Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (2): 324-333.DOI: 10.19852/j.cnki.jtcm.20240203.001
Previous Articles Next Articles
Protective effect of modified Huangqi Chifeng decoction (加味黄芪赤风汤) on immunoglobulin A nephropathy through toll-like receptor 4/myeloid differentiation factor 88/nuclear factor-kappa B signaling pathway
LI Liusheng1,2, ZHAO Mingming3,4, CHANG Meiying3,4, SI Yuan3,4, ZHAO Jinning6, YANG Bin5( ), ZHANG Yu3,4(
), ZHANG Yu3,4( )
)
- 1 Department of Oncology, Beijing Hospital of Integrated Traditional Chinese and Western Medicine, Beijing 100039, China
2 Department of Nephropathy, Xiyuan Hospital, China Academy of Chinese Medical Sciences, Beijing 100091, China
3 Department of Nephropathy, Xiyuan Hospital, China Academy of Chinese Medical Sciences, Beijing 100091, China
4 Xin-Huangpu Joint Innovation Institute of Chinese Medicine, Guangzhou 510070, China
5 Department of Pathology Xiyuan Hospital, China Academy of Chinese Medical Sciences, Beijing 100091, China
6 Department of Experimental Research Center, Xiyuan Hospital, China Academy of Chinese Medical Sciences, Beijing 100091, China
-
Received:2022-12-09Accepted:2023-05-04Online:2024-04-15Published:2024-02-03 -
Contact:Prof. ZHANG Yu, Department of Nephropathy, Xiyuan Hospital, China Academy of Chinese Medical Sciences, Beijing 100091, China.zhangyu8225@126.com ; YANG Bin, Department of Pathology, Xiyuan Hospital, China Academy of Chinese Medical Sciences, Beijing 100091, China.yangbin555@126.com Telephone: +86-13611079831 -
Supported by:Joint Innovation Fundation of JIICM: Health Management of Chronic Kidney Disease Based on Integrated Traditional Chinese And Western Medicine(2021IR009);Natural Science Foundation-funded Project: the Mechanism of Modified Huangqi Chifeng Decoction Protect Damaged Podocyte via Cross-Talking of PI3K/AKT/mTOR and AMPK/mTOR/ULK1 Signaling Pathway Regulate Autophapy(81873300);The Central Public-interest Scientific Institution Basal Research Fund of the China Academy of Chinese Medical Sciences: Comprehensive Evaluation of Clinical efficacy of Modified Huangqi Chifeng Decoction on IgA Nephropathy(ZZ11-023);The Beijing Municipal of Science and Technology Major Project: Evaluation of Clinical Efficacy of Modified Huangqi Chifeng Decoction in Treating Proteinuria in IgA Nephropathy Based on "Deficiency-Wind-Blood-Stasis-Toxicity" Mechanism in Chinese Medicine(Z191100006619063)
Cite this article
LI Liusheng, ZHAO Mingming, CHANG Meiying, SI Yuan, ZHAO Jinning, YANG Bin, ZHANG Yu. Protective effect of modified Huangqi Chifeng decoction (加味黄芪赤风汤) on immunoglobulin A nephropathy through toll-like receptor 4/myeloid differentiation factor 88/nuclear factor-kappa B signaling pathway[J]. Journal of Traditional Chinese Medicine, 2024, 44(2): 324-333.
share this article
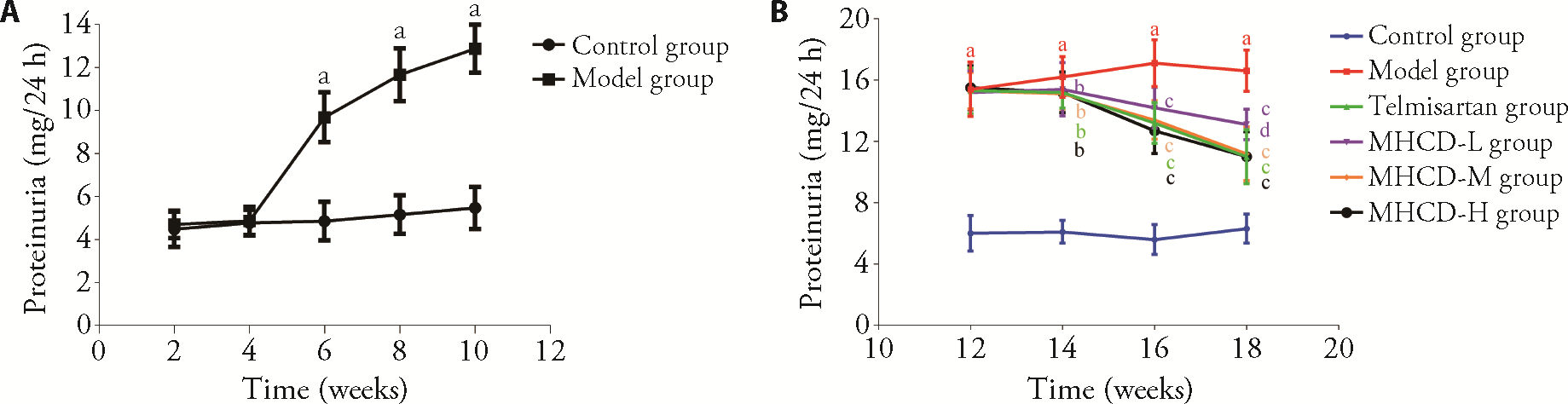
Figure 1 MHCD reduced proteinuria A: proteinuria changes at the stage of model building (n of control group = 13, n of model group = 53). B: proteinuria changes during intervention (n = 10). The telmisartan group were intragastrically administered telmisartan at a dose of 8.33 mg·kg-1·d-1 for 8 weeks; The MHCD-H, MHCD-M, and MHCD-L groups were intragastrically administered MHCD at 25.0, 12.5, and 6.25 g·kg-1·d-1, respectively, for 8 weeks; The control group and model group were given the same volumes of normal saline for 8 weeks. MHCD: modified Huangqi Chifeng decoction; MHCD-L: low-dose modified Huangqi Chifeng decoction; MHCD-M: medium-dose modified Huangqi Chifeng decoction; MHCD-H: high-dose modified Huangqi Chifeng decoction. Data represent the mean ± standard deviation using one-way analysis of variance. aP < 0.01 vs control group; bP < 0.05 and cP < 0.01 vs model group; dP < 0.05 vs MHCD-M group, MHCD-H group and telmisartan group.
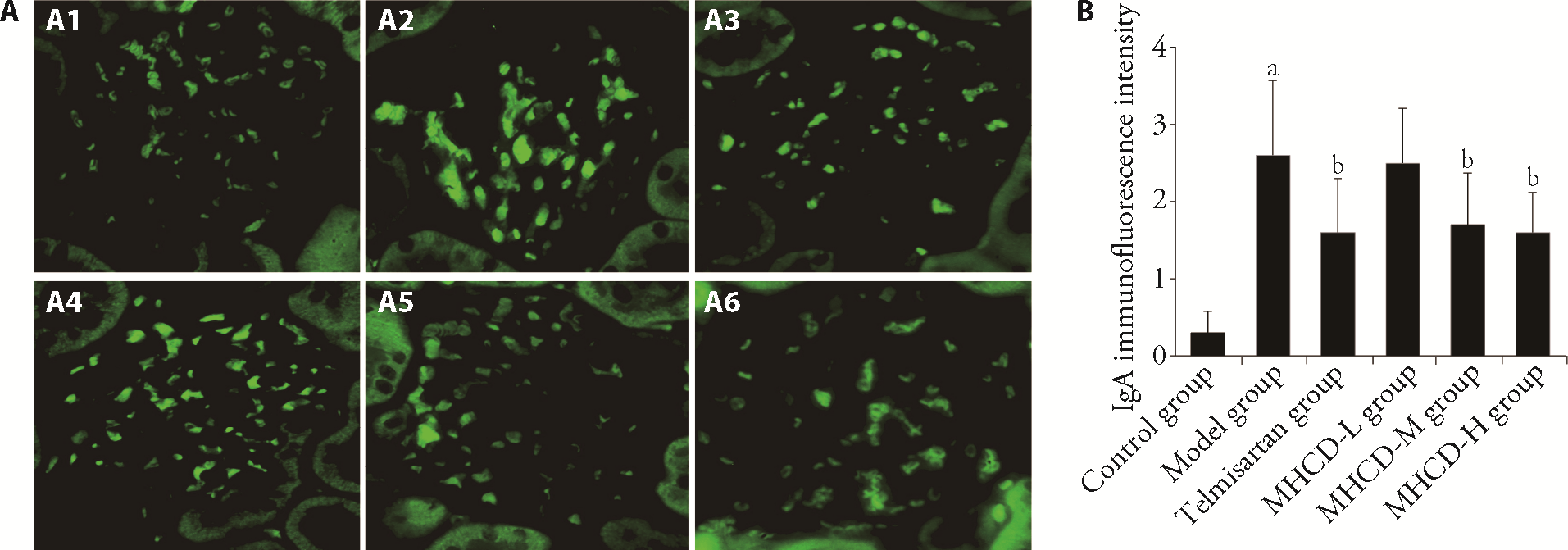
Figure 2 MHCD decreased IgA deposition (n = 10, × 400) A1-A6: images of Immunofluorescence staining. B: quantitative analysis of IgA deposition. A1, A2: selected from the control group (A1) and the model group (A2) were given the same volumes of normal saline for 8 weeks; A3: selected from the telmisartan group were intragastrically administered telmisartan at a dose of 8.33 mg·kg-1·d-1 for 8 weeks; A4: selected from the MHCD-L group were intragastrically administered MHCD at 6.25 g·kg-1·d-1 for 8 weeks; A5: selected from the MHCD-M group were intragastrically administered MHCD at 12.5 g·kg-1·d-1 for 8 weeks; A6: selected from the MHCD-H group were intragastrically administered MHCD at 25.0 g·kg-1·d-1 for 8 weeks. The Data represent the mean ± standard deviation using one-way analysis of variance. aP < 0.01 vs control group; bP < 0.05 vs model group. MHCD: modified Huangqi Chifeng decoction; MHCD-L: low-dose modified Huangqi Chifeng decoction; MHCD-M: medium-dose modified Huangqi Chifeng decoction; MHCD-H: high-dose modified Huangqi Chifeng decoction.

Figure 3 MHCD downregulated the expressions of MCP-1, IL-6, and TGF-β1 A1-A6: images of immunohistochemistry of MCP-1 (× 200); B1-B6: images of immunohistochemistry of IL-6 (× 200); C1-C6: images of immunohistochemistry of TGF-β1 (× 200); D: quantitation of MCP-1 measured by immunohistochemistry; E: quantitation of IL-6 measured by immunohistochemistry; F: quantitation of TGF-β1 measured by immunohistochemistry; G: MCP-1 was measured by qPCR; H: IL-6 was measured by qPCR; I: TGF-β1 was measured by qPCR. A1, A2, B1, B2, C1, C2: selected from the control group (A1, B1, C1) and the model group (A2, B2, C2) were given the same volumes of normal saline for 8 weeks; A3, B3, C3: selected from the telmisartan group were intragastrically administered telmisartan at a dose of 8.33 mg·kg-1·d-1 for 8 weeks; A4, B4, C4: selected from the MHCD-L group were intragastrically administered MHCD at 6.25 g·kg-1·d-1 for 8 weeks; A5, B5, C5: selected from the MHCD-M group were intragastrically administered MHCD at 12.5 g·kg-1·d-1 for 8 weeks; A6, B6, C6: selected from the MHCD-H group were intragastrically administered MHCD at 25.0 g·kg-1·d-1 for 8 weeks. MCP-1: monocyte chemotactic protein 1; IL-6: interleukin 6; TGF-β1: transforming growth factor-β 1; MHCD: modified Huangqi Chifeng decoction; MHCD-L: low-dose modified Huangqi Chifeng decoction; MHCD-M: medium-dose modified Huangqi Chifeng decoction; MHCD-H: high-dose modified Huangqi Chifeng decoction. The Data represent the mean ± standard deviation using one-way analysis of variance (n = 10). aP < 0.01 vs control group; bP < 0.05 and cP < 0.01 vs model group.

Figure 4 MHCD inhibited the TLR4/MyD88/NF-κB signaling pathway A1-A6: images of immunohistochemistry of.TLR4 (× 200); B1-B6: images of immunohistochemistry of MyD88 (× 200); C1-C6: images of immunohistochemistry of NF-κB P65 (× 200); D: quantitation of TLR4 measured by immunohistochemistry; E: quantitation of MyD88 measured by immunohistochemistry; F: quantitation of NF-κB P65 measured by immunohistochemistry; G: images of TLR4, MyD88 and NF-κB P65 analyzed by Western blotting; H: TLR4 was analyzed by Western blotting; I: MyD88 was analyzed by Western blotting; J: NF-κB P65 was analyzed by Western blotting; K: TLR4 was analyzed by qPCR; L: MyD88 was analyzed by qPCR; M: NF-κB was analyzed by qPCR. A1, A2, B1, B2, C1, C2: selected from the control group (A1, B1, C1) and the model group (A2, B2, C2) were given the same volumes of normal saline for 8 weeks; A3, B3, C3: selected from the telmisartan group were intragastrically administered telmisartan at a dose of 8.33 mg·kg-1·d-1 for 8 weeks; A4, B4, C4: selected from the MHCD-L group were intragastrically administered MHCD at 6.25 g·kg-1·d-1 for 8 weeks; A5, B5, C5: selected from the MHCD-M group were intragastrically administered MHCD at 12.5 g·kg-1·d-1 for 8 weeks; A6, B6, C6: selected from the MHCD-H group were intragastrically administered MHCD at 25.0 g·kg-1·d-1 for 8 weeks. TLR4: toll-like receptor 4; MyD88: myeloid differentiation primary response 88; NF-κB: nuclear factor kappa-B; MHCD: modified Huangqi Chifeng decoction; MHCD-L: low-dose modified Huangqi Chifeng decoction; MHCD-M: medium-dose modified Huangqi Chifeng decoction; MHCD-H: high-dose modified Huangqi Chifeng decoction. The Data represent the mean ± standard deviation using one-way analysis of variance (n = 10). aP < 0.01 and dP < 0.05 vs control group; bP < 0.05 and cP < 0.01 vs model group.
| 1. |
Roberts I. Oxford classification of immunoglobulin A nephropathy: an update. Curr Opin Nephrol Hy 2013; 22: 281-6.
DOI PMID |
| 2. |
Schena F, Nistor I. Epidemiology of IgA nephropathy: a global perspective. Semin Nephrol 2018; 38: 435-42.
DOI PMID |
| 3. |
Xu X, Ning Y, Shang WF, et al. Analysis of 4931 renal biopsy data in central China from 1994 to 2014. Ren Fail 2016; 38: 1021-30.
DOI PMID |
| 4. |
Novak J, Renfrow M, Gharavi A, Julian BA. Pathogenesis of immunoglobulin A nephropathy. Curr Opin Nephrol Hy 2013; 22: 287-94.
DOI PMID |
| 5. |
Lim BJ, Lee D, Hong SW, Jeong HJ. Toll-like receptor 4 signaling is involved in IgA-stimulated mesangial cell activation. Yonsei Med J 2011; 52: 610-5.
DOI URL |
| 6. |
Pulskens W, Rampanelli E, Teske G, et al. TLR4 promotes fibrosis but attenuates tubular damage in progressive renal injury. JASN 2010; 21: 1299-308.
DOI URL |
| 7. |
Gluba A, Banach M, Hannam S, Mikhailidis DP, Sakowicz A, Rysz J. The role of Toll-like receptors in renal diseases. Nat Rev Nephrol 2010; 6: 224-35.
DOI PMID |
| 8. | Lepenies J, Eardley KS, Kienitz T, et al. Renal TLR4 mRNA expression correlates with inflammatory marker MCP-1 and profibrotic molecule TGF-beta (1) in patients with chronic kidney disease. Nephron Clin Pract 2011; 119: c97-104. |
| 9. |
Qiang S, Lyu XW, Sun YH, Ye ZL, Kong BH, Qin ZB. Role of TLR4/MyD88/NF-κB signaling pathway in coronary microembolization-induced myocardial injury prevented and treated with nicorandil. Biomed Pharmacother 2018; 106: 776-84.
DOI PMID |
| 10. |
Chen F, Zhu XG, Sun ZQ, Ma YL. Astilbin inhibits high glucose-induced inflammation and extracellular matrix accumulation by suppressing the TLR4/MyD88/NF-κB pathway in rat glomerular mesangial cells. Front Pharmacol 2018; 9: 1187.
DOI PMID |
| 11. |
Ding Y, Yang HL, Xiang W, He XJ, Liao W, Yi ZW. CD200R1 agonist attenuates LPS-induced inflammatory response in human renal proximal tubular epithelial cells by regulating TLR4-MyD88-TAK1-mediated NF-κB and MAPK pathway. Biochem Bioph Res Co 2015; 460: 287-94.
DOI PMID |
| 12. |
Zhang J, Mi Y, Zhou R, et al. The TLR4-MyD88-NF-κB pathway is involved in sIgA-mediated IgA nephropathy. J Nephrol 2020; 33: 1251-61.
DOI |
| 13. | Jiao ZN, Zhao MM, Zhang Y, Li LS, Si Y. Single case randomized controlled research of Modified Huangqi Chifeng decoction in the treatment of proteinuria due to IgA nephropathy. Zhong Guo Yi Yao Dao Bao 2018; 15: 96-8. |
| 14. | Yu ZK, Li LS, Zhang Y, Zhao MM, Si Y, Zhang LM. Efficacy study on modified Huangqi Chifeng decoction in treatment of IgA based on real world study. Shi Jie Zhong Yi Yao 2018; 13: 2819-22. |
| 15. | Yu ZK, Yang B, Zhang Y, Li LS, Zhao JN, Hao W. Modified Huangqi Chifeng decoction inhibits excessive autophagy to protect against doxorubicin-induced nephrotic syndrome in rats via the PI3K/mTOR signaling pathway. Exp Ther Med 2018; 16: 2490-8. |
| 16. | Gao YH, Zhang Y, Li P, Liu HX, Li S, Yu ZK. Anti-renal fibrosis mechanism of modified Huangqi Chifeng decoction based on TGF-β1/Smad signal pathway. Shi Jie Zhong Xi Yi Jie He Za Zhi 2016; 36: 1486-90. |
| 17. |
Chang MY, Yang B, Li LS, et al. Modified Huangqi Chifeng decoction attenuates proteinuria by reducing podocyte injury in a rat model of immunoglobulin a nephropathy. Front Pharmacol 2021; 12: 714584.
DOI URL |
| 18. |
Zhao MM, Yang B, Li LS, et al. Efficacy of modified Huangqi Chifeng decoction in alleviating renal fibrosis in rats with IgA nephropathy by inhibiting the TGF-β1/Smad 3 signaling pathway through exosome regulation. J Ethnopharmacol 2022; 285: 114795.
DOI URL |
| 19. |
Katafuchi R, Kiyoshi Y, Oh Y, et al. Glomerular score as a prognosticator in IgA nephropathy: its usefulness and limitation. Clin Nephrol 1998; 49: 1-8.
PMID |
| 20. |
Le W, Liang S, Hu Y, et al. Long-term renal survival and related risk factors in patients with IgA nephropathy: results from a cohort of 1155 cases in a Chinese adult population. Nephrol Dial Transplant 2012; 27: 1479-85.
DOI URL |
| 21. |
Lyu J, Zhang H, Wong MG, et al. Effect of oral methylprednisolone on clinical outcomes in patients with IgA nephropathy: the TESTING randomized clinical trial. JAMA 2017; 327: 432-42.
DOI URL |
| 22. |
Chan LY, Leung JC, Tsang AW, Tang SC, Lai KN. Activation of tubular epithelial cells by mesangial-derived TNF-alpha: glomerulotubular communication in IgA nephropathy. Kidney Int 2005; 67: 602-12.
DOI PMID |
| 23. |
Masanobu, Miyazaki, Daisuke, et al. Intrarenal synthesis of IL-6 in IgA nephropathy. Nephrology 1997; 3: 421-30.
DOI URL |
| 24. |
Lee YH, Song GG. Urinary MCP-1 as a biomarker for lupus nephritis: a Meta-analysis. Z Rheumatol 2017; 76: 357-63.
DOI PMID |
| 25. |
Zhao SA, Shen HJ, Gu WZ, Liu AM, Mao JH. Evaluation of TGF-beta1 and MCP-1 expression and tubulointerstitial fibrosis in children with Henoch-Schonlein purpura nephritis and IgA nephropathy: a clinical correlation. Clinics 2017; 72: 95-102.
DOI URL |
| 26. |
Prud'homme GJ, Piccirillo CA. The inhibitory effects of transforming growth factor-beta-1 (TGF-beta1) in autoimmune diseases. J Autoimmun 2000; 14: 23-42.
PMID |
| 27. |
Yang W, Wang J, Shi L, et al. Podocyte injury and overexpression of vascular endothelial growth factor and transforming growth factor-beta 1 in adriamycin-induced nephropathy in rats. Cytokine 2012; 59: 370-6.
DOI PMID |
| 28. |
Chihara Y, Ono H, Ishimitsu T, et al. Roles of TGF-beta1 and apoptosis in the progression of glomerulosclerosis in human IgA nephropathy. Clin Nephrol 2006; 65: 385-92.
PMID |
| 29. |
Leung JC, Chan LY, Tsang AW, et al. Anti-macrophage migration inhibitory factor reduces transforming growth factor-beta 1 expression in experimental IgA nephropathy. Nephrol Dial Transplant 2004; 19: 1976-85.
DOI URL |
| 30. | Zou JN, Xiao J, Hu SS, et al. Toll-like receptor 4 signaling pathway in the protective effect of pioglitazone on experimental immunoglobulin A nephropathy. Chin Med J (Engl) 2017; 130: 906-13. |
| 31. |
Lin K, Luo W, Yang N, et al. Inhibition of MyD88 attenuates angiotensin Ⅱ-induced hypertensive kidney disease via regulating renal inflammation. Int Immunopharmacol 2022; 112: 109218.
DOI URL |
| 32. |
Chen JJ, Peng H, Chen CJ, et al. NAG-1/GDF15 inhibits diabetic nephropathy via inhibiting AGE/RAGE-mediated inflammation signaling pathways in C57BL/6 mice and HK-2 cells. Life Sci 2022; 311: 121142.
DOI URL |
| 33. |
Zhang QY, Xu SJ, Qian JC, et al. Pharmacological inhibition of MyD88 suppresses inflammation in tubular epithelial cells and prevents diabetic nephropathy in experimental mice. Acta Pharmacol Sin 2022; 43: 354-66.
DOI |
| 34. | Wang HQ, Wang SS, Chiufai K, Wang Q, Cheng XL. Umbelliferone ameliorates renal function in diabetic nephropathy rats through regulating inflammation and TLR/NF-κB pathway. Chin J Nat Med 2019; 17: 346-54. |
| 35. |
Lu SY, Zhang HL, Wei XJ, et al. 2-dodecyl-6-methoxycyclohexa-2,5-diene-1,4-dione isolated from Averrhoa carambola L. root ameliorates diabetic nephropathy by inhibiting the TLR4/MyD88/ NF-κB pathway. Diabetes Metab Syndr Obes 2019; 12: 1355-63.
DOI URL |
| 36. |
Kaur H, Chien A, Jialal I. Hyperglycemia induces toll like receptor 4 expression and activity in mouse mesangial cells: relevance to diabetic nephropathy. Am J Physiol Renal Physiol 2012; 303: F1145-50.
DOI URL |
| 37. |
Wang MH, Liu XY, Wang ZL, Xu Q. The extract of Polygala fallax Hemsl. slows the progression of diabetic nephropathy by targeting TLR4 anti-inflammation and MMP-2/9-mediated anti-fibrosis in vitro. Phytomedicine 2022; 104: 154251.
DOI URL |
| 38. |
Zhang QY, Xu SJ, Qian JC, et al. Pharmacological inhibition of MyD88 suppresses inflammation in tubular epithelial cells and prevents diabetic nephropathy in experimental mice. Acta Pharmacol Sin 2022; 43: 354-66.
DOI |
| 39. |
Guo SX, Guo LS, Fang Q, et al. Astaxanthin protects against early acute kidney injury in severely burned rats by inactivating the TLR4/MyD88/NF-κB axis and upregulating heme oxygenase-1. Sci Rep 2021; 11: 6679.
DOI PMID |
| 40. |
Zhong Y, Wu S, Yang Y, et al. LIGHT aggravates sepsis-associated acute kidney injury via TLR4-MyD88-NF-κB pathway. J Cell Mol Med 2020; 24: 11936-48.
DOI URL |
| 41. |
Wang Y, Sun C, Li CY, et al. Urinary MCP-1, HMGB1 increased in calcium nephrolithiasis patients and the influence of hypercalciuria on the production of the two cytokines. Urolithiasis 2017; 45: 159-75.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

