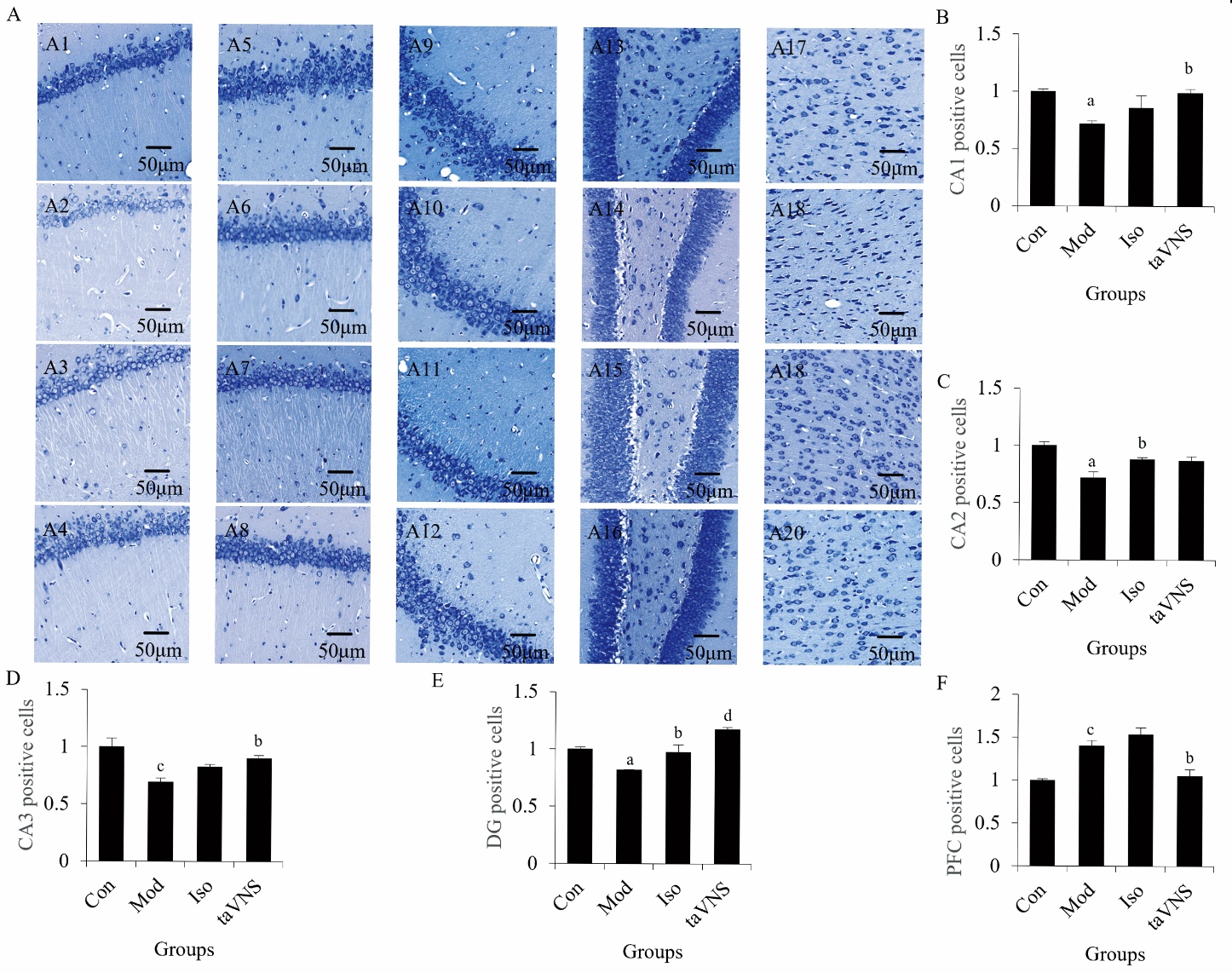
Figure 2 Effect of taVNS on the HIP and PFC in a rat model of PTSD A: Nissl staining in the hippocampal CA1, CA2, CA3, DG and PFC subregion; A1: hippocampal CA1 of Con group; A2: hippocampal CA1 of Mod group; A3: hippocampal CA1 of Iso group; A4: hippocampal CA1 of taVNS group; A5: hippocampal CA2 of Con group; A6: hippocampal CA2 of Mod group; A7: hippocampal CA2 of Iso group; A8: hippocampal CA2 of taVNS group; A9: hippocampal CA3 of Con group; A10: hippocampal CA3 of Mod group; A11: hippocampal CA3 of Iso group; A12: hippocampal CA3 of taVNS group; A13: hippocampal DG of Con group; A14: hippocampal DG of Mod group; A15: hippocampal DG of Iso group; A16: hippocampal DG of taVNS group; A17: PFC of Con group; A18: PFC of Mod group; A19: PFC of Iso group; A20: PFC of taVNS group; B: Nissl staining statistical analysis in the hippocampal CA1, subregion; C: Nissl staining statistical analysis in the hippocampal CA2 subregion; D: Nissl staining statistical analysis in the hippocampal CA3 subregion; E: Nissl staining statistical analysis in the hippocampal DG subregion; F: Nissl staining statistical analysis in the PFC region. Con: drink freely and eat freely; Mod: Con + SPS model; Iso: Con + SPS model + 14-d of anesthesia by isoflurane (30 min/d); taVNS: Con + SPS model + 14-d of taVNS anesthesia by isoflurane (30 min/d). taVNS: transcutaneous auricular vagus nerve stimulation; SPS: single prolonged stress; Iso: isoflurane; CA: cornu ammonis; DG: dentate gyrus; PFC: prefrontal cortex; HIP: hippocampal; SEM: standard error of the mean. Data are presented as mean ± SEM (n = 3 per group). Data were analyzed using one-way repeated-measures analysis of variance, followed by Tukey’s post hoc test when significant F ratios were obtained. aP < 0.05, cP < 0.01 versus the Con group; bP < 0.05, dP < 0.01 versus the Mod group.
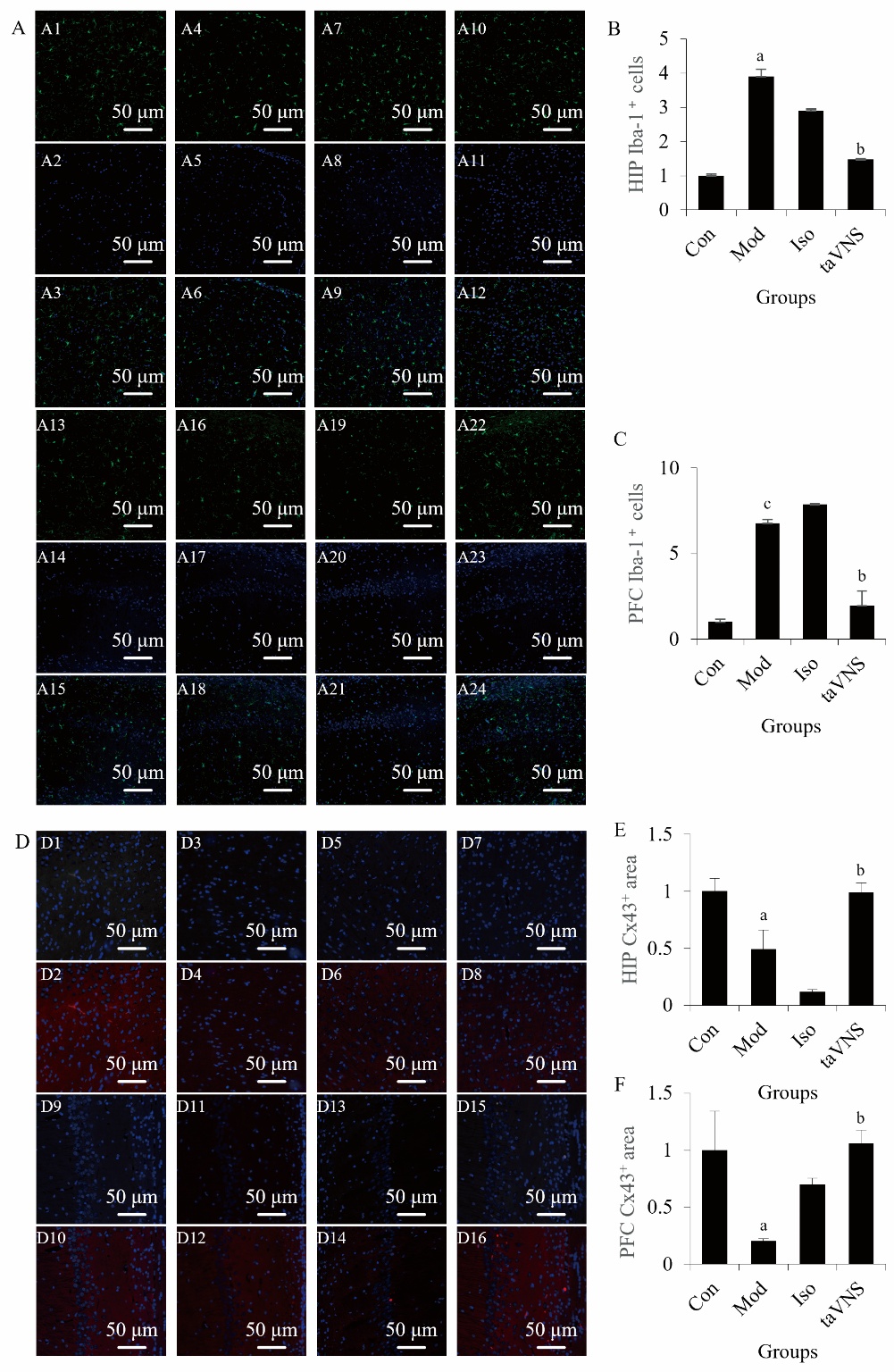
Figure 3 Effect of taVNS on microglial morphology and connexins in the HIP and PFC of a rat model of PTSD A: representative images of Iba-1 immunofluorescence. A1: Iba-1 immunofluorescence of Con group in PFC; A2: dapi of Con group in PFC; A3: merge of Con group in PFC; A4: Iba-1 immunofluorescence of Mod group in PFC; A5: dapi of Mod group in PFC; A6:merge of Mod group in PFC; A7: Iba-1 immunofluorescence of Iso group in PFC; A8: dapi of Iso group in PFC; A9: merge of Iso group in PFC; A10: Iba-1 immunofluorescence of taVNS group in PFC; A11: dapi of taVNS group in PFC; A12: merge of taVNS group in PFC; A13: Iba-1 immunofluorescence of Con group in HIP; A14: dapi of Con group in HIP; A15: merge of Con group in HIP; A16: Iba-1 immunofluorescence of Mod group in HIP; A17: dapi of Mod group in HIP; A18: merge of Mod group in HIP; A19: Iba-1 immunofluorescence of Iso group in HIP; A20: dapi of Iso group in HIP; A21: merge of Iso group in HIP; A22: Iba-1 immunofluorescence of taVNS group in HIP; A23: dapi of taVNS group in HIP; A24: merge of taVNS group in HIP; B: quantification of Iba-1-positive cells (fold of control) in the PFC; C: quantification of Iba-1-positive cells (fold of control) in the Hip; D: representative images of Cx43 immunofluorescence; D1: Cx43 immunofluorescence of Con group in PFC; D2: merge of Con group in PFC; D3: Cx43 immunofluorescence of Mod group in PFC; D4: merge of Mod group in PFC; D5: Cx43 immunofluorescence of Iso group in PFC; D6: merge of Iso group in PFC; D7: Cx43 immunofluorescence of taVNS group in PFC; D8: merge of taVNS group in PFC; E: quantification of Cx43 area (fold of control) in the PFC; F: quantification of Cx43 area (fold of control) in the Hip. Con: drink freely and eat freely; Mod: Con + SPS model; Iso: Con + SPS model + 14-d of anesthesia by isoflurane (30 min/d); taVNS: Con + SPS model + 14-d of taVNS anesthesia by isoflurane (30 min/d). taVNS: transcutaneous auricular vagus nerve stimulation; SPS: single prolonged stress; Iso: isoflurane; Iba-1: ionized calcium-binding adapter molecule 1; Cx43: connexin43; PFC: prefrontal cortex; HIP: hippocampal; SEM: standard error of the mean. Data are presented as mean ± SEM (n = 3 per group). Data were analyzed using one-way repeated-measures analysis of variance, followed by Tukey’s post hoc test when significant F ratios were obtained. aP < 0.05 versus the Con group; bP < 0.05 versus the Mod group.

Figure 4 Effect of taVNS on Nrf2-HO-1 and its downstream proteins, mRNA, and protein expression in the PFC and HIP of a rat model of post-traumatic stress disorder A: analysis of mRNA levels of NRF2 in PFC; B: analysis of mRNA levels of HO-1 in PFC; C: analysis of mRNA levels of GPX4 in PFC; D: analysis of mRNA levels of NLRP3 in PFC; E: analysis of mRNA levels of NRF2 in HIP; F: analysis of mRNA levels of HO-1 in HIP; G: analysis of mRNA levels of GPX4 in HIP; H: analysis of mRNA levels of NLRP3 in HIP; I: representative WB bands of PFC; J: representative WB bands of HIP; K: analysis of protein levels of NRF2 in PFC; L: analysis of protein levels of HO-1 in PFC; M: analysis of protein levels of GPX4 in PFC; N: analysis of protein levels of NLRP3 in PFC; O: analysis of protein levels of NRF2 in HIP; P: analysis of protein levels of HO-1 in HIP; Q: analysis of protein levels of GPX4 in HIP; R: analysis of protein levels of NLRP3 in HIP. Mod: Con + SPS model; Iso: Con + SPS model + 14-d of anesthesia by isoflurane (30 min/d); taVNS: Con + SPS model + 14-d of taVNS anesthesia by isoflurane (30 min/d). taVNS: transcutaneous auricular vagus nerve stimulation; SPS: single prolonged stress; Iso: isoflurane; PFC: prefrontal cortex; HIP: hippocampal; WB: Western blot; NRF2: nuclear factor erythroid 2-related factor 2; HO-1: heme oxygenase-1; GPX4: glutathione peroxidase 4; NLRP3: NLR family pyrin domain containing 3; SEM: standard error of the mean. Data are presented as mean ± SEM (n = 6 per group). Data were analyzed using one-way repeated-measures analysis of variance, followed by Tukey’s post hoc test when significant F ratios were obtained. aP < 0.05, dP < 0.01 versus the Con group; bP < 0.05, cP < 0.01 versus the Mod group.
| 1. |
Maercker A, Cloitre M, Bachem R, et al. Complex post-traumatic stress disorder. Lancet 2022; 400: 60-72.
DOI PMID |
| 2. |
Morina N, Ford JD, Risch AK, Morina B, Stangier U. Somatic distress among Kosovar civilian war survivors: relationship to trauma exposure and the mediating role of experiential avoidance. Soc Psychiatry Psychiatr Epidemiol 2010; 45: 1167-77.
DOI URL |
| 3. |
Morina N, Ford JD. Complex sequelae of psychological trauma among Kosovar civilian war victims. Int J Soc Psychiatry 2008; 54: 425-36.
DOI URL |
| 4. |
Cao C, Wang L, Fang R, et al. Anxiety, depression, and PTSD symptoms among high school students in china in response to the COVID-19 pandemic and lockdown. J Affect Disord 2022; 296: 126-9.
DOI URL |
| 5. |
McLaughlin KA, Koenen KC, Friedman MJ, et al. Subthreshold posttraumatic stress disorder in the world health organization world mental health surveys. Biol Psychiatry 2015; 77: 375-84.
DOI URL |
| 6. |
Steenkamp MM, Litz BT, Hoge CW, Marmar CR. Psychotherapy for military-related PTSD: a review of randomized clinical trials. JAMA 2015; 314: 489-500.
DOI PMID |
| 7. |
Shalev A, Liberzon I, Marmar C. Post-traumatic stress disorder. N Engl J Med 2017; 376: 2459-69.
DOI URL |
| 8. |
Innamorato NG, Rojo AI, García-Yagüe AJ, Yamamoto M, de Ceballos ML, Cuadrado A. The transcription factor Nrf2 is a therapeutic target against brain inflammation. J Immunol 2008; 181: 680-9.
DOI PMID |
| 9. |
Grasser LR, Javanbakht A. Treatments of posttraumatic stress disorder in civilian populations. Curr Psychiatry Rep 2019; 21: 11.
DOI |
| 10. |
Santhanam P, Teslovich T, Wilson SH, Yeh PH, Oakes TR, Weaver LK. Decreases in white matter integrity of ventro-limbic pathway linked to post-traumatic stress disorder in mild traumatic brain injury. J Neurotrauma 2019; 36: 1093-8.
DOI URL |
| 11. |
Kasai K, Yamasue H, Gilbertson MW, Shenton ME, Rauch SL, Pitman RK. Evidence for acquired pregenual anterior cingulate gray matter loss from a twin study of combat-related posttraumatic stress disorder. Biol Psychiatry 2008; 63: 550-6.
DOI URL |
| 12. |
Bremner JD. Traumatic stress: effects on the brain. Dialogues Clin Neurosci 2006; 8: 445-61.
DOI URL |
| 13. |
Ressler KJ, Berretta S, Bolshakov VY, et al. Post-traumatic stress disorder: clinical and translational neuroscience from cells to circuits. Nat Rev Neurol 2022; 18: 273-88.
DOI PMID |
| 14. |
Pitman RK, Rasmusson AM, Koenen KC, et al. Biological studies of post-traumatic stress disorder. Nat Rev Neurosci 2012; 13: 769-87.
DOI PMID |
| 15. |
Shin LM, McNally RJ, Kosslyn SM, et al. Regional cerebral blood flow during script-driven imagery in childhood sexual abuse-related PTSD: a PET investigation. Am J Psychiatry 1999; 156: 575-84.
DOI URL |
| 16. |
Gold AL, Shin LM, Orr SP, et al. Decreased regional cerebral blood flow in medial prefrontal cortex during trauma-unrelated stressful imagery in Vietnam veterans with post-traumatic stress disorder. Psychol Med 2011; 41: 2563-72.
DOI PMID |
| 17. |
Rojo AI, McBean G, Cindric M, et al. Redox control of microglial function: molecular mechanisms and functional significance. Antioxid Redox Signal 2014; 21: 1766-801.
DOI URL |
| 18. |
Wishart DS, Tzur D, Knox C, et al. HMDB: the Human Metabolome Database. Nucleic Acids Res 2007; 35: D521-6.
DOI PMID |
| 19. |
Miller MW, Sadeh N. Traumatic stress, oxidative stress and post-traumatic stress disorder: neurodegeneration and the accelerated-aging hypothesis. Mol Psychiatry 2014; 19: 1156-62.
DOI |
| 20. |
Katrinli S, Maihofer AX, Wani AH, et al. Epigenome-wide Meta-analysis of PTSD symptom severity in three military cohorts implicates DNA methylation changes in genes involved in immune system and oxidative stress. Mol Psychiatry 2022; 27: 1720-8.
DOI |
| 21. |
Peruzzolo TL, Pinto JV, Roza TH, et al. Inflammatory and oxidative stress markers in post-traumatic stress disorder: a systematic review and Meta-analysis. Mol Psychiatry 2022; 27: 3150-63.
DOI |
| 22. |
Tang C, Gao J, Li S, et al. Chlorogenic acid improves SPS-induced PTSD-like behaviors in rats by regulating the crosstalk between Nrf2 and NF-κB signaling pathway. Free Radic Biol Med 2025; 231: 136-52.
DOI URL |
| 23. |
Jîtcă G, Stoicescu R, Májai E. Cannabidiol Treatment in a predator-based animal model of PTSD: assessing oxidative stress and memory performance. Int J Mol Sci 2025; 26: 4491.
DOI URL |
| 24. |
Liu J, Litt L, Segal MR, Kelly MJ, Pelton JG, Kim M. Metabolomics of oxidative stress in recent studies of endogenous and exogenously administered intermediate metabolites. Int J Mol Sci 2011; 12: 6469-501.
DOI PMID |
| 25. | Taira J, Ogi T. Induction of antioxidant protein HO-1 through Nrf2-ARE signaling due to pteryxin in peucedanum japonicum thunb in RAW264.7 macrophage cells. Antioxidants (Basel) 2019; 8: 621. |
| 26. |
Richter-Levin G, Stork O, Schmidt MV. Animal models of PTSD: a challenge to be met. Mol Psychiatry 2019; 24: 1135-56.
DOI |
| 27. |
Koo JW, Duman RS. IL-1beta is an essential mediator of the antineurogenic and anhedonic effects of stress. Proc Natl Acad Sci U S A 2008; 105: 751-6.
DOI URL |
| 28. |
Maier SF, Watkins LR. Cytokines for psychologists: implications of bidirectional immune-to-brain communication for understanding behavior, mood, and cognition. Psychol Rev 1998; 105: 83-107.
DOI PMID |
| 29. |
Faborode OS, Dalle E, Mabandla MV. Exposure to footshock stress downregulates antioxidant genes and increases neuronal apoptosis in an Aβ(1-42) rat model of Alzheimer’s disease. Neurochem Int 2021; 150: 105170.
DOI URL |
| 30. | Muhie S, Gautam A, Yang R, et al. Molecular signatures of post-traumatic stress disorder in war-zone-exposed veteran and active-duty soldiers. Cell Rep Med 2023; 4: 101045. |
| 31. |
Austelle CW, O’Leary GH, Thompson S, et al. A comprehensive review of vagus nerve stimulation for depression. Neuromodulation 2022; 25: 309-15.
DOI URL |
| 32. |
Badran BW, Dowdle LT, Mithoefer OJ, et al. Neurophysiologic effects of transcutaneous auricular vagus nerve stimulation (taVNS) via electrical stimulation of the tragus: a concurrent taVNS/fMRI study and review. Brain Stimul 2018; 11: 492-500.
DOI URL |
| 33. |
Chen Y, Zhang Y, Wang J, et al. Anti-neuroinflammation effects of transcutaneous auricular vagus nerve stimulation against depression-like behaviors via hypothalamic α7nAchR/JAK2/STAT3/NF-κB pathway in rats exposed to chronic unpredictable mild stress. CNS Neurosci Ther 2023; 29: 2634-44.
DOI URL |
| 34. |
Wang L, Zhang J, Guo C, et al. The efficacy and safety of transcutaneous auricular vagus nerve stimulation in patients with mild cognitive impairment: a double blinded randomized clinical trial. Brain Stimul 2022; 15: 1405-14.
DOI PMID |
| 35. | Zhang S, Zhao Y, Qin Z, et al. Transcutaneous auricular vagus nerve stimulation for chronic insomnia disorder: a randomized clinical trial. JAMA Netw Open 2024; 7: e2451217. |
| 36. |
Wang JY, Zhang Y, Chen Y, et al. Mechanisms underlying antidepressant effect of transcutaneous auricular vagus nerve stimulation on CUMS model rats based on hippocampal α7nAchR/NF-κB signal pathway. J Neuroinflammation 2021; 18: 291.
DOI |
| 37. |
Siracusa R, Paola RD, Cuzzocrea S, Impellizzeri D. Fibromyalgia: pathogenesis, mechanisms, diagnosis and treatment options update. Int J Mol Sci 2021; 22: 3891.
DOI URL |
| 38. | Daskalakis NP, Iatrou A, Chatzinakos C, et al. Systems biology dissection of PTSD and MDD across brain regions, cell types, and blood. Science 2024; 384: eadh3707. |
| 39. |
Cai M, Tong L, Dong B, Hou W, Shi L, Dong H. Kelch-like ECH-associated protein 1-dependent nuclear factor-E2-related factor 2 activation in relation to antioxidation induced by sevoflurane preconditioning. Anesthesiology 2017; 126: 507-21.
DOI PMID |
| 40. |
Yang W, Wang Y, Zhang C, et al. Maresin1 protect against ferroptosis-induced liver injury through ROS inhibition and Nrf2/HO-1/GPX4 activation. Front Pharmacol 2022; 13: 865689.
DOI URL |
| 41. |
Xie P, Chen L, Wang J, Wang X, Yang S, Zhu G. Polysaccharides from polygonatum cyrtonema Hua prevent post-traumatic stress disorder behaviors in mice: mechanisms from the perspective of synaptic injury, oxidative stress, and neuroinflammation. J Ethnopharmacol 2024; 319: 117165.
DOI URL |
| 42. |
Zhang JB, Lu GD, Sun DN, et al. Pellitorine protects chronic restraint stress-induced cognitive deficits via inhibiting neural inflammation and ferroptosis. Int Immunopharmacol 2025; 162: 115166.
DOI URL |
| 43. |
Wang Z, Lai C, Shen B, et al. Effects of evodiamine on behavior and hippocampal neurons through inhibition of angiotensin-converting enzyme and modulation of the renin angiotensin pathway in a mouse model of post-traumatic stress disorder. Nutrients 2024; 16: 1957.
DOI URL |
| 44. |
Dang R, Wang M, Li X, et al. Edaravone ameliorates depressive and anxiety-like behaviors via Sirt1/Nrf2/HO-1/Gpx 4 pathway. J Neuroinflammation 2022; 19: 41.
DOI |
| 45. |
Paciello F, Pisani A, Rolesi R, et al. Oxidative stress and inflammation cause auditory system damage via glial cell activation and dysregulated expression of gap junction proteins in an experimental model of styrene-induced oto/neurotoxicity. J Neuroinflammation 2024; 21: 4.
DOI |
| 46. |
Romero-Miguel D, Casquero-Veiga M, MacDowell KS, et al. A Characterization of the effects of minocycline treatment during adolescence on structural, metabolic, and oxidative stress parameters in a maternal immune stimulation model of neurodevelopmental brain disorders. Int J Neuropsychopharmacol 2021; 24: 734-48.
DOI PMID |
| 47. |
Lu S, Ge Q, Yang M, et al. Decoupling the mutual promotion of inflammation and oxidative stress mitigates cognitive decline and depression-like behavior in rmTBI mice by promoting myelin renewal and neuronal survival. Biomed Pharmacother 2024; 173: 116419.
DOI URL |
| 48. | Jiang M, Zhang C, Chen J, et al. Unveiling aging and Alzheimer’s disease-associated dynamics of LINE1 DNA content and protein expression in mouse brains. Aging Cell 2025; 24: e70296. |
| 49. |
Morera PMA, Beserra-Filho JIA, Soares-Silva B, et al. Mixed granulocytic airway inflammation impairs aversive learning and alters neuroimmune biomarkers in mice. Sci Rep 2025; 15: 39804.
DOI |
| 50. |
Dionisie V, Ciobanu AM, Toma VA, et al. Escitalopram targets oxidative stress, Caspase-3, BDNF and MeCP2 in the hippocampus and frontal cortex of a rat model of depression induced by chronic unpredictable mild stress. Int J Mol Sci 2021; 22: 7483.
DOI URL |
| 51. | Li Y, Li L, Wang Y, et al. Cholinergic signaling to CA1 astrocytes controls fear extinction. Sci Adv 2025; 11: eads7191. |
| 52. |
Jamnicki-Abegg M, Weihrauch D, Pagel PS, et al. Isoflurane inhibits cardiac myocyte apoptosis during oxidative and inflammatory stress by activating Akt and enhancing Bcl-2 expression. Anesthesiology 2005; 103: 1006-14.
PMID |
| 53. | Qi YF, Liu Y, Liu YY, Li YY, et al. Therapeutic potential of Traditional Chinese Medicine Yisui Shengxue pills to inhibit hypoxia-inducible factor-1alpha and general control nonderepressible 2 to regulate the post-chemotherapy immune response: integrating network pharmacology and experimental validation. J Tradit Chin Med 2025; 45: 1087-97. |
| 54. | Wang RQ, Hao LY, Lu Y, et al. Mechanism analysis of Tongqiao Yizhi decoction in treating vascular dementia rats by brain tissue untargeted metabonomics and fecal 16S rRNA gene sequencing. J Tradit Chin Med 2025; 45: 759-69. |
| [1] | HU Jingnan, LIAO Man, XI Zhongwen, SONG Jing, WANG Yining, HE Tao. Identification and verification of key genes related to oxidative stress in type 2 diabetes and screening of candidate drugs from Traditional Chinese Medicine [J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 350-359. |
| [2] | NI Shuang, LIU Xiaofei, GUO Xiaoyan, GU Zuxi, WU Panqing, CONG Chao, LI Shengnan, GAO Xianwei, XU Lianwei. Mechanism of Tiaogeng decoction (调更汤) in a cognitive dysfunction mouse model [J]. Journal of Traditional Chinese Medicine, 2025, 45(5): 987-997. |
| [3] | LIU Xiaoyao, LI Jialin, WANG Weiling, FAN Qiongyin, SU Zeqi, HE Cheng, WANG Chunguo, GAO Jian, WANG Ting. Pharmacological effect and possible mechanism of Mudan Huaban recipe (牡丹化斑方) on melasma in mice induced by ultraviolet B and progesterone [J]. Journal of Traditional Chinese Medicine, 2025, 45(3): 518-527. |
| [4] | XU Bojun, TAO Tian, ZHAO Liangbin, ZHENG Hui, ZHAN huakui, GUO Julan. Bushen Tongluo recipe (补肾通络方) improves oxidative stress homeostasis, inhibits transforming growth factor/Notch signaling pathway, and regulates the lncRNA maternally expressed gene 3/miR-145 axis to delay diabetic kidney disease [J]. Journal of Traditional Chinese Medicine, 2025, 45(3): 561-570. |
| [5] | ZHENG Peng, MENG Ying, LIU Meijun, YU Di, LIU Huiying, WANG Fuchun, XU Xiaohong. Electroacupuncture inhibits hippocampal oxidative stress and autophagy in sleep-deprived rats through the protein kinase B and mechanistic target of rapamycin signaling pathway [J]. Journal of Traditional Chinese Medicine, 2024, 44(5): 974-980. |
| [6] | Zubaria Tul Ain, Iram Fatima, Sana Naseer, Sobia Kanwal, Tariq Mahmood. Assessment of phytochemicals, antioxidant, anti-hemolytic, anti-inflammatory and anti-cancer potential of flowers, leaves and stem extracts of Rosa arvensis [J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 804-812. |
| [7] | HUANG Hongmei, YANG Maojun, LI Ting, WANG Dandan, LI Ying, TANG Xiaochi, YUAN Lu, GU Shi, XU Yong. Neferine inhibits the progression of diabetic nephropathy by modulating the miR-17-5p/nuclear factor E2-related factor 2 axis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 44-53. |
| [8] | ZHANG Xiaoying, WANG Ruixuan, WANG Yiqing, XU Fanxing, YAN Tingxu, WU Bo, ZHANG Ming, JIA Ying. Spinosin protects Neuro-2a/APP695 cells from oxidative stress damage by inactivating p38 [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 868-875. |
| [9] | LIU Bingbing, LI Jieru, SI Jianchao, CHEN Qi, YANG Shengchang, JI Ensheng. Ginsenoside Rb1 alleviates chronic intermittent hypoxia-induced diabetic cardiomyopathy in db/db mice by regulating the adenosine monophosphate-activated protein kinase/Nrf2/heme oxygenase-1 signaling pathway [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 906-914. |
| [10] | ZHOU Hua, LI Hui, WANG Haihua. Potential protective effects of the water-soluble Chinese propolis on experimental ulcerative colitis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 925-933. |
| [11] | HE Jiakai, ZHANG Jinling, WANG Yu, LI Shaoyuan, FANG Jiliang, ZHANG Shuai, ZHAO Yanan, ZHAI Weihang, GAO Deqiang, LI Ran, JIANG Yuhang, CHEN Zehao, JIA Baohui, RONG Peijing. Transcutaneous auricular vagus nerve stimulation would be an alternative to implantable cervical vagus nerve stimulation in some situation [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 627-630. |
| [12] | HUANG Qiuyue, YE Hui, SHI Zongming, JIA Xiaofen, LIN Miaomiao, CHU Yingming, YU Jing, ZHANG Xuezhi. Efficacy of Qingre Huashi decoction (清热化湿方) on infection of Helicobacter pylori: inhibiting adhesion, antioxidant, and anti-inflammation [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 915-921. |
| [13] | HENG Xianpei, LI Liang, YANG Liuqin, WANG Zhita. Efficacy of Dangua Fang (丹瓜方) on endothelial cells damaged by oxidative stress [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 900-907. |
| [14] | ZHENG Wei, WANG Mingxing, LIU Shanxue, LUAN Chao, ZHANG Yanqiu, XU Duoduo, WANG Jian. Buyang Huanwu Tang (补阳还五汤) protects H2O2-induced RGC-5 cell against oxidative stress and apoptosis via reactive oxygen species-mitogen-activated protein kinase signaling pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 885-891. |
| [15] | CHEN Limei, SUN Jifei, GUO Chunlei, LI Xiaojiao, WANG Zhi, Hong Yang, FANG Jiliang. Preliminary single-arm study of brain effects during transcutaneous auricular vagus nerve stimulation treatment of recurrent depression by resting-state functional magnetic resonance imaging [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 818-824. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

