Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (5): 991-1000.DOI: 10.19852/j.cnki.jtcm.2023.05.005
Previous Articles Next Articles
Effect of Tuina along “bladder meridian” alleviating intervertebral disc degeneration by regulating the transforming growth factor-β1/Smad signaling pathway in a rabbit model
SU Chengguo1, ZHAO Xiaoyan2, YE Jiangnan1, ZHANG Xin1, JIANG Yuqing1, GUO Junjie2, ZHANG Xiyuan2, QI Wenchuan1( ), ZHU Jun1(
), ZHU Jun1( )
)
- 1 Acupuncture and Tuina School, Chengdu University of Traditional Chinese Medicine, Chengdu 610075, China
2 Clinical Medical School, Affiliated Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610075, China
-
Received:2022-03-23Accepted:2022-07-15Online:2023-10-15Published:2023-08-28 -
Contact:QI Wen-chuan, Acupuncture and Tuina School, Chengdu University of Traditional Chinese Medicine, Chengdu 610075, China.qiwenchuan@cdutcm.edu.cn ; ZHU Jun, Acupuncture and Tuina School, Chengdu University of Traditional Chinese Medicine, Chengdu, 610075, China.zhujuntcm@163.com . Telephone: +86-138 8199 6620 -
Supported by:National Natural Science Foundation of China: Based on TGF-β1/Smads Signaling Pathway to Study the Effect Mechanism of Tuina along the Bladder Meridian on Intervertebral Disc Degeneration(82004497);China Postdoctoral Science Foundation: Based on TGF-β1/RhoA/JNK Signaling Pathway to Study the Effect Mechanism of Tuina along the Bladder Meridian on Intervertebral Disc Degeneration(2021M693788)
Cite this article
SU Chengguo, ZHAO Xiaoyan, YE Jiangnan, ZHANG Xin, JIANG Yuqing, GUO Junjie, ZHANG Xiyuan, QI Wenchuan, ZHU Jun. Effect of Tuina along “bladder meridian” alleviating intervertebral disc degeneration by regulating the transforming growth factor-β1/Smad signaling pathway in a rabbit model[J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 991-1000.
share this article

Figure 1 The intervertebral disc tissues with hematoxylin and eosin (HE) staining A1, A2: HE staining results of control group; A3, A4: HE staining results of model group; A5, A6: HE staining results of Tuina group. B: Mean histopathological scores. The red arrow shows a disorganized and ruptured annulus fibrosus; the black arrow indicates greatly reduced nucleus pulposus cells; the yellow arrow shows relatively few wavy fibrocartilage lamellas; and the blue arrow indicates a slight decrease in the number of cells in the nucleus pulposus. Rabbits in the control group underwent sham surgerie. The IVD degeneration model was established in all groups except for the control group. Rabbits in the model group underwent surgeries of posterolateral annulus fibrosus puncturing (AFP). Rabbits in the Tuina group were treated with Tuina along the bladder meridian for 4 weeks. IVD: intervertebral disc. The data are shown as the mean ± standard deviation (n = 6). aP < 0.05 vs the control group; bP < 0.05 vs the model group.
| Items | Degeneration grade |
|---|---|
| I. Anulus fibrosus | 1. Normal pattern of fibrocartilage lamellae |
| 2. Slightly ruptured or irregularly-shaped fibers | |
| 3. Moderately/severely ruptured or irregularly shaped fibers | |
| II. Border between the anulus fibrosus and nucleus pulposus | 1. Normal |
| 2. Slight interruption | |
| 3. Moderate or severe interruption | |
| III. Cells of the nucleus pulposus | 1. Abundant cells |
| 2. Slight decrease in cell count | |
| 3. Moderate or severe decrease in cell count | |
| IV. Matrix of the nucleus pulposus | 1. Normal gelatinous appearance |
| 2. Slight condensation of the matrix | |
| 3. Moderate or severe condensation of the matrix |
Table 1 Histological grading scale
| Items | Degeneration grade |
|---|---|
| I. Anulus fibrosus | 1. Normal pattern of fibrocartilage lamellae |
| 2. Slightly ruptured or irregularly-shaped fibers | |
| 3. Moderately/severely ruptured or irregularly shaped fibers | |
| II. Border between the anulus fibrosus and nucleus pulposus | 1. Normal |
| 2. Slight interruption | |
| 3. Moderate or severe interruption | |
| III. Cells of the nucleus pulposus | 1. Abundant cells |
| 2. Slight decrease in cell count | |
| 3. Moderate or severe decrease in cell count | |
| IV. Matrix of the nucleus pulposus | 1. Normal gelatinous appearance |
| 2. Slight condensation of the matrix | |
| 3. Moderate or severe condensation of the matrix |
| Gene | Forward (5′-3′) | Reverse (5′-3′) |
|---|---|---|
| GAPDH | GTTCCACGGCACGGTCAAGG | CGTACTCGGCACCAGCATCAC |
| ADAMTS5 | GCCAGCATCGAGAACCACATCC | TGCTCACCTCCAGGCTCTTGTC |
| TGF-β1 | GGAACGGGCTCAACATCTACACAG | CCAGGTCCTTGCGGAAGTCAATG |
| Smad2 | ATGTCGTCCATCTTGCCGTTCAC | GCCTTCTCACACCACTTCTCTTCC |
| Smad3 | TGTCGTCCATCCTGCCCTTCAC | ACTTCTCCTCCTGCCCGTTCTG |
| Smad4 | CAGCCAGGACAGCAGCAGAATG | TGGTGGTGAGGCAAATTAGGTGTG |
Table 2 Primers for target genes
| Gene | Forward (5′-3′) | Reverse (5′-3′) |
|---|---|---|
| GAPDH | GTTCCACGGCACGGTCAAGG | CGTACTCGGCACCAGCATCAC |
| ADAMTS5 | GCCAGCATCGAGAACCACATCC | TGCTCACCTCCAGGCTCTTGTC |
| TGF-β1 | GGAACGGGCTCAACATCTACACAG | CCAGGTCCTTGCGGAAGTCAATG |
| Smad2 | ATGTCGTCCATCTTGCCGTTCAC | GCCTTCTCACACCACTTCTCTTCC |
| Smad3 | TGTCGTCCATCCTGCCCTTCAC | ACTTCTCCTCCTGCCCGTTCTG |
| Smad4 | CAGCCAGGACAGCAGCAGAATG | TGGTGGTGAGGCAAATTAGGTGTG |
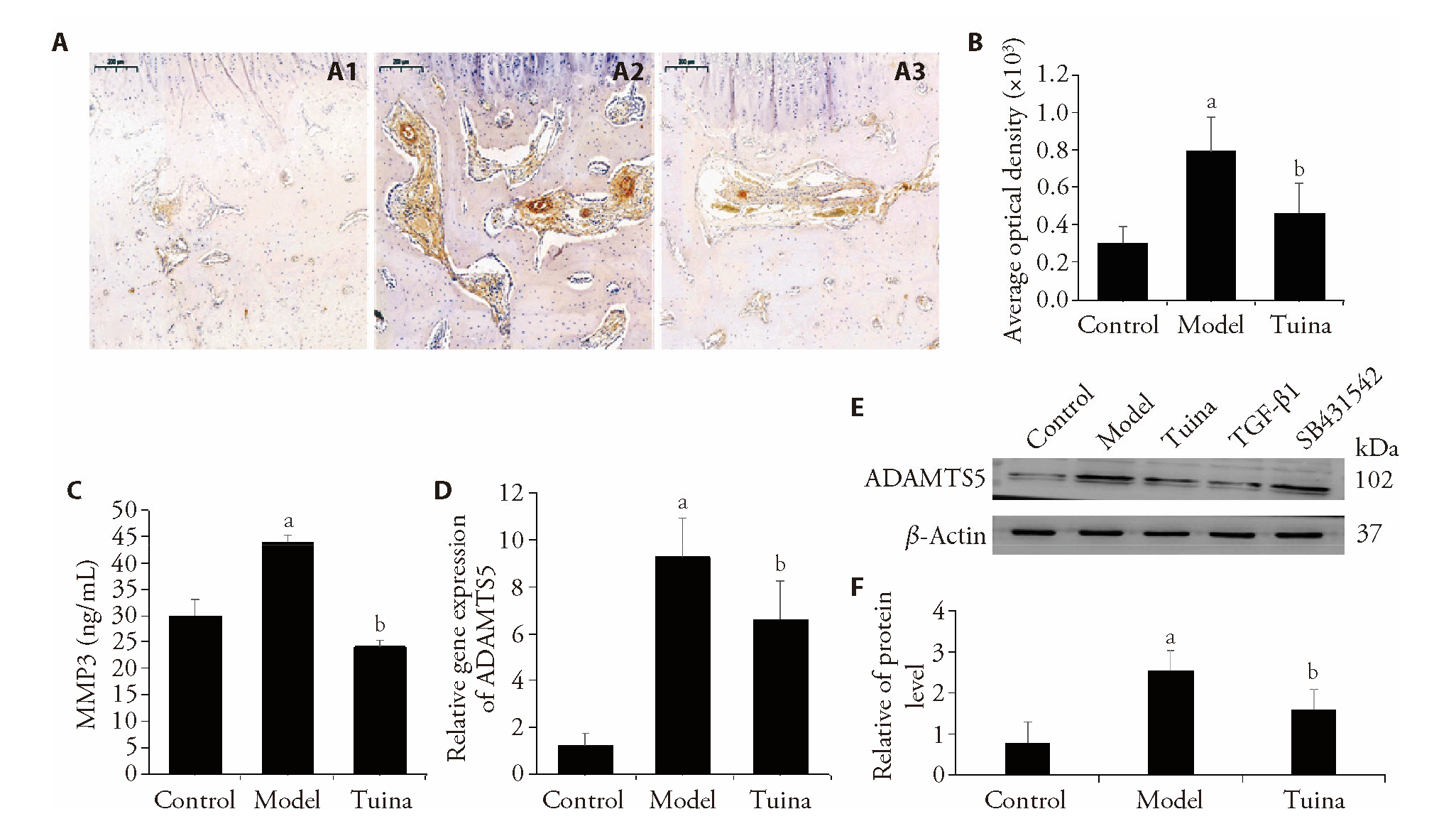
Figure 2 The expression of COL I, MMP3 and ADAMTS5 A1: Immunofluorescence staining for COL I of control group; A2: Immunofluorescence staining for COL I of model group; A3: Immunofluorescence staining for COL I of Tuina group. B: The average optical density of COL I was analyzed by Image-Pro Plus 6.0 software (n = 6). C: The concentration of MMP3 in the intervertebral disc was determined by ELISA (n = 6). D: ADAMTS5 gene expression was analyzed by real-time PCR (n = 6). E, F: The protein expression of ADAMTS5 (n = 6). Rabbits in the control group underwent sham surgeries.The IVD degeneration model was established in all groups except for the control group. Rabbits in the model group underwent surgeries of posterolateral annulus fibrosus puncturing (AFP). Rabbits in the Tuina group were treated with Tuina along the bladder meridian for 4 weeks. aP < 0.05 vs the control group; bP < 0.05 vs the model group.

Figure 3 Cell apoptosis in intervertebral discs A1-A3: terminal deoxynucleotidyl transferase-mediated nick end labeling (TUNEL) staining of control group; A4-A6: TUNEL staining of model group; A7-A9: TUNEL staining of model group. B: The number of TUNEL-positive cells (n = 6); C, D: the protein expression of Bax, caspase-3 and Bcl-2 (n = 6). The red arrow shows TUNEL-positive cells. Rabbits in the control group underwent sham surgeries. The IVD degeneration model was established in all groups except for the control group. Rabbits in the model group underwent surgeries of posterolateral annulus fibrosus puncturing (AFP). Rabbits in the Tuina group were treated with Tuina along the bladder meridian for 4 weeks. IVD: intervertebral disc. aP < 0.05 vs the control group; bP < 0.05 vs the model group.

Figure 4 Expression of the TGF-β1/Smad pathway A: the gene expression of TGF-β1, Smad2, Smad3, and Smad4 analyzed by real-time PCR (n = 6). B, C: the protein expression of TGF-β1, Smad2, Smad3, and Smad4 detected by Western blotting (n = 6). Rabbits in the control group underwent sham surgeries. The IVD degeneration model was established in all groups except for the control group. Rabbits in the model group underwent surgeries of posterolateral annulus fibrosus puncturing (AFP). Rabbits in the Tuina group were treated with Tuina along the bladder meridian for 4 weeks. Rabbits in the TGF-β1 group was injected with Recombinant TGF-β1 into the center of each IVD; while the TGF-β1 inhibitor SB431542 was injected into the SB431542 group. aP < 0.05 vs the control group; bP < 0.05 vs the model group.
| 1. |
Ma K, Chen S, Li Z, et al. Mechanisms of endogenous repair failure during intervertebral disc degeneration. Osteoarthritis Cartilage 2019; 27: 41-8.
DOI URL |
| 2. |
Vo NV, Hartman RA, Yurube T, Jacobs LJ, Sowa GA, Kang JD. Expression and regulation of metalloproteinases and their inhibitors in intervertebral disc aging and degeneration. Spine J 2013; 13: 331-41.
DOI PMID |
| 3. |
Liu H, Kang H, Song C, et al. Urolithin a inhibits the catabolic effect of tnfα on nucleus pulposus cell and alleviates intervertebral disc degeneration in vivo. Front Pharmacol 2018; 9: 1043.
DOI URL |
| 4. |
Molinos M, Almeida CR, Caldeira J, Cunha C, Gonçalves RM, Barbosa MA. Inflammation in intervertebral disc degeneration and regeneration. J R Soc Interface 2015; 12: 20150429.
DOI URL |
| 5. |
Henry N, Clouet J, Le Bideau J, Le Visage C, Guicheux J. Innovative strategies for intervertebral disc regenerative medicine: From cell therapies to multiscale delivery systems. Biotechnol Adv 2018; 36: 281-94.
DOI PMID |
| 6. |
Fontana G, See E, Pandit A. Current trends in biologics delivery to restore intervertebral disc anabolism. Adv Drug Deliv Rev 2015; 84: 146-58.
DOI URL |
| 7. |
Juch J, Maas ET, Ostelo R, et al. Effect of radiofrequency denervation on pain intensity among patients with chronic low back pain: the mint randomized clinical trials. JAMA 2017; 318: 68-81.
DOI PMID |
| 8. | Frapin L, Clouet J, Delplace V, Fusellier M, Guicheux J, Le Visage C. Lessons learned from intervertebral disc pathophysiology to guide rational design of sequential delivery systems for therapeutic biological factors. Adv Drug Deliv Rev 2019; 149-50: 49-71. |
| 9. |
Chen X, Zhu L, Wu G, Liang Z, Yang L, Du Z. A comparison between nucleus pulposus-derived stem cell transplantation and nucleus pulposus cell transplantation for the treatment of intervertebral disc degeneration in a rabbit model. Int J Surg 2016; 28: 77-82.
DOI PMID |
| 10. |
Lund T, Oxland TR. Adjacent level disk disease-is it really a fusion disease? Orthop Clin North Am 2011; 42: 529-41.
DOI URL |
| 11. | Mo Z, Li D, Zhang R, Chang M, Yang B, Tang S. Comparisons of the effectiveness and safety of Tuina, acupuncture, traction, and chinese herbs for lumbar disc herniation: a systematic review and network Meta-analysis. Evid Based Complement Alternat Med 2019; 2019: 6821310. |
| 12. | Furlan AD, Giraldo M, Baskwill A, Irvin E, Imamura M. Massage for low-back pain. Cochrane Database Syst Rev 2015; 2015: D1929. |
| 13. |
Miao Z, Tong Z, Ye J, et al. Tuina for lumbar disc herniation: a protocol for systematic review and Meta-analysis. Medicine (Baltimore) 2021; 100: e24203.
DOI URL |
| 14. | Huang LS, Huang PP, Tong XB, Li ZL, Fang YP, Liao J. Effects of chiropractics on intervertebral disk extracell matrix and metabolic enzymes in rats with cervical spondylosis. Zhen Ci Yan Jiu 2017; 42: 321-6. |
| 15. | Shu J, Li D, Tao W, Chen S. Observation on the curative effect of massage manipulation combined with core strength training in patients with chronic nonspecific low back pain. Evid Based Complement Alternat Med 2021; 2021: 7534577. |
| 16. |
Won ES, Lee H, Kang JH. Effectiveness and safety assessment of orthopedic device (LSM-01) for low back pain: a randomized, single-blinded, sham-controlled, parallel-group, pilot clinical trial. Medicine (Baltimore) 2022; 101: e28527.
DOI URL |
| 17. | Tan H, Tumilty S, Chapple C, Liu L, Othman R, Baxter GD. Acupoints sensitization in people with and without chronic low back pain: a matched-sample cross-sectional study. J Back Musculoskelet Rehabil 2023; 36: 137-46. |
| 18. |
Goo B, Kim JH, Kim EJ, et al. Thread embedding acupuncture for herniated intervertebral disc of the lumbar spine: a multicenter, randomized, patient-assessor-blinded, controlled, parallel, clinical trial. Complement Ther Clin Pract 2022; 46: 101538.
DOI URL |
| 19. |
Ding F, Shao ZW, Xiong LM. Cell death in intervertebral disc degeneration. Apoptosis 2013; 18: 777-85.
DOI PMID |
| 20. | Feng G, Zha Z, Huang Y, et al. Sustained and bioresponsive two-stage delivery of therapeutic mirna via polyplex micelle-loaded injectable hydrogels for inhibition of intervertebral disc fibrosis. Adv Healthc Mater 2018; 7: e1800623. |
| 21. | Chen S, Liu S, Ma K, Zhao L, Lin H, Shao Z. TGF-β signaling in intervertebral disc health and disease. Osteoarthritis Cartilage Online 2019; 27: 1109-17. |
| 22. |
Jin H, Shen J, Wang B, Wang M, Shu B, Chen D. TGF-β signaling plays an essential role in the growth and maintenance of intervertebral disc tissue. FEBS Lett 2011; 585: 1209-15.
DOI PMID |
| 23. |
Zieba J, Forlenza KN, Khatra JS, et al. TGFβ and BMP dependent cell fate changes due to loss of filamin b produces disc degeneration and progressive vertebral fusions. PLoS Genet 2016; 12: e1005936.
DOI URL |
| 24. |
Tian Y, Yuan W, Li J, et al. TGFβ regulates Galectin-3 expression through canonical Smad3 signaling pathway in nucleus pulposus cells: implications in intervertebral disc degeneration. Matrix Biol 2016; 50: 39-52.
DOI PMID |
| 25. |
Kim KK, Sheppard D, Chapman HA. TGF-β1 signaling and tissue fibrosis. Cold Spring Harb Perspect Biol 2018; 10: a022293.
DOI URL |
| 26. |
Xu Y, Qu X, Zhou J, et al. Pilose antler peptide-3.2KD ameliorates adriamycin-induced myocardial injury through TGF-β/SMAD signaling pathway. Front Cardiovasc Med 2021; 8: 659643.
DOI URL |
| 27. |
Bian Q, Ma L, Jain A, et al. Mechanosignaling activation of TGFβ maintains intervertebral disc homeostasis. Bone Res 2017; 5: 17008.
DOI PMID |
| 28. |
Meng XM, Nikolic-Paterson DJ, Lan HY. TGF-β: the master regulator of fibrosis. Nat Rev Nephrol 2016; 12: 325-38.
DOI |
| 29. |
Masuda K, Aota Y, Muehleman C, et al. A novel rabbit model of mild, reproducible disc degeneration by an anulus needle puncture: correlation between the degree of disc injury and radiological and histological appearances of disc degeneration. Spine (Phila Pa 1976) 2005; 30: 5-14.
DOI URL |
| 30. |
Yang H, Cao C, Wu C, et al. TGF-βl suppresses inflammation in cell therapy for intervertebral disc degeneration. Sci Rep 2015; 5: 13254.
DOI PMID |
| 31. |
Wang WJ, Yu XH, Wang C, et al. MMPs and ADAMTSs in intervertebral disc degeneration. Clin Chim Acta 2015; 448: 238-46.
DOI URL |
| 32. |
Livshits G, Popham M, Malkin I, et al. Lumbar disc degeneration and genetic factors are the main risk factors for low back pain in women: the UK Twin Spine Study. Ann Rheum Dis 2011; 70: 1740-5.
DOI PMID |
| 33. |
Takatalo J, Karppinen J, Niinimäki J, et al. Does lumbar disc degeneration on magnetic resonance imaging associate with low back symptom severity in young Finnish adults? Spine (Phila Pa 1976) 2011; 36: 2180-9.
DOI URL |
| 34. | Feng C, Yang M, Lan M, et al. ROS: Crucial intermediators in the pathogenesis of intervertebral disc degeneration. Oxid Med Cell Longev 2017; 2017: 5601593. |
| 35. | Yao C, Guo G, Huang R, et al. Manual therapy regulates oxidative stress in aging rat lumbar intervertebral discs through the SIRT1/FOXO1 pathway. Aging (Albany NY) 2022; 14: 2400-17. |
| 36. | Sudhir G, Jayabalan V, Sellayee S, Gadde S, Kailash K. Is there an interdependence between paraspinal muscle mass and lumbar disc degeneration? A MRI based study at 2520 levels in 504 patients. J Clin Orthop Trauma 2021; 22: 101576. |
| 37. |
Kader DF, Wardlaw D, Smith FW. Correlation between the MRI changes in the lumbar multifidus muscles and leg pain. Clin Radiol 2000; 55: 145-9.
DOI PMID |
| 38. |
Zhu F, Duan W, Zhong C, Ji B, Liu X. The protective effects of dezocine on interleukin-1β-induced inflammation, oxidative stress and apoptosis of human nucleus pulposus cells and the possible mechanisms. Bioengineered 2022; 13: 1399-410.
DOI PMID |
| 39. |
Freemont AJ. The cellular pathobiology of the degenerate intervertebral disc and discogenic back pain. Rheumatology (Oxford) 2009; 48: 5-10.
DOI PMID |
| 40. |
Le Maitre CL, Pockert A, Buttle DJ, Freemont AJ, Hoyland JA. Matrix synthesis and degradation in human intervertebral disc degeneration. Biochem Soc Trans 2007; 35: 652-5.
DOI URL |
| 41. |
Antoniou J, Goudsouzian NM, Heathfield TF, et al. The human lumbar endplate. Evidence of changes in biosynthesis and denaturation of the extracellular matrix with growth, maturation, aging, and degeneration. Spine (Phila Pa 1976) 1996; 21: 1153-61.
DOI URL |
| 42. | Kong M, Zhang Y, Song M, et al. Myocardin-related transcription factor a nuclear translocation contributes to mechanical overload-induced nucleus pulposus fibrosis in rats with intervertebral disc degeneration. Int J Mol Med 2021; 48. |
| 43. |
Che Z, Xueqin J, Zhang Z. LncRNA OIP5-AS1 accelerates intervertebral disc degeneration by targeting miR-25-3p. Bioengineered 2021; 12: 11201-12.
DOI PMID |
| 44. |
Le Maitre CL, Freemont AJ, Hoyland JA. Localization of degradative enzymes and their inhibitors in the degenerate human intervertebral disc. J Pathol 2004; 204: 47-54.
DOI PMID |
| 45. |
Bachmeier BE, Nerlich A, Mittermaier N, et al. Matrix metalloproteinase expression levels suggest distinct enzyme roles during lumbar disc herniation and degeneration. Eur Spine J 2009; 18: 1573-86.
DOI PMID |
| 46. |
Zhao K, Chen M, Liu T, et al. Rhizoma drynariae total flavonoids inhibit the inflammatory response and matrix degeneration via MAPK pathway in a rat degenerative cervical intervertebral disc model. Biomed Pharmacother 2021; 138: 111466.
DOI PMID |
| 47. |
Wei F, Zhong R, Zhou Z, et al. In vivo experimental intervertebral disc degeneration induced by bleomycin in the rhesus monkey. BMC Musculoskelet Disord 2014; 15: 340.
DOI URL |
| 48. |
Zhao CQ, Jiang LS, Dai LY. Programmed cell death in intervertebral disc degeneration. Apoptosis 2006; 11: 2079-88.
DOI URL |
| 49. |
Li D, Zhu B, Ding L, Lu W, Xu G, Wu J. Role of the mitochondrial pathway in serum deprivation-induced apoptosis of rat endplate cells. Biochem Biophys Res Commun 2014; 452: 354-60.
DOI URL |
| 50. |
Tu J, Li W, Zhang Y, et al. Simvastatin inhibits IL-1β-induced apoptosis and extracellular matrix degradation by suppressing the NF-kB and MAPK pathways in nucleus pulposus cells. Inflammation 2017; 40: 725-34.
DOI PMID |
| 51. |
Gruber HE, Hoelscher GL, Ingram JA, Bethea S, Zinchenko N, Hanley EJ. Variations in aggrecan localization and gene expression patterns characterize increasing stages of human intervertebral disk degeneration. Exp Mol Pathol 2011; 91: 534-9.
DOI PMID |
| 52. |
Peng B, Hao J, Hou S, et al. Possible pathogenesis of painful intervertebral disc degeneration. Spine (Phila Pa 1976) 2006; 31: 560-6.
DOI URL |
| 53. |
Bian Q, Jain A, Xu X, et al. Excessive activation of TGFβ by spinal instability causes vertebral endplate sclerosis. Sci Rep 2016; 6: 27093.
DOI PMID |
| 54. |
Qin Z, Xia W, Fisher GJ, Voorhees JJ, Quan T. YAP/TAZ regulates TGF-β/Smad3 signaling by induction of Smad7 via AP-1 in human skin dermal fibroblasts. Cell Commun Signal 2018; 16: 18.
DOI |
| 55. |
Heldin CH, Moustakas A. Role of Smads in TGFβ signaling. Cell Tissue Res 2012; 347: 21-36.
DOI URL |
| 56. |
Moustakas A, Heldin CH. The regulation of TGFbeta signal transduction. Development 2009; 136: 3699-714.
DOI PMID |
| 57. |
He W, Li AG, Wang D, et al. Overexpression of Smad7 results in severe pathological alterations in multiple epithelial tissues. Embo J 2002; 21: 2580-90.
DOI PMID |
| 58. |
Hu HH, Chen DQ, Wang YN, et al. New insights into TGF-β/Smad signaling in tissue fibrosis. Chem Biol Interact 2018; 292: 76-83.
DOI URL |
| 59. |
Chen SJ, Ning H, Ishida W, et al. The early-immediate gene EGR-1 is induced by transforming growth factor-beta and mediates stimulation of collagen gene expression. J Biol Chem 2006; 281: 21183-97.
DOI URL |
| 60. |
Ghosh AK, Yuan W, Mori Y, Varga J. Smad-dependent stimulation of type I collagen gene expression in human skin fibroblasts by TGF-beta involves functional cooperation with p300/CBP transcriptional coactivators. Oncogene 2000; 19: 3546-55.
DOI PMID |
| 61. |
Yang H, Yuan C, Wu C, et al. The role of TGF-β1/Smad2/3 pathway in platelet-rich plasma in retarding intervertebral disc degeneration. J Cell Mol Med 2016; 20: 1542-9.
DOI PMID |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

