Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (2): 339-349.DOI: 10.19852/j.cnki.jtcm.2026.02.007
• Original Articles • Previous Articles Next Articles
Moxibustion alleviates autophagy and inhibits ferroptosis to improve cardiac function in rats with post-myocardial infarction heart failure
GAO Bing1, LIU Pan1, LI Lan2, GONG Tiantian2, ZHU Ling2, LI Lingji1, XIA Ran1,3, MA Qiang2,3, HU Jing1, WANG Jing1,3,4( )
)
- 1
College of Traditional Chinese Medicine ,Anhui University of Chinese Medicine Hefei 230012, China
2Acupuncture and moxibustion and Massage College ,Anhui University of Chinese Medicine Hefei 230012, China
3Key Laboratory of Xin’an Medical Ministry of Education ,Anhui University of Chinese Medicine Hefei 230012, China
4Center for Xin’an Medicine and Modernization of Traditional Chinese Medicine of IHM Hefei 230012, China
-
Received:2024-11-03Accepted:2025-05-15Online:2026-04-15Published:2026-04-04 -
Contact:Dr. WANG Jing, College of Traditional Chinese Medicine, Anhui University of Chinese Medicine, Hefei 230012, China. Wangjing2161@126.com; Telephone: +86-13865912237 -
Supported by:National Natural Science Foundation of China: Research on the Cardiomyoprotective Effect Mechanism of Moxibustion Regulating the Mammalian Target of Rapamycin Signaling Pathway to Inhibit Autophagy(81574084);Natural Science Research Project of Colleges and Universities in Anhui Province, China: Exploring the Mechanism of Moxibustion Anti-chronic Heart Failure Fibrosis Based on MicroRNA-21/Phosphatase and Tensin Homolog/mTOR Signaling Pathway-mediated Circular RNA PAN3 Regulation of Cardiomyocyte Autophagy, and Investigating the Mechanism of Moxibustion in Preventing and Treating Chronic Heart Failure Myocardial Fibrosis Based on Transient Receptor Potential Vanilloid 1-regulated Calcitonin Gene-Related Peptide-mediated Vascular Endothelial Growth Factor Endothelial Nitric Oxide Synthase Signaling Pathway(kj2021a0570);Natural Science Research Project of Colleges and Universities in Anhui Province, China: Exploring the Mechanism of Moxibustion Anti-chronic Heart Failure Fibrosis Based on MicroRNA-21/Phosphatase and Tensin Homolog/mTOR Signaling Pathway-mediated Circular RNA PAN3 Regulation of Cardiomyocyte Autophagy, and Investigating the Mechanism of Moxibustion in Preventing and Treating Chronic Heart Failure Myocardial Fibrosis Based on Transient Receptor Potential Vanilloid 1-regulated Calcitonin Gene-Related Peptide-mediated Vascular Endothelial Growth Factor Endothelial Nitric Oxide Synthase Signaling Pathway(2023AH050796);Special Project of Xin'an Medical and Traditional Chinese Medicine Modernization Research Institute of Great Health Research Institute: Research on the Academic Thoughts of Xin'an Medical Expert Wu Yiding in Treating Fever with Moxibustion and the Application of Moxibustion in the Treatment of Epidemic Viral Pneumonia(2023CXMMTCM022)
Cite this article
GAO Bing, LIU Pan, LI Lan, GONG Tiantian, ZHU Ling, LI Lingji, XIA Ran, MA Qiang, HU Jing, WANG Jing. Moxibustion alleviates autophagy and inhibits ferroptosis to improve cardiac function in rats with post-myocardial infarction heart failure[J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 339-349.
share this article
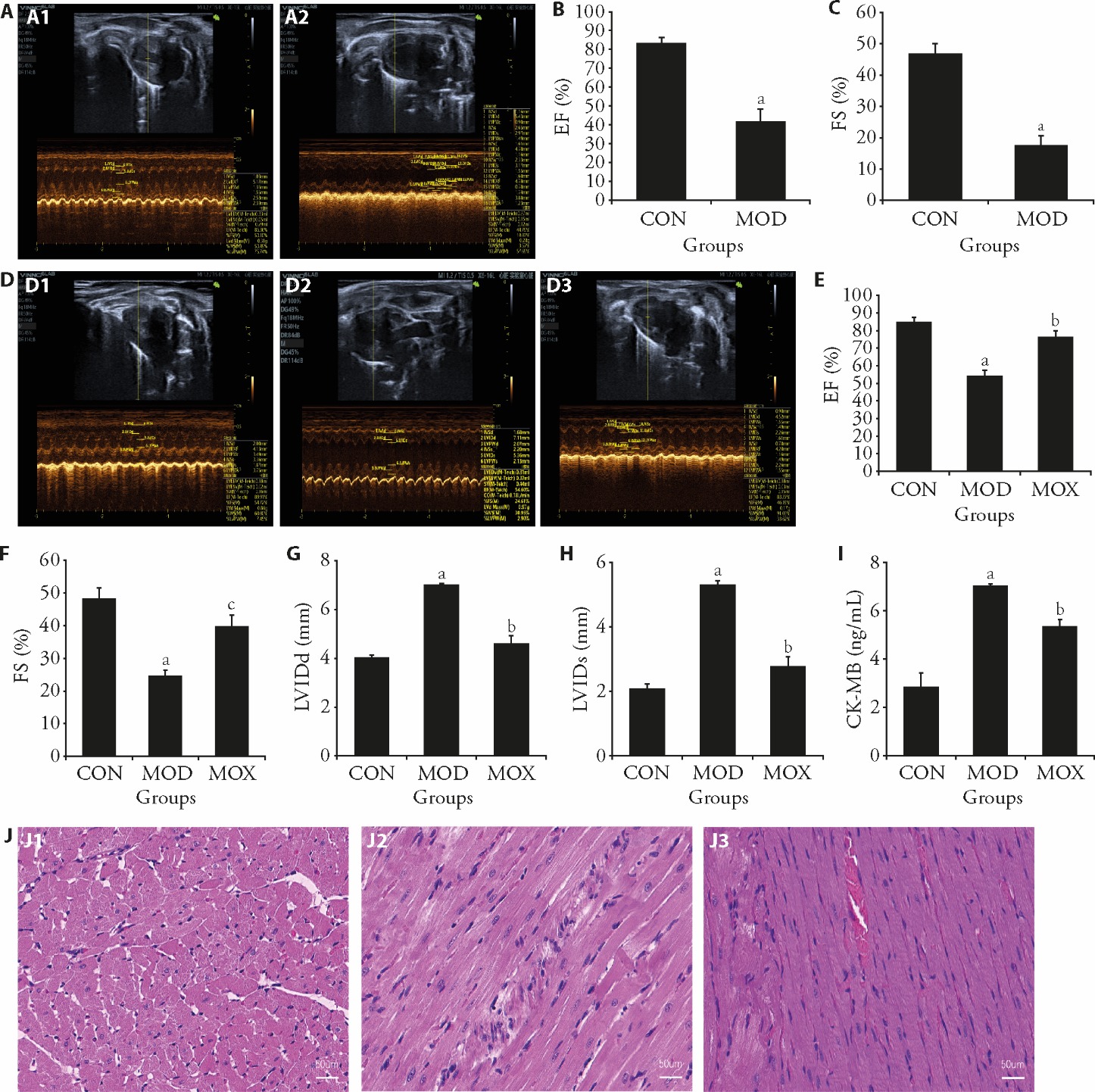
Figure 1 Protective effect of moxibustion on myocardial injury in rats with post-MI HF A: identification model of echocardiography; A1: CON group; A2: MOD group; B: EF values; C: FS values; D: three groups of rat echocardiography; D1: CON group; D2: MOD group; D3: MOX group; E: EF values; F: FS values; G: LVIDd vales; H: LVIDs values; I: comparison of ck-MB content in 3 groups of rats; J: histochemical sections of the left ventricle were stained with HE (scale bar, 50 μm); J1: CON group; J2: MOD group; J3: MOX group. CON: control group; MOD: model group, heart failure after myocardial infarction; MOX: moxibustion group, moxibustion at acupoints Feishu (BL13) and Xinshu (BL15), once daily for 30 min, q.d. 4 weeks. MI: myocardial infarction; HF: heart failure; EF: left ventricular ejection fraction; FS: fractional shortening; LVIDd: left ventricular internal dimension in diastole; LVIDs: left ventricular internal dimension systole; ck-MB: the creatine kinase-myocardial band; HE: hematoxylin and eosin staining. Data were processed by one-way analysis of variance and values are expressed as mean ± standard deviation (n = 3). Compared with the Control group, aP < 0.01; compared with the Model group, bP < 0.01, cP < 0.05.
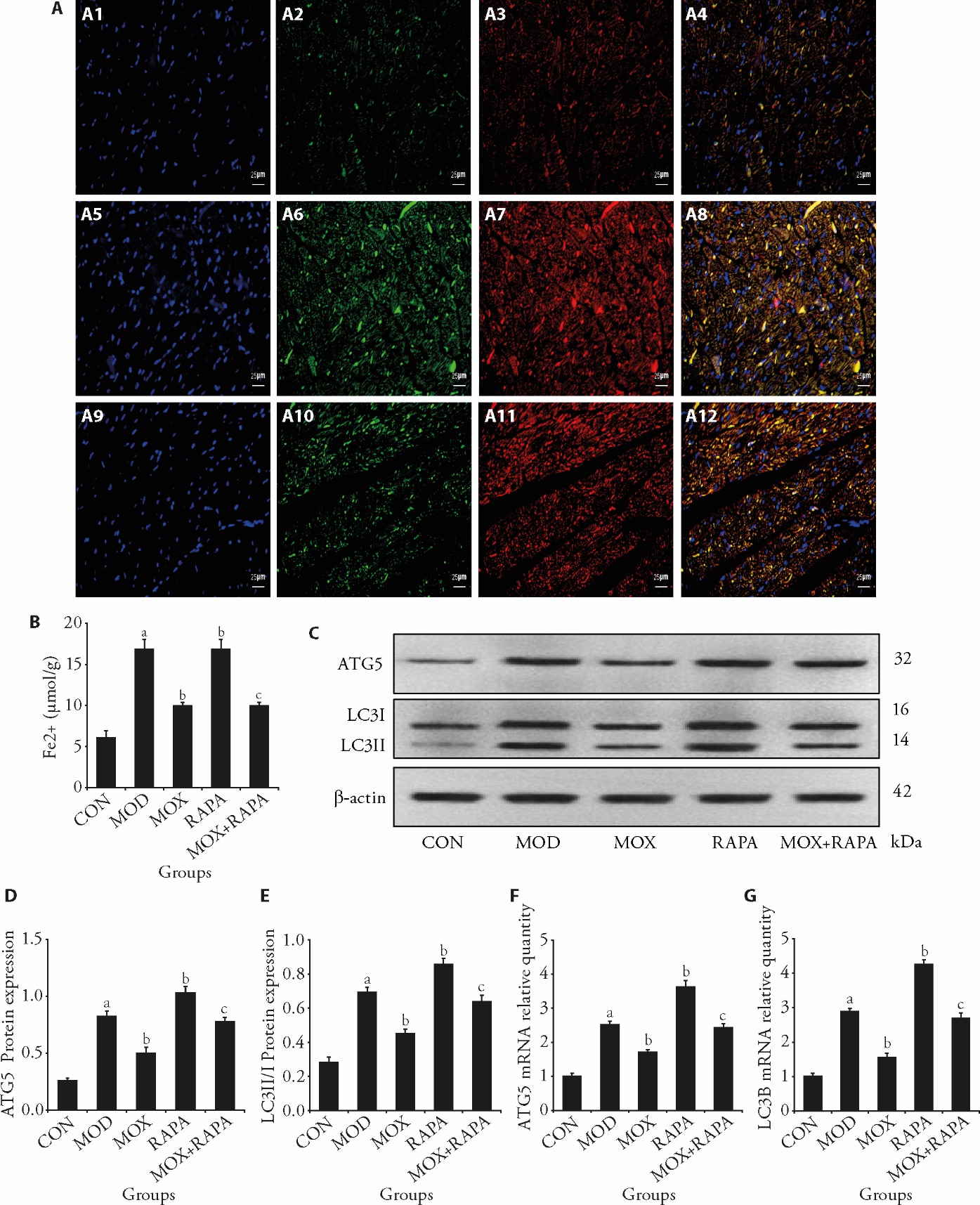
Figure 2 Moxibustion reduces autophagy and ferritinophagy caused by post-MI HF A: immunofluorescence co-localization intensity of LC3B and NCOA4 in myocardial tissues (scale bar, 50 μm); A1: DAPI staining in CON group; A2: LC3B fluorescence staining in CON group; A3: NCOA4 fluorescence staining in CON group; A4: LC3B-NCOA4 co localization in CON group; A5: DAPI staining in MOD group; A6: LC3B fluorescence staining in MOD group; A7: NCOA4 fluorescence staining in MOD group; A8: LC3B-NCOA4 co localization in MOD group; A9: DAPI staining in MOX group; A10: LC3B fluorescence staining in MOX group; A11: NCOA4 fluorescence staining in MOX group; A12: LC3B-NCOA4 co localization in MOX group; B: content of Fe2+ in myocardial tissue; C: representative Western blot bands showing protein expression of ATG5, LC3Ⅱ/I, and β-actin in the myocardium of rats; D: quantitative analysis of ATG5 protein expression normalized to β-actin; E: quantitative analysis of LC3II/I protein expression normalized to β-actin; F: ATG5 mRNA expression levels across experimental groups; G: LC3B mRNA expression levels across experimental groups. CON: control group; MOD: model group, heart failure after myocardial infarction; MOX: moxibustion group, moxibustion at acupoints Feishu (BL13) and Xinshu (BL15), once daily for 30 min, q.d. 4 weeks; RAPA: rapamycin group, 1 mg/kg, q.d. 4 weeks; MOX + RAPA: moxibustion + rapamycin group, combined moxibustion and rapamycin intervention, q.d. 4 weeks. MI: myocardial infarction; HF: heart failure; LC3B: microtubule-associated proteins 1A/1B light chain 3; NCOA4: nuclear receptor coactivator 4; DAPI: 4′, 6-diamidino-2-phenylindole; Fe2+: ferrous iron; ATG5: autophagy-related protein 5; LC3II/I: microtubule-associated protein 1A/1B light chain 3. Data were processed by one-way analysis of variance and values are expressed as mean ± standard deviation (n = 3). Compared with the Control group, aP < 0.01; compared with the Model group, bP < 0.01; compared with the rapamycin group, cP < 0.01.

Figure 3 Moxibustion inhibits post-MI HF myocardial injury-induced ferroptosis in cardiac tissue A: transmission electron microscopy observation of myocardial cells; A1: CON group; A2: MOD group; A3: MOX group; A4: RAPA group; A5: MOX + RAPA group; B: representative Western blot bands showing protein expression of NCOA4, GPX4, FTH1, and β-actin in the myocardium of rats; C: quantitative analysis of NCOA4 protein expression normalized to β-actin; D: quantitative analysis of GPX4 protein expression normalized to β-actin; E: quantitative analysis of FTH1 protein expression normalized to β-actin; F: myocardial GSH content assayed by commercial kit; G: myocardial MDA activity assayed by commercial kit; H: myocardial SOD activity assayed by commercial kit. CON: control group; MOD: model group, heart failure after myocardial infarction; MOX: moxibustion group, moxibustion at acupoints Feishu (BL13) and Xinshu (BL15), once daily for 30 min, q.d. 4 weeks; RAPA: rapamycin group, 1 mg/kg, q.d. 4 weeks; MOX + RAPA: moxibustion + rapamycin group, combined moxibustion and rapamycin intervention, q.d. 4 weeks. MI: myocardial infarction; HF: heart failure; GPX4: glutathione peroxidase 4; NCOA4: nuclear receptor coactivator 4; FTH1: ferritin, heavy polypeptide 1; GSH: glutathione; MDA: malondialdehyde; SOD: superoxide dismutase. Data were processed by one-way analysis of variance and values are expressed as mean ± standard deviation (n = 3). Compared with the Control group, aP < 0.01; compared with the Model group, bP < 0.01; compared with the rapamycin group, cP < 0.01.
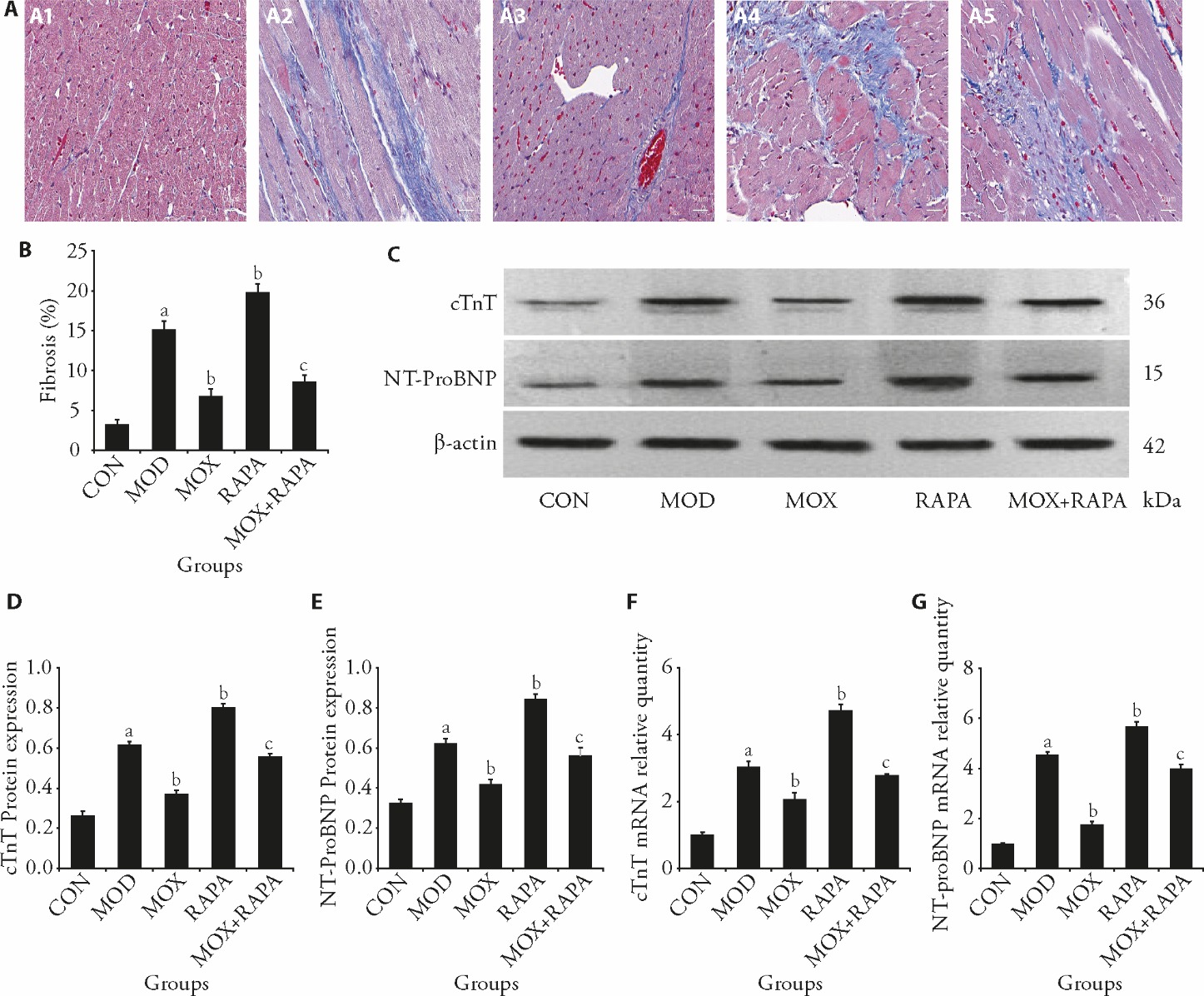
Figure 4 Moxibustion attenuates post-MI HF myocardial fibrosis and improves cardiac function A: Masson’s trichrome staining of tissue samples from rats after 8 weeks (scale bar, 50 μm); A1: CON group; A2: MOD group; A3: MOX group; A4: RAPA group; A5: MOX + RAPA group; B: the areas of cardiac fibrosis were quantified by Masson’s trichrome staining; C: representative Western blot bands showing protein expression of cTnT and NT-ProBNP, and β-actin in the myocardium of rats; D: quantitative analysis of cTnT protein expression normalized to β-actin; E: quantitative analysis of NT-ProBNP protein expression normalized to β-actin; F: cTnT mRNA expression levels across experimental groups; G: NT-ProBNP mRNA expression levels across experimental groups. CON: control group; MOD: model group, heart failure after myocardial infarction; MOX: moxibustion group, moxibustion at acupoints Feishu (BL13) and Xinshu (BL15), once daily for 30 min, q.d. 4 weeks; RAPA: rapamycin group, 1 mg/kg, q.d. 4 weeks; MOX + RAPA: moxibustion + rapamycin group, combined moxibustion and rapamycin intervention, q.d. 4 weeks; MI: myocardial infarction; HF: heart failure; cTnT: cardiac troponin T; NT-ProBNP: N-terminal prohormone of brain natriuretic peptide. Data were processed by one-way analysis of variance and values are expressed as mean ± standard deviation (n = 3). Compared with the Control group, aP < 0.01; compared with the Model group, bP < 0.01; compared with the rapamycin group, cP < 0.01.
| 1. | Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: a report of the American college of cardiology/American heart association joint committee on clinical practice guidelines. Circulation 2022; 145: e895-1032. |
| 2. | Branch of Cardiovascular Physicians, Chinese Medical Doctor Association, China Cardiovascular Health Alliance, The Expert Consensus Working Group on the Prevention and Treatment of Heart Failure After Myocardial Infarction. 2020 Expert consensus on the prevention and treatment of heart failure after myocardial infarction. Zhong Guo Xun Huan Za Zhi 2020; 35: 1166-80. |
| 3. |
Gerber Y, Weston SA, Enriquez-Sarano M, et al. Atherosclerotic burden and heart failure after myocardial infarction. JAMA Cardiol 2016; 1: 156-62.
DOI PMID |
| 4. | Zhang ZY, Jia ZZ, Song YW, et al. Optimized new Shengmai powder inhibits myocardial fibrosis in heart failure by regulating the rat sarcoma/rapidly accelerated fibrosarcoma/mitogen-activated protein kinase kinase/extracellular regulated protein kinases signaling pathway. J Tradit Chin Med 2024; 44: 448-57. |
| 5. |
Emmons-Bell S, Johnson C, Roth G. Prevalence, incidence and survival of heart failure: a systematic review. Heart 2022; 108: 1351-60.
DOI PMID |
| 6. |
Zhang Q, Li Z, Li Q, et al. Control of NAD+ homeostasis by autophagic flux modulates mitochondrial and cardiac function. EMBO J 2024; 43: 362-90.
DOI |
| 7. |
Qin W, Cao L, Massey IY. Role of PI3K/Akt signaling pathway in cardiac fibrosis. Mol Cell Biochem 2021; 476: 4045-59.
DOI PMID |
| 8. |
Mancias JD, Wang X, Gygi SP, Harper JW, Kimmelman AC. Quantitative proteomics identifies NCOA4 as the cargo receptor mediating ferritinophagy. Nature 2014; 509: 105-9.
DOI |
| 9. |
He JH, Li ZW, Xia PP, et al. Ferroptosis and ferritinophagy in diabetes complications. Mol Metab 2022; 60: 101470.
DOI URL |
| 10. |
Zheng HY, Shi L, Tong CC, Liu YN, Hou MX. circSnx 12 is involved in ferroptosis during heart failure by targeting miR-224-5p. Front Cardiovasc Med 2021; 8: 656093.
DOI URL |
| 11. |
Chen X, Yu C, Kang R, Kroemer G, Tang DL. Cellular degradation systems in ferroptosis. Cell Death Differ 2021; 28: 1135-48.
DOI PMID |
| 12. |
Zhang YX, Qu YY, Cai RP, et al. Atorvastatin ameliorates diabetic nephropathy through inhibiting oxidative stress and ferroptosis signaling. Eur J Pharmacol 2024; 976: 176699.
DOI URL |
| 13. |
Tian Y, Lu J, Hao XQ, et al. FTH1 inhibits ferroptosis through ferritinophagy in the 6-OHDA model of Parkinson’s disease. Neurotherapeutics 2020; 17: 1796-812.
DOI PMID |
| 14. |
Rui TY, Wang HC, Li QQ, et al. Deletion of ferritin H in neurons counteracts the protective effect of melatonin against traumatic brain injury-induced ferroptosis. J Pineal Res 2021; 70: 12704.
DOI PMID |
| 15. |
Jaladat AM, Alizadeh Vaghasloo M, Atarzadeh F, et al. Similarities and differences between kaiy in persian medicine and moxibustion in Chinese medicine. J Integr Med 2023; 21: 354-60.
DOI URL |
| 16. | Yang WX, Liang MZ, Xu RM. Clinical efficacy of Fuling Sini decoction combined with moxibustion therapy in the treatment of heart-Yang deficiency type chronic heart failure in the elderly. Zhong Guo Lao Nian Xue Za Zhi 2023; 43: 3077-81. |
| 17. | Wang Z, Xia R, Gao B, et al. Efficacy observation of moxibustion plus medication for chronic heart failure of blood stasis due to Qi deficiency syndrome. Shanghai Zhen Jiu Za Zhi 2023; 42: 797-801. |
| 18. | Guo Yi. Experimental acupuncture and moxibustion. Beijing: China Traditional Chinese Medicine Press, 2021: 189. |
| 19. | Li QL, Ma Q, Li Y, et al. Effect of moxibustion on cardiac function and expression of autophagy-related proteins of cardiomyocytes in chronic heart failure rats. Zhen Ci Yan Jiu 2020; 45: 259-63+74. |
| 20. |
Xia R, Wang W, Gao B, et al. Moxibustion alleviates chronic heart failure by regulating mitochondrial dynamics and inhibiting autophagy. Exp Ther Med 2022; 23: 359.
DOI PMID |
| 21. | National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide for the care and use of laboratory animals. 8th ed. Washington (DC): National Academies Press (US), 2011: 4-6. |
| 22. |
Zhang L, Gan ZK, Han LN, et al. Protective effect of heme oxygenase-1 on wistar rats with heart failure through the inhibition of inflammation and amelioration of intestinal microcirculation. J Geriatr Cardiol 2015; 12: 353-65.
DOI PMID |
| 23. | Xu L. Experimental study about prevention of anthracycline-induced myocardial injury by ginger moxibustion. Zhong Guo Xian Dai Yi Sheng 2016; 54: 139-41. |
| 24. | Liu SS, Li YB, Shi H, et al. Observation on the clinical effect of heat sensitive moxibustion on patients with heart failure and diuretic resistance. Jiangxi Zhong Yi Yao Da Xue Xue Bao 2023; 35: 65-8. |
| 25. | Li Q, Wang W, Ma Q, et al. Moxibustion improves chronic heart failure by inhibiting autophagy and inflammation via upregulation of mTOR expression. Evid Based Complement Alternat Med 2021; 2021: 6635876. |
| 26. |
Wang W, Li QL, Ma Q, et al. Effects of moxibustion at bilateral Feishu (BL13) and Xinshu (BL15) combined with benazepril on myocardial cells apoptosis index and apoptosis-related proteins cytochrome C and apoptosis-inducing factor in rats with chronic heart failure. J Tradit Chin Med 2022; 42: 227-33.
DOI |
| 27. |
Zhang H, Zhabyeyev P, Wang S, Oudit GY. Role of iron metabolism in heart failure: from iron deficiency to iron overload. Biochim Biophys Acta Mol Basis Dis 2019; 1865: 1925-37.
DOI URL |
| 28. |
Gao MH, Monian P, Pan QH, Zhang W, Xiang J, Jiang XJ. Ferroptosis is an autophagic cell death process. Cell Res 2016; 26: 1021-32.
DOI PMID |
| 29. | Fan ZY, Cai LL, Wang SN, Wang J, Chen BH. Baicalin prevents myocardial ischemia/reperfusion injury through inhibiting ACSL4 mediated ferroptosis. Front Pharmacol 2021; 4: 628988. |
| 30. |
Hou W, Xie Y, Song X, et al. Autophagy promotes ferroptosis by degradation of ferritin. Autophagy 2016; 12: 1425-8.
DOI PMID |
| 31. | Xiao Y, Ding L, Chen H, et al. Grain-moxibustion may protect myocardium by reducing oxidative stress in doxorubicin-induced cardiomyopathy rats. Zhen Ci Yan Jiu 2016; 41: 502-8. |
| 32. |
Hajri T, Gharib M, Fungwe T, M’Koma A. Very low-density lipoprotein receptor mediates triglyceride-rich lipoprotein-induced oxidative stress and insulin resistance. Am J Physiol Heart Circ Physiol 2024; 327: H733-48.
DOI URL |
| 33. |
Sahebkar A, Foroutan Z, Katsiki N, Jamialahmadi T, Mantzoros CS. Ferroptosis, a new pathogenetic mechanism in cardiometabolic diseases and cancer: is there a role for statin therapy? Metabolism 2023; 146: 155659.
DOI URL |
| 34. |
Fang YY, Chen XC, Tan QY, Zhou HH, Xu J, Gu Q. Inhibiting ferroptosis through disrupting the NCOA4-FTH1 interaction: a new mechanism of action. ACS Cent Sci 2021; 7: 980-9.
DOI URL |
| 35. |
Zeidan RS, Han SM, Leeuwenburgh C, Xiao R. Iron homeostasis and organismal aging. Ageing Res Rev 2021; 72: 101510.
DOI URL |
| 36. | Zeng Z, Huang H, Zhang J, et al. HDM induce airway epithelial cell ferroptosis and promote inflammation by activating ferritinophagy in asthma. FASEB J 2022; 36: e22359. |
| 37. |
Laukaitiene J, Gujyte G, Kadusevicius E. Cardiomyocyte damage: ferroptosis relation to ischemia-reperfusion injury and future treatment options. Int J Mol Sci 2023; 24: 12846.
DOI URL |
| 38. |
Sumneang N, Siri-Angkul N, Kumfu S, Chattipakorn SC, Chattipakorn N. The effects of iron overload on mitochondrial function, mitochondrial dynamics, and ferroptosis in cardiomyocytes. Arch Biochem Biophys 2020; 680: 108241.
DOI URL |
| 39. |
Wang XC, Wei TT, Luo JL, et al. Iron overload-dependent ferroptosis aggravates LPS-induced acute lung injury by impairing mitochondrial function. Inflammation 2024; 47: 2013-26.
DOI PMID |
| 40. |
Huang FZ, Liu XM, Liu JJ, et al. Phosphatidylethanolamine aggravates angiotensin II-induced atrial fibrosis by triggering ferroptosis in mice. Front Pharmacol 2023; 14: 1148410.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

