Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (1): 103-112.DOI: 10.19852/j.cnki.jtcm.20231215.003
• Original articles • Previous Articles Next Articles
Taohong Siwu decoction (桃红四物汤) ameliorates atherosclerosis in rats possibly through toll-like receptor 4/myeloid differentiation primary response protein 88/nuclear factor-κB signal pathway
CHANG Fengjin1, ZHOU Peng2, LI Guoying2, ZHANG Weizhi2, ZHANG Yanyan1, PENG Daiyin3( ), CHEN Guangliang3(
), CHEN Guangliang3( )
)
- 1 Department of Pharmacy, the First Affiliated Hospital of Anhui University of Chinese Medicine, Hefei 230031, China
2 School of Integrated Traditional and Western Medicine, Anhui University of Chinese Medicine, Hefei 230012, China
3 Anhui Province Key Laboratory of Chinese Medicinal Formula, Hefei 230012, China; School of Pharmacy, Anhui University of Chinese Medicine, Hefei 230012, China
4 School of Integrated Traditional and Western Medicine, Anhui University of Chinese Medicine, Hefei 230012, China; Anhui Province Key Laboratory of Chinese Medicinal Formula, Hefei 230012, China
-
Received:2022-11-11Accepted:2023-02-17Online:2024-02-15Published:2023-12-15 -
Contact:CHEN Guangliang, School of Integrated Traditional and Western Medicine, Anhui University of Chinese Medicine, Hefei 230012, China; Anhui Province Key Laboratory of Chinese Medicinal Formula, Hefei 230012, China. chen_guangl@126.com;PENG Daiyin, Anhui Province Key Laboratory of Chinese Medicinal Formula, Hefei 230012, China; School of Pharmacy, Anhui University of Chinese Medicine, Hefei 230012, China. pengdy@ahtcm.edu.cn. Telephone: +86-551-62838661 -
Supported by:National Natural Science Foundation of China: Investigating the Mechanism of Total Saponins in Treating Gouty Arthritis Based on Toll-like Receptor /Myeloid Differentiation Factor 88/Nuclear Factor-Kappa B Signal Pathway and Nacht Leucine-Rich Repeat Protein 3-Inflammasome(81573670);Study on the Material Basis and Pathway of Taohong Siwu Decoction in Regulating the Release of Platelet Alpha Granules in Postpartum Blood Stasis(81473387);Study on the Regulatory Mechanism of Taohong Siwu Decoction on Experimental Cerebral Ischemia Angiogenesis Based on the Messenger Effect of Platelet Microparticles(81503291);Investigating the Material Basis and Molecular Mechanism of Taohong Siwu Decoction Against Vascular Dementia Based on Microdialysis Technology and NOD-Like Receptor Protein 3 Inflammasome-Vascular Endothelial Cell Interaction(81903953);Anhui Province Key Research and Development Program: Research on the Development and Preparation of Taohong Siwu Granules(1704a0802141)
Cite this article
CHANG Fengjin, ZHOU Peng, LI Guoying, ZHANG Weizhi, ZHANG Yanyan, PENG Daiyin, CHEN Guangliang. Taohong Siwu decoction (桃红四物汤) ameliorates atherosclerosis in rats possibly through toll-like receptor 4/myeloid differentiation primary response protein 88/nuclear factor-κB signal pathway[J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 103-112.
share this article
| Gene | Accession No. | Sense primer (5′-3′) | Antisense primer (5′-3′) |
|---|---|---|---|
| TLR4 | NM_019178.2 | ATCGGTGGTCAGTGTGCTTGTG | AAAGCTGAAAGCGGGGCACT |
| MyD88 | NM_198130.2 | TCCAACGCTGTCCTGTCTGCAT | TGCCACCTCAAGCAAGGCAAA |
| NF-κB | NM_001276711.1 | ACGCGGTTACGGGAGATGTGAA | TCACGGCCAAGTGCAAAGGTGT |
| β-actin | NM_031144.3 | TGGCTACAGCTTCACCACCACA | TCGGAACCGCTCATTGCCGATA |
Table 1 Primer sequence in qRT-PCR
| Gene | Accession No. | Sense primer (5′-3′) | Antisense primer (5′-3′) |
|---|---|---|---|
| TLR4 | NM_019178.2 | ATCGGTGGTCAGTGTGCTTGTG | AAAGCTGAAAGCGGGGCACT |
| MyD88 | NM_198130.2 | TCCAACGCTGTCCTGTCTGCAT | TGCCACCTCAAGCAAGGCAAA |
| NF-κB | NM_001276711.1 | ACGCGGTTACGGGAGATGTGAA | TCACGGCCAAGTGCAAAGGTGT |
| β-actin | NM_031144.3 | TGGCTACAGCTTCACCACCACA | TCGGAACCGCTCATTGCCGATA |

Figure 1 Effects of TSD on Atherosclerotic Lesions in AS rats (Sudan IV staining and HE staining) A: Sudan IV staining of aortas in different groups. A1: Control group, A2: Model group, A3: TSD-H group, A4: TSD-M group, A5: TSD-L group, A6: AT group. B: proportion of plaque area to total intimal area in different groups. C: HE staining of aortas (×200) in different groups. C1: Control group, C2: Model group, C3: TSD-H group, C4: TSD-M group, C5: TSD-L group, C6: AT group. Control group: the rats in the control group were fed with standard rat chow. Model group: the rats in model group were fed with a high-fat diet containing 84% standard rat chow, 5% yolk powder, 5% lard, 5% white sugar and 1% cholesterol for 9 subsequent weeks and performed intraperitoneal injection of VD2 at the dose of 6.0×105 IU/kg at the beginning of the experiment, then VD2 at the dose of 1.0×105 IU/kg was injected at 3th, 6th and 9th weeks. TSD-H, TSD-M, TSD-L group (20, 10, 5 g/kg): the rats in the TSD groups were treated in the same way as rats in the model group, from the 9th week, the drugs were administered intragastrically once a day for 8 weeks. AT group: the rats in the AT group were treated in the same way as rats in the model group, from the 9th week, the drugs were administered intragastrically once a day for 8 weeks at a dose of 2 mg/kg. TSD: Taohong Siwu decoction; TSD-H: Taohong Siwu decoction high dose group; TSD-M: Taohong Siwu decoction middle dose group; TSD-L: Taohong Siwu decoction low dose group; VD2: vitamin D2; AS: atherosclerosis; AT: atorvastatin group; HE: Hematoxylin and eosin. Data were expressed as mean ± standard deviation (n = 6). aP < 0.01 compared with control group; bP < 0.01, cP < 0.05, compared with model group.
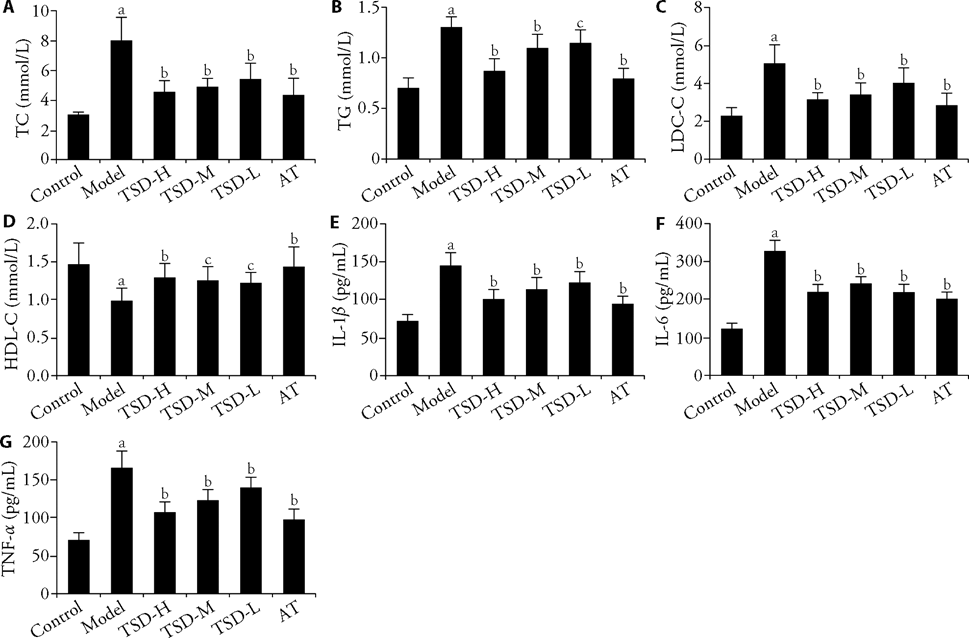
Figure 2 Effects of TSD on Dyslipidemia and Serum Inflammatory Cytokines in AS rats (serum lipid profiles and serum inflammatory cytokine levels) A-D: effects of TSD on cyslipidemia in AS rats (serum lipid profiles). A: TC in serum, B: TG in serum, C: LDL-C in serum, D: HDL-C in serum. E-G: effects of TSD on serum inflammatory cytokines in AS rats (serum inflammatory cytokine levels). E: IL-1β in serum, F: IL-6 in serum, G: TNF-α in serum. Control group: the rats in the control group were fed with standard rat chow. Model group: the rats in model group were fed with a high-fat diet containing 84% standard rat chow, 5% yolk powder, 5% lard, 5% white sugar and 1% cholesterol for 9 subsequent weeks and performed intraperitoneal injection of VD2 at the dose of 6.0×105 IU/kg at the beginning of the experiment, then VD2 at the dose of 1.0×105 IU/kg was injected at 3th, 6th and 9th weeks. TSD-H, TSD-M, TSD-L group (20, 10, 5 g/kg): the rats in the TSD groups were treated in the same way as rats in the model group, from the 9th week, the drugs were administered intragastrically once a day for 8 weeks. AT group: the rats in the AT group were treated in the same way as rats in the model group, from the 9th week, the drugs were administered intragastrically once a day for 8 weeks at a dose of 2 mg/kg. TSD: Taohong Siwu decoction; TSD-H: Taohong Siwu decoction high dose group; TSD-M: Taohong Siwu decoction middle dose group; TSD-L: Taohong Siwu decoction low dose group; VD2: vitamin D2; AT: atorvastatin group; AS: atherosclerosis; TC: total cholestero; TG: triglyceride; LDL-C: low density lipoprotein-cholesterol; HDL-C: high density lipid-cholesterol; IL-1β: interleukin-1β; IL-6: interleukin-6; TNF-α: tumor necrosis factor-α. Data were expressed as mean ± standard deviation (n = 10). aP < 0.01 compared with control group; bP < 0.01, cP < 0.05, compared with model group.
| Chemical | Vina score | Cavity score | Center (x, y, z) | Size (x, y, z) |
|---|---|---|---|---|
| Native ligand | -5.7 | 254 | 0, 2, 16 | 21, 21, 21 |
| Paeoniflorin | -6.7 | 254 | 0, 2, 16 | 23, 23, 23 |
| Verbascoside | -6.6 | 254 | 0, 2, 16 | 27, 27, 27 |
| Laetrile | -5.6 | 254 | 0, 2, 16 | 24, 24, 24 |
| Hydroxysafflor yellow | -5.3 | 254 | 0, 2, 16 | 25, 25, 25 |
| Ferulic acid | -5.0 | 254 | 0, 2, 16 | 19, 19, 19 |
| Oshaic lactone | -5.0 | 254 | 0, 2, 16 | 18, 18, 18 |
| Gallic acid | -4.9 | 254 | 0, 2, 16 | 17, 17, 17 |
| 5-hydroxymethyl furfuraldehyde | -4.1 | 254 | 0, 2, 16 | 16, 16, 16 |
Table 2 Docking of main components of TSD with TLR4
| Chemical | Vina score | Cavity score | Center (x, y, z) | Size (x, y, z) |
|---|---|---|---|---|
| Native ligand | -5.7 | 254 | 0, 2, 16 | 21, 21, 21 |
| Paeoniflorin | -6.7 | 254 | 0, 2, 16 | 23, 23, 23 |
| Verbascoside | -6.6 | 254 | 0, 2, 16 | 27, 27, 27 |
| Laetrile | -5.6 | 254 | 0, 2, 16 | 24, 24, 24 |
| Hydroxysafflor yellow | -5.3 | 254 | 0, 2, 16 | 25, 25, 25 |
| Ferulic acid | -5.0 | 254 | 0, 2, 16 | 19, 19, 19 |
| Oshaic lactone | -5.0 | 254 | 0, 2, 16 | 18, 18, 18 |
| Gallic acid | -4.9 | 254 | 0, 2, 16 | 17, 17, 17 |
| 5-hydroxymethyl furfuraldehyde | -4.1 | 254 | 0, 2, 16 | 16, 16, 16 |
| Chemical | Vina score | Cavity score | 8Center (x, y, z) | Size (x, y, z) |
|---|---|---|---|---|
| Native ligand | -7.1 | 554 | -7, 7, 9 | 27, 27, 27 |
| Verbascoside | -6.8 | 554 | -7, 7, 9 | 27, 27, 27 |
| Paeoniflorin | -6.8 | 554 | -7, 7, 9 | 23, 23 23 |
| Hydroxysafflor yellow | -6.8 | 554 | -7, 7, 9 | 25, 25, 25 |
| Laetrile | -6.6 | 554 | -7, 7, 9 | 24, 24, 24 |
| Ferulic acid | -5.3 | 554 | -7, 7, 9 | 19, 19, 19 |
| Oshaic lactone | -4.9 | 554 | -7, 7, 9 | 18, 24, 18 |
| Gallic acid | -4.8 | 554 | -7, 7, 9 | 17, 24, 17 |
| 5-hydroxymethyl furfuraldehyde | -3.8 | 554 | -7, 7, 9 | 16, 24, 16 |
Table 3 Docking of main components of TSD with MyD88
| Chemical | Vina score | Cavity score | 8Center (x, y, z) | Size (x, y, z) |
|---|---|---|---|---|
| Native ligand | -7.1 | 554 | -7, 7, 9 | 27, 27, 27 |
| Verbascoside | -6.8 | 554 | -7, 7, 9 | 27, 27, 27 |
| Paeoniflorin | -6.8 | 554 | -7, 7, 9 | 23, 23 23 |
| Hydroxysafflor yellow | -6.8 | 554 | -7, 7, 9 | 25, 25, 25 |
| Laetrile | -6.6 | 554 | -7, 7, 9 | 24, 24, 24 |
| Ferulic acid | -5.3 | 554 | -7, 7, 9 | 19, 19, 19 |
| Oshaic lactone | -4.9 | 554 | -7, 7, 9 | 18, 24, 18 |
| Gallic acid | -4.8 | 554 | -7, 7, 9 | 17, 24, 17 |
| 5-hydroxymethyl furfuraldehyde | -3.8 | 554 | -7, 7, 9 | 16, 24, 16 |
| Chemical | Vina score | Cavity score | Center (x, y, z) | Size (x, y, z) |
|---|---|---|---|---|
| Native ligand | -5.6 | 408 | 7, 1, 6 | 25, 25, 25 |
| Hydroxysafflor yellow | -6.1 | 408 | 7, 1, 6 | 25, 25, 25 |
| Laetrile | -5.9 | 408 | 7, 1, 6 | 24, 24, 24 |
| Verbascoside | -5.8 | 408 | 7, 1, 6 | 27, 27, 27 |
| Paeoniflorin | -5.2 | 408 | 7, 1, 6 | 23, 23, 23 |
| Gallic acid | -5.2 | 408 | 7, 1, 6 | 17, 17, 17 |
| Oshaic lactone | -5.1 | 408 | 7, 1, 6 | 18, 18, 18 |
| Ferulic acid | -4.7 | 408 | 7, 1, 6 | 19, 19, 19 |
| 5-hydroxymethyl furfuraldehyde | -4.4 | 408 | 7, 1, 6 | 16, 16, 16 |
Table 4 Docking of main components of TSD with NF-κB
| Chemical | Vina score | Cavity score | Center (x, y, z) | Size (x, y, z) |
|---|---|---|---|---|
| Native ligand | -5.6 | 408 | 7, 1, 6 | 25, 25, 25 |
| Hydroxysafflor yellow | -6.1 | 408 | 7, 1, 6 | 25, 25, 25 |
| Laetrile | -5.9 | 408 | 7, 1, 6 | 24, 24, 24 |
| Verbascoside | -5.8 | 408 | 7, 1, 6 | 27, 27, 27 |
| Paeoniflorin | -5.2 | 408 | 7, 1, 6 | 23, 23, 23 |
| Gallic acid | -5.2 | 408 | 7, 1, 6 | 17, 17, 17 |
| Oshaic lactone | -5.1 | 408 | 7, 1, 6 | 18, 18, 18 |
| Ferulic acid | -4.7 | 408 | 7, 1, 6 | 19, 19, 19 |
| 5-hydroxymethyl furfuraldehyde | -4.4 | 408 | 7, 1, 6 | 16, 16, 16 |

Figure 3 Effects of TSD on the mRNA expression levels of TLR4/MyD88/NF-κB signaling pathway components in aorta of AS rats Control group: the rats in the control group were fed with standard rat chow. Model group: the rats in model group were fed with a high-fat diet containing 84% standard rat chow, 5% yolk powder, 5% lard, 5% white sugar and 1% cholesterol for 9 subsequent weeks and performed intraperitoneal injection of VD2 at the dose of 6.0×105 IU/kg at the beginning of the experiment, then VD2 at the dose of 1.0 × 105 IU/kg was injected at 3th, 6th and 9th weeks. TSD-H, TSD-M, TSD-L group (20, 10, 5 g/kg): the rats in the TSD groups were treated in the same way as rats in the model group, from the 9th week, the drugs were administered intragastrically once a day for 8 weeks. AT group: the rats in the AT group were treated in the same way as rats in the model group, from the 9th week, the drugs were administered intragastrically once a day for 8 weeks at a dose of 2 mg/kg.TSD: Taohong Siwu decoction; TSD-H: Taohong Siwu decoction high dose group; TSD-M: Taohong Siwu decoction middle dose group; TSD-L: Taohong Siwu decoction low dose group; VD2: vitamin D2; AT: atorvastatin group; mRNA: messenger RNA; TLR4: toll-like receptor 4; MyD88: myeloid differentiation factor 88; NF-κB: nuclear factor-kappa B. Data were expressed as mean ± standard deviation (n = 3). aP < 0.01, compared with control group; bP < 0.01, compared with model group.
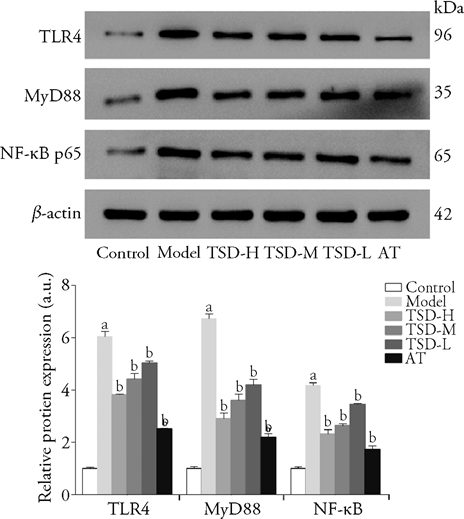
Figure 4 Effect of TSD on the protien expression levels of TLR4/MyD88/NF-κB signaling pathway components in aorta of AS rats Control group: the rats in the control group were fed with standard rat chow. Model group: the rats in model group were fed with a high-fat diet containing 84% standard rat chow, 5% yolk powder, 5% lard, 5% white sugar and 1% cholesterol for 9 subsequent weeks and performed intraperitoneal injection of VD2 at the dose of 6.0 × 105 IU/kg at the beginning of the experiment, then VD2 at the dose of 1.0 × 105 IU/kg was injected at 3th, 6th and 9th weeks. TSD-H, TSD-M, TSD-L group (20, 10, 5 g/kg): the rats in the TSD groups were treated in the same way as rats in the model group, from the 9th week, the drugs were administered intragastrically once a day for 8 weeks. AT group: the rats in the AT group were treated in the same way as rats in the model group, from the 9th week, the drugs were administered intragastrically once a day for 8 weeks at a dose of 2 mg/kg. TSD: Taohong Siwu decoction; TSD-H: Taohong Siwu decoction high dose group; TSD-M: Taohong Siwu decoction middle dose group; TSD-L: Taohong Siwu decoction low dose group; VD2: vitamin D2; AT: atorvastatin group; TLR4: toll-like receptor 4; MyD88: myeloid differentiation factor 88; NF-κB: nuclear factor-kappa B. Data were expressed as mean ± standard deviation (n = 3). aP < 0.01, compared with control group; bP < 0.01, compared with model group.
| 1. |
Timmis A, Townsend N, Gale CP, et al. European society of cardiology: cardiovascular disease statistic 2019. Eur Heart J 2020; 41: 12-85.
DOI PMID |
| 2. |
Leong DP, Joseph PG, McKee M, et al. Reducing the global burden of cardiovascular disease, part 2: prevention and treatment of cardiovascular disease. Circ Res 2017; 121: 695-710.
DOI PMID |
| 3. | Hu SS. Report on cardiovascular health and diseases in China 2021:an updated summary. J Geriatr Cardiol 2023; 20: 1-32. |
| 4. |
Lusis AJ. Atherosclerosis. Nature 2000; 407: 233-41.
DOI |
| 5. |
Moriya J. Critical roles of inflammation in atherosclerosis. J Cardiol 2019; 73: 22-7.
DOI PMID |
| 6. |
Lu Z, Zhang X, Li Y, et al. TLR4 antagonist reduces early-stage atherosclerosis in diabetic apolipoprotein E-deficient mice. J Endocrinol 2013; 216: 61-71.
DOI PMID |
| 7. | Roshan M, Amos T, Pace NP. The role of TLR2, TLR4, and TLR9 in the pathogenesis of atherosclerosis. Int J Inflam 2016; 1532832. |
| 8. |
Hayashi C, Papadopoulos G, Gudino CV, et al. Protective role for TLR4 signaling in atherosclerosis progression as revealed by infection with a common oral pathogen. J Immunol 2012; 189: 3681-88.
PMID |
| 9. |
BjoRkbacka H, Kunjathoor VV, Moore K J, et al. Reduced atherosclerosis in MyD88-null mice links elevated serum cholesterol levels to activation of innate immunity signaling pathways. Nat Med 2004; 10: 416-21.
PMID |
| 10. |
Monaco C, Paleolog E. Nuclear factor κB: a potential therapeutic target in atherosclerosis and thrombosis. Cardiovasc Res 2004; 61: 671-82.
DOI URL |
| 11. |
Li L, Yang N, Nin L, et al. Chinese herbal medicine formula Taohong Siwu decoction protects against cerebral ischemia-reperfusion injury via PI3K/Akt and the Nrf2 signaling pathway. J Nat Med 2015; 69:76-85.
DOI URL |
| 12. |
Ma Q, Li PL, Hua YL, et al. Effects of Tao-Hong-Si-Wu decoction on acute blood stasis in rats based on a LC-Q/TOF-MS metabolomics and network approach. Biomed Chromatogr 2018; 32: e4144.
DOI URL |
| 13. | Chen FF, Wang MM, Xia WW, et al. Taohong Siwu decoction promotes angiogenesis after cerebral ischaemia in rats via platelet microparticles. Chin J Nat Med 2020; 18: 620-7. |
| 14. |
Deng J, Li C, Wang H, et al. Amygdalin mediates relieved atherosclerosis in apolipoprotein E deficient mice through the induction of regulatory T cells. Biochem Biophys Res Commun 2011; 411: 523-9.
DOI URL |
| 15. |
Wang Y, Jia Q, Zhang Y, et al. Amygdalin attenuates atherosclerosis and plays an anti-inflammatory role in ApoE knock-out mice and bone marrow-derived macrophages. Front Pharmacol 2020; 11: 590929.
DOI URL |
| 16. |
Li H, Jiao Y, Xie M. Paeoniflorin ameliorates atherosclerosis by suppressing TLR4-mediated NF-κB activation. Inflammation 2017; 40: 2042-51.
DOI PMID |
| 17. |
Zhang X, Wang D, Ren X, et al. System Bioinformatic approach through molecular docking, network pharmacology and microarray data analysis to determine the molecular mechanism underlying the effects of Rehmanniae Radix Praeparata on cardiovascular diseases. Curr Protein Pept Sci 2019; 20: 964-75.
DOI URL |
| 18. |
Wu YC, Hsieh CL. Pharmacological effects of Radix Angelica Sinensis (Danggui) on cerebral infarction. Chin Med 2011; 6: 32.
DOI URL |
| 19. |
Tu Y, Xue Y, Guo D, et al. Carthami flos: a review of its ethnopharmacology, pharmacology and clinical applications. Rev Bras Farmacogn 2015; 25: 553-66.
DOI URL |
| 20. | Bu Y, Zhao P, Ke W. Clinical study of Taohong Siwu Tang for diabetes of phlegm and blood stasis type with carotid atherosclerosis. Xin Zhong Yi 2018; 50: 103-5. |
| 21. |
Saikia S, Bordoloi M. Molecular docking: challenges, advances and its use in drug discovery perspective. Curr Drug Targets 2019; 20: 501-21.
DOI PMID |
| 22. |
Huang S, Chen Y, Pan L, et al. Exploration of the potential mechanism of Taohong Siwu decoction for the treatment of breast cancer based on network pharmacology and in vitro experimental verification. Front Oncol 2021; 11: 731522.
DOI URL |
| 23. |
Kim HM, Park BS, Kim JI, et al. Crystal structure of the TLR4-MD-2 complex with bound endotoxin antagonist Eritoran. Cell 2007; 130: 906-17.
PMID |
| 24. |
Bovijn C, Desmet AS, Uyttendaele I, et al. Identification of binding sites for myeloid differentiation primary response gene 88 (MyD88) and toll-like receptor 4 in MyD88 adapter-like (Mal). J Biol Chem 2013; 288: 12054-66.
DOI PMID |
| 25. |
Qin J, Clore GM, Kennedy WMP, et al. Solution structure of human thioredoxin in a mixed disulfide intermediate complex with its target peptide from the transcription factor NF kappa B. Structure 1995; 3: 289-97.
PMID |
| 26. |
Liu Y, Grimm M, Dai W, et al. CB-Dock: a web server for cavity detection-guided protein-ligand blind docking. Acta Pharmacol Sin 2020; 41: 138-44.
DOI PMID |
| 27. |
Taleb S. Inflammation in atherosclerosis. Arch Cardiovasc Dis 2016; 109: 708-715.
DOI PMID |
| 28. |
Falk E. Pathogenesis of atherosclerosis. J Am Coll Cardiol 2006; 47: C7-12.
DOI PMID |
| 29. | Ivanova EA, Myasoedova VA, Melnichenko AA, et al. Small dense low-density lipoprotein as biomarker for atherosclerotic diseases. Oxid Med Cell Longev 2017; 2017: 1273042. |
| 30. |
Rhoads JP, Major AS. How oxidized low-density lipoprotein activates inflammatory responses. Crit Rev Immunol 2018; 38: 333-42.
DOI PMID |
| 31. |
Li B, Xia Y, Hu B. Infection and atherosclerosis: TLR-dependent pathways. Cell Mol Life Sci 2020; 77: 2751-69.
DOI PMID |
| 32. | Zhang Y, Ren P, Kang Q, et al. Effect of tetramethylpyrazine on atherosclerosis and SCAP/SREBP-1c signaling pathway in ApoE-/- mice fed with a high-fat diet. Evid-Based Compl Alt 2017; 2017: 3121989. |
| 33. |
Chen Z, Gao X, Jiao Y, et al. Tanshinone ⅡA exerts anti-inflammatory and immune-regulating effects on vulnerable atherosclerotic plaque partially via the TLR4/MyD88/NF-κB signal pathway. Front Pharmacol 2019; 10: 850.
DOI URL |
| 34. |
Xie X, Shi X, Liu M. The roles of TLR gene polymorphisms in atherosclerosis: a systematic review and Meta-analysis of 35 317 subjects. Scand J Immunol 2017; 86: 50-8.
DOI PMID |
| 35. |
Xu XH, Shah PK, Faure E, et al. Toll-like receptor-4 is expressed by macrophages in murine and human lipid-rich atherosclerotic plaques and upregulated by oxidized LDL. Circulation 2001; 104: 3103-8.
DOI PMID |
| 36. |
Bayer AL, Alcaide P. MyD88: At the heart of inflammatory signaling and cardiovascular disease. J Mol Cell Cardiol 2021; 161: 75-85.
DOI PMID |
| 37. | Liu T, Zhang L, Joo D, et al. NF-κB signaling in inflammation. Signal Transduct Target Ther 2017; 2: 17023. |
| 38. |
Gragnano F, Calabro P. Role of dual lipid-lowering therapy in coronary atherosclerosis regression: evidence from recent studies. Atherosclerosis 2018; 269: 219-28.
DOI PMID |
| 39. |
Kouhpeikar H, Delbari Z, Sathyapalan T, et al. The effect of statins through mast cells in the pathophysiology of atherosclerosis: a review. Curr Atheroscler Rep 2020; 22: 19.
DOI PMID |
| 40. |
Li H, Dai M, Jia W. Paeonol attenuates high-fat-diet-induced atherosclerosis in rabbits by anti-inflammatory activity. Planta Med 2009; 75: 7-11.
DOI PMID |
| 41. |
Wu H, Song A, Hu W, et al. The anti-atherosclerotic effect of paeonol against vascular smooth muscle cell proliferation by up-regulation of autophagy via the AMPK/mTOR signaling pathway. Front Pharmacol 2018; 8: 948.
DOI URL |
| 42. | Kosmas CE, Martinez I, Sourlas A, et al. High-density lipoprotein (HDL) functionality and its relevance to atherosclerotic cardiovascular disease. Drugs in Context 2018; 7: 212525. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

