Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (6): 1081-1091.DOI: 10.19852/j.cnki.jtcm.20231018.002
• Research Articles • Previous Articles Next Articles
Bornyl acetate extracted from Sharen (Fructus Amomi) inhibits proliferation, invasion and induces apoptosis by suppressing phosphatidylinositol-3-kinase/protein kinase B signaling in colorectal cancer
LI Xiaohua1, DUAN Zhihang1, YUE Jianjun1, ZHANG Yongyu1, LI Yihang2, LIU Shifang2, NIE Qu1, YANG Depo2( ), ZHANG Lixia3(
), ZHANG Lixia3( )
)
- 1 School of Traditional Dai-Thai Medicine, West Yunnan University of Apllied Science, Jinghong 666100, China
2 Institute of Medicinal Plant Development, Chinese Academy of Medical Sciences, Jinghong 666100, China
3 Yunnan Branch, Institute of Medicinal Plant Development, Chinese Academy of Medical Sciences, Yunnan Key Laboratory of Southern Medicine Utilization, Jinghong 666100, China
-
Received:2022-08-23Accepted:2022-11-15Online:2023-10-25Published:2023-11-01 -
Contact:YANG Depo, School of Pharmaceutical Science, Sun Yat-sen University, Guangzhou 510120, China. lssydp@mail.sysu.edu.cn; ZHANG Lixia, Yunnan Branch, Institute of Medicinal Plant Development, Chinese Academy of Medical Sciences, Yunnan Key Laboratory of Southern Medicine Utilization, Jinghong 666100, China; 13988194288@163.com. Telephone: +86-13640706628; +86-13988194288 -
Supported by:Underground Ecological Planting Technology and Base Establishment of Sharen (Fructus Amomi) in the forest(2017YFC1701102);Characteristic Dai-Medicine Resource ERC of West Yunnan University of Apllied Science(2022KYPT0004);Study on the symbiotic system of Sharen(Fructus Amomi);weevil pollination and its "push-pull" pollination mechanism(82260736);Major Science and Technology Special Plan of Yunnan Province(202102AA100020);Scientific and Technological Talents and Platform Plan of Yunnan Province(202105AG070011)
Cite this article
LI Xiaohua, DUAN Zhihang, YUE Jianjun, ZHANG Yongyu, LI Yihang, LIU Shifang, NIE Qu, YANG Depo, ZHANG Lixia. Bornyl acetate extracted from Sharen (Fructus Amomi) inhibits proliferation, invasion and induces apoptosis by suppressing phosphatidylinositol-3-kinase/protein kinase B signaling in colorectal cancer[J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1081-1091.
share this article
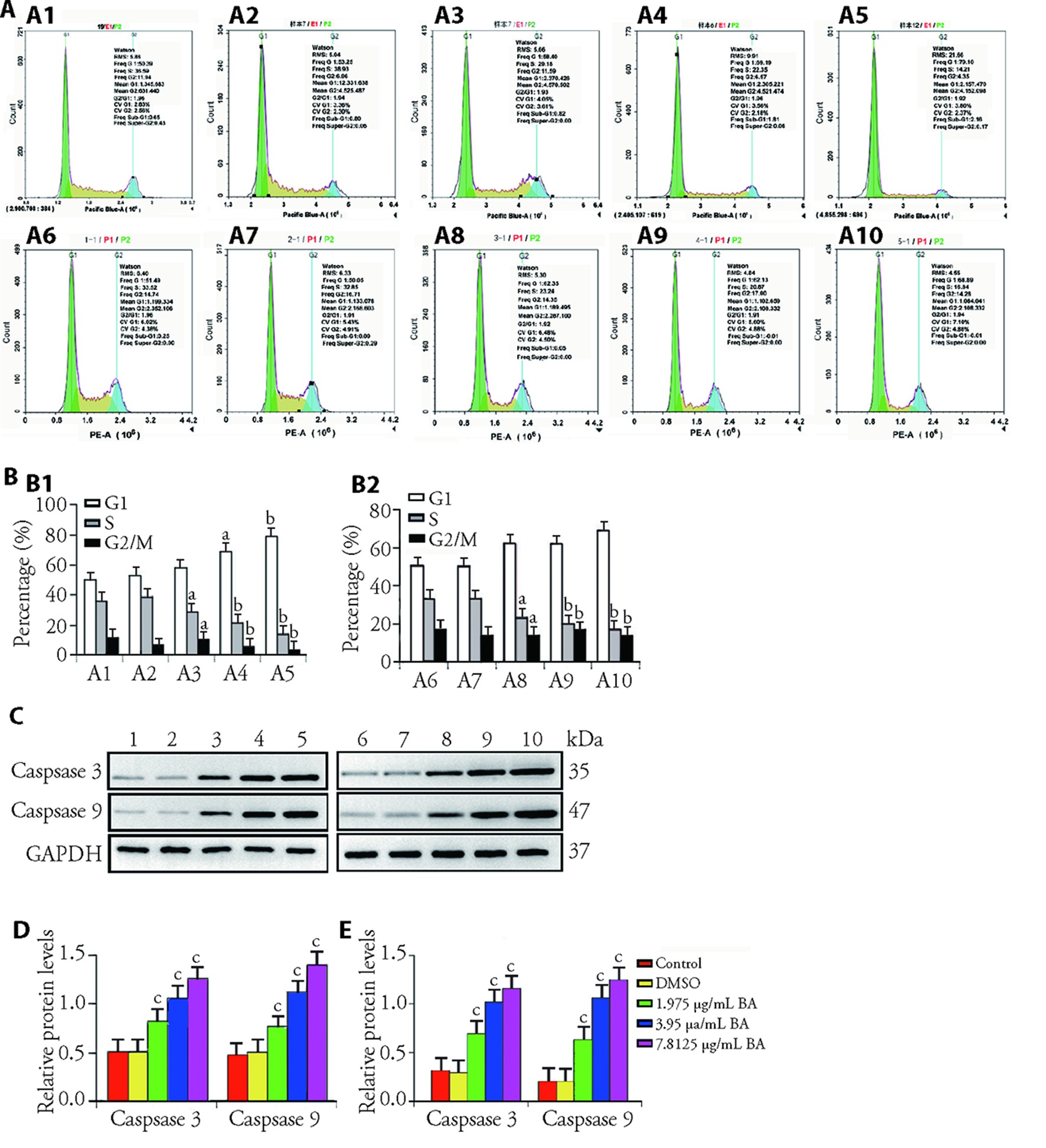
Figure 1 BA induces apoptosis and blocks cell cycle of CRC A, B: effect of DMSO and increasing concentration of BA on SW480 and HT29 cells cycle arrest. A1: the percentage of SW480 cell at G1/S/G2/M phases of control group; A2: the percentage of SW480 cell at G1/S/G2/M phases of DMSO group; A3: the percentage of SW480 cell at G1/S/G2/M phases of BA low-dose group; A4: the percentage of SW480 cell at G1/S/G2/M phases of BA middle-dose group; A5:the percentage of SW480 cell at G1/S/G2/M phases of BA high-dose group; A6: the percentage of HT29 cell at G1/S/G2/M phases of control group; A7: the percentage of HT29 cell at G1/S/G2/M phases of 0.2% DMSO group; A8: the percentage of HT29 cell at G1/S/G2/M phases of BA low-dose group; A9: the percentage of HT29 cell at G1/S/G2/M phases of BA middle-dose group; A10: the percentage of HT29 cell at G1/S/G2/M phases of BA high-dose group; B1: ratio of G1, S and G2/M period of SW480 cell; B2: ratio of G1, S and G2/M period of HT29 cell (n = 6). C: protein expression image of cleaved-caspase 3 and caspase 9 of SW480 and HT29. 1: protein expression image of cleaved-caspase 3 and caspase 9 of SW480 cell in control group; 2: protein levels of cleaved-caspase 3 and caspase 9 of SW480 cell in DMSO group; 3: protein expression image of cleaved-caspase 3 and caspase 9 of SW480 cell in BA low-dose group; 4: protein expression image of cleaved-caspase 3 and caspase 9 of SW480 cell treated in BA middle-dose; 5: protein expression image of cleaved-caspase 3 and caspase 9 of SW480 cell in BA high-dose group; 6: protein expression image of cleaved-caspase 3 and caspase 9 of HT29 cell in control group; 7: protein levels of cleaved-caspase 3 and caspase 9 of HT29 cell in DMSO group; 8: protein expression image of cleaved-caspase 3 and caspase 9 of HT29 cell in BA low-dose group; 9: protein expression image of cleaved-caspase 3 and caspase 9 of HT29 cell treated in BA middle-dose; 10: protein expression image of cleaved-caspase 3 and caspase 9 of HT29 cell in BA high-dose group. D: relative protein level of cleaved-caspase 3 and caspase 9 of SW480 cell (n = 6). E: relative protein level of cleaved-caspase 3 and caspase 9 of HT29 cell (n = 6). Control group: the cells were cultured in a complete medium containing DMEM, 10% FBS, and 1% penicillin-streptomycin solution; DMSO group: the cells were treated with 0.2% DMSO; BA low-dose group: the cells were given 1.975 μg/mL BA; BA middle-dose group: the cells were given 3.95 μg/mL BA; BA high-dose group: the cells were given 7.8125 μg/mL BA. BA: bornyl acetate; CRC: colorectal cancer; DMSO: dimethyl sulfoxide; DMEM: dulbecco's modified eagle medium; FBS: fetal bovine serum; G1: gap1, the gap time from the completion of mitosis to the start of DNA replication; S: synthesis phase, the period of DNA replication; G2/M: gap2/mitosis, the ratio of the period from the completion of DNA replication to the beginning of mitosis and mitosis. Data are presented as mean ± standard error of mean. aP < 0.05, bP < 0.01, and cP < 0.001, compared with control group.
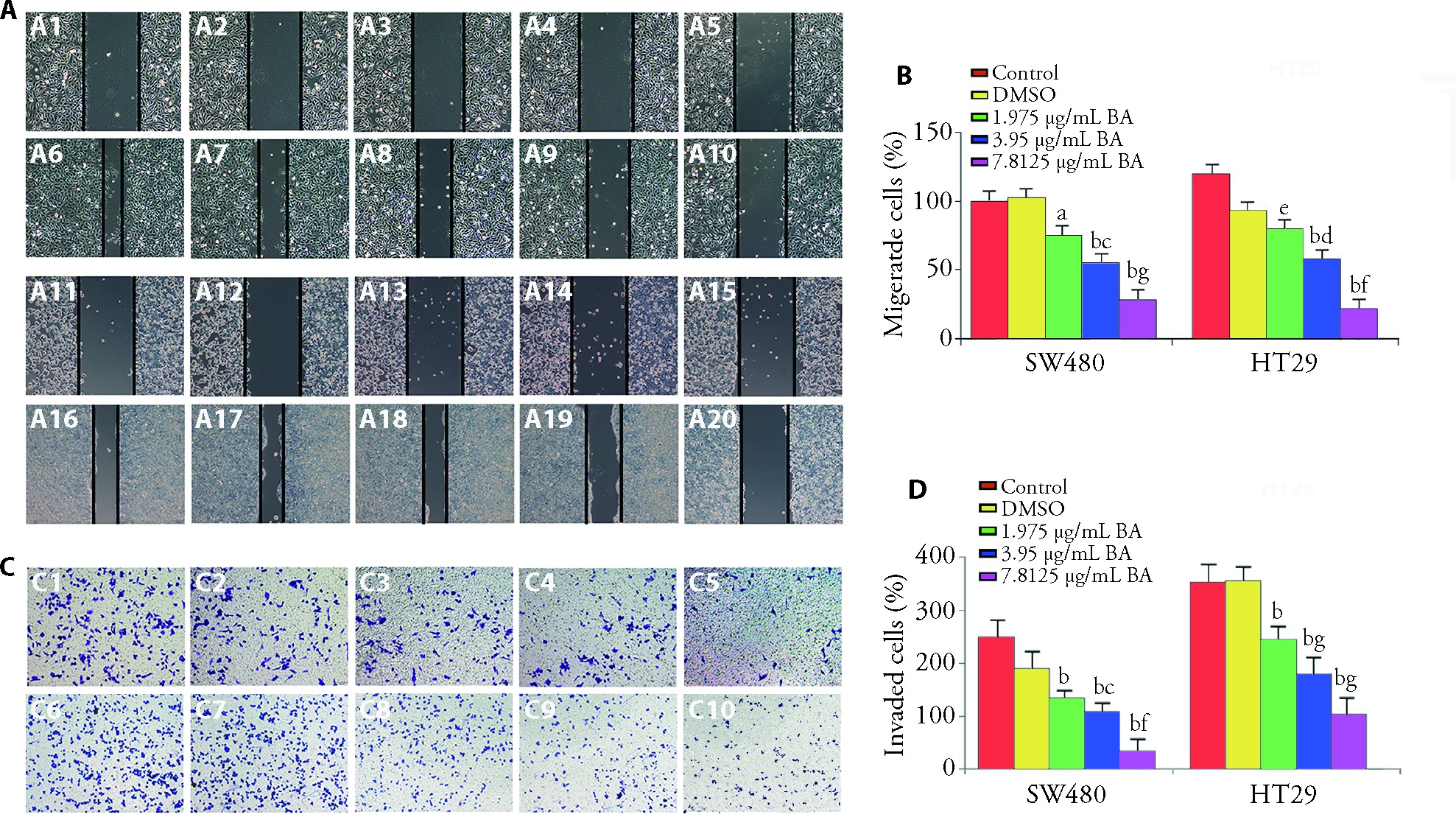
Figure 2 BA inhibits migration and invasion of CRCs A: SW480 and HT29 cells migration ability after treated with increasing concentration of BA at 0 and 24 h, × 40. A1: the migration ability of SW480 cell at 0 h in control group; A2: the migration ability of SW480 cell at 0 h in DMSO group; A3: the migration ability of SW480 cell at 0 h in BA low-dose group; A4: the migration ability of SW480 cell at 0 h in BA middle-dose group; A5: the migration ability of SW480 cell at 0 h in BA high-dose group; A6: the migration ability of SW480 cell at 24h in control group; A7: the migration ability of SW480 cell at 24 h in DMSO group; A8: the migration ability of SW480 cell at 24 h in BA low-dose group; A9: the migration ability of SW480 cell at 24 h in BA middle-dose group; A10: the migration ability of SW480 cell at 24 h in BA high-dose group; A11: the migration ability of HT29 cell at 0 h in control group; A12: the migration ability of HT29 cell at 0 h in DMSO group; A13: the migration ability of HT29 cell at 0 h in BA low-dose group; A14: the migration ability of HT29 cell at 0 h in BA middle-dose group; A15: the migration ability of HT29 cell at 0 h in BA high-dose group; A16: the migration ability of HT29 cell at 24h in control group; A17: the migration ability of HT29 cell at 24 h in DMSO group; A18: the migration ability of HT29 cell at 24 h in BA low-dose group; A19: the migration ability of HT29 cell at 24 h in BA middle-dose group; A20: the migration ability of HT29 cell at 24 h in BA high-dose group. B: the migration cells percentage of SW480 and HT29 (n = 6). C: the invasion ability cells of SW480 and HT29 cells treated with increasing concentrations of BA, ×40. C1: the invasion ability of SW480 cell at 24 h of control group; C2: the invasion ability of SW480 cell at 24 h of DMSO group; C3: the invasion ability of SW480 cell at 24 h of BA low-dose group; C4: the invasion ability of SW480 cell at 24 h of BA middle-dose group; C5: the invasion ability of SW480 cell at 24 h of BA high-dose group; C6: the invasion ability of HT29 cell at 24h of control group; C7: the invasion ability of HT29 cell at 24 h of DMSO group; C8: the invasion ability of HT29 cell at 24 h of BA low-dose group; C9: the invasion ability of HT29 cell at 24 h of BA middle-dose group; C10: the invasion ability of HT29 cell at 24 h of BA high-dose group. D: the invaded cell percentage of SW480 and HT29 (n = 6). Control group: the cells were cultured in a complete medium containing DMEM, 10% FBS, and 1% penicillin-streptomycin solution; DMSO group: the cells were treated with 0.2% DMSO; BA low-dose group: the cells were given 1.975 μg/mL BA; BA middle-dose group: the cells were given 3.95 μg/mL BA; the high-dose group: the cells were given 7.8125 μg/mL BA. BA: bornyl acetate; CRCs: colorectal cancers; DMSO: dimethyl sulfoxide; DMEM: dulbecco's modified eagle medium; FBS: fetal bovine serum. Data are presented as mean ± standard error of mean. aP < 0.05, bP < 0.001, and eP < 0.01, compared with control group; cP < 0.05 and dP < 0.001, compared with low-dose group; gP < 0.001 and fP < 0.01, compared with middle-dose group.

Figure 3 BA inhibits SW480 cell migration and invasion via inhibiting PI3K/AKT pathway A: the migration ability of SW480 cell treated with different administration, × 40. A1: the migration ability at 0 h in control group; A2: the migration ability at 0 h in 0.2% DMSO group; A3: the migration ability at 0 h in 7.8125 μg/mL BA group; A4: the migration ability at 0 h in 7.8125 μg/mL BA+LY294002 group; A5: the migration ability at 0 h in 7.8125 μg/mL BA + 740Y-P group; A6: the migration ability at 24 h in control group; A7: the migration ability at 24 h in DMSO group; A8: the migration ability at 24 h in 7.8125 μg/mL BA group; A9: the migration ability at 24 h in 7.8125 μg/mL BA + LY294002 group; A10: the migration ability at 24 h in 7.8125 μg/mL BA + 740Y-P group. B: the migration cells percentage of SW480 (n = 6). C: The invasion ability of SW480 cells treated with different administration at 24 h. ×40. C1: the invasion ability of control group; C2: the invasion ability of DMSO group; C3: the invasion ability of 7.8125 μg/mL BA group; C4: the invasion ability of 7.8125 μg/mL BA + LY294002 group; C5: the invasion ability of 7.8125 μg/mL BA + 740Y-P group. D: the invaded cells percentage of SW480 (n = 6). E: the P85, phosphorylation of P85, AKT and phosphorylation of AKT protein levels of SW480 cells treated with different administration by WB method. ×40. 1: control group; 2: DMSO group; 3: 7.8125 μg/mL BA group; 4: 7.8125 μg/mL BA + LY294002 group; 5: 7.8125 μg/mL BA + 740Y-P group. F: the relative protein levels of SW480cells (n = 6). Control group: the cells were cultured in a complete medium containing DMEM, 10% FBS, and 1% penicillin-streptomycin solution; DMSO group: the cells were treated with 0.2% DMSO. BA: bornyl acetate; PI3K/AKT: phosphatidylinositol-3-kinase/protein kinase B; DMSO: dimethyl sulfoxide; DMEM: dulbecco's modified eagle medium; FBS: fetal bovine serum; WB: Western blotting; 740Y-P: 740YPDGFR; LY294002: 2-morpholino-8-phenyl-4-oxo-4H-1-benzopyran. Data are presented as mean ± standard error of mean. aP < 0.001, compared with control group; bP < 0.001, cP < 0.05, and dP < 0.01, compared with 7.8125 μg/mL BA group; eP < 0.001, compared with 7.8125 μg/mL BA + 740Y-P group.
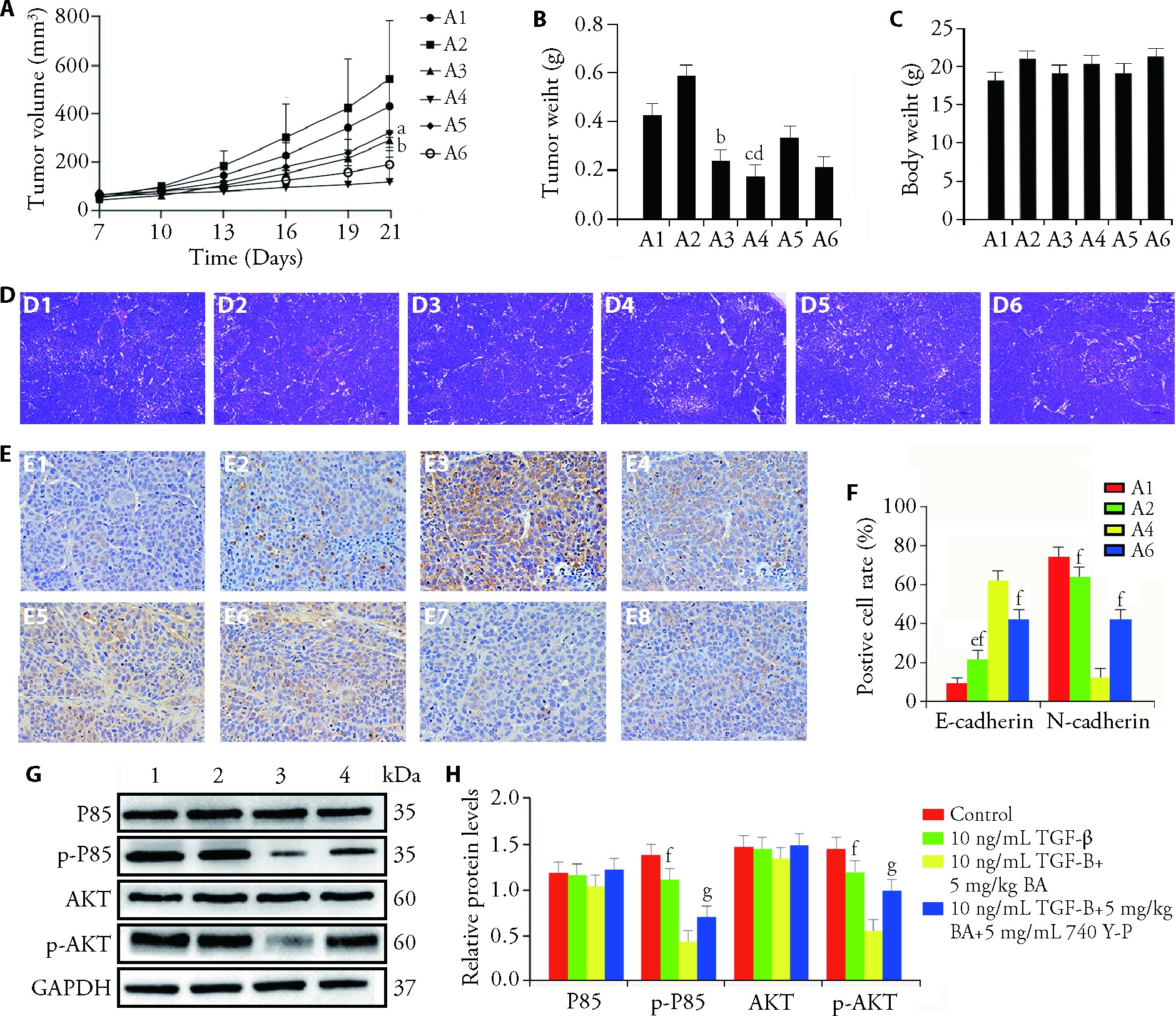
Figure 4 BA represses tumor growth in vivo SW480 cells were pretreated with TGF-β prior to their transplantation into mice, and then intraperitoneally injected with 0.2 mL BA (5 mg/kg, 2 mg/kg) or intraperitoneally co-injected with 0.2 mL BA (5, 2 mg/kg) and 2 mg/mL 740 Y-P. A-C: The tumors volume, tumor size and body weight of mice in different groups, × 10. A1: control group; A2: TGF-β group; A3: low-dose BA group; A4: high-dose BA group; A5: low-dose BA + PI3K group; A6: high-dose BA + PI3K group. D-F: the pathology of tumors was analyzed by HE and IHC staining, × 40. D1: hepatic histological of control group; D2: hepatic histological of 10 ng/mL TGF-β group; D3: hepatic histological of low-dose BA group; D4: hepatic histological of High-dose BA group; D5: hepatic histological of low-dose BA + PI3K group; D6: hepatic histological of high-dose BA + PI3K group. E1: E-cadherin protein expression level of control group; E2: E-cadherin protein expression level of TGF-β group; E3: E-cadherin protein expression level of high-dose BA group; E4: E-cadherin protein expression level of high-dose BA + PI3K group; E5: N-cadherin protein expression level of control group; E6: N-cadherin protein expression level of TGF-β group; E7: N-cadherin protein expression level of high-dose BA group; E8: N-cadherin protein expression level of high-dose BA + PI3K group. G, H: the protein levels of P85, AKT as well as phosphorylation of P85 and AKT analyzed by WB. 1: control group; 2:10 ng/mL TGF-β group; 3: high-dose BA group; 4: high-dose BA + PI3K group (n = 3). Control group: mice were injected with 200 μL SW480 cells at a density of 5.0×106 cells/mL until tumor growth to 100 mm3, and then administered intraperitoneal injection of 0.2 mL saline each day for 14 d. The remaining mice were injected with 200 μL SW480 cells pretreated with 10 ng/mL TGF-β until the tumors grew to 100 mm3. TGF-β group: mice were administered intraperitoneal injection of 0.2 mL saline per day for 14 d; low-dose BA group: mice received intraperitoneal injection of 0.1 mL BA (2 mg/kg) for 14 d; high-dose BA group: mice received intraperitoneal injection of 0.2 mL BA (5 mg/kg) for 14 d; low-dose BA + PI3K group: mice received intraperitoneal injection of 0.1 mL BA (2 mg/kg) and 0.1 mL 740 Y-P (10 mg/kg) for 14 d; high-dose BA + PI3K group: mice received intraperitoneal injection of 0.2 mL BA (5 mg/kg) and 0.1 mL 740 Y-P (10 mg/kg) for 14 d. BA: bornyl acetate; TGF-β: transforming growth factor beta; 740 Y-P: 740YPDGFR; PI3K: phosphatidylinositol-3-kinase; P85: PI3 kinase p85 antibody; AKT: protein kinase B; WB: western blotting; HE: hematoxylin and eosin; IHC: immunochemistry. Data are presented as mean ± standard error of mean. aP < 0.05, bP < 0.01 and cP < 0.001, compared with TGF-β group; dP < 0.05, compared with high-dose BA group; eP < 0.05, compared with control group; fP < 0.001 and gP < 0.001, compared with high-dose BA + PI3K group.
| 1. |
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018; 68: 394-424.
DOI URL |
| 2. |
Brody H. Colorectal cancer. Nature 2015; 521: S1.
DOI |
| 3. |
Gu MJ, Huang QC, Bao CZ, et al. Attributable causes of colorectal cancer in China. BMC Cancer 2018; 18: 38.
DOI URL |
| 4. |
Patel SG, Ahnen DJ. Colorectal cancer in the young. Curr Gastroenterol Rep 2018; 20: 15.
DOI PMID |
| 5. |
Oncology TL. Colorectal cancer: a disease of the young? Lancet Oncol 2017; 18: 413.
DOI PMID |
| 6. |
Zhang H, Wang Z, Wu JZ, Ma R, Feng JF. Long noncoding RNAs predict the survival of patients with colorectal cancer as revealed by constructing an endogenous RNA network using bioinformation analysis. Cancer Med 2019; 8: 863-73.
DOI URL |
| 7. |
Kuipers EJ, Grady WM, Lieberman D, et al. Colorectal cancer. Nat Rev Dis Primers 2015; 1: 15065.
DOI PMID |
| 8. |
Suo SZ, Lai YF, Li M, et al. Phytochemicals, pharmacology, clinical application, patents, and products of Amomi fructus. Food Chem Toxicol 2018; 119: 31-6.
DOI PMID |
| 9. | Chen L AoH, Ye Q, Li HX. Chemical constituents analysis of volatile oil from different parts of the fruit of Amomum villosum by GC-MS. Zhong Guo Shi Yan Fang Ji Xue 2014: 80-3. |
| 10. | State Pharmacopoeia Committee of China. Chinese pharmacopoeia Vol.I. Beijing: the Medicine Science and Technology Press of China, 2020: 264. |
| 11. | Zhang J, Lu Q, Zhang XF, Dai YP, Zu LH. Discussion on pharmacopoeia standard and drug site of Amomi Fructus based on quality investigation of commercial samples. Zhong Guo Shi Yan Fang Ji Xue 2022; 28: 139-44. |
| 12. |
Yang H, Zhao RG, Chen H, Jia P, Bao L, Tang H. Bornyl acetate has an anti-inflammatory effect in human chondrocytes via induction of IL-11. IUBMB Life 2014; 66: 854-9.
DOI PMID |
| 13. | Yang L, Liu JL, Li YZ, Qi GY. Bornyl acetate suppresses ox-LDL-induced attachment of THP-1 monocytes to endothelial cells. Biomed Pharmacother 2018: 103234-9. |
| 14. |
Shahwar D, Ullah S, Khan MA, Ahmad N, Saeed A, Ullah S. Anticancer activity of Cinnamon tamala leaf constituents towards human ovarian cancer cells. Pak J Pharm Sci 2015; 28: 969-72.
PMID |
| 15. |
Li J, Wang SX. Synergistic enhancement of the antitumor activity of 5-fluorouracil by bornyl acetate in SGC-7901 human gastric cancer cells and the determination of the underlying mechanism of action. J buon 2016; 21: 108-17.
PMID |
| 16. |
Xie YB, Shi XF, Sheng K, et al. PI3K/Akt signaling transduction pathway, erythropoiesis and glycolysis in hypoxia (review). Mol Med Rep 2019; 19: 783-91.
DOI PMID |
| 17. | Ediriweera MK, Tennekoon KH, Samarakoon SR. Role of the PI3K/AKT/mTOR signaling pathway in ovarian cancer: biological and therapeutic significance. Semin Cancer Biol 2019: 59147-60. |
| 18. | Ma ZL, Zhang WW, Wu YR, et al. Cyclophilin A inhibits A 549 cell oxidative stress and apoptosis by modulating the PI3K/Akt/mTOR signaling pathway. Biosci Rep 2021;41: BSR20203219. |
| 19. |
Xu JC, Chen TY, Liao LT, et al. NETO2 promotes esophageal cancer progression by inducing proliferation and metastasis via PI3K/AKT and ERK pathway. Int J Biol Sci 2021; 17: 259-70.
DOI URL |
| 20. | Hong SZ, Cai WJ, Huang ZC, et al. Ginsenoside Rg3 enhances the anticancer effect of 5‑FU in colon cancer cells via the PI3K/AKT pathway. Oncol Rep 2020; 44: 1333-42. |
| 21. |
Cheng HY, Jiang X, Zhang Q, et al. Naringin inhibits colorectal cancer cell growth by repressing the PI3K/AKT/mTOR signaling pathway. Exp Ther Med 2020; 19: 3798-804.
DOI PMID |
| 22. | Pei XD, Yao HL, Shen LQ, et al. α-Cyperone inhibits the proliferation of human cervical cancer HeLa cells via ROS-mediated PI3K/Akt/mTOR signaling pathway. Eur J Pharmacol 2020; 883: 173355. |
| 23. |
Chen XJ, Liu JQ, Zhang QL, et al. Exosome-mediated transfer of miR-93-5p from cancer-associated fibroblasts confer radioresistance in colorectal cancer cells by downregulating FOXA1 and upregulating TGFB3. J Exp Clin Cancer Res 2020; 39: 65.
DOI PMID |
| 24. |
Han QR, Ma Y, Wang H, et al. Resibufogenin suppresses colorectal cancer growth and metastasis through RIP3-mediated necroptosis. J Transl Med 2018; 16: 201.
DOI PMID |
| 25. |
Toulany M, Rodemann HP. Phosphatidylinositol 3-kinase/Akt signaling as a key mediator of tumor cell responsiveness to radiation. Semin Cancer Biol 2015: 35: 180-90.
DOI PMID |
| 26. | Ma K, Zhang C, Li W. TGF-β is associated with poor prognosis and promotes osteosarcoma progression via PI3K/Akt pathway activation. Cell Cycle 2020; 19: 2327-39. |
| 27. |
Wang KL, Chen Q, Shao YY, et al. Anticancer activities of TCM and their active components against tumor metastasis. Biomed Pharmacother 2021; 133: 111044.
DOI PMID |
| 28. |
Liu YL, Chen YC, Lin LF, Li H. Gambogic acid as a candidate for cancer therapy: a review. Int J Nanomedicine 2020: 15: 10385-99.
DOI URL |
| 29. |
Gao C, Zhou YL, Jiang ZL, et al. Cytotoxic and chemosensitization effects of Scutellarin from traditional Chinese herb Scutellaria altissima L. in human prostate cancer cells. Oncol Rep 2017; 38: 1491-9.
DOI URL |
| 30. | Saunders IT, Mir H, Kapur N, Singh S. Emodin inhibits colon cancer by altering BCL-2 family proteins and cell survival pathways. Cancer Cell Int 2019: 19: 98. |
| 31. | Chen X, Guo H, Li FX, D Fan. Physcion 8-O-β-glucopyranoside suppresses the metastasis of breast cancer in vitro and in vivo by modulating DNMT1. Pharmacol Rep 2017; 69: 36-44. |
| 32. |
Ateba SB, Mvondo MA, Ngeu ST, et al. Natural terpenoids against female breast cancer: a 5-year recent research. Curr Med Chem 2018; 25: 3162-213.
DOI PMID |
| 33. | Sharma SH, Thulasingam S, Nagarajan S. Terpenoids as anti-colon cancer agents-A comprehensive review on its mechanistic perspectives. Eur J Pharmacol 2017: 795169-78. |
| 34. |
Zakaria KN, Amid A, Zakaria Z, Jamal P, Ismail A. Anti-proliferative activity of triterpenes isolated from Clinicanthus nutans on Hep-G2 liver cancer cells. Asian Pac J Cancer Prev 2019; 20: 563-7.
DOI URL |
| 35. |
Yang TY, Wu YJ, Chang CI, Chiu CC, Wu ML. The Effect of bornyl cis-4-hydroxycinnamate on melanoma cell apoptosis is associated with mitochondrial dysfunction and endoplasmic reticulum stress. Int J Mol Sci 2018; 19: 1370.
DOI URL |
| 36. |
Yang TY, Wu ML, Chang CI, Liu CI, Cheng TC, Wu YJ. Bornyl cis-4-hydroxycinnamate suppresses cell metastasis of melanoma through FAK/PI3K/Akt/mTOR and MAPK signaling pathways and inhibition of the epithelial-to-mesenchymal transition. Int J Mol Sci 2018; 19: 2152.
DOI URL |
| 37. |
Namshir J, Shatar A, Khandaa O, Tserennadmid R, Shiretorova VG, Nguyen MC. Antimicrobial, antioxidant and cytotoxic activity on human breast cancer cells of essential oil from Pinus sylvestris. var mongolica needle. Mongolian J Chem 2020; 21: 19-26.
DOI URL |
| 38. |
Karan T, Yildiz I, Aydin A, Erenler R. Inhibition of various cancer cells proliferation of bornyl acetate and essential oil from Inula graveolens (Linnaeus) Desf. Rec Nat Prod 2018; 12: 273-83.
DOI URL |
| 39. |
LaFargue CJ, Dal MGZ, Sood AK, Coleman RL. Exploring and comparing adverse events between PARP inhibitors. Lancet Oncol 2019; 20: 15-28.
DOI PMID |
| 40. |
Ibrahim A, Hirschfeld S, Cohen MH, Griebel DJ, Williams GA, Pazdur R. FDA drug approval summaries: oxaliplatin. Oncologist 2004; 9: 8-12.
PMID |
| 41. |
Grothey A, Goldberg RM. A review of oxaliplatin and its clinical use in colorectal cancer. Expert Opin Pharmacother 2004; 5: 2159-70.
PMID |
| 42. | Rejhová A, Opattová A, Čumová A, Slíva D, P Vodička. Natural compounds and combination therapy in colorectal cancer treatment. Eur J Med Chem 2018: 144582-94. |
| 43. |
Slezakova S, Ruda-Kucerova J. Anticancer activity of artemisinin and its derivatives. Anticancer Res 2017; 37: 5995-6003.
PMID |
| 44. |
Narayanankutty A. PI3K/Akt/mTOR pathway as a therapeutic target for colorectal cancer: a review of preclinical and clinical evidence. Curr Drug Targets 2019; 20:1217-26.
DOI PMID |
| 45. | Lawrence MS, Stojanov P, Mermel CH, et al. Discovery and saturation analysis of cancer genes across 21 tumour types Nature 2014; 505: 495-501. |
| 46. | Temiz TK, Altun A, Turgut N, Balci E. Investigation of the effects of drugs effective on PI3K-AKT signaling pathway in colorectal cancer alone and in combination. Cumhur Dent J. 2014; 36: 167-77. |
| 47. |
Hoxhaj G, Manning BD. The PI3K-AKT network at the interface of oncogenic signalling and cancer metabolism. Nat Rev Cancer 2020; 20: 74-88.
DOI PMID |
| 48. |
Costa RLB, Han HS, Gradishar WJ. Targeting the PI3K/AKT/mTOR pathway in triple-negative breast cancer: a review. Breast Cancer Res Treat 2018; 169: 397-406.
DOI |
| 49. |
Janku F, Yap TA, Meric-Bernstam F. Targeting the Targeting the PI3K pathway in cancer: are we making headway? Nat Rev Clin Oncol 2018; 15: 273-91.
DOI PMID |
| 50. |
Song M, Bode AM, Dong Z, Lee MH. AKT as a Therapeutic target for cancer. Cancer Res 2019; 79: 1019-31.
DOI PMID |
| 51. |
Amirani E, Hallajzadeh J, Asemi Z, Mansournia MA, Yousefi B. Effects of chitosan and oligochitosans on the phosphatidylinositol 3-kinase-AKT pathway in cancer therapy. Int J Biol Macromol 2020; 164: 456-67.
DOI PMID |
| 52. | Sun DD, Zhang F, Qian J, et al. 4'-hydroxywogonin inhibits colorectal cancer angiogenesis by disrupting PI3K/AKT signaling. Chem Biol Interact 2018; 29626-33. |
| 53. | Li QY, Lai ZJ, Yan ZK, et al. Hedyotis diffusa Willd inhibits proliferation and induces apoptosis of 5‑FU resistant colorectal cancer cells by regulating the PI3K/AKT signaling pathway. Mol Med Rep 2018; 17: 358-65. |
| 54. |
Liu L, Gao HW, Wang HB, et al. Catalpol promotes cellular apoptosis in human HCT116 colorectal cancer cells via microRNA-200 and the downregulation of PI3K-Akt signaling pathway. Oncol Lett 2017; 14: 3741-7.
DOI PMID |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

