Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (2): 303-314.DOI: 10.19852/j.cnki.jtcm.20230510.001
Previous Articles Next Articles
Yemazhui (Herba Eupatorii Lindleyani) ameliorates lipopolysaccharide-induced acute lung injury via modulation of the toll-like receptor 4/nuclear factor kappa-B/nod-like receptor family pyrin domain-containing 3 protein signaling pathway and intestinal flora in rats
REN Li1, HAI Yang2, YANG Xue1, LUO Xianqin1( )
)
- 1 Chongqing Key Laboratory of Traditional Chinese Medicine for Prevention and Cure of Metabolic Diseases, College of Traditional Chinese Medicine, Chongqing Medical University, Chongqing 400016, China
2 College of Basic Medicine, Chongqing Medical University, Chongqing 400016, China
-
Received:2022-11-11Accepted:2023-05-06Online:2024-04-15Published:2023-05-10 -
Contact:LUO Xianqin, Chongqing Key Laboratory of Traditional Chinese Medicine for Prevention and Cure of Metabolic Diseases, College of Traditional Chinese Medicine, Chongqing Medical University, Chongqing 400016, China.lxq_0203@163.com Telephone: +86-13983844070 -
Supported by:Natural Science Foundation Project of Chongqing Municipality: a Metabolome-based Study on the Protective Mechanism of Yemazhui (Herba Eupatorii Lindleyani) Sesquiterpene Lactones Against Acute Lung Injury(cstc2021jcyj-msxmX0365);Science and Technology Research Program of Chongqing Municipal Education Commission: a Cytokine Storm-based Study of the Protective Effect of Yemazhui (Herba Eupatorii Lindleyani) Extract Intervention on COVID-19 Lung Injury(KJZD-K202215101)
Cite this article
REN Li, HAI Yang, YANG Xue, LUO Xianqin. Yemazhui (Herba Eupatorii Lindleyani) ameliorates lipopolysaccharide-induced acute lung injury via modulation of the toll-like receptor 4/nuclear factor kappa-B/nod-like receptor family pyrin domain-containing 3 protein signaling pathway and intestinal flora in rats[J]. Journal of Traditional Chinese Medicine, 2024, 44(2): 303-314.
share this article
| Gene | Forward primer (5’-3’) | Reverse primer (5’-3’) |
|---|---|---|
| TNF-α | ATGGGCTCCCTCTCATCAGTTCC | CCTCCGCTTGGTGGTTTGCTAC |
| IL-1β | AATCTCACAGCAGCATCTCGACAAG | TCCACGGGCAAGACATAGGTAGC |
| IL-6 | ACTTCCAGCCAGTTGCCTTCTTG | TGGTCTGTTGTGGGTGGTATCCTC |
| GAPDH | GACATGCCGCCTGGAGAAAC | A GCCCAGGATGCCCTTTA GT |
Table 1 Primer sequences used for qRT-PCR
| Gene | Forward primer (5’-3’) | Reverse primer (5’-3’) |
|---|---|---|
| TNF-α | ATGGGCTCCCTCTCATCAGTTCC | CCTCCGCTTGGTGGTTTGCTAC |
| IL-1β | AATCTCACAGCAGCATCTCGACAAG | TCCACGGGCAAGACATAGGTAGC |
| IL-6 | ACTTCCAGCCAGTTGCCTTCTTG | TGGTCTGTTGTGGGTGGTATCCTC |
| GAPDH | GACATGCCGCCTGGAGAAAC | A GCCCAGGATGCCCTTTA GT |

Figure 1 Main chemical components of HEL and histopathological changes in lung tissue Base peak chromatogram from UPLC-Q-TOF/MS analysis of HEL under A: positive ion mode and B: negative ion mode. C: representative photomicrographs of HE staining of lung tissues in each group (magnification, × 100). C1: control group (distilled water by gavage for a week); C2: lipopolysaccharide group (distilled water by gavage for a week); C3: dexamethasone group (5 mg/kg by gavage for a week); C4: Yemazhui (Herba Eupatorii Lindleyani) low-dose group (6 g/kg by gavage for a week); C5: Yemazhui (Herba Eupatorii Lindleyani) medium-dose group (18 g/kg by gavage for a week); C6: Yemazhui (Herba Eupatorii Lindleyani) high-dose group (54 g/kg by gavage for a week). HEL: Yemazhui (Herba Eupatorii Lindleyani); UPLC-Q-TOF/MS: ultra-performance liquid chromatography-quadrupole-time of flight-mass spectrometry; HE: hematoxylin and eosin staining.
| Group | n | Lung coefficient (%) | Wet/dry weight ratio (%) | Histopathological score |
|---|---|---|---|---|
| Control | 9 | 1.13±0.18 | 11.14±2.58 | 1.00±0.50 |
| LPS | 9 | 1.36±0.18a | 17.07±3.71a | 8.05±0.69d |
| Dex | 9 | 1.33±0.12b | 11.36±1.94c | 6.50±0.92c |
| HEL-L | 9 | 1.21±0.20c | 12.290±1.81b | 7.20±0.68 |
| HEL-M | 9 | 1.24±0.14b | 11.24±2.38c | 6.05±1.23e |
| HEL-H | 9 | 1.21±0.24c | 11.66±3.78b | 6.30±1.55c |
Table 2 Changes in lung coefficient, wet/dry weight ratio of lung tissue and histopathological score in animals 24 h after induction of the injury ($\bar{x}±s$)
| Group | n | Lung coefficient (%) | Wet/dry weight ratio (%) | Histopathological score |
|---|---|---|---|---|
| Control | 9 | 1.13±0.18 | 11.14±2.58 | 1.00±0.50 |
| LPS | 9 | 1.36±0.18a | 17.07±3.71a | 8.05±0.69d |
| Dex | 9 | 1.33±0.12b | 11.36±1.94c | 6.50±0.92c |
| HEL-L | 9 | 1.21±0.20c | 12.290±1.81b | 7.20±0.68 |
| HEL-M | 9 | 1.24±0.14b | 11.24±2.38c | 6.05±1.23e |
| HEL-H | 9 | 1.21±0.24c | 11.66±3.78b | 6.30±1.55c |
| Group | n | Leukocyte (%) | Neutrophils (%) | Lymphocytes (%) | MPO activity (U/mg tissue) |
|---|---|---|---|---|---|
| Control | 6 | 4.77±0.91 | 2.00±0.51 | 2.64±0.92 | 1.06±0.51 |
| LPS | 6 | 8.50±2.09a | 3.11±1.55 | 5.41±1.32e | 3.59±2.33a |
| Dex | 6 | 4.14±2.15b | 1.94±0.79 | 0.81±0.55f | 1.46±0.24c |
| HEL-L | 6 | 5.80±1.36c | 1.30±0.35d | 3.67±0.81c | 1.92±0.30 |
| HEL-M | 6 | 5.03±2.35d | 1.28±0.59d | 3.65±1.85c | 1.23±0.69d |
| HEL-H | 6 | 4.90±1.06d | 2.02±1.01 | 2.28±0.84f | 1.70±0.16c |
Table 3 Changes in leukocytes, neutrophils, lymphocytes count in plasma and MPO activity in lung tissue ($\bar{x}±s$)
| Group | n | Leukocyte (%) | Neutrophils (%) | Lymphocytes (%) | MPO activity (U/mg tissue) |
|---|---|---|---|---|---|
| Control | 6 | 4.77±0.91 | 2.00±0.51 | 2.64±0.92 | 1.06±0.51 |
| LPS | 6 | 8.50±2.09a | 3.11±1.55 | 5.41±1.32e | 3.59±2.33a |
| Dex | 6 | 4.14±2.15b | 1.94±0.79 | 0.81±0.55f | 1.46±0.24c |
| HEL-L | 6 | 5.80±1.36c | 1.30±0.35d | 3.67±0.81c | 1.92±0.30 |
| HEL-M | 6 | 5.03±2.35d | 1.28±0.59d | 3.65±1.85c | 1.23±0.69d |
| HEL-H | 6 | 4.90±1.06d | 2.02±1.01 | 2.28±0.84f | 1.70±0.16c |
| Group | n | TNF-α | IL-1β | IL-6 |
|---|---|---|---|---|
| Control | 6 | 129.8±6.4 | 14.0±1.0 | 58.1±2.5 |
| LPS | 6 | 185.9±14.4a | 17.4±1.2c | 77.5±4.1a |
| Dex | 6 | 137.1±15.1b | 13.4±1.2b | 61.7±4.5d |
| HEL-L | 6 | 138.5±13.2b | 14.2±1.0d | 65.2±4.9e |
| HEL-M | 6 | 130.7±12.4b | 14.2±1.1d | 53.1±7.6b |
| HEL-H | 6 | 130.3±20.6b | 14.3±1.2d | 66.3±8.1f |
Table 4 Changes in TNF-α, IL-1β and IL-6 in BALF (ng/L, $\bar{x}±s$)
| Group | n | TNF-α | IL-1β | IL-6 |
|---|---|---|---|---|
| Control | 6 | 129.8±6.4 | 14.0±1.0 | 58.1±2.5 |
| LPS | 6 | 185.9±14.4a | 17.4±1.2c | 77.5±4.1a |
| Dex | 6 | 137.1±15.1b | 13.4±1.2b | 61.7±4.5d |
| HEL-L | 6 | 138.5±13.2b | 14.2±1.0d | 65.2±4.9e |
| HEL-M | 6 | 130.7±12.4b | 14.2±1.1d | 53.1±7.6b |
| HEL-H | 6 | 130.3±20.6b | 14.3±1.2d | 66.3±8.1f |
| Group | n | TNF-α | IL-1β | IL-6 |
|---|---|---|---|---|
| Control | 6 | 223.9±13.6 | 13.6±1.4 | 58.5±6.6 |
| LPS | 6 | 262.5±14.3a | 20.1±3.2d | 98.5±14.4d |
| Dex | 6 | 235.1±11.3b | 14.4±1.5e | 67.6±6.4c |
| HEL-L | 6 | 204.6±8.4c | 16.1±1.5b | 83.6±5.8b |
| HEL-M | 6 | 214.0±23.6c | 12.3±1.8c | 73.9±9.1e |
| HEL-H | 6 | 196.9±11.9c | 10.9±1.9c | 60.9±7.2c |
Table 5 Changes in TNF-α, IL-1β and IL-6 in serum (ng/L, $\bar{x}±s$)
| Group | n | TNF-α | IL-1β | IL-6 |
|---|---|---|---|---|
| Control | 6 | 223.9±13.6 | 13.6±1.4 | 58.5±6.6 |
| LPS | 6 | 262.5±14.3a | 20.1±3.2d | 98.5±14.4d |
| Dex | 6 | 235.1±11.3b | 14.4±1.5e | 67.6±6.4c |
| HEL-L | 6 | 204.6±8.4c | 16.1±1.5b | 83.6±5.8b |
| HEL-M | 6 | 214.0±23.6c | 12.3±1.8c | 73.9±9.1e |
| HEL-H | 6 | 196.9±11.9c | 10.9±1.9c | 60.9±7.2c |
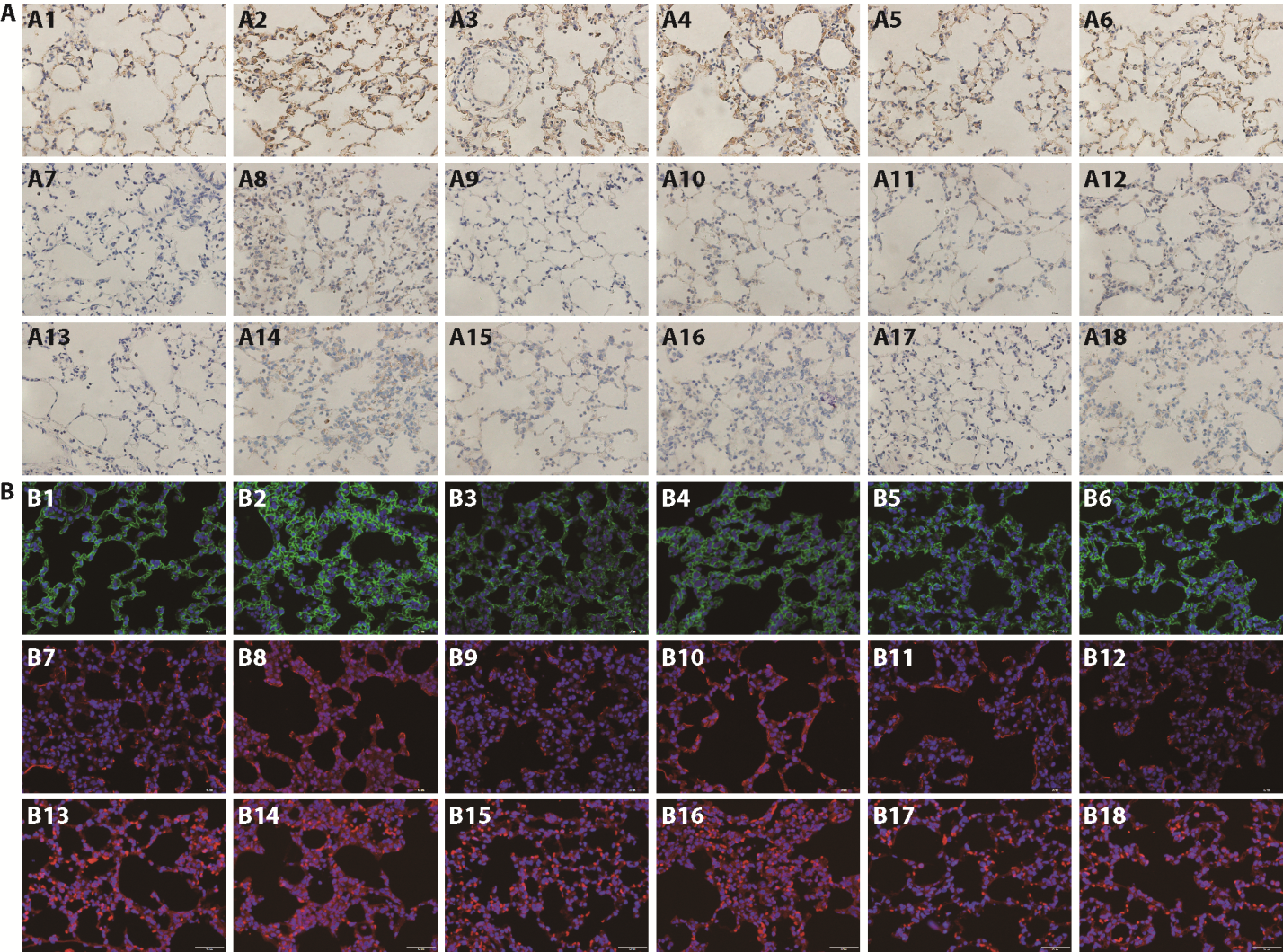
Figure 2 Immunohistochemical and immunofluorescent results visualizing TLR4, NF-κBp65, NLRP3 in lung tissue A: A1-A6: microscopic images of TLR4 for Control, LPS, Dex, HEL-L, HEL-M and HEL-H groups as determined by immunohistochemical assays. A7-A12: microscopic images of NF-κBp65 for Control, LPS, Dex, HEL-L, HEL-M and HEL-H groups as determined by immunohistochemical assays. A13-A18: microscopic images of NLRP3 for Control, LPS, Dex, HEL-L, HEL-M and HEL-H groups as determined by immunohistochemical assays. Scale bar: 50 μm. B: B1-B6: confocal merge images of TLR4 for Control, LPS, Dex, HEL-L, HEL-M and HEL-H groups as determined by immunofluorescence assays. B7-B12: confocal merge images of NF-κBp65 for Control, LPS, Dex, HEL-L, HEL-M and HEL-H groups as determined by immunofluorescence assays. B13-B18: confocal merge images of NLRP3 for Control, LPS, Dex, HEL-L, HEL-M and HEL-H groups as determined by immunofluorescence assays. Scale bar: 50 μm. Control: control group (distilled water by gavage for a week); LPS: lipopolysaccharide group (distilled water by gavage for a week); Dex: dexamethasone group (5 mg/kg by gavage for a week); HEL-L: Yemazhui (Herba Eupatorii Lindleyani) low-dose group (6 g/kg by gavage for a week); HEL-M: Yemazhui (Herba Eupatorii Lindleyani) medium-dose group rats (18 g/kg by gavage for a week); HEL-H: Yemazhui (Herba Eupatorii Lindleyani) high-dose group rats (54 g/kg by gavage for a week). HEL: Yemazhui (Herba Eupatorii Lindleyani); TLR4: toll-like receptor 4; NFκBp65: nuclear factor kappa-B p65; NLRP3: nod-like receptor family pyrin domain-containing 3 protein; LPS: lipopolysaccharide.
| Group | n | TLR4 | NF-κBp65 | NLRP3 |
|---|---|---|---|---|
| Control | 6 | 5.1±0.8 | 1.4±0.2 | 1.9±0.3 |
| LPS | 6 | 9.4±0.8a | 4.7±0.3a | 4.6±0.4a |
| Dex | 6 | 6.0±0.5b | 1.9±0.2b | 3.1±0.6c |
| HEL-L | 6 | 7.1±0.5b | 3.2±0.2b | 3.7±0.2d |
| HEL-M | 6 | 5.9±0.2b | 2.2±0.2b | 2.9±0.4b |
| HEL-H | 6 | 6.4±0.5b | 2.4±0.4b | 3.4±0.4e |
Table 6 Average integrated optical density of TLR4, NF-κBp65 and NLRP3 in lung tissue (Area%, $\bar{x}±s$)
| Group | n | TLR4 | NF-κBp65 | NLRP3 |
|---|---|---|---|---|
| Control | 6 | 5.1±0.8 | 1.4±0.2 | 1.9±0.3 |
| LPS | 6 | 9.4±0.8a | 4.7±0.3a | 4.6±0.4a |
| Dex | 6 | 6.0±0.5b | 1.9±0.2b | 3.1±0.6c |
| HEL-L | 6 | 7.1±0.5b | 3.2±0.2b | 3.7±0.2d |
| HEL-M | 6 | 5.9±0.2b | 2.2±0.2b | 2.9±0.4b |
| HEL-H | 6 | 6.4±0.5b | 2.4±0.4b | 3.4±0.4e |
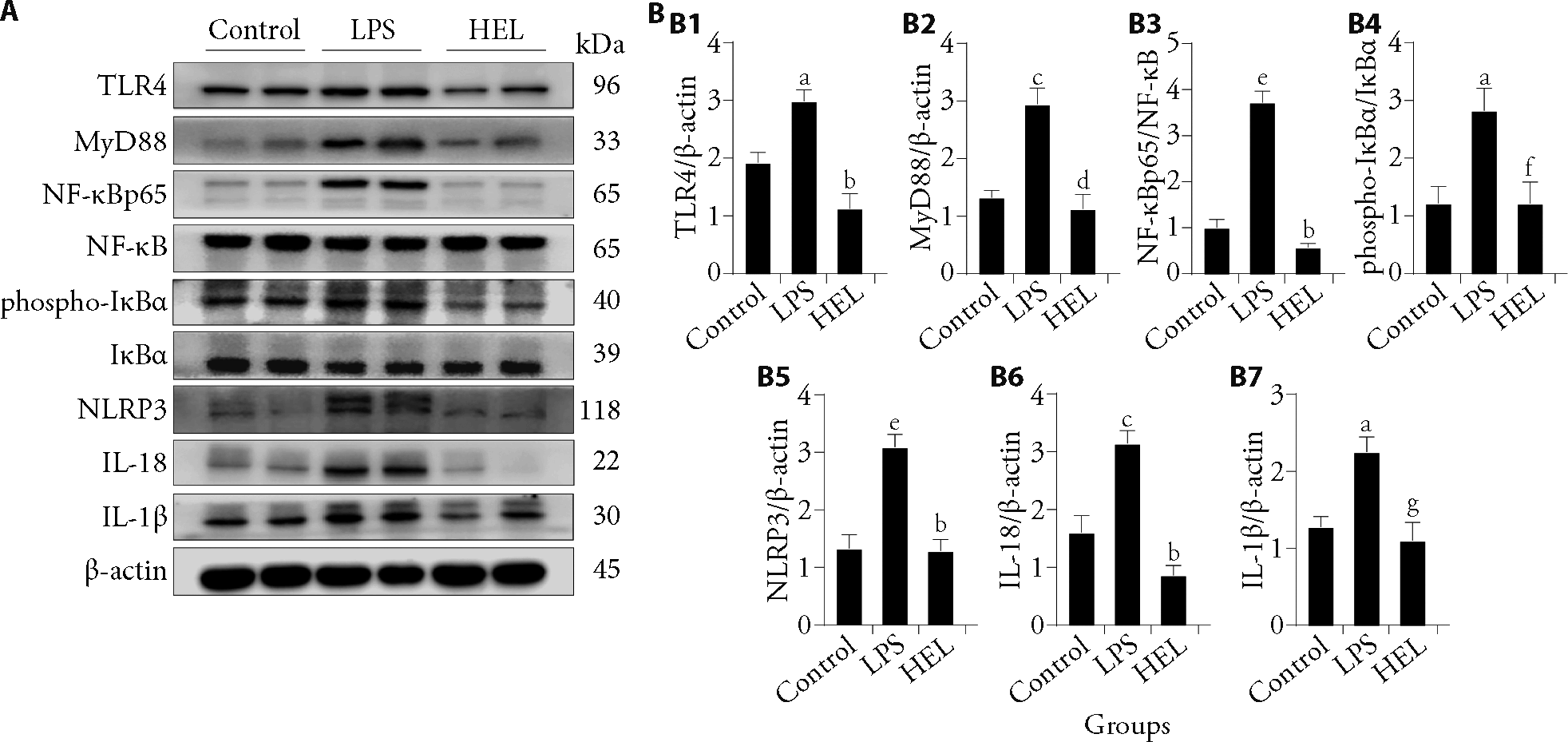
Figure 3 HEL inhibited the TLR4/NF-κB/NLRP3 signaling pathway in vivo A: Western blotting representative images of TLR4, MyD88, NF-κBp65, NF-κB, phospho-IκBα, IκBα, NLRP3, IL-18 and IL-1β proteins. B: B1: Protein expression levels of TLR4; B2: protein expression levels of MyD88; B3: protein expression levels of NF-κBp65; B4: protein expression levels of phospho-IκBα; B5: protein expression levels of NLRP3; B6: protein expression levels of IL-18; B7: protein expression levels of IL-1β. The data were expressed as the mean ± standard deviation (n = 6). Control: control group (distilled water by gavage for a week); LPS: lipopolysaccharide group (distilled water by gavage for a week for a week); HEL: Yemazhui (Herba Eupatorii Lindleyani) medium-dose group rats (18 g/kg by gavage for a week). HEL: Yemazhui (Herba Eupatorii Lindleyani); TLR4: toll-like receptor 4; MyD88: myeloid differentiation primary response protein 88; NF-κB: nuclear factor kappa-B; NF-κBp65: nuclear factor kappa-B p65; IκBα: inhibitor kappa B alpha; phospho-IκBα phosphorylated inhibitor kappa B alpha; NLRP3: nod-like receptor family pyrin domain-containing 3 protein; IL-18: interleukin 18; IL-1β: interleukin-1beta. Compared with the control group, aP < 0.01, cP < 0.001, eP < 0.0001; compared with the LPS group, bP < 0.0001, dP < 0.001, fP < 0.05, gP < 0.01.
| Group | n | TNF-α | IL-1β | IL-6 |
|---|---|---|---|---|
| Control | 6 | 1.19±0.25 | 1.14±0.38 | 0.53±0.19 |
| LPS | 6 | 5.89±2.37a | 2.12±0.91c | 2.19±1.25a |
| HEL | 6 | 1.89±1.11b | 1.20±0.17b | 0.89±0.30b |
Table 7 Levels of TNF-α, IL-1β, and IL-6 mRNA in lung tissue ($\bar{x}±s$)
| Group | n | TNF-α | IL-1β | IL-6 |
|---|---|---|---|---|
| Control | 6 | 1.19±0.25 | 1.14±0.38 | 0.53±0.19 |
| LPS | 6 | 5.89±2.37a | 2.12±0.91c | 2.19±1.25a |
| HEL | 6 | 1.89±1.11b | 1.20±0.17b | 0.89±0.30b |
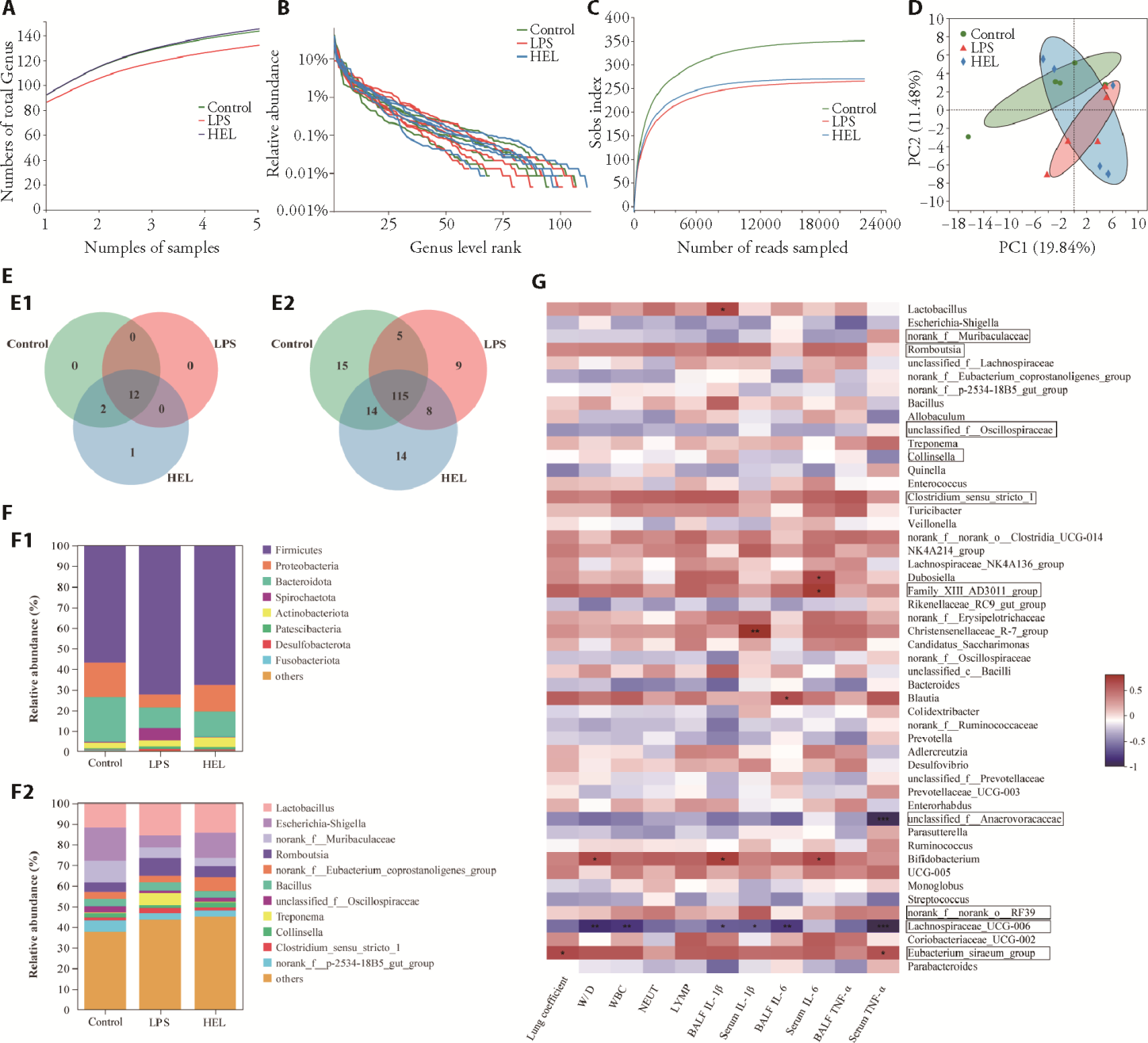
Figure 4 Intestinal flora analysis of HEL on the cecal contents in vivo A: pan chart by ASV analysis. B: rank abundance chart. C: sobs index represents α diversity. D: PCA analysis of gut bacterial community based on the genus level. E1: venn chart based on OTU at the phylum levels; E2: venn chart based on OTU at the genus levels. F1: stacking diagram of community composition structure at the phylum levels; F2: stacking diagram of community composition structure at the genus levels. G: correlation between intestinal flora and lung injury index or Inflammatory cytokines. The above data were analyzed in control, LPS and HEL groups (n = 6). Control: control group (distilled water by gavage for a week for a week); LPS: lipopolysaccharide group (distilled water by gavage for a week); HEL: Yemazhui (Herba Eupatorii Lindleyani) medium-dose group rats (18 g/kg by gavage for a week). HEL: Yemazhui (Herba Eupatorii Lindleyani), ASV: amplicon sequence variants, PCA: principal component analysis, OTU: operational taxonomic units.
| 1. |
Confalonieri M, Salton F, Fabiano F. Acute respiratory distress syndrome. Eur Respir Rev 2017; 26: 160116.
DOI URL |
| 2. |
Thompson BT, Chambers RC, Liu KD. Acute Respiratory Distress Syndrome. N Engl J Med 2017; 377: 562-72.
DOI URL |
| 3. |
Zhang H, Wang H, Lu D, et al. Berberine inhibits cytosolic phospholipase a2 and protects against LPS-induced lung injury and lethality independent of the α2-adrenergic receptor in mice. Shock 2008; 29: 617-22.
DOI URL |
| 4. |
Mokra D, Kosutova P. Biomarkers in acute lung injury. Resp Physiol Neurobi 2015; 209: 52-8.
DOI URL |
| 5. |
Zheng J, Lu C, Yang M, et al. Lung-targeted delivery of cepharanthine by an erythrocyte-anchoring strategy for the treatment of acute lung injury. Pharmaceutics 2022; 14: 1820.
DOI URL |
| 6. | Mokra D. Acute lung injury-from pathophysiology to treatment. Physiol Res 2020; 69: S353-66. |
| 7. |
Mokra D, Mikolka P, Kosutova P, Mokry J. Corticosteroids in acute lung injury: the dilemma continues. Int J Mol Sci 2019; 20: 4765.
DOI URL |
| 8. |
Chen H, Bai C, Wang X. The value of the lipopolysaccharide-induced acute lung injury model in respiratory medicine. Expert Rev Resp Med 2010; 4: 773-83.
DOI PMID |
| 9. | Han X, Wu YC, Meng M, Sun QS, Gao SM, Sun H. Linarin prevents LPS‑induced acute lung injury by suppressing oxidative stress and inflammation via inhibition of TXNIP/NLRP3 and NF‑κB pathways. Int J Mol Med 2018; 42: 1460-72. |
| 10. |
Yang X, An X, Wang C, et al. Protective effect of oxytocin on ventilator-induced lung injury through NLRP3-mediated pathways. Front Pharmacol 2021; 12: 722907.
DOI URL |
| 11. |
Kelley N, Jeltema D, Duan Y, He Y. The NLRP3 inflammasome: an overview of mechanisms of activation and regulation. Int J Mol Sci 2019; 20: 3328.
DOI URL |
| 12. |
Wu Y, Wang Y, Gong S, et al. Ruscogenin alleviates LPS-induced pulmonary endothelial cell apoptosis by suppressing TLR4 signaling. Biomed Pharmacother 2020; 125: 109868.
DOI PMID |
| 13. | Yin L, Zhang G, Yan X, Wang Y, Xu Y, Yang Y. An in vivo and in vitro evaluation of the mutual interactions between the lung and the large intestine. Evid Based Complement Alternat Med 2013; 2013: 1-9. |
| 14. |
Dickson RP, Singer BH, Newstead MW, et al. Enrichment of the lung microbiome with gut bacteria in sepsis and the acute respiratory distress syndrome. Nat Microbiol 2016; 1: 16113.
DOI PMID |
| 15. |
Zhang D, Li S, Wang N, Tan H, Zhang Z, Feng Y. The cross-talk between gut microbiota and lungs in common lung diseases. Front Microbiol 2020; 11: 301.
DOI PMID |
| 16. |
Wang X, Ma S, Lai F, Wang Y, Lou C. Traditional applications, phytochemistry, and pharmacological activities of Eupatorium lindleyanum DC.: a comprehensive review. Front Pharmacol 2020; 8: 577124.
DOI URL |
| 17. |
Ding Z, Zhong R, Xia T, et al. Advances in research into the mechanisms of Chinese Materia Medica against acute lung injury. Biomed Pharmacother 2020; 122: 109706.
DOI PMID |
| 18. |
Wang X, Xu J, Tao Y, Fan X, Shen X, Tian S. Eupatorium lindleyanum DC. sesquiterpene fraction F1012-2 regulates p53/NF-κB signaling pathways in human breast cancer. Arch Biol Sci 2022; 74: 291-9.
DOI URL |
| 19. |
Li C, Chen S, Sha J, et al. Extraction and purification of total flavonoids from Eupatorium lindleyanum DC. and evaluation of their antioxidant and enzyme inhibitory activities. Food Sci Nutr 2021; 9: 2349-63.
DOI URL |
| 20. |
Wang F, Zhong H, Fang S, et al. Potential anti-inflammatory sesquiterpene lactones from Eupatorium lindleyanum. Planta Med 2018; 84: 123-8.
DOI PMID |
| 21. | Shen X, Hu H, Xu J, Tian S. Prediction targets of a novel sesquiterpene lactone from Eupatorium lindleyanum DC. against viral pneumonia using computational pharmacology. Nat Prod Commun 2022; 17: 1934578X-2211314X. |
| 22. |
Liu J, Huang X, Hu S, He H, Meng Z. Dexmedetomidine attenuates lipopolysaccharide induced acute lung injury in rats by inhibition of caveolin-1 downstream signaling. Biomed Pharmacother 2019; 118: 109314.
DOI URL |
| 23. |
Chimenti L, Camprubí-Rimblas M, Guillamat-Prats R, et al. Nebulized Heparin attenuates pulmonary coagulopathy and inflammation through alveolar macrophages in a rat model of acute lung injury. Thromb Haemostasis 2017; 117: 2125-34.
DOI PMID |
| 24. | Chinese Pharmacopoeia Commission. Pharmacopoeia of the People's Republic of China. Beijing: China Medical Science Press, 2020: 326. |
| 25. |
Li Z, Liu J, Zhang D, et al. Nuciferine and paeoniflorin can be quality markers of Tangzhiqing tablet, a Chinese traditional patent medicine, based on the qualitative, quantitative and dose-exposure-response analysis. Phytomedicine 2018; 44: 155-63.
DOI PMID |
| 26. |
Ludden TM. Nonlinear pharmacokinetics: clinical implications. Clin Pharmacokinet 1991; 6: 429-46.
DOI URL |
| 27. |
Matute-Bello G, Frevert CW, Martin TR. Animal models of acute lung injury. Am J Physiol Lung Cell Mol Physiol 2008; 295: L379-99.
DOI URL |
| 28. |
Lei J, Wei Y, Song P, et al. Cordycepin inhibits LPS-induced acute lung injury by inhibiting inflammation and oxidative stress. Eur J Pharmacol 2018; 818: 110-4.
DOI PMID |
| 29. |
Chu C, Ren H, Xu N, Xia L, Chen D, Zhang J. Eupatorium lindleyanum DC. sesquiterpenes fraction attenuates lipopolysaccharide-induced acute lung injury in mice. J Ethnopharmacol 2016; 185: 263-71.
DOI PMID |
| 30. |
Chu C, Yao S, Chen J, et al. Eupatorium lindleyanum DC. flavonoids fraction attenuates lipopolysaccharide-induced acute lung injury in mice. Int Immunopharmacol 2016; 39: 23-33.
DOI PMID |
| 31. | Orecchioni M, Ghosheh Y, Pramod AB, Ley K. Macrophage polarization: different gene signatures in M1 (LPS+) vs classically and M2 (LPS-) vs alternatively activated macrophages. Front Immunol 2019; 101084. |
| 32. |
Zhao K, Li X, Yang J, et al. Protective effect of Amomum Roxb. essential oils in lipopolysaccharide-induced acute lung injury mice and its metabolomics. J Ethnopharmacol 2022; 290: 115119.
DOI URL |
| 33. |
Wu J, Yan Z, Schwartz DE, Yu J, Malik AB, Hu G. Activation of NLRP3 inflammasome in alveolar macrophages contributes to mechanical stretch-induced lung inflammation and injury. J Immunol 2013; 190: 3590-9.
DOI PMID |
| 34. |
Sharif H, Wang L, Wang WL, et al. Structural mechanism for NEK7-licensed activation of NLRP3 inflammasome. Nature 2019; 570: 338-43.
DOI |
| 35. |
Patel MN, Carroll RG, Galván-Peña S, et al. Inflammasome priming in sterile inflammatory disease. Trends Mol Med 2017; 23: 165-80.
DOI PMID |
| 36. | Liu P, Wang P, Tian D, Liu J, Chen G, Liu S. Study on Traditional Chinese Medicine theory of lung being con- nected with large intestine. J Tradit Chin Med 2012; 1: 1-2. |
| 37. |
Wedgwood S, Gerard K, Halloran K, et al. Intestinal dysbiosis and the developing lung: the role of toll-like receptor 4 in the gut-lung axis. Front Immunol 2020; 11: 357.
DOI PMID |
| 38. |
Tang J, Xu L, Zeng Y, Gong F. Effect of gut microbiota on LPS-induced acute lung injury by regulating the TLR4/NF-kB signaling pathway. Int Immunopharmacol 2021; 91: 107272.
DOI URL |
| 39. |
Zhou B, Yuan Y, Zhang S, et al. Intestinal flora and disease mutually shape the regional immune system in the intestinal tract. Front Immunol 2020; 11: 575.
DOI PMID |
| 40. |
Yildirim E, Ilina L, Laptev G, et al. The structure and functional profile of ruminal microbiota in young and adult reindeers (Rangifer tarandus) consuming natural winter-spring and summer-autumn seasonal diets. Peer J 2021; 9: e12389.
DOI URL |
| 41. |
Guo Y, Huang S, Zhao L, Zhang J, Ji C, Ma Q. Pine (Pinus massoniana Lamb.) needle extract supplementation improves performance, egg quality, serum parameters, and the gut microbiome in laying hens. Front Nutr 2022; 9: 810462.
DOI URL |
| 42. |
Zhao Z, Fei K, Bai H, Wang Z, Duan J, Wang J. Metagenome association study of the gut microbiome revealed biomarkers linked to chemotherapy outcomes in locally advanced and advanced lung cancer. Thorac Cancer 2021; 12: 66-78.
DOI PMID |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

