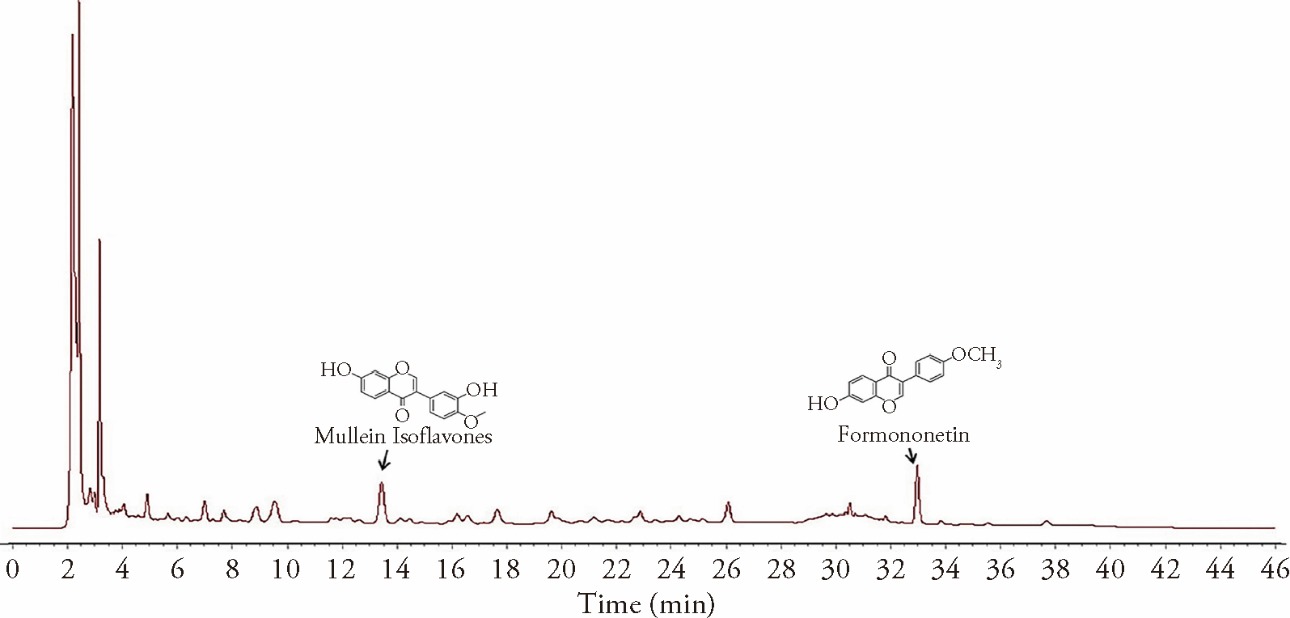
Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (6): 1072-1080.DOI: 10.19852/j.cnki.jtcm.2023.06.005
• Research Articles • Previous Articles Next Articles
Qiteng Xiaozhuo granules (芪藤消浊颗粒) medicated serum inhibits excessive proliferation and promotes apoptosis of human glomerular mesangial cells by targeting fat mass and obesity associated proteins
ZHUANG Xingxing1,2,3, LIU Tao4,5, WEI Liangbing6, SONG Junmei7, GAO Jiarong8,9( )
)
- 1 Department of Pharmacy, the First Affiliated Hospital of Anhui University of Chinese Medicine, Hefei 230012, China
2 Department of Pharmacy, Chaohu Hospital of Anhui Medical University, Chaohu 238000, China
3 School of Pharmacy, Anhui University of Chinese Medicine, Hefei 230013, China
4 Department of Pharmacy, the First Affiliated Hospital of Anhui University of Chinese Medicine, Hefei 230012, Chin
5 School of Pharmacy, Anhui University of Chinese Medicine, Hefei 230013, China
6 Department of Pharmacy, the First Affiliated Hospital of Anhui University of Chinese Medicine, Hefei 230012, China
7 School of Biological and Environmental Engineering, Chaohu University, Chaohu 238000, China
8 Department of Pharmacy, the First Affiliated Hospital of Anhui University of Chinese Medicine, Hefei 230012, China
9 School of Pharmacy, Anhui University of Chinese Medicine, Hefei 230013, China
-
Received:2022-09-23Accepted:2022-12-24Online:2023-10-25Published:2023-09-01 -
Contact:GAO Jiarong, Department of Pharmacy, the First Affiliated Hospital of Anhui University of Chinese Medicine, Hefei 230012, China. zyfygjr2006@163.com. Telephone: +86-551-62838556 -
Supported by:Lnc NONRATG001910.2 Competitively Binds miR-339 to Regulate the Mechanism of Syk Involvement in the Pathogenesis of Chronic Nephritis and the Intervention of Qiteng Xiaozhuo Particles(81973546);Study on the Mechanism of FTO-Mediated FOXO6 mRNA m6A Modification Regulating the Proliferation and Apoptosis of Glomerular Mesangial Cells(2022AH050747);Expression and Target Analysis of miR-155-5p in Kidney Tissue of CGN Rats(XLY-201912)
Cite this article
ZHUANG Xingxing, LIU Tao, WEI Liangbing, SONG Junmei, GAO Jiarong. Qiteng Xiaozhuo granules (芪藤消浊颗粒) medicated serum inhibits excessive proliferation and promotes apoptosis of human glomerular mesangial cells by targeting fat mass and obesity associated proteins[J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1072-1080.
share this article
| Latin name | Chinese name | Medicinal part | Weight (g) |
|---|---|---|---|
| Radix Astragali Mongolici | Huangqi | Root | 6.0 |
| Herba Leonuri Japonici | Yimucao | Herb | 3.0 |
| Semen Coicis | Yiyiren | Seed | 3.0 |
| Herba Hedyotdis | Baihuasheshecao | Herb | 3.0 |
| Rhizoma Imperatae | Baimaogen | Rhizoma | 3.0 |
| Periostracum Cryptotympanae | Chantui | Shell | 2.0 |
| Radix et Rhizoma Tripterygii | Leigongteng | Root | 2.0 |
Table 1 The composition and dosage of QTXZG
| Latin name | Chinese name | Medicinal part | Weight (g) |
|---|---|---|---|
| Radix Astragali Mongolici | Huangqi | Root | 6.0 |
| Herba Leonuri Japonici | Yimucao | Herb | 3.0 |
| Semen Coicis | Yiyiren | Seed | 3.0 |
| Herba Hedyotdis | Baihuasheshecao | Herb | 3.0 |
| Rhizoma Imperatae | Baimaogen | Rhizoma | 3.0 |
| Periostracum Cryptotympanae | Chantui | Shell | 2.0 |
| Radix et Rhizoma Tripterygii | Leigongteng | Root | 2.0 |
| Gene | Amplicon Size (bp) | Forward primer (5'→3') | Reverse primer (5'→3') |
|---|---|---|---|
| FTO | 148 | AGACACCTGGTTTGGCGATA | GTTCCTGTTGAGCACTCTGC |
| PCNA | 169 | AGCCGAAACCAGCTAGACTT | ACCGCTGGAGCTAATATCCC |
| Cyclin D1 | 93 | ATGAACTACCTGGACCGCTT | CTTAGAGGCCACGAACATGC |
| Bax | 103 | GCCTCCTCTCCTACTTTGGG | CCTCAGCCCATCTTCTTCCA |
| Bcl2 | 94 | TCTGCGAATACCGGACTGAA | CAGGAATCCCAACCGGAGAT |
| β-actin | 96 | CCCTGGAGAAGAGCTACGAG | GGAAGGAAGGCTGGAAGAGT |
Table 2 Primers used for the RT-qPCR
| Gene | Amplicon Size (bp) | Forward primer (5'→3') | Reverse primer (5'→3') |
|---|---|---|---|
| FTO | 148 | AGACACCTGGTTTGGCGATA | GTTCCTGTTGAGCACTCTGC |
| PCNA | 169 | AGCCGAAACCAGCTAGACTT | ACCGCTGGAGCTAATATCCC |
| Cyclin D1 | 93 | ATGAACTACCTGGACCGCTT | CTTAGAGGCCACGAACATGC |
| Bax | 103 | GCCTCCTCTCCTACTTTGGG | CCTCAGCCCATCTTCTTCCA |
| Bcl2 | 94 | TCTGCGAATACCGGACTGAA | CAGGAATCCCAACCGGAGAT |
| β-actin | 96 | CCCTGGAGAAGAGCTACGAG | GGAAGGAAGGCTGGAAGAGT |
| Concentration of medicated serum (%) | 24 h | 48 h | 72 h |
|---|---|---|---|
| Blank serum | 1.0000 | 1.0000 | 1.0000 |
| 2.5% QTXZG medicated serum | 0.9787±0.0741 | 0.4603±0.0459 | 0.3743±0.0297 |
| 5% QTXZG medicated serum | 0.8970±0.0670 | 0.8717±0.0216 | 0.7953±0.0181 |
| 10% QTXZG medicated serum | 0.8390±0.0582 | 0.7297±0.0023 | 0.6823±0.0575 |
| 15% QTXZG medicated serum | 0.7970±0.0574 | 0.6617±0.0447 | 0.6007±0.0465 |
| 20% QTXZG medicated serum | 0.7140±0.0335 | 0.6103±0.0180 | 0.5297±0.0064 |
| 40% QTXZG medicated serum | 0.6330±0.0223 | 0.4947±0.0360 | 0.4483±0.0135 |
Table 3 Cell viability detected by CCK8 after intervention with different time and concentration of QTXZG medicated serum ($\bar{x}$ ± s)
| Concentration of medicated serum (%) | 24 h | 48 h | 72 h |
|---|---|---|---|
| Blank serum | 1.0000 | 1.0000 | 1.0000 |
| 2.5% QTXZG medicated serum | 0.9787±0.0741 | 0.4603±0.0459 | 0.3743±0.0297 |
| 5% QTXZG medicated serum | 0.8970±0.0670 | 0.8717±0.0216 | 0.7953±0.0181 |
| 10% QTXZG medicated serum | 0.8390±0.0582 | 0.7297±0.0023 | 0.6823±0.0575 |
| 15% QTXZG medicated serum | 0.7970±0.0574 | 0.6617±0.0447 | 0.6007±0.0465 |
| 20% QTXZG medicated serum | 0.7140±0.0335 | 0.6103±0.0180 | 0.5297±0.0064 |
| 40% QTXZG medicated serum | 0.6330±0.0223 | 0.4947±0.0360 | 0.4483±0.0135 |
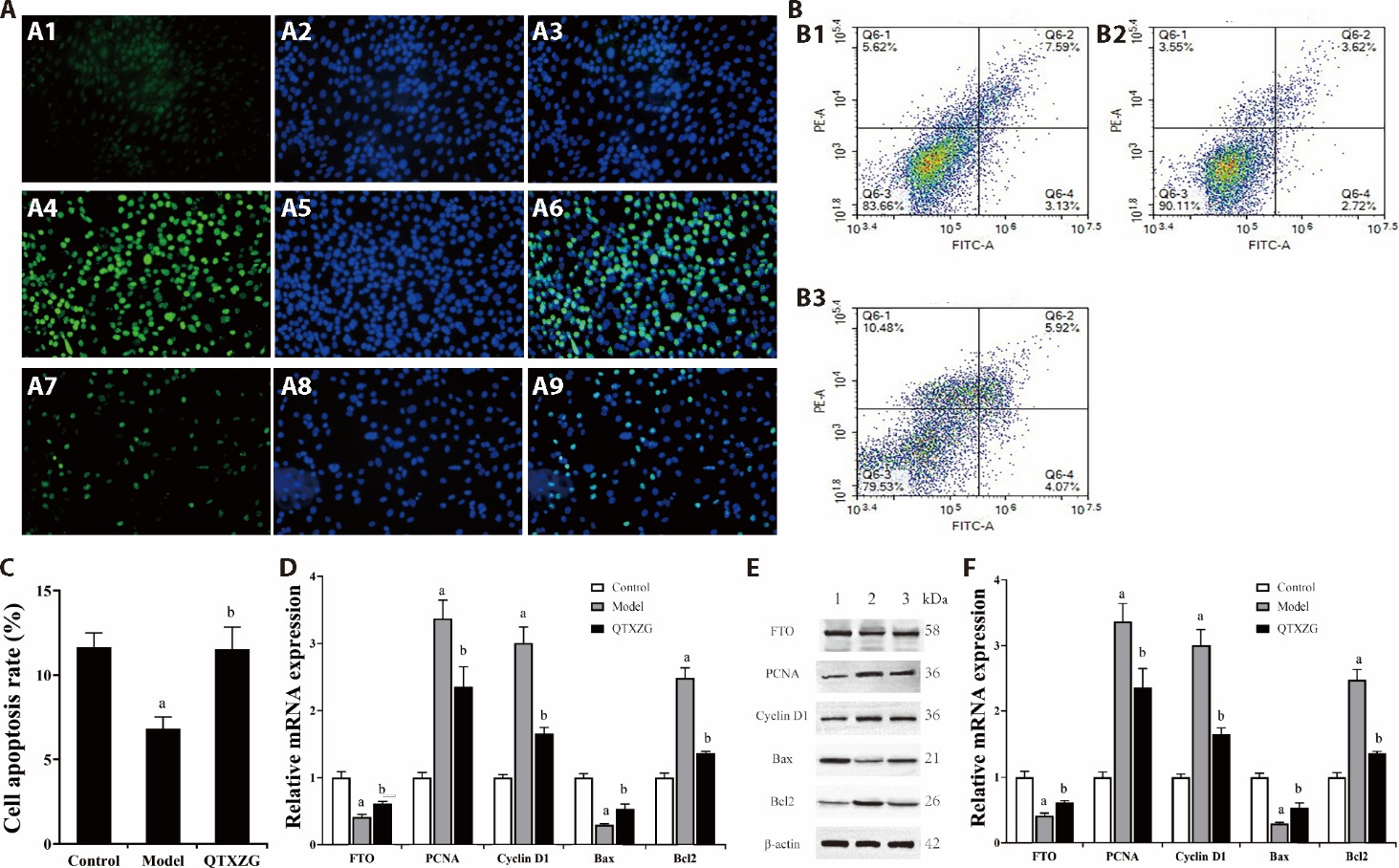
Figure 2 QTXZG medicated serum inhibited excessive proliferation and promoted apoptosis of HGMCs A: representative images for the EdU proliferation assay (× 100 magnification, each sample was repeated three times); the proliferating cells were fluorescently stained with EdU (Green). the nuclei were stained with hoechst 33342 (Blue). the higher the cell proliferation rate, the brighter the green in the plot; A1: EdU of the control group; A2: hoechst of the control group; A3: merge of the control group; A4: EdU of the model group; A5: hoechst of the model group; A6: merge of the model group; A7: EdU of the QTXZG group; A8: hoechst of the QTXZG group; A9: merge of the QTXZG group. B: apoptosis plots of flow cytometry; B1: control group; B2: model group; B3: QTXZG group. C: the rate of cell apoptosis in each group, the ratio of the second quadrant plus the fourth quadrant represents the ratio of cell apoptosis, n = 3. D: the mRNA expression levels of FTO, PCNA, Cyclin D1, Bax, and Bcl2 were measured by RT-qPCR. E: representative blot images of FTO, PCNA, Cyclin D1, Bax, and Bcl2 measured by western blot (1-3: Control, Model, QTXZG). F: protein expression levels of FTO, PCNA, Cyclin D1, Bax, and Bcl2 measured by Western blot. The control group and model group were treated with blank serum, and the QTXZG group were treated with 20% QTXZG medicated serum. QTXZG: Qiteng Xiaozhuo granules; HGMCs: human glomerular mesangial cells; EdU: 5-ethynyl-2′-deoxyuridine; FTO: fat mass and obesity associated proteins; PCNA: proliferating cell nuclear antigen; Bcl2: B-cell lymphoma 2; Bax: BCL2 assaciated X; RT-qPCR: real-time quantitative polymerase chain reaction. The graphs represent the average of 3 individual experiments, and the error bars denote the standard deviation. aP < 0.05 compared to the control group; bP < 0.05 compared to the model group.

Figure 3 FTO overexpression inhibited excessive proliferation and promoted apoptosis of HGMCs A: representative images for the EdU proliferation assay (× 100 magnification, each sample was repeated three times). The proliferating cells were fluorescently stained with EdU (Green). The nuclei were stained with hoechst 33342 (Blue). the higher the cell proliferation rate, the brighter the green in the plot; A1: EdU of the control group; A2: hoechst of the control group; A3: merge of the control group; A4: EdU of the model group; A5: hoechst of the model group; A6: merge of the model group; A7: EdU of the OE + NC group; A8: hoechst of the OE + NC group; A9: merge of the OE + NC group; A10: EdU of the OE + FTO group; A11: hoechst of the OE + FTO group; A12: merge of the OE + FTO group. B: apoptosis plots of flow cytometry; B1: control group; B2: model group; B3: OE + NC group; B4: OE + FTO group. C: the rate of cell apoptosis in each group, the ratio of the second quadrant plus the fourth quadrant represents the ratio of cell apoptosis, n = 3. D: the mRNA expression levels of FTO, PCNA, Cyclin D1, Bax, and Bcl2 were measured by RT-qPCR. E: representative blot images of FTO, PCNA, Cyclin D1, Bax, and Bcl2 measured by Western blot (1-4: Control, Model, OE + NC, OE + FTO). F: protein expression levels of FTO, PCNA, Cyclin D1, Bax, and Bcl2 measured by Western blot (1-4: Control, Model, OE + NC, OE + FTO). The control group and model group were treated with blank serum, the OE + NC group and OE + FTO group were treated with 20% QTXZG medicated serum. HGMCs: human glomerular mesangial cells; EdU: 5-ethynyl-2′-deoxyuridine; FTO: fat mass and obesity associated proteins; PCNA: proliferating cell nuclear antigen; Bcl2: B-cell lymphoma 2; Bax: BCL2 assaciated X; RT-qPCR: real-time quantitative polymerase chain reaction; OE: overexpression; NC: negative control. The graphs represent the average of 3 individual experiments, and the error bars denote the standard deviation. aP < 0.05 compared to the control group; bP < 0.05 compared to the OE + NC group.

Figure 4 FB23-2 can significantly attenuate the effect of QTZXG medicated serum in inhibiting excessive proliferation and promoting apoptosis of HGMCs A: representative images for the EdU proliferation assay (× 100 magnification, each sample was repeated three times). The proliferating cells were fluorescently stained with EdU (Green). The nuclei were stained with hoechst 33342 (Blue). The higher the cell proliferation rate, the brighter the green in the plot; A1: EdU of the control group; A2: hoechst of the control group; A3: merge of the control group; A4: EdU of the model group; A5: hoechst of the model group; A6: merge of the model group; A7: EdU of the QTXZG + FB23-2 group; A8: hoechst of the QTXZG + FB23-2 group; A9: merge of the QTXZG + FB23-2 group; A10: EdU of the QTXZG group; A11: hoechst of the QTXZG group; A12: merge of the QTXZG group. B: apoptosis plots of flow cytometry; B1: control group; B2: model group; B3: QTXZG + FB23-2 group; B4: QTXZG group. C: the rate of cell apoptosis in each group, the ratio of the second quadrant plus the fourth quadrant represents the ratio of cell apoptosis, n = 3. D: the mRNA expression levels of FTO, PCNA, Cyclin D1, Bax, and Bcl2 were measured by RT-qPCR. E: representative blot images of FTO, PCNA, Cyclin D1, Bax, and Bcl2 measured by Western blot (1-4: Control, Model, QTXZG + FB23-2, QTXZG).F: protein expression levels of FTO, PCNA, Cyclin D1, Bax, and Bcl2 measured by Western blot. The control group and model group were treated with blank serum, the QTXZG + FB23-2 group and QTXZG group were treated with 20% QTXZG medicated serum. QTXZG: Qiteng Xiaozhuo granules; HGMCs: human glomerular mesangial cells; EdU: 5-ethynyl-2′-deoxyuridine; FTO: fat mass and obesity associated proteins; PCNA: proliferating cell nuclear antigen; Bcl2: B-cell lymphoma 2; Bax: BCL2 assaciated X; RT-qPCR: real-time quantitative polymerase chain reaction. The graphs represent the average of 3 individual experiments, and the error bars denote the standard deviation. aP < 0.05 compared to the control group; bP < 0.05, compared to the QTXZG + FB23-2 group.
| Group | n | Total m6A (ng) | m6A (%) |
|---|---|---|---|
| Control | 3 | 0.3650±0.0071 | 0.1460±0.0028 |
| Model | 3 | 0.6535±0.0325a | 0.2614±0.0130a |
| QTXZG | 3 | 0.5449±0.0279b | 0.2179±0.0112b |
Table 4 Overall percentage of m6A in RNA changes after the intervention of QTXZG medicated serum ($\bar{x}$ ± s)
| Group | n | Total m6A (ng) | m6A (%) |
|---|---|---|---|
| Control | 3 | 0.3650±0.0071 | 0.1460±0.0028 |
| Model | 3 | 0.6535±0.0325a | 0.2614±0.0130a |
| QTXZG | 3 | 0.5449±0.0279b | 0.2179±0.0112b |
| 1. |
Wei LB, Gao JR, Gao YC, Liu XC, Jiang H, Qin XJ. Effect of the Traditional Chinese Medicine Qiteng Xiaozhuo granules on chronic glomerulonephritis rats studied by using long noncoding RNAs expression profiling. Gene 2020; 728: 144279.
DOI URL |
| 2. |
Gao JR, Jiang NN, Jiang H, et al. Effects of Qiteng Xiaozhuo granules on circRNA expression profiles in rats with chronic glomerulonephritis. Drug Des Devel Ther 2019; 13: 1901-13.
DOI URL |
| 3. |
Gao JR, Qin XJ, Jiang H, Wang T, Song JM, Xu SZ. The effects of Qiteng Xiaozhuo granules, Traditional Chinese Medicine, on the expression of genes in chronic glomerulonephritis rats. J Ethnopharmacol 2016; 193: 140-9.
DOI PMID |
| 4. |
Liu T, Gao YC, Qin XJ, Gao JR. Exploring the mechanism of Jianpi Qushi Huayu formula in the treatment of chronic glomerulonephritis based on network pharmacology. Naunyn Schmiedebergs Arch Pharmacol 2021; 394: 2451-70.
DOI |
| 5. |
Avraham S, Korin B, Chung JJ, Oxburgh L, Shaw AS. The mesangial cell-the glomerular stromal cell. Nat Rev Nephrol 2021; 17: 855-64.
DOI |
| 6. |
Kurogi Y. Mesangial cell proliferation inhibitors for the treatment of proliferative glomerular disease. Med Res Rev 2003; 23: 15-31.
PMID |
| 7. |
Tsuboi N, Utsunomiya Y, Kawamura T, et al. Shedding of growth-suppressive gangliosides from glomerular mesangial cells undergoing apoptosis. Kidney Int 2003; 63: 936-46.
DOI PMID |
| 8. | Scindia YM, Deshmukh US, Bagavant H. Mesangial pathology in glomerular disease: targets for therapeutic intervention. Adv Drug Deliv Rev. 2010; 62: 1337-43. |
| 9. | Wu L, Li O, Zhu F, et al. Krϋppel-like factor 15 suppresses renal glomerular mesangial cell proliferation via enhancing P53 SUMO1 conjugation. J Cell Mol Med 2021; 25: 5691-706. |
| 10. | Shan HJ, Gu WX, Duan G, Chen HL. Fat mass and obesity associated (FTO)-mediated N6-methyladenosine modification of Krüppel-like factor 3 (KLF3) promotes osteosarcoma progression. Bioengineered 2022; 13: 8038-50. |
| 11. | Yang X, Mei C, Ma X, Du J, Wang J, Zan L. m6A methylases regulate myoblast proliferation, apoptosis and differentiation. Animals (Basel) 2022; 12: 773. |
| 12. | Zhuang XX, Zhou L, Miao RH, Ni SD, Li M. Pharmacokinetic interaction between Asari Radix et Rhizoma and Dried Ginger (Zingiber officinalis) in rats. Curr Pharm Analy 2022; 18: 63-70. |
| 13. |
Liu T, Zhuang XX, Qin XJ, Wei LB, Gao JR. Alteration of N6-methyladenosine epitranscriptome profile in lipopolysaccharide-induced mouse mesangial cells. Naunyn Schmiedebergs Arch Pharmacol 2022; 395: 445-58.
DOI |
| 14. |
Lee IT, Shih RH, Lin CC, Chen JT, Yang CM. Role of TLR4/NADPH oxidase/ROS-activated p38 MAPK in VCAM-1 expression induced by lipopolysaccharide in human renal mesangial cells. Cell Commun Signal 2012; 10: 33.
DOI PMID |
| 15. | Chao S, Xu Q, Dong S, Guo M, Liu X, Cheng X. Polygala fallax Hemsl combined with compound Sanqi granules relieves glomerulonephritis by regulating proliferation and apoptosis of glomerular mesangial cells. J Int Med Res 2020; 48: 300060519894124. |
| 16. |
Huang Y, Su R, Sheng Y, et al. Small-molecule targeting of oncogenic FTO demethylase in acute myeloid leukemia. Cancer Cell 2019; 35: 677-91.
DOI PMID |
| 17. |
Jia G, Fu Y, Zhao X, et al. N6-methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nat Chem Biol 2011; 7: 885-7.
DOI PMID |
| 18. |
Zhao X, Yang Y, Sun BF, et al. FTO-dependent demethylation of N6-methyladenosine regulates mRNA splicing and is required for adipogenesis. Cell Res 2014; 24: 1403-19.
DOI PMID |
| 19. |
Niu Y, Lin Z, Wan A, et al. RNA N6-methyladenosine demethylase FTO promotes breast tumor progression through inhibiting BNIP3. Mol Cancer 2019; 18: 46.
DOI PMID |
| 20. |
Wani SM, Gani A, Mir SA, Masoodi FA, Khanday FA. β-Glucan: a dual regulator of apoptosis and cell proliferation. Int J Biol Macromol 2021; 182: 1229-37.
DOI PMID |
| 21. |
Ye K, Wei Q, Gong Z, et al. Effect of norcantharidin on the proliferation, apoptosis, and cell cycle of human mesangial cells. Ren Fail 2017; 39: 458-64.
DOI PMID |
| 22. | Zhang CJ, Zhao D, Yin X, et al. Effects of 1,25(OH)2D3 on proliferation and apoptosis of human glomerular mesangial cells. Am J Transl Res 2016; 8: 2659-66. |
| 23. |
Liang X, Xu W. miR-181a-5p regulates the proliferation and apoptosis of glomerular mesangial cells by targeting KLF6. Exp Ther Med 2020; 20: 1121-8.
DOI PMID |
| [1] | LI Xi, LIN Xiangquan, CHEN Dongdong, LIU Hui. B-cell lymphoma-2 phosphorylation at Ser70 site-related autophagy mediates puerarin-inhibited the apoptosis of MC3T3-E1 cells during osteoblastogenesis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 27-34. |
| [2] | SU Chengguo, ZHAO Xiaoyan, YE Jiangnan, ZHANG Xin, JIANG Yuqing, GUO Junjie, ZHANG Xiyuan, QI Wenchuan, ZHU Jun. Effect of Tuina along “bladder meridian” alleviating intervertebral disc degeneration by regulating the transforming growth factor-β1/Smad signaling pathway in a rabbit model [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 991-1000. |
| [3] | ZHU Bin, YU Ning, WANG Lei, TIAN Yue, WU Mingfen, ZHAO Zhigang. Synergistic effect of schizandrin A and DNase I knockdown on high glucose induced beta cell apoptosis by decreasing intracellular calcium concentration [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 661-666. |
| [4] | JIANG Yiqian, ZHOU Xibin, PU Wenyuan, ZHOU Chunxiang. Sanwu Baisan decoction (三物白散) inhibits colorectal cancer progression in mice by remodeling gut microbiota and tumorigenesis [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 466-473. |
| [5] | WANG Tianqi, LI Zining, CHEN Ting, CHEN Rui, JIN Ya, Oduro Patrick kwabena, ZHANG Han, WANG Yi. Zuogui (左归丸) and Yougui pills (右归丸) improve perimenopausal syndrome via regulation of apoptosis in mice [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 474-483. |
| [6] | XIE Jing, BI Zheng, WANG Sihai, SHEN Guoming, FANG Zhaohui. Danzhi Jiangtang capsule (丹蛭降糖胶囊) reduces renal injury in rats with diabetes induced by high fat diet and streptozotocin via downregulating toll-like receptor 4-nuclear factor-κB pathway and apoptosis [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 312-321. |
| [7] | DING Yajie, LIU Feng, LI Zhaoyan, Xu Yan, CAO Nida, ZHANG Guangao, WANG Rui, ZHAO Aiguang. Efficacy of luteolin on the human gastric cancer cell line MKN45 and underlying mechanism [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 34-41. |
| [8] | JIANG Wen, ZHANG Wei, ZHANG Yuxiang, YANG Hao, PAN Xiaomei, CHEN Qiang, CHEN Junhui. Tilianin extracted from Xiangqinglan (Herba Dracocephali Moldovicae) inhibits apoptosis induced by mitochondrial pathway and endoplasmic reticulum stress in H9c2 cells after oxygen-glucose deprivation/reoxygenation [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 42-50. |
| [9] | YANG Ye, ZHANG Aihui, LI Anmao. Celastrol promotes apoptotic cell death in children neuroblastoma cells through caspases dependent pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 877-884. |
| [10] | ZHENG Wei, WANG Mingxing, LIU Shanxue, LUAN Chao, ZHANG Yanqiu, XU Duoduo, WANG Jian. Buyang Huanwu Tang (补阳还五汤) protects H2O2-induced RGC-5 cell against oxidative stress and apoptosis via reactive oxygen species-mitogen-activated protein kinase signaling pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 885-891. |
| [11] | YANG Mengzhe, ZHANG Beibei, LIANG Zhenqiang, CHENG Nannan, Lü Anqiao, YANG Jianyu, GUO Xingzhe, BAI Xianyu, HUANG Yuanjiao, JIAO Aijun, XU Ning. Sanguinarine suppresses cell proliferation, migration and invasion in nasopharyngeal carcinoma via inhibiting mTOR signaling [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 687-692. |
| [12] | DING Jiamin, XING Yifeng, CHEN Zuoliang, CHEN Wanlu, MA Zhongxiong, XIE Yunde, ZHOU Lin. Qilan preparation (芪蓝颗粒) inhibits proliferation and induces apoptosis by down-regulating microRNA-21 in human Tca8113 tongue squamous cell carcinoma cells [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 693-700. |
| [13] | LI Miao, ZHENG Jialu, WANG Shuangshuang, CHEN Lei, PENG Xiao, CHEN Jinfang, AN Hongmei, HU Bing. Tenglong Buzhong granules (藤龙补中颗粒) inhibits the growth of SW620 human colon cancer in vivo [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 701-706. |
| [14] | YANG Liping, YU Xinglin, ZHANG Chao, CHEN Pu, DUAN Xiaohua. Yajieshaba prevents lipopolysaccharide-induced intestinal barrier injury via anti-inflammatory and anti-apoptosis [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 707-714. |
| [15] | YIN Xiuping, ZHANG Xiaotong, ZHU Rongjia, SONG Ping. Effect of astragaloside IV on the immunoregulatory function of adipose-derived mesenchymal stem cells from patients with psoriasis vulgaris [J]. Journal of Traditional Chinese Medicine, 2022, 42(4): 513-519. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||