Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (2): 268-276.DOI: 10.19852/j.cnki.jtcm.20240203.005
Previous Articles Next Articles
Emodin suppresses alkali burn-induced corneal inflammation and neovascularization by the vascular endothelial growth factor receptor 2 signaling pathway
ZHENG Xueying1, GUO Liang1, LAI Siyi3,4, LI Fengyue1, LIANG Mingli1, LIU Wanting1, MENG Chun2( ), LIU Guanghui3,4(
), LIU Guanghui3,4( )
)
- 1 Department of Bioengineering, College of Biological Science and Biotechnology, Fuzhou University, Fuzhou 350104, China
2 Department of Bioengineering, College of Biological Science and Biotechnology, Fuzhou University, Fuzhou 350104, China; Eye Institute of Integrated Chinese and Western Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou 350004, China
3 Department of Ophthalmology, Affiliated People's Hospital (Fujian Provincial People's Hospital), Fujian University of Traditional Chinese Medicine, Fuzhou 350004, China
4 Eye Institute of Integrated Chinese and Western Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou 350004, China
-
Received:2022-11-22Accepted:2023-04-27Online:2024-04-15Published:2024-02-03 -
Contact:Prof. MENG Chun, Department of Bioengineering, College of Biological Science and Biotechnology, Fuzhou University, Fuzhou 350108, China; Eye Institute of Integrated Chinese and Western Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou 350004, China.mengchun@fzu.edu.cn Telephone: +86-591-83947028; Dr. LIU Guanghui, Department of Ophthalmology, Affiliated People's Hospital (Fujian Provincial People's Hospital), Fujian University of Traditional Chinese Medicine, Fuzhou 350004, China; Eye Institute of Integrated Chinese and Western Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou 350004, China.latiny@gmail.com -
Supported by:Fujian Major Research Grants for Young and Middle-aged Health Professionals(Research and Development of Anti-Keratitis Protein Drug Sgp130)(2021ZQNZD012);National Natural Science Foundation of China(Study on Mechanism of Yijing Decoction in Preventing Microvascular Damage of Early Diabetic Retinopathy based on MMPs/TIMPs)(81774369)
Cite this article
ZHENG Xueying, GUO Liang, LAI Siyi, LI Fengyue, LIANG Mingli, LIU Wanting, MENG Chun, LIU Guanghui. Emodin suppresses alkali burn-induced corneal inflammation and neovascularization by the vascular endothelial growth factor receptor 2 signaling pathway[J]. Journal of Traditional Chinese Medicine, 2024, 44(2): 268-276.
share this article
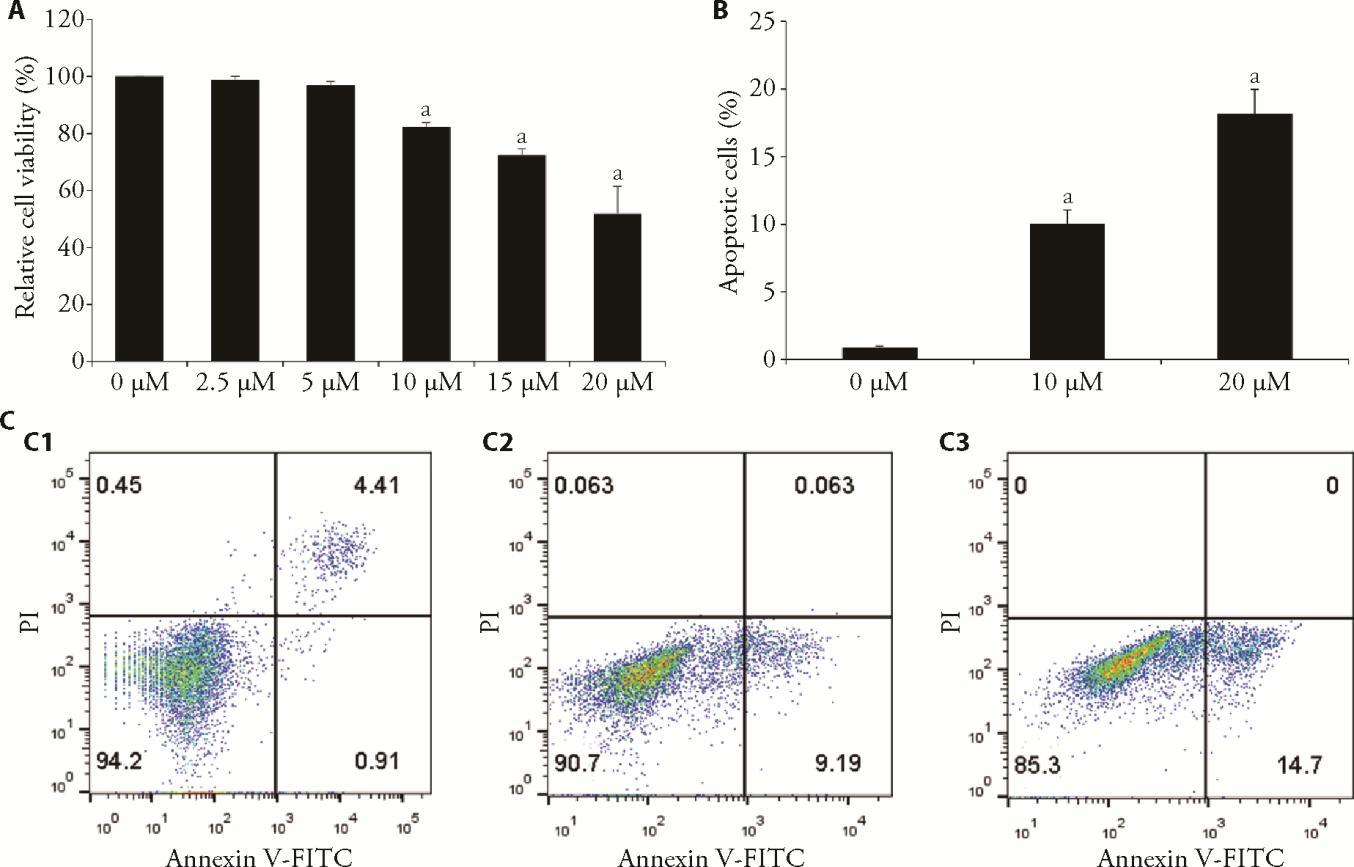
Figure 1 Apoptosis and cell viability with exposure to different doses of emodin were identified using flow cytometry and the CCK-8 assay A: HUVEC were incubated with different concentrations (0-20 μM, 24 h) of emodin, CCK-8 assay was applied to determine cells viability; B: the rate of cell apoptosis in each group, the ratio of the second quadrant plus the fourth quadrant represents the ratio of cell apoptosis; C: apoptosis was assessed by flow cytometry assay. C1: 0 μM emodin group; C2: 10 μM emodin group; C3: 20 μM emodin group. CCK8: cell-counting-kit-8; HUVEC: human umbilical vein endothelial cell. The mean ± standard deviation is indicated by each value (n = 3). aP < 0.05, compared with 0 μM emodin treatment group.

Figure 2 Emodin inhibited migration, invasion, and tube formation of HUVEC A: HUVEC were treated with various concentrations of (0, 10 and 20 μM) emodin 24 h, and the migration of HUVEC was determined by transwell migration assay; B: HUVEC were treated with various concentrations of emodin (0, 10 and 20 μM), and the migration of HUVEC was determined transwell invasion assay; C: HUVEC were treated with various concentrations of emodin (0, 10 and 20 μM), and the migration of HUVEC was determined by matrigel tube-formation assay; D: results of quantitative analysis of HUVEC transwell migration assay. E: results of quantitative analysis of HUVEC transwell invasion assay. F: results of quantitative analysis of HUVEC matrigel tube-formation assay. A1, B1, C1: cells treated with 0 μM emodin 24 h; A2, B2, C2: cells treated with 10 μM emodin 24 h; A3, B3, C3: cells treated with 20 μM emodin 24 h. HUVEC: human umbilical vein endothelial cells. The mean ± standard deviation is indicated by each value (n = 3). aP < 0.01, bP < 0.05, compared with 0 μM emodin group.

Figure 3 In mouse corneas, emodin inhibits alkali burn-induced neovascularization and inflammatory cell infiltration A: slit-lamp microscopy was used to detect CNV on Days 7 and 14; A1: pictures of corneas of mouse 7 d after treatment of corneal alkali burns with PBS eye drops. A2: pictures of corneas of mouse 14 d after treatment of corneal alkali burns with PBS eye drops. A3: pictures of corneas of mouse 7 d after treatment of corneal alkali burns with emodin eye drops. A4: pictures of corneas of mouse 14 d after treatment of corneal alkali burns with emodin eye drops. B: corneal inflammatory cell infiltration in various groups of mouse (hematoxylin-eosin staining, × 100); B1: normal mouse corneas without any treatment; B2: treatment of mouse corneal alkali burns with PBS drops for 14 d; B3: treatment of mouse corneal alkali burns with emodin drops for 14 d; C: statistical analysis of the CNV-covered area at various time points; D: quantification of inflammatory cell infiltration on 14 d. CNV: corneal neovascularization. PBS: phosphate buffered saline. The mean ± standard deviation is indicated by each value (n = 3). aP < 0.05, bP < 0.01, compared with PBS group.

Figure 4 Effect of emodin on the expression of CD31 and VEGFR2 signaling pathway after corneal alkali burns A: expression of CD31 in mouse cornea by immunofluorescence detection; A1: normal mouse corneas without any treatment; A2: treatment of mouse corneal alkali burns with PBS drops for 14 d; A3: treatment of mouse corneal alkali burns with emodin drops for 14 d; B: quantitative analysis of CD31 expression. Control: blank control group mice, without any treatment. PBS: negative control group mice treated with phosphate buffered saline eye drops. Emodin: experimental group mice, treated with emodin eye drops; C: quantitative analysis of VEGFR2, pVEGFR2, pSTAT3, tSTAT3, pPI3K, tPI3K, pAkt, and tAkt; D: representative Western blot of VEGFR2, pVEGFR2, pSTAT3, tSTAT3, pPI3K, tPI3K, pAkt, and tAkt after treatment of VEGF-stimulated HUVEC with PBS and 20 μM emodin for 24 h; E: VEGFR2 expression bands in alkali-burned mouse corneas from the emodin treatment and control groups on 14 d; F: quantitative analysis of VEGFR2 in alkali-burned mouse corneas from the emodin treatment and control groups on 14 d. CD31: platelet endothelial cell adhesion molecule-1; VEGF: vascular endothelial growth factor; VEGFR2: vascular endothelial growth factor receptor 2; p-VEGFR2: phospho-vascular endothelial growth factor receptor 2. STAT3: signal transducer and activator of transcription 3; p-STAT3: phospho-signal transducer and activator of transcription 3; PI3K: phosphoinosmde-3-kinase; p-PI3K: phospho-phosphoinosmde-3-kinase; Akt: total protein kinase B; p-Akt: phospho-protein kinase B. The mean ± standard deviation is indicated by each value (n = 3). aP < 0.05, bP < 0.01, compared with PBS group.
| 1. |
Sharma N, Kaur M, Agarwal T, Sangwan VS, Vajpayee RB. Treatment of acute ocular chemical burns. Surv Ophthalmol 2018; 63: 214-35.
DOI PMID |
| 2. |
Bakunowicz-Łazarczyk A, Urban B. Assessment of therapeutic options for reducing alkali burn-induced corneal neovascularization and inflammation. Adv Med Sci 2016; 61: 101-12.
DOI URL |
| 3. |
Arima T, Uchiyama M, Nakano Y, et al. Peroxisome proliferator-activated receptor alpha agonist suppresses neovascularization by reducing both vascular endothelial growth factor and angiopoietin-2 in corneal alkali burn. Sci Rep 2017; 7: 17763.
DOI PMID |
| 4. |
Peach CJ, Mignone VW, Arruda MA, et al. Molecular pharmacology of VEGF-A isoforms: binding and signalling at VEGFR2. Int J Mol Sci 2018; 19: 1264.
DOI URL |
| 5. |
Claesson-Welsh L, Welsh M. VEGFA and tumour angiogenesis. J Intern Med 2013; 273: 114-27.
DOI PMID |
| 6. |
Dong X, Fu J, Yin X, et al. Emodin: a review of its pharmacology, toxicity and pharmacokinetics. Phytother Res 2016; 30: 1207-18.
DOI PMID |
| 7. |
Xia S, Ni Y, Zhou Q, et al. Emodin attenuates severe acute pancreatitis via antioxidant and anti-inflammatory activity. Inflammation 2019; 42: 2129-38.
DOI |
| 8. |
Du HX, Zhu JQ, Chen J, Zhou HF, Yang JH, Wan HT. Revealing the therapeutic targets and molecular mechanisms of emodin-treated coronavirus disease 2019 via a systematic study of network pharmacology. Aging 2021; 13: 14571-89.
DOI URL |
| 9. |
Ji C, Xin G, Duan F, Huang W, Tan T. Study on the antibacterial activities of emodin derivatives against clinical drug-resistant bacterial strains and their interaction with proteins. Ann Transl Med 2020; 8: 92.
DOI PMID |
| 10. |
Ding Y, Liu P, Chen ZL, et al. Emodin attenuates lipopolysaccharide-induced acute liver injury via inhibiting the tlr 4 signaling pathway in vitro and in vivo. Front Pharmacol 2018; 9: 962.
DOI PMID |
| 11. |
Sun Y, Wang X, Zhou Q, et al. Inhibitory effect of emodin on migration, invasion and metastasis of human breast cancer MDA-MB-231 cells in vitro and in vivo. Oncol Rep 2015; 33: 338-46.
DOI URL |
| 12. |
Dai G, Ding K, Cao Q, et al. Emodin suppresses growth and invasion of colorectal cancer cells by inhibiting VEGFR2. Eur J Pharmacol 2019; 859: 172525.
DOI URL |
| 13. |
Xu K, Li B, Zhang S, et al. DCZ3301, An aryl-guanidino agent, inhibits ocular neovascularization via PI3K/AKT and ERK1/ 2 signaling pathways. Exp Eye Res 2020; 201: 108267.
DOI URL |
| 14. | Herbert SP, Stainier DY. Molecular control of endothelial cell behaviour during blood vessel morphogenesis. Nat Rev Mol Cell Biol 2011; 12: 551-64. |
| 15. |
Cha TL, Chuang MJ, Tang SH, et al. Emodin modulates epigenetic modifications and suppresses bladder carcinoma cell growth. Mol Carcinog 2015; 54: 167-77.
DOI URL |
| 16. |
Jia X, Yu F, Wang J, et al. Emodin suppresses pulmonary metastasis of breast cancer accompanied with decreased macrophage recruitment and M2 polarization in the lungs. Breast Cancer Res Treat 2014; 148: 291-302.
DOI URL |
| 17. |
Jelassi B, Anchelin M, Chamouton J, et al. Anthraquinone emodin inhibits human cancer cell invasiveness by antagonizing P2X7 receptors. Carcinogenesis 2013; 34: 1487-96.
DOI PMID |
| 18. | Chen GL, Zhang JJ, Kao X, Wei LW, Liu ZY. Emodin ameliorates lipopolysaccharides-induced corneal inflammation in rats. Int J Ophthalmol 2015; 8: 665-9. |
| 19. |
Caligiuri G. CD31 as a therapeutic target in atherosclerosis. Circ Res 2020; 126: 1178-89.
DOI PMID |
| 20. |
Jackson JR, Bolognese B, Hillegass L, et al. Pharmacological effects of SB 220025, a selective inhibitor of P38 mitogen-activated protein kinase, in angiogenesis and chronic inflammatory disease models. J Pharmacol Exp Ther 1998; 284: 687-92.
PMID |
| 21. |
Chen WL, Chen YM, Chu HS, et al. Mechanisms controlling the effects of bevacizumab (avastin) on the inhibition of early but not late formed corneal neovascularization. PLoS One 2014; 9: e94205.
DOI URL |
| 22. |
Arulanandam R, Batenchuk C, Angarita FA, et al. VEGF-mediated induction of prd1-bf1/blimp1 expression sensitizes tumor vasculature to oncolytic virus infection. Cancer Cell 2015; 28: 210-24.
DOI PMID |
| 23. |
Kamran MZ, Gude RP. Pentoxifylline inhibits melanoma tumor growth and angiogenesis by targeting STAT3 signaling pathway. Biomed Pharmacother 2013; 67: 399-405.
DOI PMID |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

