Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (5): 723-731.DOI: 10.19852/j.cnki.jtcm.2022.05.006
• Research Articles • Previous Articles Next Articles
Huangqi decoction (黄芪汤) attenuates renal interstitial fibrosis via transforming growth factor-β1/mitogen-activated protein kinase signaling pathways in 5/6 nephrectomy mice
ZHAO Jie1,3, WANG Li3, CAO Ai-li3, WANG Yun-man2, CHI Yang-feng2, WANG Yi2, WANG Hao2,3( ), PENG Wen2,3(
), PENG Wen2,3( )
)
- 1 Department of Chinese Medicine/Department of Chinese Integrative Medicine, The First Affiliated Hospital, Anhui Medical University, Hefei 230022, China
2 Department of Nephrology, Putuo Hospital affiliated to Shanghai University of Chinese Medicine, Shanghai 200062, China
3 Laboratory of Renal Disease, Putuo Hospital, Shanghai University of Chinese Medicine, Shanghai 200062, China
-
Received:2021-07-12Accepted:2021-10-14Online:2022-10-15Published:2022-09-02 -
Contact:WANG Hao,PENG Wen -
About author:Prof. WANG Hao, Department of Nephrology, Laboratory of Renal Disease, Putuo Hospital, Shanghai University of Chinese Medicine, Shanghai 200062, China. wang402hao@163.com
Prof. PENG Wen, Department of Nephrology, Laboratory of Renal Disease, Putuo Hospital, Shanghai University of Chinese Medicine, Shanghai 200062, China. pengwen_01@vip.sina.com;
-
Supported by:National Natural Science Fund(81903994);Youth Development of the First Affiliated Hospital of Anhui Medical University(2793);Budget Project of Shanghai University of Chinese Medicine(2019LK095)
Cite this article
ZHAO Jie, WANG Li, CAO Ai-li, WANG Yun-man, CHI Yang-feng, WANG Yi, WANG Hao, PENG Wen. Huangqi decoction (黄芪汤) attenuates renal interstitial fibrosis via transforming growth factor-β1/mitogen-activated protein kinase signaling pathways in 5/6 nephrectomy mice[J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 723-731.
share this article
| Group | Week 1 | Week 4 | Week 8 | Week 12 | Week 16 |
|---|---|---|---|---|---|
| Sh | 116.9±6.4 | 119.8±6.8 | 125.8±7.6 | 125.3±7.1 | 125.7±2.5 |
| Nx | 112.8±7.0 | 129.9±7.5 | 150.5±2.3a | 151.9±6.5a | 155.0±4.8a |
| En | 110.4±6.4 | 121.4±6.5 | 130.0±5.2b | 121.8±4.0b | 122.9±3.9c |
| Hl | 114.0±5.3 | 139.9±8.9 | 140.8±6.4 | 141.5±7.4 | 147.9±7.1 |
| Hm | 118.3±6.6 | 136.0±10.0 | 143.4±7.9 | 142.6±5.5 | 147.7±3.3 |
| Hh | 115.8±6.8 | 128.3±9.3 | 138.9±10.1 | 142.0±7.2 | 145.3±8.6 |
Table 1 Effect of Huangqi decotion on the blood pressure of the mice (mean, n = 6)
| Group | Week 1 | Week 4 | Week 8 | Week 12 | Week 16 |
|---|---|---|---|---|---|
| Sh | 116.9±6.4 | 119.8±6.8 | 125.8±7.6 | 125.3±7.1 | 125.7±2.5 |
| Nx | 112.8±7.0 | 129.9±7.5 | 150.5±2.3a | 151.9±6.5a | 155.0±4.8a |
| En | 110.4±6.4 | 121.4±6.5 | 130.0±5.2b | 121.8±4.0b | 122.9±3.9c |
| Hl | 114.0±5.3 | 139.9±8.9 | 140.8±6.4 | 141.5±7.4 | 147.9±7.1 |
| Hm | 118.3±6.6 | 136.0±10.0 | 143.4±7.9 | 142.6±5.5 | 147.7±3.3 |
| Hh | 115.8±6.8 | 128.3±9.3 | 138.9±10.1 | 142.0±7.2 | 145.3±8.6 |
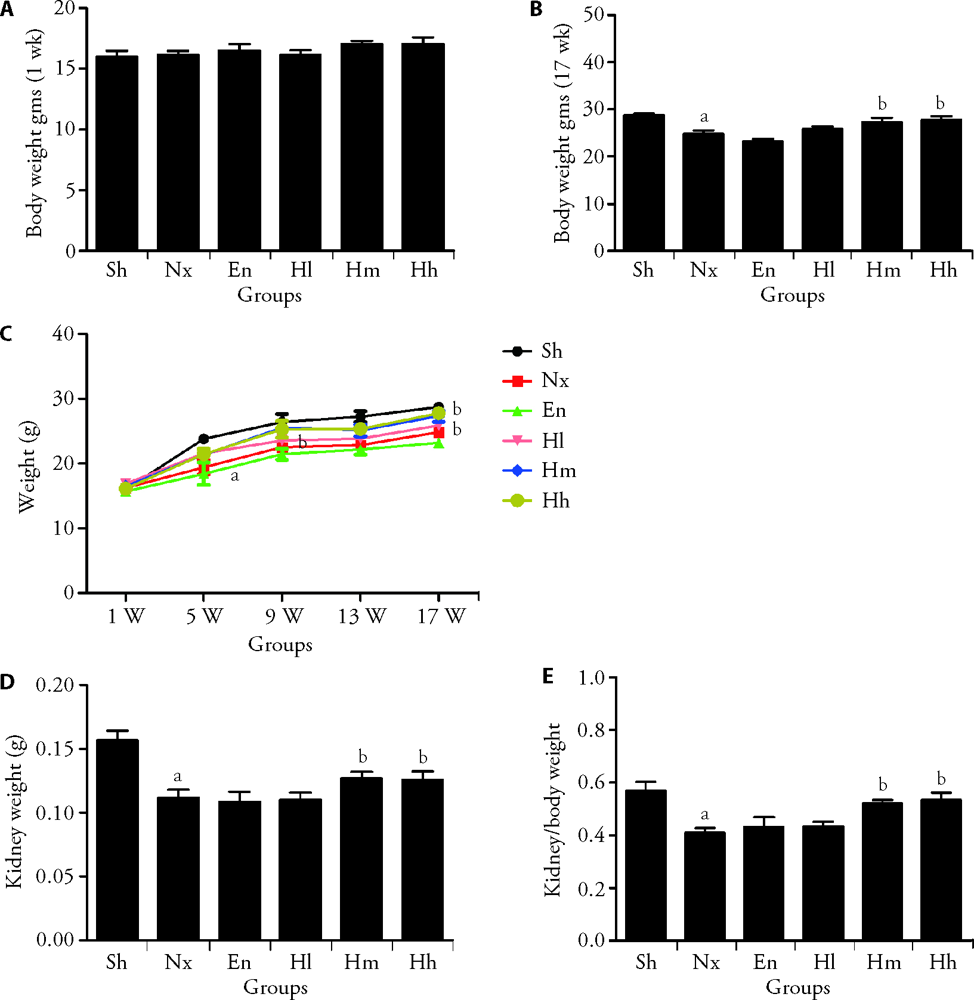
Figure 1 Effect of Huangqi decotion on weight,kidney index and blood pressure changes. A: the body weight during the first week; B: the body weight at the end of week17; C: the body weight during the whole process; D: the weight of the remanent kidney; E: the kidney index of the mice. Sh group was the control group; Nx group was the 5/6 nephrectomy model group; En group was the control group by Enalapri; Hl group was the Low dosing of Huangqi decotion group (Hl, 0.12g /kg, n = 20); Hm group was the Medium dosing of Huangqi decotion group (Hm, 0.36g/kg, n = 20); HH group was the High dosing of Huangqi decotion group (HH, 1.08g/kg, n = 20). Sh: Sham group; En: Enalapri group; Nx: 5/6 nephrectomy model group; Hh: High dosing of Huangqi decotion group; Hm: Medium dosing of Huangqi decotion group; Hl: Low dosing of Huangqi decotion group. aP < 0.05, compared with Sham group; bP < 0.01 or cP < 0.05, compared with Nx group, n = 8-11.
| Group | Week 1 | Week 4 | Week 8 | Week 12 | Week 16 |
|---|---|---|---|---|---|
| Sh | 4.0±0.4 | 3.8±0.4 | 3.5±0.2 | 3.4±0.3 | 3.9±0.3 |
| Nx | 4.0±0.4 | 1.3±0.2a | 1.3±0.2a | 16.9±0.7a | 16.8±0.6a |
| En | 3.9±0.3 | 1.9±0.3 | 1.4±0.1 | 10.6±0.6b | 9.4±0.2c |
| Hl | 4.0±0.4 | 1.0±0.2 | 1.4±0.2 | 17.2±0.7 | 16.4±0.6 |
| Hm | 3.9±0.3 | 1.0±0.1 | 2.5±0.2b | 16.4±0.6 | 15.8±0.5 |
| Hh | 3.9±0.2 | 1.6±0.2 | 2.8±0.2c | 16.8±0.5 | 13.8±0.5b |
Table 2 Effect of Huangqi decotion on 24 h urinary protein (s, n = 6)
| Group | Week 1 | Week 4 | Week 8 | Week 12 | Week 16 |
|---|---|---|---|---|---|
| Sh | 4.0±0.4 | 3.8±0.4 | 3.5±0.2 | 3.4±0.3 | 3.9±0.3 |
| Nx | 4.0±0.4 | 1.3±0.2a | 1.3±0.2a | 16.9±0.7a | 16.8±0.6a |
| En | 3.9±0.3 | 1.9±0.3 | 1.4±0.1 | 10.6±0.6b | 9.4±0.2c |
| Hl | 4.0±0.4 | 1.0±0.2 | 1.4±0.2 | 17.2±0.7 | 16.4±0.6 |
| Hm | 3.9±0.3 | 1.0±0.1 | 2.5±0.2b | 16.4±0.6 | 15.8±0.5 |
| Hh | 3.9±0.2 | 1.6±0.2 | 2.8±0.2c | 16.8±0.5 | 13.8±0.5b |
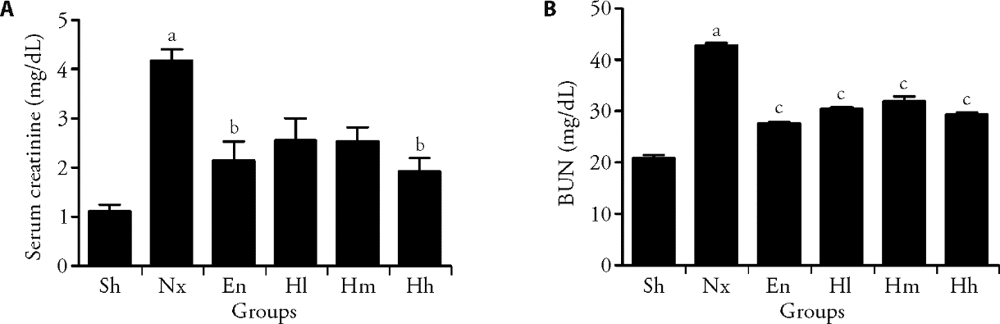
Figure 2 Effect of Huangqi decotion on renal function. A: effect of Huangqi decotion on serum creatinine; B: effect of Huangqi decotion on blood urea nitrogen. Sh group was the control group; Nx group was the 5/6 nephrectomy model group; En group was the control group by Enalapri; Hl group was the Low dosing of Huangqi decotion group (Hl, 0.12g /kg, n = 20); Hm group was the Medium dosing of Huangqi decotion group (Hm, 0.36g/kg, n = 20); HH group was the High dosing of Huangqi decotion group (HH, 1.08g/kg, n = 20). Sh: Sham group; En: Enalapri group; Nx: 5/6 nephrectomy model group; Hh: High dosing of Huangqi decotion group; Hm: Medium dosing of Huangqi decotion group; Hl: Low dosing of Huangqi decotion group. aP < 0.05, compared with Sham group; bP < 0.01 or cP < 0.05, compared with Nx group, n = 8-11.

Figure 3 Huangqi treatment attenuates renal damage in a dosage-dependent manner in Nx mice A: representative PAS-stained kidney sections from Sham, Nx, enalapril, Hl, Hm, Hh groups; B: histopathological scores and inflammatory cells count within the tubulointerstitium from sections similar to those shown in (A); C: representative Masson's modified trichrome stained kidney sections from Sham, Nx, enalapril, Hl, Hm, Hh groups; D: quantitative analysis of the extent of renal interstitial expansion (area %) from sections similar to those shown in. Sh group was the control group; Nx group was the 5/6 nephrectomy model group; En group was the control group by Enalapri; Hl group was the Low dosing of Huangqi decotion group (Hl, 0.12g /kg, n = 20); Hm group was the Medium dosing of Huangqi decotion group (Hm, 0.36g/kg, n = 20); HH group was the High dosing of Huangqi decotion group (HH, 1.08g/kg, n = 20). Sh: Sham group; En: Enalapri group; Nx: 5/6 nephrectomy model group; Hh: High dosing of Huangqi decotion group; Hm: Medium dosing of Huangqi decotion group; Hl: Low dosing of Huangqi decotion group. aP < 0.05, compared with Sham group; bP < 0.01 or cP < 0.05, compared with Nx group, n = 8-11. Magnification: (× 400).
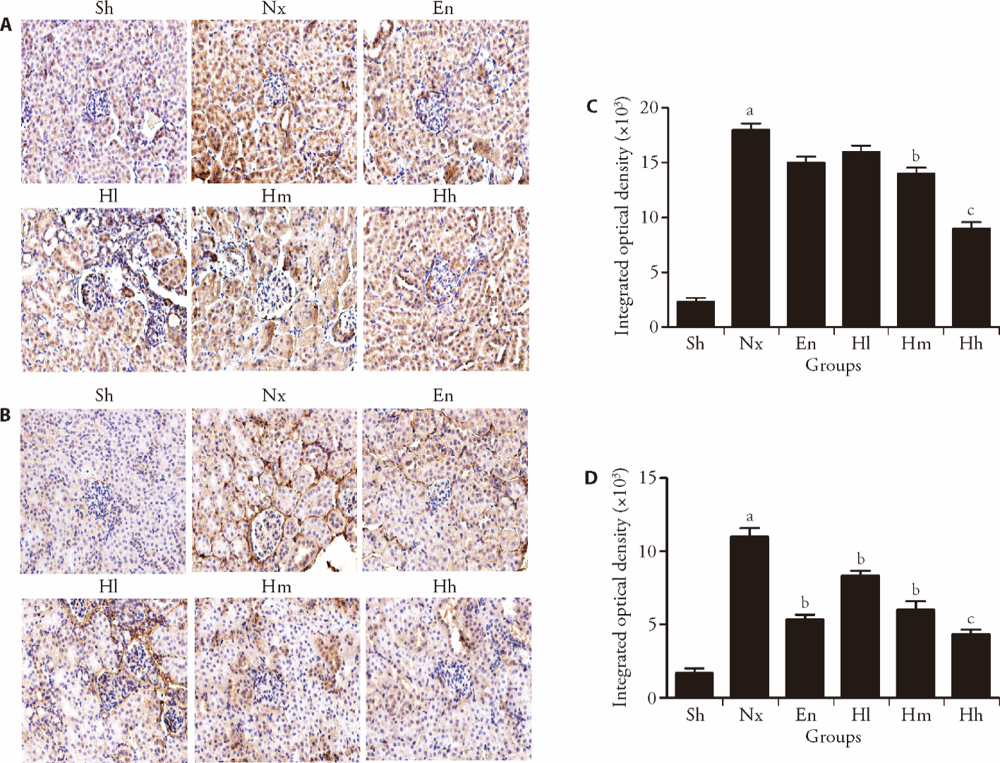
Figure 4 Huangqi decoction decreases the Nx kidneys' CTGF and Collagen III expression by immunohistochemistry A, C: representative photomicrographs of CTGF and Collagen III immunohistochemistry on kidney sections from the Sham, Nx, enalapril, Hl, Hm, Hh groups; B, D: quantitative analysis of CTGF and Collagen III expression from sections similar to those shown in A, C. Sh group was the control group; Nx group was the 5/6 nephrectomy model group; En group was the control group by Enalapri; Hl group was the Low dosing of Huangqi decotion group (Hl, 0.12g /kg, n = 20); Hm group was the Medium dosing of Huangqi decotion group (Hm, 0.36g/kg, n = 20); HH group was the High dosing of Huangqi decotion group (HH, 1.08g/kg, n = 20). Sh: Sham group; En: Enalapri group; Nx: 5/6 nephrectomy model group; Hh: High dosing of Huangqi decotion group; Hm: Medium dosing of Huangqi decotion group; Hl: Low dosing of Huangqi decotion group. aP < 0.05, compared with Sham group; bP < 0.01 or cP < 0.05, compared with Nx group, n = 8-11. Magnification: (× 400).

Figure 5 Huangqi decotion alleviates matrix accumulation in Nx kidneys A: protein expressions of TGF-β1, FSP-1 and α-SMA by Western Blot; B-D: statistical analyses versus A. E: representative photomicrographs of FSP-1by immunohistochemistry on kidney sections from the Sham, Nx, enalapril, Hl, Hm, Hh groups; F: quantitative analysis of FSP-1 expression from sections similar to those shown in E. Sh group was the control group; Nx group was the 5/6 nephrectomy model group; En group was the control group by Enalapri; Hl group was the Low dosing of Huangqi decotion group (Hl, 0.12g /kg, n = 20); Hm group was the Medium dosing of Huangqi decotion group (Hm, 0.36g/kg, n = 20); HH group was the High dosing of Huangqi decotion group (HH, 1.08g/kg, n = 20).Sh: Sham group; En: Enalapri group; Nx: 5/6 nephrectomy model group; Hh: High dosing of Huangqi decotion group; Hm: Medium dosing of Huangqi decotion group; Hl: Low dosing of Huangqi decotion group. aP < 0.05, compared with Sham grou;. bP < 0.01 or cP < 0.05, compared with Nx group, n = 8-11. Magnification: (× 400).
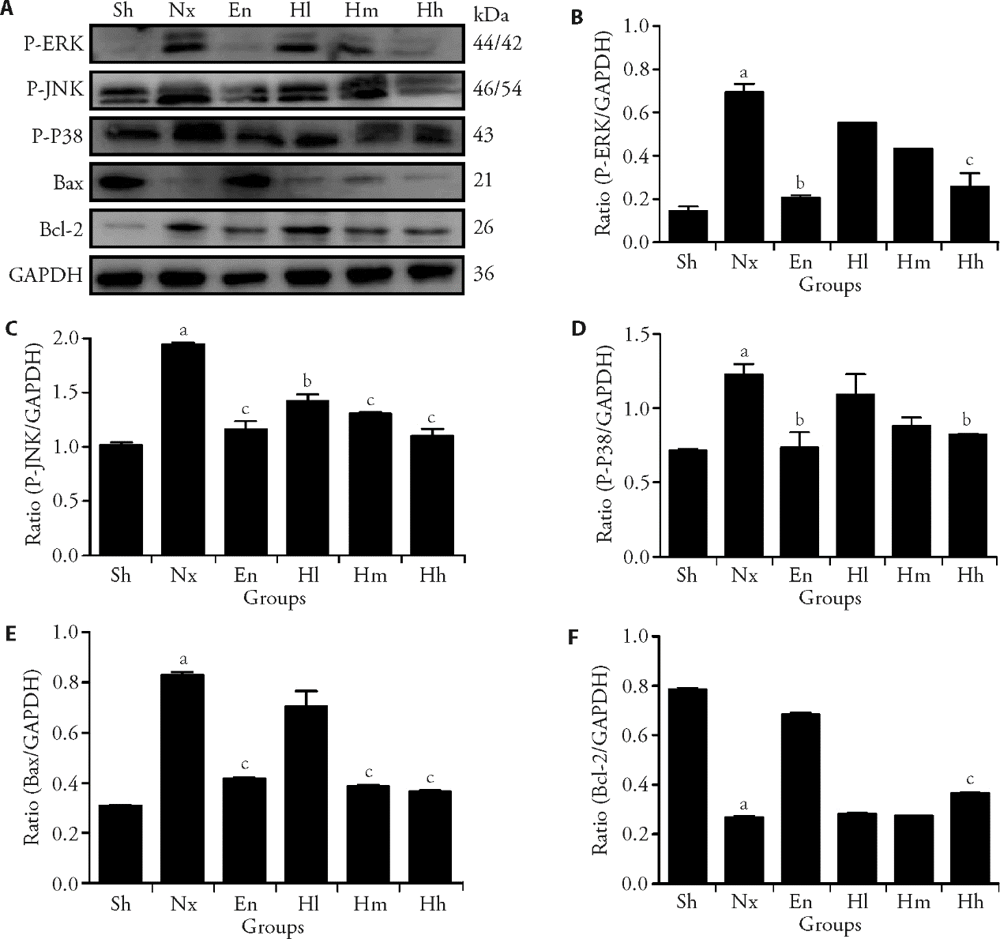
Figure 6 Huangqi decotion downregulats MAPK signaling pathway on kidney sections A: protein expressions of P-P38, P-JNK, P-ERK, Bax and Bcl-2 by Western Blot; B-F: statistical analyses versus A. Sh group was the control group; Nx group was the 5/6 nephrectomy model group; En group was the control group by Enalapri; Hl group was the Low dosing of Huangqi decotion group (Hl, 0.12g /kg, n = 20); Hm group was the Medium dosing of Huangqi decotion group (Hm, 0.36g/kg, n = 20); HH group was the High dosing of Huangqi decotion group (HH, 1.08g/kg, n = 20).Sh: Sham group; En: Enalapri group; Nx: 5/6 nephrectomy model group; Hh: High dosing of Huangqi decotion group; Hm: Medium dosing of Huangqi decotion group; Hl: Low dosing of Huangqi decotion group. aP < 0.05, compared with Sham group. bP < 0.01 or cP < 0.05, compared with Nx group, n = 8-11.
| 1. |
Kawada T. Carotid intima-media thickness and cardiovascular risk in patients with diabetes mellitus type 2 and chronic kidney disease. Ren Fail 2020; 42: 330-3322.
DOI URL |
| 2. |
Liang S, Wu YS, Li DY, et al. Autophagy and renal fibrosis. Aging Dis 2022; 13: 712-31.
DOI |
| 3. |
Lee JW, An JN, Hwang JH, et al. p38 MAPK activity is associated with the histological degree of interstitial fibrosis in IgA nephropathy patients. PLoS One 2019; 14: p. e0213981.
DOI URL |
| 4. |
Shu SS, Liu H, Yang JZ, et al. Targeted inhibition of ZAK ameliorates renal interstitial fibrosis. Transl Res 2022; 246: 49-65.
DOI URL |
| 5. |
Zhang X, Lu H, Xie SS, et al. Resveratrol suppresses the myofibroblastic phenotype and fibrosis formation in kidneys via proliferation-related signalling pathways. Br J Pharmacol 2019; 176: 4745-59.
DOI URL |
| 6. |
Hong WL, Zhang G, Lu H, et al. Epithelial and interstitial Notch 1 activity contributes to the myofibroblastic phenotype and fibrosis. Cell Commun Signal 2019; 12; 17: 145.
DOI URL |
| 7. |
Zhao J, Wang L, Cao AL, et al. Huangqi decoction ameliorates renal fibrosis via tgf-beta/smad signaling pathway in vivo and in vitro. Cell Physiol Biochem 2016; 38: 1761-74.
DOI PMID |
| 8. | Chu S, Mao XD, Wang L, et al. Effects of Huang Qi decoction on endothelial dysfunction induced by homocysteine. Evid Based Complement Alternat Med 2016; 2016: 7272694. |
| 9. |
Zhou X, Sun X, Gong X, et al. Astragaloside IV from Astragalus membranaceus ameliorates renal interstitial fibrosis by inhibiting inflammation via TLR4/NF-small ka, CyrillicB in vivo and in vitro. Int Immunopharmacol 2017; 42: 18-24.
DOI URL |
| 10. |
Che X, Wang Q, Xie Y, et al. Astragaloside IV suppresses transforming growth factor-beta1 induced fibrosis of cultured mouse renal fibroblasts via inhibition of the MAPK and NF-kappaB signaling pathways. Biochem Biophys Res Commun 2015; 464: 1260-6.
DOI URL |
| 11. | Li Z, Deng W, Cao A, et al. Huangqi decoction inhibits hyperglycemia-induced podocyte apoptosis by down-regulated Nox4/p53/Bax signaling in vitro and in vivo. Am J Transl Res 2019; 11: 3195-3212. |
| 12. |
Wang M, Chen DQ, Wang MC, et al. Poricoic acid ZA, a novel RAS inhibitor attracted from Wolfiporia extensa, attenuates tubulo-interstitial fibrosis and podocyte injury by inhibiting TGF-β/Smad signaling pathway. Phytomedicine 2017; 36: 243-253.
DOI URL |
| 13. |
Zhang Y, Zhang D, Zhang M. Inhibition mechanism of compound ethanol extracts from Wuweizi (Fructus Schisandrae Chinensis) on renal interstitial fibrosis in diabetic nephropathy model mice. J Tradit Chin Med 2012; 32: 669-73.
DOI URL |
| 14. | Chen ZJ, Ma F, Sun XM, et al. Renoprotective Effect of a Chinese herbal formula, Qidan Dihuang decoction, on streptozotocin-induced diabetes in rat. Evid Based Complement Alternat Med 2018; 2018: 7321086. |
| 15. | Liu D. and Lv LL. New understanding on the role of proteinuria in progression of chronic kidney disease. Adv Exp Med Biol 2019; 1165: 487-500. |
| 16. | Yang Z, He LJ, Sun SR. Role of endothelial cells in renal fibrosis. Adv Exp Med Biol 2019; 1165: 145-63. |
| 17. |
Qiu ZZ, He JM, Zhang HX, et al. Renoprotective effects of pirfenidone on chronic renal allograft dysfunction by reducing renal interstitial fibrosis in a rat model. Life Sci 2019; 233: 116666.
DOI URL |
| 18. |
de Frutos S, Luengo A, García-Jérez A, et al. Chronic kidney disease induced by an adenine rich diet upregulates integrin linked kinase (ILK) and its depletion prevents the disease progression. Biochim Biophys Acta Mol Basis Dis 2019; 1865: 1284-97.
DOI URL |
| 19. |
Higashi AY, Aronow BJ, Dressler GR. Expression profiling of fibroblasts in chronic and acute disease models reveals novel pathways in kidney fibrosis. J Am Soc Nephrol 2019; 30: 80-94.
DOI PMID |
| 20. |
Liu BC, Tang TT, Lv LL, et al. Renal tubule injury: a driving force toward chronic kidney disease. Kidney Int 2018; 93: 568-79.
DOI URL |
| 21. |
Liu M, Ning X, Li R, et al. Signalling pathways involved in hypoxia-induced renal fibrosis. J Cell Mol Med 2017; 21: 1248-59.
DOI URL |
| 22. | Li ZL, Liu BC. Hypoxia and renal tubulointerstitial fibrosis. Adv Exp Med Biol 2019; 1165: 467-85. |
| 23. |
Vanhove T, Goldschmeding R, Kuypers D. Kidney fibrosis: origins and interventions. Transplantation 2017; 101: 713-26.
DOI PMID |
| 24. |
Suárez-Álvarez B, Liapis H, Anders HJ. Links between coagulation, inflammation, regeneration, and fibrosis in kidney pathology. Lab Invest 2016; 96: 378-90.
DOI PMID |
| 25. | Su H, Wan C, Song A, et al. Oxidative stress and renal fibrosis: mechanisms and therapies. Adv Exp Med Biol 2019; 1165: 585-604. |
| 26. |
Chen Y, Lin L, Tao X, et al. The role of podocyte damage in the etiology of ischemia-reperfusion acute kidney injury and post-injury fibrosis. BMC Nephrol 2019; 20: 106.
DOI PMID |
| 27. |
Chung S, Kim S, Son M, et al. Inhibition of p300/CBP-associated factor attenuates renal tubulointerstitial fibrosis through modulation of NF-kB and Nrf2. Int J Mol Sci 2019; 20: 1554.
DOI URL |
| 28. | Yin Q, Liu H. Connective tissue growth factor and renal fibrosis. Adv Exp Med Biol 2019; 1165: 365-80. |
| 29. |
Gewin LS. Renal fibrosis: primacy of the proximal tubule. Matrix Biol 2018; 68-69: 248-62.
DOI URL |
| 30. |
Cruz-Solbes AS, Youker K. Epithelial to mesenchymal transition (EMT) and endothelial to mesenchymal transition (EndMT): role and implications in kidney fibrosis. Results Probl Cell Differ 2017; 60: 345-72.
DOI PMID |
| 31. |
Annaldas S, Saifi MA, Khurana A, et al. Nimbolide ameliorates unilateral ureteral obstruction-induced renal fibrosis by inhibition of TGF-beta and EMT/Slug signalling. Mol Immunol 2019; 112: 247-55.
DOI URL |
| 32. |
Wang S, Zhou Y, Zhang Y, et al. Roscovitine attenuates renal interstitial fibrosis in diabetic mice through the TGF-beta1/p 38 MAPK pathway. Biomed Pharmacother 2019; 115: 108895.
DOI URL |
| 33. |
Sugiyama N, Kohno M, Yokoyama T. Inhibition of the p38 MAPK pathway ameliorates renal fibrosis in an NPHP2 mouse model. Nephrol Dial Transplant 2012; 27: 1351-8..
DOI URL |
| 34. |
Ghasemian M, Rajabibazl M, Sahebi U, et al. Long non-coding RNA MIR4435-2HG: a key molecule in progression of cancer and non-cancerous disorders. Cancer Cell Int 2022; 22: 215.
DOI URL |
| 35. | Zeng X, Cai G, Liang T, et al. Rhubarb and Astragalus capsule attenuates renal interstitial fibrosis in rats with unilateral ureteral obstruction by alleviating apoptosis through regulating transforming growth factor beta1 (TGF-β1)/p38 mitogen-activated protein kinases (p38 MAPK) pathway. Med Sci Monit 2020; 26: e920720-1-14. |
| 36. |
Lindqvist LM, Heinlein M, Huang DC, et al. Prosurvival Bcl-2 family members affect autophagy only indirectly, by inhibiting Bax and Bak. Proc Natl Acad Sci USA 2014; 111: 8512-17.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

