Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (1): 22-33.DOI: 10.19852/j.cnki.jtcm.2026.01.003
• Original Articles • Previous Articles Next Articles
Therapeutic effect and anti-inflammatory mechanism of modified Shoutai pills (改良寿胎丸) against lipopolysaccharide-induced miscarriage in mice
LI Junwei1,2,3,4, LIU Xinyue1,2,3,4, MOU Xuan1,2,3,4, XIE Hongliang1,2,3,4, GAO Yating5, ZHANG Aolin1,2,3,4, HE Tao1,2,3,4, YEUNG Tszching6, TANG Chao7, WU Xiaoke8, FAN Xiaohui1,2,3,4, WANG Chichiu6( ), LI Lu1,2,3,4,6(
), LI Lu1,2,3,4,6( )
)
- 1 College of Pharmaceutical Sciences & Women's Hospital, School of Medicine, Zhejiang University, Hangzhou 310058, China
2 State Key Laboratory of Chinese Medicine Modernization, College of Pharmaceutical Sciences, Zhejiang University, Hangzhou 310058, China
3 International Science and Technology Cooperation Base of Chinese Medicine Modernization and Big Health, Innovation Center of Yangtze River Delta, Zhejiang University, Jiaxing 314102, China
4 Modern Chinese Medicine and Reproductive Health Joint Innovation Center, Innovation Center of Yangtze River Delta, Zhejiang University, Jiaxing 314102, China
5 College of Basic Medical Sciences, Zhejiang Chinese Medical University, Hangzhou 310053, China
6 Department of Obstetrics and Gynaecology; Li Ka Shing Institute of Health Sciences; School of Biomedical Sciences; and Sichuan University-Chinese University of Hong Kong Joint Reproductive Medicine Laboratory, the Chinese University of Hong Kong, Hong Kong 999077, China
7 National Clinical Research Center for Child Health of the Children's Hospital, Zhejiang University School of Medicine, Hangzhou 310052, China
8 Department of Obstetrics and Gynecology, First Affiliated Hospital, Heilongjiang University of Chinese Medicine, Harbin 150040, China
-
Accepted:2025-10-30Online:2026-02-15Published:2026-01-28 -
Contact:Prof. LI Lu, College of Pharmaceutical Sciences & Women's Hospital, School of Medicine, Zhejiang University, Hangzhou 310058, China; State Key Laboratory of Chinese Medicine Modernization, College of Pharmaceutical Sciences, Zhejiang University, Hangzhou 310058, China; International Science and Technology Cooperation Base of Chinese Medicine Modernization and Big Health, Innovation Center of Yangtze River Delta, Zhejiang University, Jiaxing 314102, China; Modern Chinese Medicine and Reproductive Health Joint Innovation Center, Innovation Center of Yangtze River Delta, Zhejiang University, Jiaxing 314102, China; Department of Obstetrics and Gynaecology; Li Ka Shing Institute of Health Sciences; School of Biomedical Sciences; and Sichuan University-Chinese University of Hong Kong Joint Reproductive Medicine Laboratory, the Chinese University of Hong Kong, Hong Kong 999077, China.luciali@zju.edu.cn ; Prof. WANG Chichiu, Department of Obstetrics and Gynaecology; Li Ka Shing Institute of Health Sciences; School of Biomedical Sciences; and Sichuan University-Chinese University of Hong Kong Joint Reproductive Medicine Laboratory, the Chinese University of Hong Kong, Hong Kong 999077, China.ccwang@cuhk.edu.hk ; Telephone: +86-571-88208596 -
Supported by:‘Pioneer’ R&D Program of Zhejiang: Research on Key Technologies for the Development of Traditional Chinese Medicine New Drugs(2023C03004);National Key Research and Development Program of China: Mechanism Study and Clinical Exploration of Electroacupuncture Promoting Immune Normalization, Supporting the Body and Inhibiting Cancer, and Synergistic Programmed Death Receptor 1 /Programmed Cell Death Ligand 1 Monoclonal Antibody therapy for Intestinal and Biliary Tumors(2023YFC3504600);Zhejiang Province Traditional Chinese Medicine Science and Technology Project: Clinical Metabolomics Based Discovery of Effective Markers for Nourishing Yin of Radix Ophiopogonis in the Treatment of Gestational Diabetes(GZY-ZJKJ-24076);‘Pioneer’ R&D Program of Zhejiang: Analysis of the Complex System of Traditional Chinese Medicine and Development of New Chinese Medicine Drugs - Analysis of the Complex Cross Organ Action Mode of Traditional Chinese Medicine for Anti-Coronary Heart Disease and Blood Stasis Syndrome and Development of New Drugs(2024C03106);Health and Medicinal Research Fund from Health Burden, Hong Kong Special Administrative Region of the People's Republic of China: Chinese Versus Western Medicine for Threatened Miscarriage: Abridged Secondary Publication(15160971);Transverse Research Project of Zhejiang University: Development of Traditional Chinese Medicine Big Health Formula for Reproductive Health(2023-KYY-A070350007)
Cite this article
LI Junwei, LIU Xinyue, MOU Xuan, XIE Hongliang, GAO Yating, ZHANG Aolin, HE Tao, YEUNG Tszching, TANG Chao, WU Xiaoke, FAN Xiaohui, WANG Chichiu, LI Lu. Therapeutic effect and anti-inflammatory mechanism of modified Shoutai pills (改良寿胎丸) against lipopolysaccharide-induced miscarriage in mice[J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 22-33.
share this article
| Item | Control | LPS | MSTP_L | MSTP_M | MSTP_H |
|---|---|---|---|---|---|
| Weight of pregnant mice | 31.48±0.83 | 30.28±3.10 | 30.30±1.89 | 31.91±1.30 | 31.67±1.45 |
| Weight of the pregnant uterus | 2.36±0.26 | 1.58±0.55 | 2.02±0.77 | 1.98±0.50 | 1.99±0.55 |
| Litter size per pregnancy | 14.38±2.07 | 14.75±1.48 | 14.20±3.71 | 14.44±2.24 | 14.56±2.13 |
| Resorption rate | 1.59±2.97 | 94.36±9.36a | 66.47±29.73 | 66.83±42.04 | 55.88±30.13b |
Table 1 Effect of MSTP on LPS-induced miscarriage in mice
| Item | Control | LPS | MSTP_L | MSTP_M | MSTP_H |
|---|---|---|---|---|---|
| Weight of pregnant mice | 31.48±0.83 | 30.28±3.10 | 30.30±1.89 | 31.91±1.30 | 31.67±1.45 |
| Weight of the pregnant uterus | 2.36±0.26 | 1.58±0.55 | 2.02±0.77 | 1.98±0.50 | 1.99±0.55 |
| Litter size per pregnancy | 14.38±2.07 | 14.75±1.48 | 14.20±3.71 | 14.44±2.24 | 14.56±2.13 |
| Resorption rate | 1.59±2.97 | 94.36±9.36a | 66.47±29.73 | 66.83±42.04 | 55.88±30.13b |
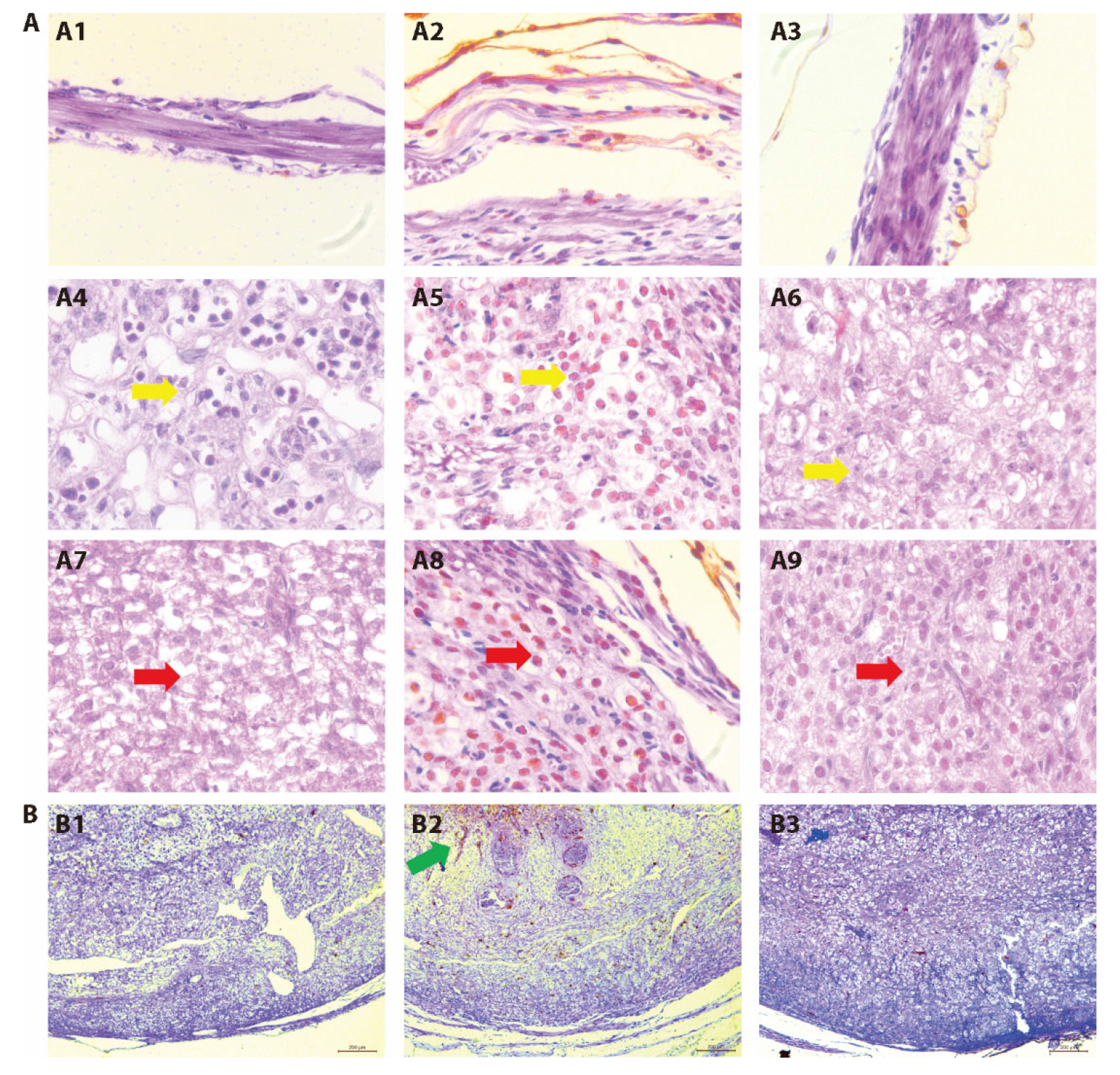
Figure 1 Effect of MSTP on pregnancy tissue morphology in LPS-abortive mice A: representative HE staining images of myometrium (A1-A3), placenta (A4-A6), and decidua (A7-A9) (× 20). A1, A4, A7: control group; A2, A5, A8: LPS group; A3, A6, A9: MSTP_H group. The yellow arrow refers to the placental cell nuclei and changes in red blood cells. The red arrow refers to decidual nuclei and staining changes. B: representative terminal deoxynucleotidyl transferase dUTP nick-end labeling staining images (× 4). B1: control group; B2: LPS group; B3: MSTP_H group. The green arrow refers to the apoptotic cells. Control group treated with oral distilled water (0.3 mL) from E1 to E9; LPS group treated with oral distilled water (0.3 mL) from E1 to E9 and LPS on E9 (0.04 μg/kg); MSTP_H group treated with MSTP from E1 to E9 (4.4 g/kg) and LPS on E9 (0.04 μg/kg). dUTP: deoxyuridine triphosphate; LPS: lipopolysaccharide; MSTP_H: modified Shoutai pills_High dose.
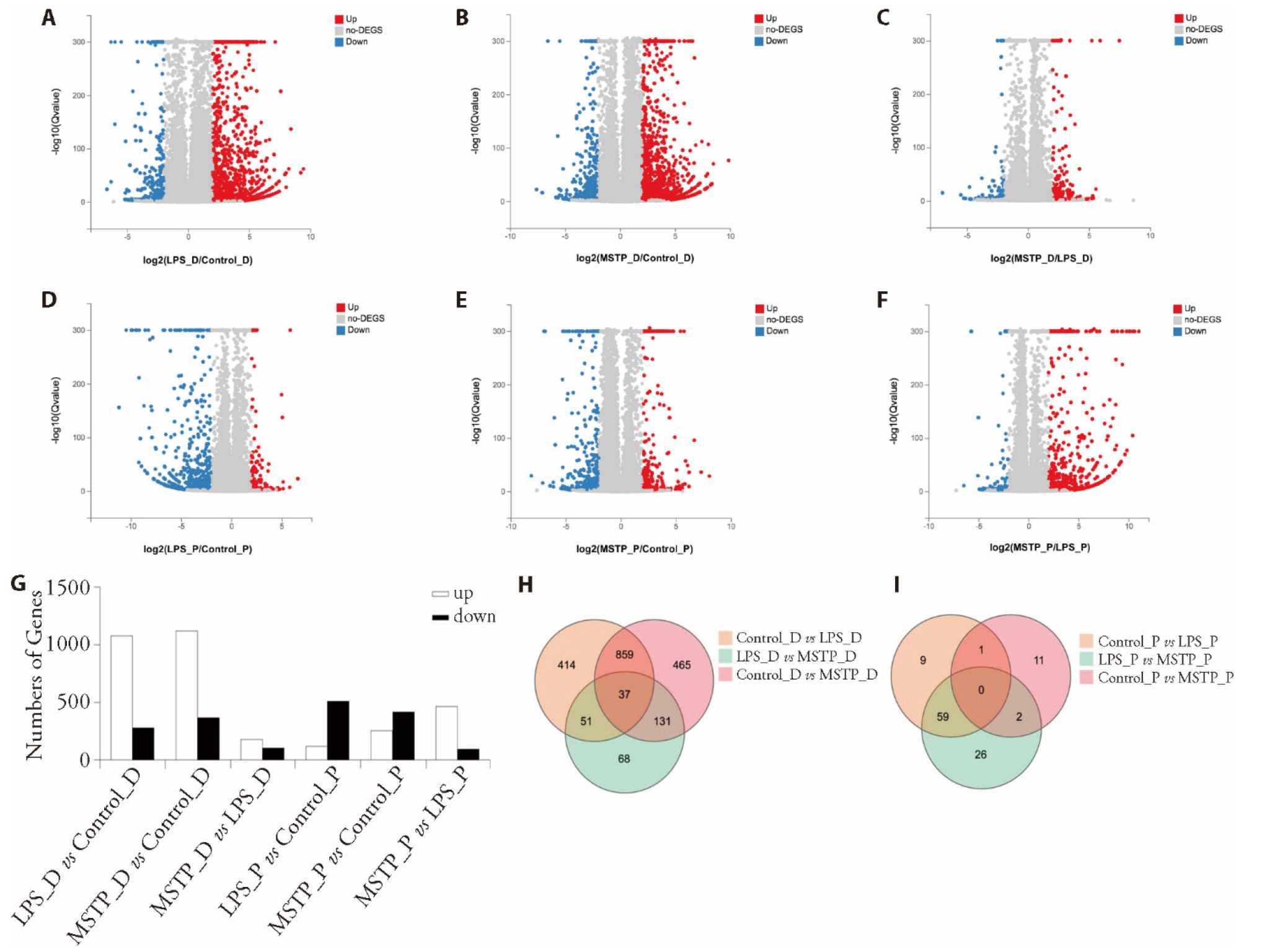
Figure 2 DEGs in decidual and placental tissues A: volcano plots of DEGs in decidual tissues in LPS group vs control group; B: volcano plots of DEGs in decidual tissues in MSTP group vs control group; C: volcano plots of DEGs in decidual tissues in MSTP group vs LPS group; D: volcano plots of DEGs in placental tissues in LPS group vs control group; E: volcano plots of DEGs in decidual tissues in MSTP group vs control group; F: volcano plots of DEGs in decidual tissues in MSTP group vs LPS group; (G) number of upregulated and downregulated genes in LPS group vs control group, MSTP group vs control group, and MSTP group vs LPS group in decidual and placental tissues; H: venn diagram of DEGs in LPS group vs control group, MSTP group vs control group, and MSTP group vs LPS group in decidual tissues; I: venn diagram of DEGs in LPS group vs control group, MSTP group vs control group, and MSTP group vs LPS group in placental tissues. Control (control_D and control_P) group treated with oral distilled water (0.3 mL) from E1 to E9; LPS (LPS_D and LPS_P) group treated with oral distilled water (0.3 mL) from E1 to E9 and LPS on E9 (0.04 μg/kg); MSTP (MSTP_D and MSTP_P) group treated with MSTP from E1 to E9 (4.4 g/kg) and LPS on E9 (0.04 μg/kg). MSTP: modified Shoutai pills; D: decidua; P: placenta; DEGs: differentially expressed genes.
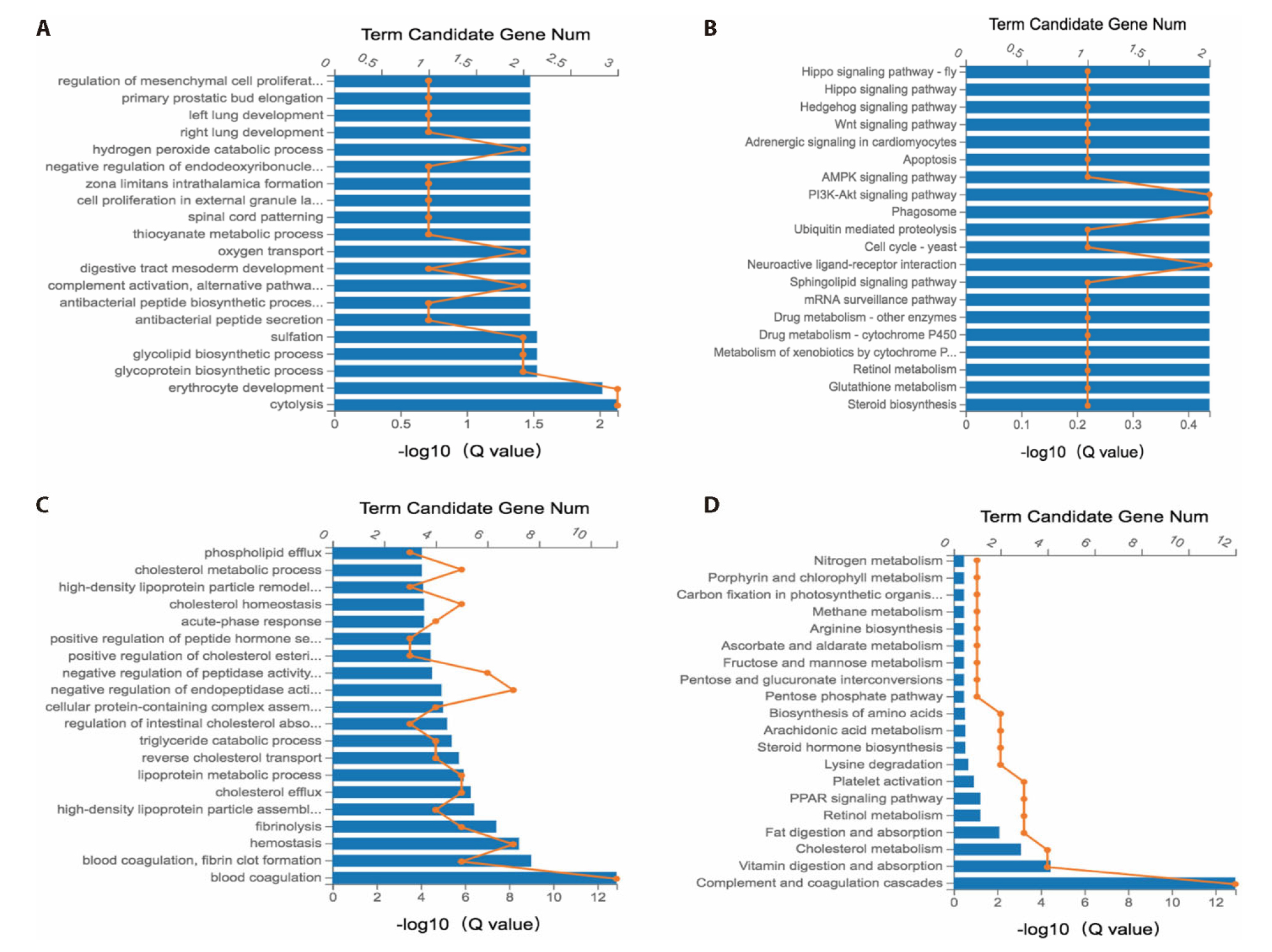
Figure 3 GO and KEGG analysis of DEGs in decidual and placental tissues from mice A: GO analysis of 51 DEGs in decidual tissues; B: KEGG analysis of 51 DEGs in decidual tissues; C: GO analysis of 59 genes in placental tissues. D: KEGG analysis of 59 genes in placental tissues. GO: gene ontology; KEGG: kyoto encyclopedia of genes and genomes; DEGs: differentially expressed genes.
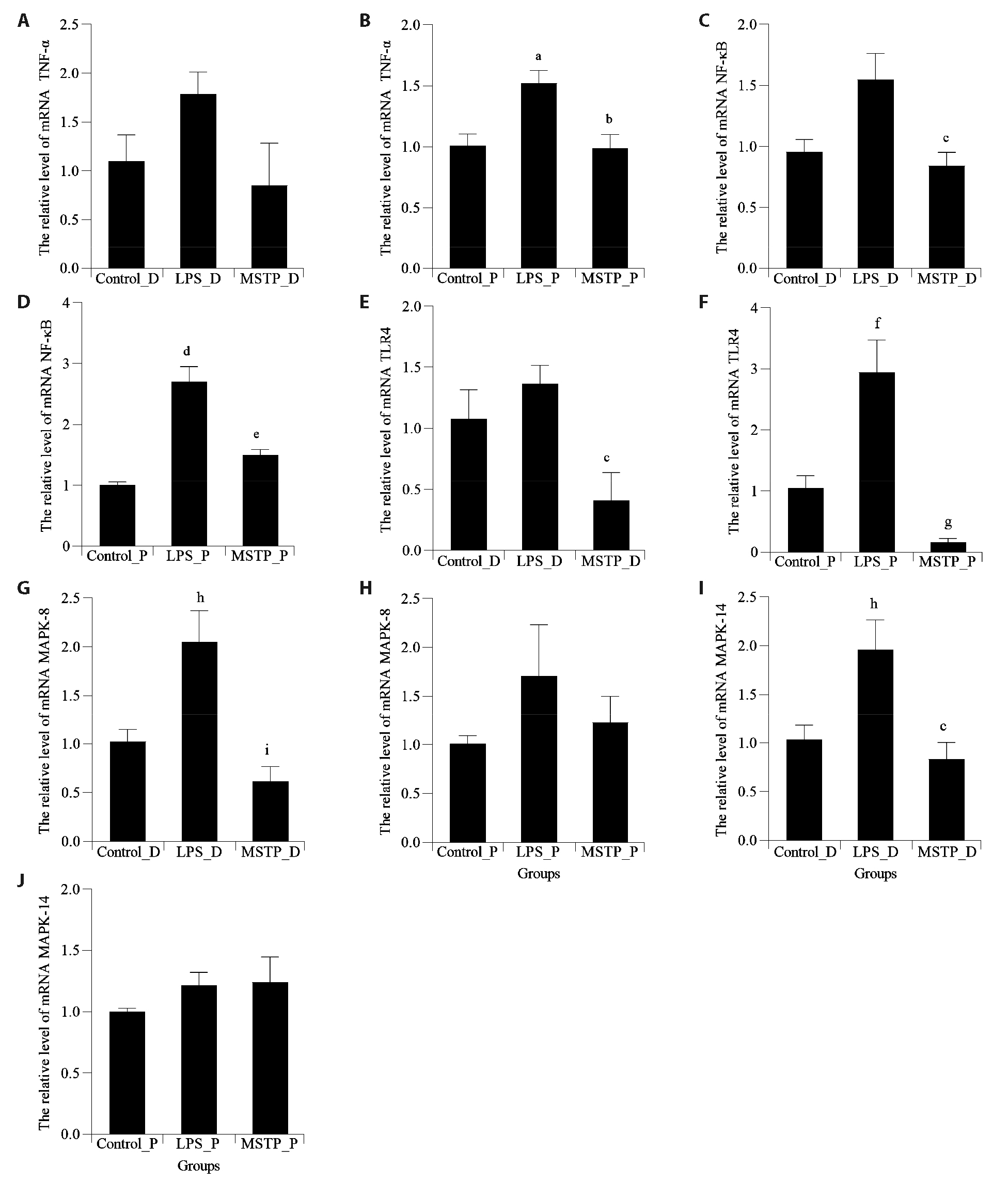
Figure 4 Expression levels of TNF-α, TLR4, NF-κB, JNK1, and p38 in decidual and placental tissues of the mice A: relative expression level of TNF-α by qRT-PCR analysis in decidual tissues; B: relative expression level of TNF-α by qRT-PCR analysis in placental tissues; C: relative expression level of NF-κB by qRT-PCR analysis in decidual tissues; D: relative expression level of NF-κB by qRT-PCR analysis in placental tissues; E: relative expression level of TLR4 by qRT-PCR analysis in decidual tissues; F: relative expression level of TLR4 by qRT-PCR analysis in placental tissues; G: relative expression level of JNK1 (MAPK-8) by qRT-PCR analysis in decidual tissues; H: relative expression level of MAPK-8 by qRT-PCR analysis in placental tissues; I: relative expression level of p38 (MAPK-14) by qRT-PCR analysis in decidual tissues; J: relative expression level of MAPK-14 by qRT-PCR analysis in placental tissues; Control_D and Control_P treated with oral distilled water (0.3 mL) from E1 to E9, LPS_D and LPS_P treated with oral distilled water (0.3 mL) from E1 to E9 and LPS on E9 (0.04 μg/kg), MSTP_D and MSTP_P treated with MSTP from E1 to E9 (4.4 g/kg) and LPS on E9 (0.04 μg/kg). D: decidua; P: placenta; DEGs: differentially expressed genes; TNF-α: tumor necrosis factor-α; NF-κB: nuclear factor kappa-B; TLR4: toll-like receptor 4; JNK-1 (MAPK-8): c-Jun N-terminal kinase 1; qRT-PCR: quantitative real-time polymerase chain reaction; p38 (MAPK-14): mitogen-activated protein kinases 14. Differences in means between groups were examined using one-way analysis of variance. Compared with the Control_P group, aP < 0.05; compared with the LPS_P group, bP < 0.05; compared with the LPS_D group, cP < 0.05; compared with the Control_P group, dP < 0.0001; compared with the LPS_P group, eP < 0.01; compared with the Control_P group, fP < 0.01; compared with the LPS_P group, gP < 0.001; compared with the Control_D group, hP < 0.05; compared with the LPS_D group, iP < 0.01.
| 1. |
Quenby S, Gallos ID, Dhillon-Smith RK, et al. Miscarriage matters: the epidemiological, physical, psychological, and economic costs of early pregnancy loss. Lancet 2021; 397: 1658-67.
DOI PMID |
| 2. |
Tessema GA, Håberg SE, Pereira G, et al. Interpregnancy interval and adverse pregnancy outcomes among pregnancies following miscarriages or induced abortions in Norway (2008-2016): a cohort study. PLoS Med 2022; 19: e1004129.
DOI URL |
| 3. |
Blomqvist L, Nyström HF, Hellgren M, et al. Preconceptual thyroid peroxidase antibody positivity in women with recurrent pregnancy losses may contribute to an increased risk for another miscarriage. Clin Endocrinol (Oxf) 2023; 98: 259-69.
DOI URL |
| 4. |
Okoth K, Subramanian A, Chandan JS, et al. Long term miscarriage-related hypertension and diabetes mellitus. Evidence from a United Kingdom population-based cohort study. PLoS One 2022; 17: e0261769.
DOI URL |
| 5. |
Belmaker I, Anca ED, Rubin LP, et al. Adverse health effects of exposure to plastic, microplastics and their additives: environmental, legal and policy implications for Israel. Isr J Health Policy Res 2024; 13: 44.
DOI |
| 6. |
Giri N, Alter BP, Savage SA, et al. Gynaecological and reproductive health of women with telomere biology disorders. Br J Haematol 2021; 193: 1238-46.
DOI URL |
| 7. |
Al Balawi AN, Alblwi NAN, Soliman R, et al. Impact of Vitamin D deficiency on immunological and metabolic responses in women with recurrent pregnancy loss: focus on VDBP/HLA-G1/CTLA-4/ENTPD1/adenosine-fetal-maternal conflict crosstalk. BMC Pregnancy Childbirth 2024; 24: 709.
DOI |
| 8. |
Ng KYB, Wellstead S, Cheong Y, et al. A randomised controlled trial of a personalised lifestyle coaching application in modifying periconceptional behaviours in women suffering from reproductive failures (iPLAN trial). BMC Womens Health 2018; 18: 196.
DOI PMID |
| 9. |
McLindon LA, James G, Beckmann MM, et al. Progesterone for women with threatened miscarriage (STOP trial): a placebo-controlled randomized clinical trial. Hum Reprod 2023; 38: 560-8.
DOI URL |
| 10. | Farren J, Jalmbrant M, Falconieri N, et al. Posttraumatic stress, anxiety and depression following miscarriage and ectopic pregnancy: a multicenter, prospective, cohort study. Am J Obstet Gynecol 2020; 222: 367.e1-e22. |
| 11. | Li L, Dou L, Leung PC, et al. Chinese herbal medicines for unexplained recurrent miscarriage. Cochrane Database Syst Rev 2016; 2016: Cd010568. |
| 12. |
Nahas R, Saliba W, Elias A, et al. The prevalence of thrombophilia in women with recurrent fetal loss and outcome of anticoagulation therapy for the prevention of miscarriages. Clin Appl Thromb Hemost 2018; 24: 122-8.
DOI URL |
| 13. |
Yamada H, Deguchi M, Saito S, et al. Intravenous immunoglobulin treatment in women with four or more recurrent pregnancy losses: a double-blind, randomised, placebo-controlled trial. EClinicalMedicine 2022; 50: 101527.
DOI URL |
| 14. |
Snoek KM, Steegers-Theunissen RPM, Hazebroek EJ, et al. The effects of bariatric surgery on periconception maternal health: a systematic review and Meta-analysis. Hum Reprod Update 2021; 27: 1030-55.
DOI PMID |
| 15. | Ghosh J, Papadopoulou A, Devall AJ, et al. Methods for managing miscarriage: a network Meta-analysis. Cochrane Database Syst Rev 2021; 2021: CD012602. |
| 16. |
Lee DYW, Li QY, Liu J, et al. Traditional Chinese herbal medicine at the forefront battle against COVID-19: clinical experience and scientific basis. Phytomedicine 2021; 80: 153337.
DOI URL |
| 17. |
Ruan LJ, Cai Y, Yin Y, et al. Shoutai pills for treating recurrent miscarriage: a systematic review and Meta-analysis of the safety and clinical efficacy in 11 randomized controlled trials. Front Pharmacol 2025; 16: 1540073.
DOI URL |
| 18. |
Yang Z, Chen K, Zhang Y, et al. Study on pharmacokinetic and tissue distribution of hyperin, astragalin, kaempferol-3-O-β-D-glucuronide from rats with multiple administrations of Semen Cuscutae processed with salt solution with effect of treating recurrent spontaneous abortion. Front Pharmacol 2024; 15: 1440810.
DOI URL |
| 19. |
Li HF, Shen QH, Li XQ, et al. The efficacy of Traditional Chinese Medicine shoutai pill combined with western medicine in the first trimester of pregnancy in women with unexplained recurrent spontaneous abortion: a systematic review and Meta-analysis. Biomed Res Int 2020; 2020: 7495161.
DOI URL |
| 20. | Zhang XC (Qing dynasty). Yi Xue Zhong Zhong Can Xi Lu. Fuzhou: Fujian Science and Technology Publishing House, 2003: 5. |
| 21. |
Zhang J, Chen L, Zheng CH, et al. Effect of shoutai pills on th1/th2 cytokines in serum and endometrium of rats with stimulated ovulation. Curr Med Sci 2019; 39: 285-90.
DOI PMID |
| 22. | Huang CS, Guan YC, Liu XY. Treatment of 31 cases of recurrent spontaneous abortion with deficiency of spleen and kidney by integrated Traditional Chinese and Western Medicine. Jiangxi Zhong Yi Yao 2016; 47: 50-2. |
| 23. | Guo M, Ma ML, Chen P. Clinical efficacy of modified Shoutai Pill combined with progesterone in the treatment of recurrent spontaneous abortion. Hubei Min Zu Da Xue Xue Bao 2018; 35: 7-9. |
| 24. | Feng YQ. Clinical effects of Shoutai pills combined with Antai Fuyuan drink on patients with unexplained recurrent spontaneous abortion due to kidney deficiency and blood stasis pattern. Zhong Cheng Yao 2019; 41: 2908-13. |
| 25. |
Li D, Jiang M, Song Y, et al. Preventive vs therapeutic effects of Shoutai Wan: maintaining an acidic microenvironment at the maternal-fetal interface to promote angiogenesis and minimize pregnancy loss in RSA mice. J Ethnopharmacol 2025; 342: 119345.
DOI URL |
| 26. | Xu L, Fu ZH, Xia JQ, et al. Determination of flavonoids in Shoutai pills by ultra performance liquid chromatography. Beijing Zhong Yi Yao Da Xue Xue Bao 2014; 33: 766-9. |
| 27. |
Wu C, Wu C, Peng L, et al. Multi-omics approaches for the understanding of therapeutic mechanism for Huangqi Longdan granule against ischemic stroke. Pharmacol Res 2024; 205: 107229.
DOI URL |
| 28. |
Deng Y, Zhong G, Jin T, et al. Mechanism exploration of Wenshen Jianpi decoction on renoprotection in diabetic nephropathy via transcriptomics and metabolomics. Phytomedicine 2025; 139: 156446.
DOI URL |
| 29. |
Liu X, Zhang F, Wang Z, et al. Altered gut microbiome accompanying with placenta barrier dysfunction programs pregnant complications in mice caused by graphene oxide. Ecotoxicol Environ Saf 2021; 207: 111143.
DOI URL |
| 30. |
Chang S, Yin T, He F, et al. CaMK4 promotes abortion-related Th17 cell imbalance by activating akt/mtor signaling pathway. Am J Reprod Immunol 2020; 84: e13315.
DOI URL |
| 31. | Chen Q. Methodology of Traditional Chinese Medicine pharmacology research. Beijing: People's Health Publishing House 2010: 28. |
| 32. | Wei W, Wu XM, Li YJ. Methodology of pharmacological experiment. Beijing: People's Health Publishing House, 2010: 69-73. |
| 33. | Huang JH, Huang XH, Chen ZY, et al. Dose conversion among different animals and healthy volunteers in pharmacological study. Zhong Guo Lin Chuang Yao Li Xue Yu Zhi Liao Xue 2004: 1069-72. |
| 34. |
Li Z, Pan H, Yang J, et al. Xuanfei Baidu formula alleviates impaired mitochondrial dynamics and activated NLRP 3 inflammasome by repressing NF-κB and MAPK pathways in LPS-induced ALI and inflammation models. Phytomedicine 2023; 108: 154545.
DOI URL |
| 35. |
Gong B, Guo D, Zheng C, et al. Complement C3a activates astrocytes to promote medulloblastoma progression through TNF-α. J Neuroinflamm 2022; 19: 1-17.
DOI |
| 36. |
Sun Q, Liu YH, Teng XJ, et al. Immunosuppression participated in complement activation-mediated inflammatory injury caused by 4-octylphenol via TLR7/IκBα/NF-κB pathway in common carp (Cyprinus carpio) gills. Aquatic Toxicol 2022; 249: 106211.
DOI URL |
| 37. |
Xie HL, Zhang AL, Mou X, et al. Chinese herbal medicine for threatened miscarriage: an updated systematic review and Meta-analysis. Front Pharmacol 2023; 14: 1083746.
DOI URL |
| 38. |
Gao F, Zhou C, Qiu W, et al. Total flavonoids from Semen Cuscutae target MMP9 and promote invasion of EVT cells via Notch/AKT/MAPK signaling pathways. Sci Rep 2018; 8: 17342.
DOI |
| 39. | Qin M, Huang Q, Yang X, et al. Taxillus chinensis (DC.) Danser: a comprehensive review on botany, traditional uses, phytochemistry, pharmacology, and toxicology. Chin Med 2022; 17: 1-29. |
| 40. |
Sun X, Zhang Y, Yang Y, et al. Qualitative and quantitative analysis of furofuran lignans, iridoid glycosides, and phenolic acids in Radix Dipsaci by UHPLC-Q-TOF/MS and UHPLC-PDA. J Pharm Biomed Anal 2018; 154: 40-7.
DOI URL |
| 41. |
Zhang Q, Wei W, Jin X, et al. Traditional uses, phytochemistry, pharmacology, quality control and clinical studies of Cimicifugae Rhizoma: a comprehensive review. Chin Med 2024; 19: 66.
DOI |
| 42. | Wu HY, Sun JM, Zhang H. The research progress of Chinese medicine Asini Corii Colla. Jilin Zhong Yi Yao 2016; 36: 57-60. |
| 43. | Jing X, Jiang H, Du HH, et al. Research progress on dioscorea opposita in China. Anhui Nong Ye Ke Xue 2016; 44: 114-7. |
| 44. |
Cui L, Guo J, Wang Z, et al. Meloxicam inhibited oxidative stress and inflammatory response of LPS-stimulated bovine endometrial epithelial cells through Nrf2 and NF-κB pathways. Int Immunopharmacol 2023; 116: 109822.
DOI URL |
| 45. |
Ryu JK, Kim SJ, Rah SH, et al. Reconstruction of LPS transfer cascade reveals structural determinants within LBP, CD14, and TLR4-MD2 for efficient LPS recognition and transfer. Immunity 2017; 46: 38-50.
DOI URL |
| 46. |
Müller MM, Baldauf C, Hornischer S, et al. Staphylococcus aureus induces tolerance in human monocytes accompanied with expression changes of cell surface markers. Front Immunol 2023; 14: 1046374.
DOI URL |
| 47. |
Feng J, Gao P, Wu T, et al. Imbalance polarization of M1/M2 macrophages in miscarried uterus. PLoS One 2024; 19: e0304590.
DOI URL |
| 48. | Yao YL, Xu XH, Jin LP. Macrophage polarization in physiological and pathological pregnancy. Front Immunol 2019; 10. |
| 49. |
Woon EV, Greer O, Shah N, et al. Number and function of uterine natural killer cells in recurrent miscarriage and implantation failure: a systematic review and Meta-analysis. Hum Reprod Update 2022; 28: 548-82.
DOI PMID |
| 50. | Bonney EA. Mapping out p38MAPK. Am J Reprod Immnol 2017; 77: 12652. |
| 51. |
Wu TT, Yang X, Zhu HP, et al. Regulatory effects of the p38 mitogen-activated protein kinase-myosin light chain kinase pathway on the intestinal epithelial mechanical barrier and the mechanism of modified Pulsatilla decoction (加味白头翁汤) in the treatment of ulcerative colitis. J Tradit Chin Med 2024; 44: 885-95.
DOI |
| 52. | Huang HY, Zhu SM, Zhong SW, et al. Fuzheng Xuanfei Huashi prescription (扶正宣肺化湿方) suppresses inflammation in lipopolysaccharide-induced lung injury in mice via toll-like recptor 4/nuclear transcription factor κB and cyclooxygenase-2/prostaglandin E 2 pathway. J Tradit Chin Med 2025; 45: 272-80. |
| 53. |
Piccinni M-P, Raghupathy R, Saito S, et al. Cytokines, hormones and cellular regulatory mechanisms favoring successful reproduction. Front Immunol 2021; 12: 717808.
DOI URL |
| 54. |
Luo K, Zhong YM, Guo YD, et al. Moxibustion inhibits the macrophage M1 polarization toll-like receptor 4/myeloid differentiation factor 88/nuclear factor kappa B signaling pathway by regulating T-cell immunoglobulin and mucin-containing protein-3 in rheumatoid arthritis. J Tradit Chin Med 2024; 44: 1227-35.
DOI |
| 55. | Liu GM, Sun JH, Li JL, et al. Clinical and animal experimental research on kidney deficiency syndrome: a review. Zhong Guo Shi Yan Fang Ji Xue Za Zhi 2024; 30: 269-80. |
| 56. |
You Z, Yang ZZ, Cao S, et al. The novel klf4/big1 regulates lps-mediated neuro-inflammation and migration in BV2 cells via PI3K/Akt/NF-kB signaling pathway. Neuroscience 2022; 488: 102-11.
DOI URL |
| 57. | Yang CY, Luo J, Peng WJ, et al. Huaiyu pill (槐榆片) alleviates inflammatory bowel disease in mice via blocking toll like receptor 4/myeloid differentiation primary response gene 88/ nuclear factor kappa B subunit 1 pathway. J Tradit Chin Med 2024; 44: 916-25. |
| 58. | Gao F, Wu H. Shoutai pill for threatened miscarriage: Meta-analysis of randomized controlled trials. Zhong Hua Zhong Yi Yao Xue Kan 2016; 34: 2637-42. |
| 59. | Guan MX, Wang Y, Zhu MY, et al. Research progress on Shoutai pill in treatment of early threatened abortion. Zhong Hua Zhong Yi Yao Xue Kan 2025; 43: 96-100. |
| 60. |
Li Y, Zhang D, Xu L, et al. Cell-cell contact with proinflammatory macrophages enhances the immunotherapeutic effect of mesenchymal stem cells in two abortion models. Cell Mol Immunol 2019; 16: 908-20.
DOI PMID |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

