Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (1): 42-50.DOI: 10.19852/j.cnki.jtcm.20221111.001
• Original articles • Previous Articles Next Articles
Tilianin extracted from Xiangqinglan (Herba Dracocephali Moldovicae) inhibits apoptosis induced by mitochondrial pathway and endoplasmic reticulum stress in H9c2 cells after oxygen-glucose deprivation/reoxygenation
JIANG Wen1, ZHANG Wei2, ZHANG Yuxiang3, YANG Hao1, PAN Xiaomei1, CHEN Qiang1( ), CHEN Junhui1(
), CHEN Junhui1( )
)
- 1 Department of Pharmacy, the Sixth Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, China
2 Department of Anesthesiology, the First Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, China
3 Department of Laboratory, the Sixth Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, China
-
Received:2021-12-01Accepted:2022-02-27Online:2023-02-15Published:2023-01-10 -
Contact:CHEN Qiang,CHEN Junhui -
About author:CHEN Junhui, Department of Pharmacy, the Sixth Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, China. wensmile1987@126.com. Telephone:+86-9912660042; +86-991-2662027
CHEN Qiang, Department of Pharmacy, the Sixth Affiliated Hospital of Xinjiang Medical University, Urumqi 830000, China. cq201007@sina.com.
-
Supported by:Mechanisms of the Protective Effects of Tilianin Against Myocardium Ischemia/Reperfusion Injury Through Ox-CaMKII-mediated Mitochondrial Regulating Pathway(81760045);Study on the Mechanism of Tilianin Anti-atherosclerosis Effect of Tealis via the Reverse Cholesterol Transport Pathway(2018D01C317);Study on the Mechanism of Tilianin Mediate Mitochondrial Function to Regulate Endoplasmic Reticulum Stress and Participate in Anti-myocardial Ischemia/Reperfusion Injury(320.6750.2020-04-32)
Cite this article
JIANG Wen, ZHANG Wei, ZHANG Yuxiang, YANG Hao, PAN Xiaomei, CHEN Qiang, CHEN Junhui. Tilianin extracted from Xiangqinglan (Herba Dracocephali Moldovicae) inhibits apoptosis induced by mitochondrial pathway and endoplasmic reticulum stress in H9c2 cells after oxygen-glucose deprivation/reoxygenation[J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 42-50.
share this article
| Group | n | Survival rate (100%) | ||||
|---|---|---|---|---|---|---|
| 3 h hypoxia/3 h reoxygenation | 6 h hypoxia/3 h reoxygenation | 12 h hypoxia/3hreoxygenation | ||||
| Control | 5 | 100.0±13.0 | 100.0±8.4 | 100.0±2.7 | ||
| OGD/R model | 5 | 92.6±4.0 | 84.1±7.4a | 42.4±3.0a | ||
Table 1 Effects of different hypoxia time on the survival of OGD/R-induced H9C2 cell model ($\bar{x}\pm s$)
| Group | n | Survival rate (100%) | ||||
|---|---|---|---|---|---|---|
| 3 h hypoxia/3 h reoxygenation | 6 h hypoxia/3 h reoxygenation | 12 h hypoxia/3hreoxygenation | ||||
| Control | 5 | 100.0±13.0 | 100.0±8.4 | 100.0±2.7 | ||
| OGD/R model | 5 | 92.6±4.0 | 84.1±7.4a | 42.4±3.0a | ||
| Group | n | LDH activity (U/L) | ||
|---|---|---|---|---|
| 3 h hypoxia/3 h reoxygenation | 6h hypoxia/3 h reoxygenation | 12 h hypoxia/3 hreoxygenation | ||
| Control | 3 | 116±12 | 134±3 | 158±5 |
| OGD/R model | 3 | 129±5 | 185±7a | 227±15a |
Table 2 Effects of different hypoxia time on LDH activity of OGD/R-induced H9C2 cell model ($\bar{x}\pm s$)
| Group | n | LDH activity (U/L) | ||
|---|---|---|---|---|
| 3 h hypoxia/3 h reoxygenation | 6h hypoxia/3 h reoxygenation | 12 h hypoxia/3 hreoxygenation | ||
| Control | 3 | 116±12 | 134±3 | 158±5 |
| OGD/R model | 3 | 129±5 | 185±7a | 227±15a |
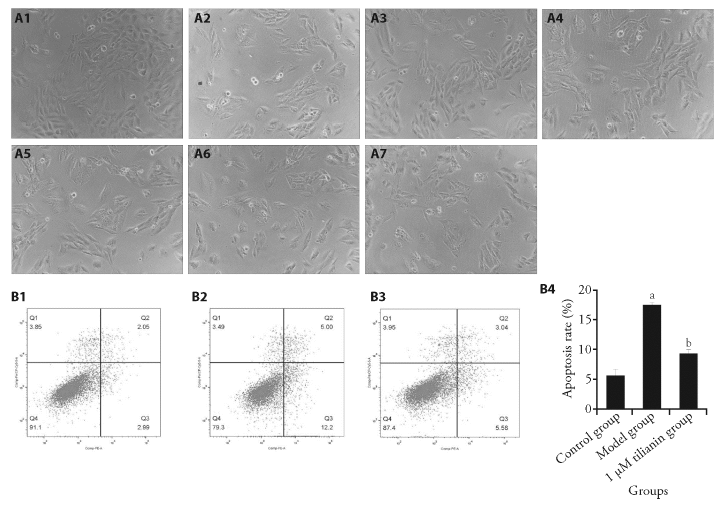
Figure 1 Effect of tilianin on cell morphology and cellular apoptosis A: the cells were intervened with different concentrations of tilianin as indicated. Cell morphology was observed under an inverted microscope (×100). A1: control; A2: model; A3: 1 μM tilianin; A4: 3 μM tilianin; A5: 10 μM tilianin; A6: 20 μM tilianin; A7: 30 μM tilianin. B: the apoptosis rats of the cells of the control, model, and 1 μM tilianin intervention groups, respectively, were detected with flow cytometry. B1-B3: representative flow cytometry results were shown. B1: control group; B2: model group; B3: 1 μM tilianin group; B4: statistical analysis of the apoptosis rates were shown. The H9c2 rat cardiomyocytes were divided into control group: cells were cultured normally without any drug treatment; model group: cells were cultured for 6 h under normal conditions, and cultured for 6 h in serum free and glucose free medium under anoxic conditions; model+tilianin group: cells were cultured under normal culture conditions, pre intervened with tilianin (1, 3, 10, 20 and 30 μM) for 6 h, then cultured with serum free and glucose free hypoxia medium for 6 h (maintaining drug concentration), and then cultured with normal reoxygenation for 3 h (maintaining drug concentration). aP < 0.05, compared with control group; bP < 0.05, compared with model group.
| Group | n | Survival rate (%) | LDH activity (U/L) |
|---|---|---|---|
| Control | 5 | 100±6 | 75±11 |
| OGD/R model | 5 | 80±10a | 136±14a |
| 1 μM tilianin | 5 | 95±12b | 86±10b |
| 3 μM tilianin | 5 | 96±9b | 106±7abc |
| 10 μM tilianin | 5 | 83±5ac | 117±5abc |
| 20 μM tilianin | 5 | 82±6ac | 123±4ac |
| 30 μM tilianin | 5 | 74±5ac | 143±9ac |
Table 3 Effect of tilianin on the survival and LDH activity in OGD/R-induced H9C2 cell model ($\bar{x}\pm s$)
| Group | n | Survival rate (%) | LDH activity (U/L) |
|---|---|---|---|
| Control | 5 | 100±6 | 75±11 |
| OGD/R model | 5 | 80±10a | 136±14a |
| 1 μM tilianin | 5 | 95±12b | 86±10b |
| 3 μM tilianin | 5 | 96±9b | 106±7abc |
| 10 μM tilianin | 5 | 83±5ac | 117±5abc |
| 20 μM tilianin | 5 | 82±6ac | 123±4ac |
| 30 μM tilianin | 5 | 74±5ac | 143±9ac |

Figure 2 Effect of tilianin on mitochondrial functions The effects of tilianin on the mitochondrial membrane potential Rh123 fluorescence intensity (A), Mitosox ROS fluorescence intensity (B), and mitochondrial calcium ion FLuo-3 fluorescence intensity (C) were detected with flow cytometry. A1, B1, C1: control group; A2, B2, C2: model group; A3, B3, C3: 1 μM tilianin group; A4, B4, C4: statistical analysis were shown. The H9c2 rat cardiomyocytes were divided into control group: cells were cultured normally without any drug treatment; model group: cells were cultured for 6 h under normal conditions, and cultured for 6 h in serum free and glucose free medium under anoxic conditions; model + tilianin group: cells were cultured under normal culture conditions, pre intervened with tilianin (1, 3, 10, 20 and 30 μM) for 6 h, then cultured with serum free and glucose free hypoxia medium for 6 h (maintaining drug concentration), and then cultured with normal reoxygenation for 3 h (maintaining drug concentration). ROS: reactive oxygen species. aP < 0.05, compared with control group; bP < 0.05, compared with model group.
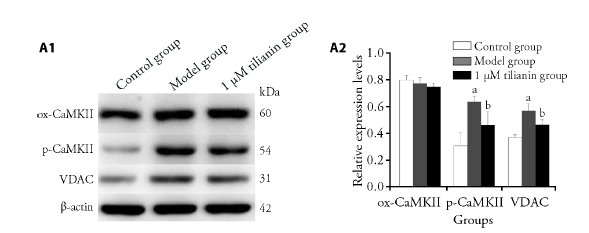
Figure 3 Effects of tilianin on expression levels ox-CaMKII, p-CaMKII, and VDAC A1: representative Western blot results of the protein expression levels were shown. A2: quantitative Western blot result of the protein expression levels were shown. The H9c2 rat cardiomyocytes were divided into control group: cells were cultured normally without any drug treatment; model group: cells were cultured for 6 h under normal conditions, and cultured for 6 h in serum free and glucose free medium under anoxic conditions; model + tilianin group: cells were cultured under normal culture conditions, pre intervened with tilianin (1, 3, 10, 20 and 30 μM) for 6 h, then cultured with serum free and glucose free hypoxia medium for 6 h (maintaining drug concentration), and then cultured with normal reoxygenation for 3 h (maintaining drug concentration). ox-CaMKII: oxidized Calmodulin-dependent protein kinase II; p-CaMKII: p-Calmodulin-dependent protein kinase II; VDAC: voltage-dependent anion selective channel. aP < 0.05, compared with control group; bP < 0.05, compared with model group.
| Group | n | SDH activity (milliunit/μL) | Succinate contents (ng/μL) |
|---|---|---|---|
| Control | 3 | 0.0067±0.0021 | 0.9743±0.4029 |
| OGD/R model | 3 | 0.0113±0.0012a | 2.0843±0.3303a |
| 1 μM tilianin | 3 | 0.0080±0.0017b | 1.3787±0.2779b |
Table 4 Analysis of mitochondrial SDH activity and succinate contents
| Group | n | SDH activity (milliunit/μL) | Succinate contents (ng/μL) |
|---|---|---|---|
| Control | 3 | 0.0067±0.0021 | 0.9743±0.4029 |
| OGD/R model | 3 | 0.0113±0.0012a | 2.0843±0.3303a |
| 1 μM tilianin | 3 | 0.0080±0.0017b | 1.3787±0.2779b |
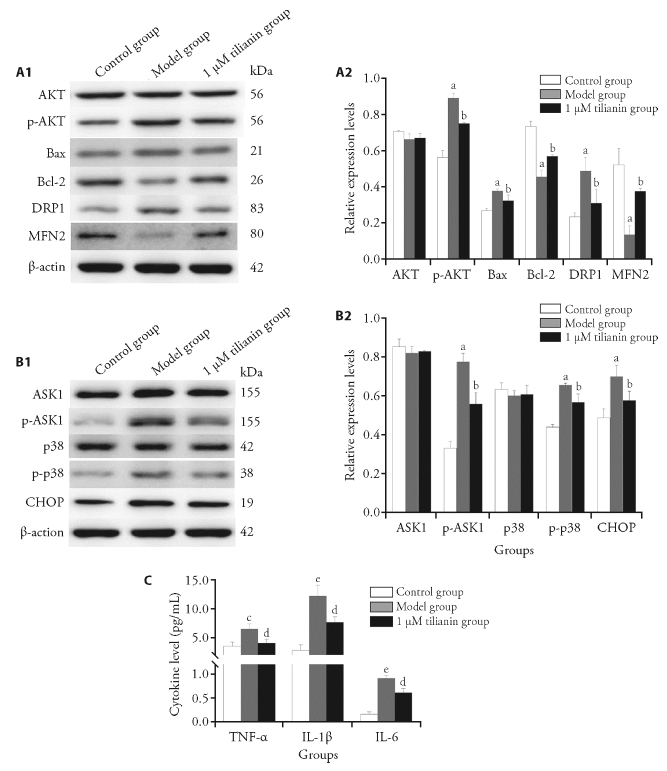
Figure 4 Analysis of expression levels of michondrial pathway-related proteins and endoplasmic reticulum stress-related proteins as well as cytokine levels The expression levels of michondrial pathway-related proteins (A) and endoplasmic reticulum stress-related proteins (B) were detected. C: the levels of TNF-α, IL-1β and IL-6 in the culture supernatant were detected with ELISA. A1, B1: representative Western blot results of the protein expression levels were shown. A2, B2: quantitative Western blot result of the protein expression levels were shown. The H9c2 rat cardiomyocytes were divided into control group: cells were cultured normally without any drug treatment; model group: cells were cultured for 6 h under normal conditions, and cultured for 6 h in serum free and glucose free medium under anoxic conditions; model + tilianin group: cells were cultured under normal culture conditions, pre intervened with tilianin (1, 3, 10, 20 and 30 μM) for 6 h, then cultured with serum free and glucose free hypoxia medium for 6 h (maintaining drug concentration), and then cultured with normal reoxygenation for 3 h (maintaining drug concentration). AKT: protein kinase B; p-AKT: phosphorylated protein kinase B; Bax: B-cell lymphoma-2-associated X; Bcl-2: B-cell lymphoma-2; DRP1: dynamin-related protein 1; MFN2: mitochondrial protein 2; ASK1: apoptosis signal-regulated kinase 1; p-ASK1: phosphorylated-apoptosis signal-regulated kinase 1; p38: mitogen-activated protein kinases; p-p38: phosphorylated-mitogen-activated protein kinases; CHOP: C/EBP homologous protein; TNF- α: tumor necrosis factor- α; IL-1β: interleukin-1β; IL-6: interleukin-6; ELISA: enzyme-linked immunosorbent assay. aP < 0.05, compared with control group; bP < 0.05, compared with model group. cP<0.01, eP < 0.001, compared with control group; dP < 0.01, compared with model group.
| 1 | Gao J, Guo Y, Liu Y, et al. Protective effect of FBXL 10 in myocardial ischemia reperfusion injury via inhibiting endoplasmic reticulum stress. Respir Med 2020; 161: 105852. |
| 2 |
Zhu W, Liu F, Wang L, et al. pPolyHb protects myocardial H9C2 cells against ischemia-reperfusion injury by regulating the pink1-parkin-mediated mitochondrial autophagy pathway. Artif Cells Nanomed Biotechnol 2019; 47: 1248-55.
DOI PMID |
| 3 | Ling H, Gray CB, Zambon AC, et al. Ca2+/calmodulin-dependent protein kinase II δ mediates myocardial ischemia/reperfusion injury through nuclear factor-κB. Circ Res 2013; 112: 935-44. |
| 4 | Abdelwahid E, Stulpinas A, Kalvelyte A. Effective agents targeting the mitochondria and apoptosis to protect the heart. Curr Pharm Des 2017; 23: 1153-66. |
| 5 |
Wu H, Tang Q, Yang J, et al. Atorvastatin ameliorates myocardial ischemia/reperfusion injury through attenuation of endoplasmic reticulum stress-induced apoptosis. Int J Clin Exp Med 2014; 7: 4915-23.
PMID |
| 6 |
Cao L, Chen Y, Zhang Z, et al. Endoplasmic reticulum stress-induced NLRP1 inflammasome activation contributes to myocardial ischemia/reperfusion injury. Shock 2019; 51: 511-8.
DOI PMID |
| 7 | Tan ME, He CH, Jiang W, et al. Development of solid lipid nanoparticles containing total flavonoid extract from Dracocephalum moldavica L. and their therapeutic effect against myocardial ischemia-reperfusion injury in rats. Int J Nanomed 2017; 12: 3253-65. |
| 8 | García-Díaz JA, Navarrete-Vázquez G, García-Jiménez S, et al. Antidiabetic, antihyperlipidemic and anti-inflammatory effects of tilianin in streptozotocin-nicotinamide diabetic rats. Biomed Pharmacother 2016; 83: 667-75. |
| 9 |
Akanda MR, Uddin MN, Kim IS, et al. The biological and pharmacological roles of polyphenol flavonoid tilianin. Eur J Pharmacol 2019; 842: 291-7.
DOI PMID |
| 10 | Zeng C, Jiang W, Zheng R, et al. Cardioprotection of tilianin ameliorates myocardial ischemia-reperfusion injury: role of the apoptotic signaling pathway. PLoS One 2018; 13: e0193845. |
| 11 | De Los Santos S, García-Pérez V, Hernández-Reséndiz S, et al. (-)-Epicatechin induces physiological cardiac growth by activation of the PI3K/AKT pathway in mice. Mol Nutr Food Res 2017; 61: 1600343. |
| 12 | Bell JR, Raaijmakers AJ, Janssens JV, et al. CaMKIIδ and cardiomyocyte Ca (2+) signalling new perspectives on splice variant targeting. Clin Exp Pharmacol Physiol 2015; 42: 1327-32. |
| 13 | Mattiazzi A, Bassani RA, Escobar AL, et al. Chasing cardiac physiology and pathology down the CaMKII cascade. Am J Physiol Heart Circ Physiol 2015; 308: H1177-91. |
| 14 | Joiner ML, Koval OM, Li J, et al. CaMKII determines mitochondrial stress responses in heart. Nature 2012; 491: 269-73. |
| 15 |
Tan W, Colombini M. VDAC closure increases calcium ion flux. Biochim Biophys Acta 2007; 1768: 2510-5.
DOI PMID |
| 16 | Heslop KA, Rovini A, Hunt EG, et al. JNK activation and translocation to mitochondria mediates mitochondrial dysfunction and cell death induced by VDAC opening and sorafenib in hepatocarcinoma cells. Biochem Pharmacol 2020; 171: 113728. |
| 17 | Liao YX, Zhang ZP, Zhao J, Liu JP. Effects of fibronectin 1 on cell proliferation, senescence and apoptosis of human glioma cells through the PI3K/AKT signaling pathway. Cell Physiol Biochem 2018; 48: 1382-96. |
| 18 | Xu L, Jiang X, Wei F, et al. Leonurine protects cardiac function following acute myocardial infarction through anti‑apoptosis by the PI3K/AKT/GSK3β signaling pathway. Mol Med Rep 2018; 18: 1582-90. |
| 19 |
Guan L, Che Z, Meng X, et al. MCU up-regulation contributes to myocardial ischemia-reperfusion injury through calpain/opa-1-mediated mitochondrial fusion/mitophagy inhibition. J Cell Mol Med 2019; 23: 7830-43.
DOI PMID |
| 20 | Bhat TA, Chaudhary AK, Kumar S, et al. Endoplasmic reticulum-mediated unfolded protein response and mitochondrial apoptosis in cancer. Biochim Biophys Acta Rev Cancer 2017; 1867: 58-66. |
| 21 |
Sano R, Reed JC. ER stress-induced cell death mechanisms. Biochim Biophys Acta 2013; 1833: 3460-70.
DOI PMID |
| 22 | Baig MH, Baker A, Ashraf GM, et al. ASK1 and its role in cardiovascular and other disorders: available treatments and future prospects. Expert Rev Proteomics 2019; 16: 857-70. |
| 23 | Matsuzawa A, Nishitoh H, Tobiume K, et al. Physiological roles of ASK1-mediated signal transduction in oxidative stress- and endoplasmic reticulum stress-induced apoptosis: advanced findings from ASK1 knockout mice. Antioxid Redox Signal 2002; 4: 415-25. |
| 24 | Xu W, Zhang L, Zhang Y, et al. TRAF1 exacerbates myocardial ischemia reperfusion injury via ASK1-JNK/p38 signaling. J Am Heart Assoc 2019; 8: e012575. |
| 25 | Zhang R, Xu L, Zhang D et al. Cardioprotection of Ginkgolide B on myocardial ischemia/reperfusion-induced inflammatory injury via regulation of A20-NF-κB pathway. Front Immunol 2018; 9: 2844. |
| 26 | Zhi W, Li K, Wang H, et al. Melatonin elicits protective effects on OGD/R-insulted H9c2 cells by activating PGC-1α/Nrf2 signaling. Int J Mol Med 2020; 45: 1294-304. |
| 27 | Ma L, Liu H, Xie Z, et al. Ginsenoside Rb3 protects cardiomyocytes against ischemia-reperfusion injury via the inhibition of JNK-mediated NF-κB pathway: a mouse cardiomyocyte model. PLoS One 2014; 9: e103628. |
| 28 |
Shen W, Anwaier G, Cao Y, et al. Atheroprotective mechanisms of tilianin by inhibiting inflammation through down-regulating NF-κB pathway and foam cells formation. Front Physiol 2019; 10: 825.
DOI PMID |
| [1] | LI Xi, LIN Xiangquan, CHEN Dongdong, LIU Hui. B-cell lymphoma-2 phosphorylation at Ser70 site-related autophagy mediates puerarin-inhibited the apoptosis of MC3T3-E1 cells during osteoblastogenesis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 27-34. |
| [2] | ZHUANG Xingxing, LIU Tao, WEI Liangbing, SONG Junmei, GAO Jiarong. Qiteng Xiaozhuo granules (芪藤消浊颗粒) medicated serum inhibits excessive proliferation and promotes apoptosis of human glomerular mesangial cells by targeting fat mass and obesity associated proteins [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1072-1080. |
| [3] | SU Chengguo, ZHAO Xiaoyan, YE Jiangnan, ZHANG Xin, JIANG Yuqing, GUO Junjie, ZHANG Xiyuan, QI Wenchuan, ZHU Jun. Effect of Tuina along “bladder meridian” alleviating intervertebral disc degeneration by regulating the transforming growth factor-β1/Smad signaling pathway in a rabbit model [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 991-1000. |
| [4] | ZHU Bin, YU Ning, WANG Lei, TIAN Yue, WU Mingfen, ZHAO Zhigang. Synergistic effect of schizandrin A and DNase I knockdown on high glucose induced beta cell apoptosis by decreasing intracellular calcium concentration [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 661-666. |
| [5] | JIANG Yiqian, ZHOU Xibin, PU Wenyuan, ZHOU Chunxiang. Sanwu Baisan decoction (三物白散) inhibits colorectal cancer progression in mice by remodeling gut microbiota and tumorigenesis [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 466-473. |
| [6] | WANG Tianqi, LI Zining, CHEN Ting, CHEN Rui, JIN Ya, Oduro Patrick kwabena, ZHANG Han, WANG Yi. Zuogui (左归丸) and Yougui pills (右归丸) improve perimenopausal syndrome via regulation of apoptosis in mice [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 474-483. |
| [7] | WU Haiyang, WANG Ying, HAN Wei, LI Huihui, JI Haisheng, LIU Xiuxiu. Protective effect of Tongdu Tiaoshen acupuncture combined with Xiaoxuming decoction (小续命汤) on dopaminergic neurons in Parkinson’s disease model [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 484-493. |
| [8] | XIE Jing, BI Zheng, WANG Sihai, SHEN Guoming, FANG Zhaohui. Danzhi Jiangtang capsule (丹蛭降糖胶囊) reduces renal injury in rats with diabetes induced by high fat diet and streptozotocin via downregulating toll-like receptor 4-nuclear factor-κB pathway and apoptosis [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 312-321. |
| [9] | DING Yajie, LIU Feng, LI Zhaoyan, Xu Yan, CAO Nida, ZHANG Guangao, WANG Rui, ZHAO Aiguang. Efficacy of luteolin on the human gastric cancer cell line MKN45 and underlying mechanism [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 34-41. |
| [10] | LI Han, HUANG Xiaomin, CAI Haiyang, HEROK George, HE Jing, SU Yixun, LI Weihong, YI Chenju, OLIVER Brian G, CHEN Hui. Mitochondrial dysfunction in a rat model and the related risk of metabolic disorders [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 95-104. |
| [11] | YANG Ye, ZHANG Aihui, LI Anmao. Celastrol promotes apoptotic cell death in children neuroblastoma cells through caspases dependent pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 877-884. |
| [12] | ZHENG Wei, WANG Mingxing, LIU Shanxue, LUAN Chao, ZHANG Yanqiu, XU Duoduo, WANG Jian. Buyang Huanwu Tang (补阳还五汤) protects H2O2-induced RGC-5 cell against oxidative stress and apoptosis via reactive oxygen species-mitogen-activated protein kinase signaling pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 885-891. |
| [13] | ZHU Lingyan, WEI Yihong, WANG Youhua, YANG Jianmei, LI Jiawei, CAO Min, ZHOU Duan. Protective efficacy of Shenge San (参蛤散) on mitochondria in H9c2 cardiomyocytes [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 892-899. |
| [14] | HENG Xianpei, LI Liang, YANG Liuqin, WANG Zhita. Efficacy of Dangua Fang (丹瓜方) on endothelial cells damaged by oxidative stress [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 900-907. |
| [15] | YANG Mengzhe, ZHANG Beibei, LIANG Zhenqiang, CHENG Nannan, Lü Anqiao, YANG Jianyu, GUO Xingzhe, BAI Xianyu, HUANG Yuanjiao, JIAO Aijun, XU Ning. Sanguinarine suppresses cell proliferation, migration and invasion in nasopharyngeal carcinoma via inhibiting mTOR signaling [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 687-692. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

