Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (1): 34-41.DOI: 10.19852/j.cnki.jtcm.2023.01.005
• Original articles • Previous Articles Next Articles
Efficacy of luteolin on the human gastric cancer cell line MKN45 and underlying mechanism
DING Yajie, LIU Feng, LI Zhaoyan, Xu Yan, CAO Nida, ZHANG Guangao, WANG Rui, ZHAO Aiguang( )
)
- Department of Oncology Medicine, Longhua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai 200032, China
-
Received:2021-06-12Accepted:2021-09-25Online:2023-02-15Published:2023-01-10 -
Contact:ZHAO Aiguang -
About author:Prof. ZHAO Aiguang, Department of Oncology Medicine, Longhua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai 200032, China. aiguang@hotmail.com. Telephone:+86-21-64385700
-
Supported by:The National Key R & D Program(2017YFC1700605);The National Science and Technology Major Special Project(2017ZX09304001);The State Administration of Traditional Chinese Medicine, and the State Clinical Laboratory of Traditional Chinese Medicine(JDZX2015068)
Cite this article
DING Yajie, LIU Feng, LI Zhaoyan, Xu Yan, CAO Nida, ZHANG Guangao, WANG Rui, ZHAO Aiguang. Efficacy of luteolin on the human gastric cancer cell line MKN45 and underlying mechanism[J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 34-41.
share this article
| Group | n | G0/G1 | S | G2/M |
|---|---|---|---|---|
| 0 | 3 | 76.12±1.74 | 7.12±0.51 | 14.12±0.07 |
| 10 μmol /L | 3 | 41.12±0.96 | 20.48±1.58a | 16.09±0.66 |
| 30 μmol/L | 3 | 43.55±2.53 | 44.15±2.00a | 12.30±0.03 |
| 50 μmol/L | 3 | 54.44±0.97 | 55.44±0.97a | 4.29±0.48 |
Table1 Effect of luteolin on cell cycle of gastric cancer MKN45 ($\bar{x}\pm s$)
| Group | n | G0/G1 | S | G2/M |
|---|---|---|---|---|
| 0 | 3 | 76.12±1.74 | 7.12±0.51 | 14.12±0.07 |
| 10 μmol /L | 3 | 41.12±0.96 | 20.48±1.58a | 16.09±0.66 |
| 30 μmol/L | 3 | 43.55±2.53 | 44.15±2.00a | 12.30±0.03 |
| 50 μmol/L | 3 | 54.44±0.97 | 55.44±0.97a | 4.29±0.48 |
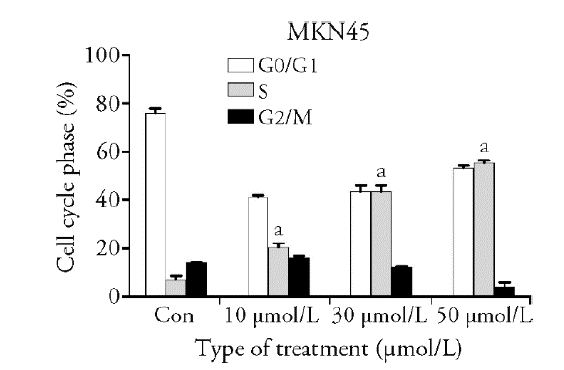
Figure 1 Effect of luteolin on cell cycle of gastric cancer MKN45 A: evaluation of cell cycle, using flow cytometry and PI staining, in MKN45 cells treated with varying concentrations of luteolin (0, 10, 30, and 50 μmol/L) for 48 h. B: the ratio of cells per cell cycle phase of MKN45 cells treated with varying concentrations of luteolin (0, 10, 30, and 50 μmol/L). Compared with the control, aP < 0.001. G: Gap phase. S: Synthesis phase. M: mitotic. Con: control.

Figure 2 Luteolin induces apoptosis of gastric cancer MKN45 cells A: Evaluation of cell apoptosis, using flow cytometry and DAPI staining, in MKN45 cells treated with varying concentrations of luteolin (0, 10, 30, and 50 μmol/L) for 48 h. B: The percentage of total apoptotic MKN45 cells for varying concentrations of luteolin (0, 10, 30, and 50 μmol/L). Compared with the control, aP < 0.001. Con: control.

Figure 3 Effect of luteolin on apoptotic morphology of MKN45 gastric cancer cells MKN45 cells treated with varying concentrations of luteolin (0, 10, 30, and 50 μmol/L) for 48 h and stained by DAPI, and observed the changes of cell morphology under a fluorescence microscope (× 200).

Figure 4 Effect of luteolin on mitochondrial membrane potential of MKN45 gastric cancer A: evaluation of mitochondrial membrane potential, using flow cytometry and JC-l staining, in MKN45 cells treated with varying concentrations of luteolin (0, 10, 30, and 50 μmol/L) for 48 h. B: The percentage of JC-l positive MKN45 cells for varying concentrations of luteolin (0, 10, 30, and 50 μmol/L). Compared with the control, aP < 0.001. Con: control.
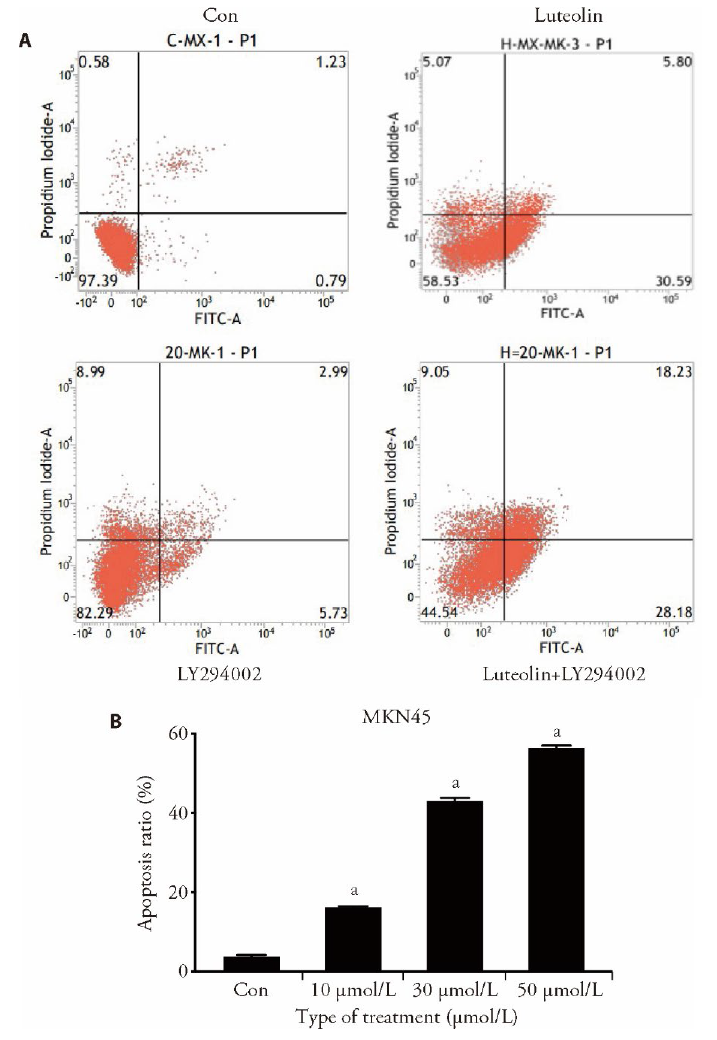
Figure 5 Effect of luteolin on apoptosis of MKN45 cells A: Evaluation of cell apoptosis, using flow cytometry and DAPI staining, in MKN45 cells treated with varying concentrations of Luteolin (20 μmol/L), LY294002 inhibitor (20 μmol/L), and luteolin (20 μmol/L) + LY294002 inhibitor (20 μ mol/L) for 48 h. B: The percentage of total apoptotic MKN45 cells for varying concentrations of Luteolin (20 μmol/L), LY294002 inhibitor (20 μmol/L), and luteolin (20 μmol/L) + LY294002 inhibitor (20 μ mol/L). Compared with the control, aP < 0.001. Con: control.
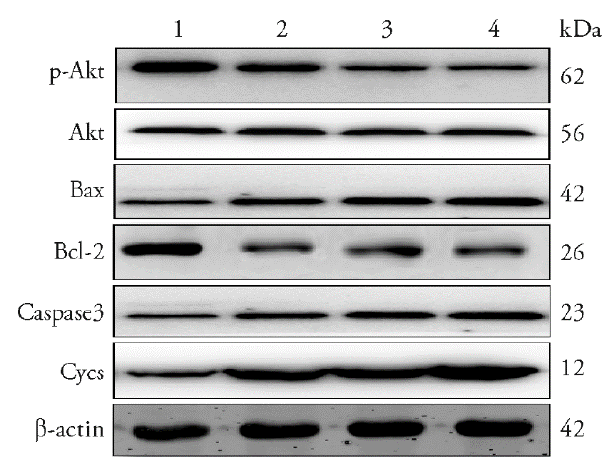
Figure 6 Effect of luteolin on apoptotic protein of MKN45 gastric cancer cells 1: Con; 2: Luteolin; 3: LY294002; 4: LY294002 + Luteolin. Expression of PI3K/Akt signalling pathway-related and apoptosis-related proteins was determined using western blot, in MKN45 cells treated with varying concentrations of Luteolin (20 μmol/L), LY294002 inhibitor (20 μmol/L), and luteolin (20 μmol/L) + LY294002 inhibitor (20 μmol/L) for 48 h. Con: control. Bcl: B-cell lymphoma. Bax: Bcl-2 associated X protein.
| Group | n | Con | Luteolin | LY294002 | LY294002+ Luteolin |
|---|---|---|---|---|---|
| Akt | 3 | 1.023±0.021 | 1.044±0.027 | 1.033±0.012 | 1.013±0.022 |
| p-Akt | 3 | 1.152±0.018 | 1.033±0.031a | 0.932±0.014a | 0.872±0.011ad |
| Bax | 3 | 0.824±0.010 | 1.020±0.013a | 1.031±0.010a | 1.142±0.010ad |
| Bcl-2 | 3 | 1.140±0.009 | 0.980±0.018c | 0.871±0.038c | 0.821±0.027be |
| Caspase3 | 3 | 0.843±0.067 | 1.012±0.08c | 1.060±0.046c | 1.243±0.014be |
| Cycs | 3 | 0.612±0.053 | 1.021±0.002a | 1.043±0.014a | 1.342±0.012ad |
Table 2 Luteolin quantitative analysis of the detection of apoptotic protein of mkn45 gastric cancer cells by Western blotting ($\bar{x}\pm s$)
| Group | n | Con | Luteolin | LY294002 | LY294002+ Luteolin |
|---|---|---|---|---|---|
| Akt | 3 | 1.023±0.021 | 1.044±0.027 | 1.033±0.012 | 1.013±0.022 |
| p-Akt | 3 | 1.152±0.018 | 1.033±0.031a | 0.932±0.014a | 0.872±0.011ad |
| Bax | 3 | 0.824±0.010 | 1.020±0.013a | 1.031±0.010a | 1.142±0.010ad |
| Bcl-2 | 3 | 1.140±0.009 | 0.980±0.018c | 0.871±0.038c | 0.821±0.027be |
| Caspase3 | 3 | 0.843±0.067 | 1.012±0.08c | 1.060±0.046c | 1.243±0.014be |
| Cycs | 3 | 0.612±0.053 | 1.021±0.002a | 1.043±0.014a | 1.342±0.012ad |
| 1 | Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin 2019; 69: 7-34. |
| 2 |
Valastyan S, Weinberg RA. Tumor metastasis: molecular insights and evolving paradigms. Cell 2011; 147: 275-92.
DOI PMID |
| 3 | Allemani C, Weir HK, Carreira H, et al. Global surveillance of cancer survival 1995-2009: analysis of individual data for 25, 676, 887 patients from 279 population-based registries in 67 countries (CONCORD-2). Lancet 2015; 385: 977-1010. |
| 4 | Dai ZJ, Gao J, Ji ZZ, et al. Matrine induces apoptosis in gastric carcinoma cells via alteration of Fas/FasL and activation of caspase-3. J Ethnopharmacol 2009; 123: 91-6. |
| 5 |
Davern M, Lysaght J. Cooperation between chemotherapy and immunotherapy in gastroesophageal cancers-ScienceDirect. Cancer Lett 2020; 495: 89-99.
DOI PMID |
| 6 | Wu B, Zhang Q, Shen W, et al. Anti-proliferative and chemo-sensitizing effects of luteolin on human gastric cancer AGS cell line. Mol Cell Biochem 2008; 313: 125-32. |
| 7 | Wu H, Huang M, Liu Y, et al. Luteolin induces apoptosis by up-regulating mir-34a in human gastric cancer cells. Technol Cancer Res Treat 2015; 14: 747-55. |
| 8 | Tan J, Tian Y, Cai R, et al. Antiproliferative and proapoptotic effects of a protein component purified from Aspongopus chinensis Dallas on cancer cells in vitro and in vivo. Evid Based Complement Alternat Med 2019; 2019: 8934794. |
| 9 | Yao X, Jiang W, Yu D, et al. Luteolin inhibits proliferation and induces apoptosis of human melanoma cells in vivo and in vitro by suppressing MMP-2 and MMP-9 through the PI3K/AKT pathway. Food Funct 2019; 10: 703-12. |
| 10 |
Lim D, Cho H, Kim J, et al. Luteolin decreases IGF-II production and downregulates insulin-like growth factor-I receptor signaling in HT-29 human colon cancer cells. BMC Gastroenterol 2012; 12: 9.
DOI PMID |
| 11 | Chen KC, Hsu WH, Ho JY, et al. Flavonoids luteolin and quercetin inhibit RPS19 and contributes to metastasis of cancer cells through c-Myc reduction. J Food Drug Anal 2018; 26: 1180-91. |
| 12 |
Imran M, Rauf A, Abu-Izneid T, et al. Luteolin, a flavonoid, as an anticancer agent: a review. Biomed Pharmacother 2019; 112: 108612.
DOI |
| 13 | Kou Y, Tong B, Wu W, et al. Berberine improves chemo-sensitivity to cisplatin by enhancing cell apoptosis and repressing PI3K/AKT/mTOR signaling pathway in gastric cancer. Front Pharmacol 2020; 11: 616251. |
| 14 | Hassan M, Watari H, AbuAlmaaty A, et al. Apoptosis and molecular targeting therapy in cancer. Biomed Res Int 2014; 2014: 150845. |
| 15 | Du L, Fei Z, Song S, et al. Antitumor activity of Lobaplatin against esophageal squamous cell carcinoma through caspase-dependent apoptosis and increasing the Bax/Bcl-2 ratio. Biomed Pharma-cother 2017; 95: 447-52. |
| 16 |
Huang L, Jin K, Lan H. Luteolin inhibits cell cycle progression and induces apoptosis of breast cancer cells through downregulation of human telomerase reverse transcriptase. Oncol Lett 2019; 17: 3842-50.
DOI PMID |
| 17 |
Hoxhaj G, Manning BD. The PI3K-AKT network at the interface of oncogenic signalling and cancer metabolism. Nat Rev Cancer 2020; 20: 74-88.
DOI PMID |
| 18 |
Faes S, Dormond O. PI3K and AKT: unfaithful partners in cancer. Int J Mol Sci 2015; 16: 21138-52.
DOI PMID |
| 19 | Liu Q, Dong H, Sun W, et al. Apoptosis initiation of β-ionone in SGC-7901 gastric carcinoma cancer cells via a PI3K-AKT pathway. Arch Toxicol 2013; 87: 481-90. |
| 20 | Yin J, Ji Z, Hong Y, et al. Sh-MARCH8 inhibits tumorigenesis via PI3K pathway in gastric cancer. Cell Physiol Biochem 2018; 49: 306-21. |
| 21 |
Sathe A, Nawroth R. Targeting the PI3K/AKT/mTOR pathway in bladder cancer. Methods Mol Biol 2018; 1655: 335-50.
DOI PMID |
| 22 | Lu X, Li Y, Li X, et al. Luteolin induces apoptosis in vitro through suppressing the MAPK and PI3K signaling pathways in gastric cancer. Oncol Lett 2017; 14: 1993-2000. |
| 23 | Hasibuan PAZ, Harahap U, Sitorus P, et al. The anticancer activities of Vernonia amygdalina Delile. leaves on 4T1 breast cancer cells through phosphoinositide 3-kinase (PI3K) pathway. Heliyon 2020; 6: e04449. |
| 24 | Yan W, Ma X, Zhao X, et al. Baicalein induces apoptosis and autophagy of breast cancer cells via inhibiting PI3K/AKT pathway in vivo and vitro. Drug Des Devel Ther 2018; 12: 3961-72. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

