Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (2): 350-359.DOI: 10.19852/j.cnki.jtcm.2026.02.008
• Original Articles • Previous Articles Next Articles
Identification and verification of key genes related to oxidative stress in type 2 diabetes and screening of candidate drugs from Traditional Chinese Medicine
HU Jingnan1,2, LIAO Man1,2, XI Zhongwen1,2, SONG Jing1,2, WANG Yining1,2, HE Tao3( )
)
- 1
Department of Pharmacy ,Hebei Provincial Hospital of Chinese Medicine Shijiazhuang 050011, China
2Department of Analysis and Testing Center ,Hebei Industrial Technology Institute for Traditional Chinese Medicine Preparation Shijiazhuang 050011, China
3Department of Anesthesiology ,Hebei Provincial Hospital of Chinese Medicine Shijiazhuang 050011, China
-
Received:2025-02-05Accepted:2025-06-06Online:2026-04-15Published:2026-04-04 -
Contact:HE Tao, Department of Anesthesiology, Hebei Provincial Hospital of Chinese Medicine, Shijiazhuang 050011, China. zest2030@163.com; Telephone: +86-311-69095606 -
Supported by:Scientific Research Project of Hebei Administration of Traditional Chinese Medicine: Study on the Pharmacodynamic Material Basis and Mechanism of Cyanotis arachnoidea in the Treatment of Type 2 Diabetes Mellitus(2024009)
Cite this article
HU Jingnan, LIAO Man, XI Zhongwen, SONG Jing, WANG Yining, HE Tao. Identification and verification of key genes related to oxidative stress in type 2 diabetes and screening of candidate drugs from Traditional Chinese Medicine[J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 350-359.
share this article

Figure 1 GO and KEGG enrichment analyses of genes in the turquoise and brown modules A: GO enrichment analysis of turquoise module genes; B: KEGG pathway enrichment analysis of turquoise module genes; C: GO enrichment analysis of brown module genes; D: KEGG pathway enrichment analysis of brown module genes. GO: gene ontology; KEGG: kyoto encyclopedia of genes and genomes.
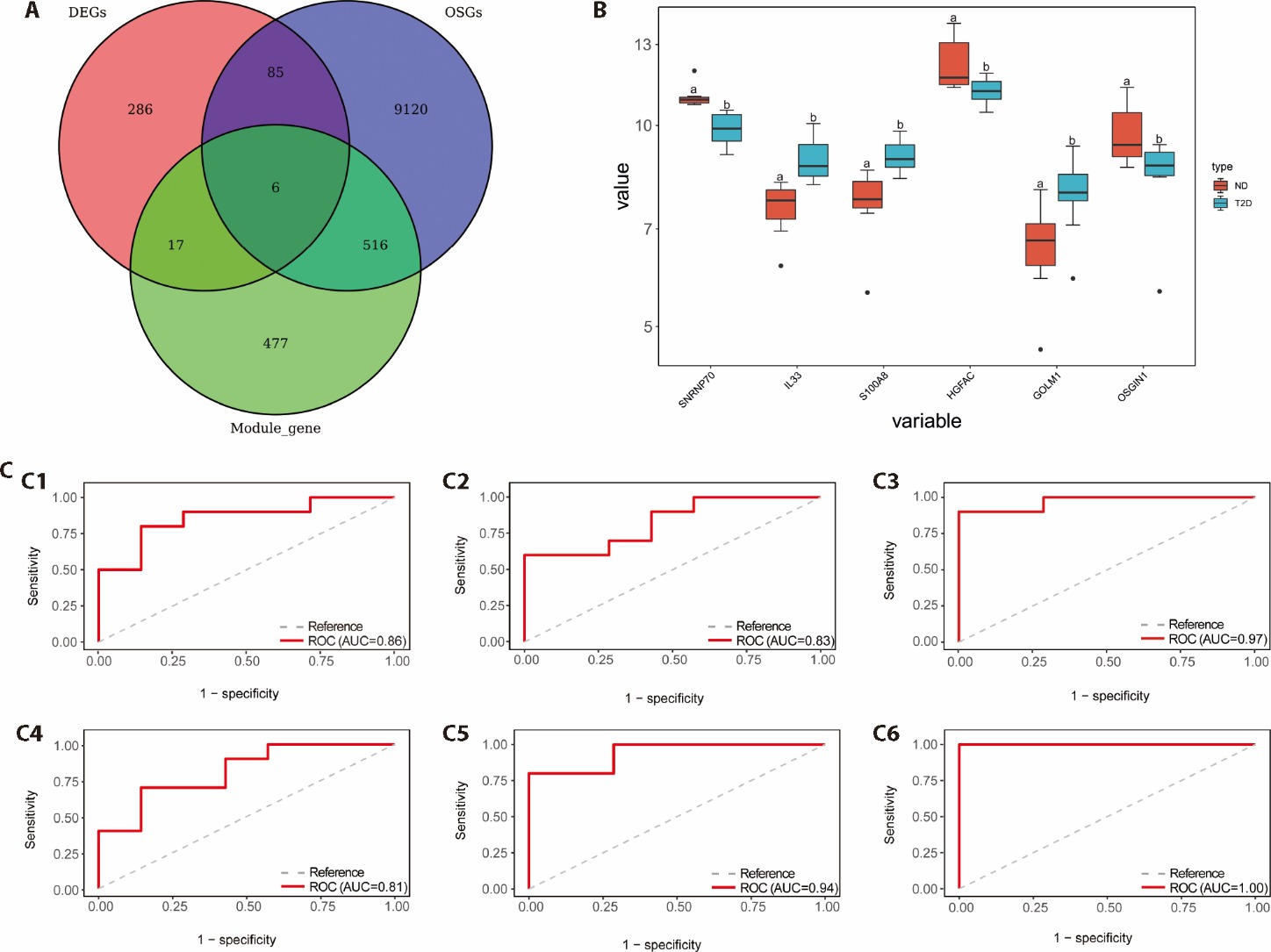
Figure 2 Identification, expression, and diagnostic evaluation of T2DM-related hub genes. A: venn diagram showing the intersection of DEGs, WGCNA module genes, and OS-related genes; B: boxplots illustrating the expression levels of the six hub genes in liver tissues from healthy individuals (ND) and T2DM patients. The differences between groups were compared through the Wilcoxon test; C: ROC curves evaluating the diagnostic performance of the six hub genes; C1: ROC curve of GOLM1; C2: ROC curve of HGFAC; C3: ROC curve of IL33; C4: ROC curve of OSG1N1; C5: ROC curve of S100AB; C6: ROC curve of SNRNP70. DEGs: differentially expressed genes; OSGs: oxidative stress genes; ND: non-diabetes; T2DM: type 2 diabetes mellitus; ROC: receiver operating characteristic. aP < 0.01, bP < 0.05, compared to the ND group.
| Item | Control group (n = 10) | T2DM group (n = 10) |
|---|---|---|
| IL33 | 1.01±0.17 | 2.02±0.10a |
| S100A8 | 1.01±0.16 | 2.50±0.09a |
| GOLM1 | 1.01±0.13 | 1.48±0.07a |
| SNRNP70 | 1.01±0.14 | 0.2 3± 0.03a |
| HGFAC | 1.01±0.13 | 0.35±0.05a |
| OSGIN1 | 1.01±0.16 | 0.94±0.08a |
Table 1 Relative mRNA expression levels of six key genes in the liver tissues of control and T2DM rats ($ \bar{x} \pm s$)
| Item | Control group (n = 10) | T2DM group (n = 10) |
|---|---|---|
| IL33 | 1.01±0.17 | 2.02±0.10a |
| S100A8 | 1.01±0.16 | 2.50±0.09a |
| GOLM1 | 1.01±0.13 | 1.48±0.07a |
| SNRNP70 | 1.01±0.14 | 0.2 3± 0.03a |
| HGFAC | 1.01±0.13 | 0.35±0.05a |
| OSGIN1 | 1.01±0.16 | 0.94±0.08a |
| Item | Control group (n = 10) | T2DM group (n = 10) | T2DM+WED group (n = 10) |
|---|---|---|---|
| FBG | 106.770±3.562 | 162.590±7.020a | 125.370±4.364b |
| FSI | 0.238±0.037 | 1.423±0.115a | 0.540±0.080b |
| GSP | 2.502±0.204 | 8.640±0.210a | 4.045±0.183b |
| HOMA-IR | 0.063±0.011 | 0.570±0.039a | 0.167±0.023b |
Table 2 Effect of Wedelolactone on glucose metabolism and insulin resistance in T2DM rats ($ \bar{x} \pm s$)
| Item | Control group (n = 10) | T2DM group (n = 10) | T2DM+WED group (n = 10) |
|---|---|---|---|
| FBG | 106.770±3.562 | 162.590±7.020a | 125.370±4.364b |
| FSI | 0.238±0.037 | 1.423±0.115a | 0.540±0.080b |
| GSP | 2.502±0.204 | 8.640±0.210a | 4.045±0.183b |
| HOMA-IR | 0.063±0.011 | 0.570±0.039a | 0.167±0.023b |
| Item | Control group (n = 10) | T2DM group (n = 10) | T2DM+WED group (n = 10) |
|---|---|---|---|
| AST | 29.570±8.080 | 130.650±13.689a | 68.879±5.036b |
| ALT | 58.617±6.476 | 185.643±13.449a | 96.032±6.992b |
| ALP | 0.106±0.007 | 0.207±0.007a | 0.140±0.006b |
| LDH | 167.580±32.556 | 367.489±26.839a | 172.511±15.211b |
Table 3 Effect of Wedelolactone on liver function parameters in T2DM rats ($ \bar{x} \pm s$)
| Item | Control group (n = 10) | T2DM group (n = 10) | T2DM+WED group (n = 10) |
|---|---|---|---|
| AST | 29.570±8.080 | 130.650±13.689a | 68.879±5.036b |
| ALT | 58.617±6.476 | 185.643±13.449a | 96.032±6.992b |
| ALP | 0.106±0.007 | 0.207±0.007a | 0.140±0.006b |
| LDH | 167.580±32.556 | 367.489±26.839a | 172.511±15.211b |
| Item | Control group (n = 10) | T2DM group (n = 10) | T2DM + WED group (n = 10) |
|---|---|---|---|
| TG | 1.19±0.14 | 2.76±0.18a | 1.85±0.11b |
| TC | 542.66±80.28 | 1705.43±131.27a | 986.21±88.39b |
| LDL-C | 28.96±0.96 | 45.74±0.81a | 33.17±0.53b |
| HDL-C | 1.17±0.08 | 0.52±0.07a | 0.96±0.06b |
Table 4 Effects of Wedelolactone on serum lipid profiles in T2DM rats ($ \bar{x} \pm s$)
| Item | Control group (n = 10) | T2DM group (n = 10) | T2DM + WED group (n = 10) |
|---|---|---|---|
| TG | 1.19±0.14 | 2.76±0.18a | 1.85±0.11b |
| TC | 542.66±80.28 | 1705.43±131.27a | 986.21±88.39b |
| LDL-C | 28.96±0.96 | 45.74±0.81a | 33.17±0.53b |
| HDL-C | 1.17±0.08 | 0.52±0.07a | 0.96±0.06b |
| Item | Control group (n = 10) | T2DM group (n = 10) | T2DM+WED group (n = 10) |
|---|---|---|---|
| GSH | 8.88±0.56 | 4.94±0.35a | 7.35±0.47b |
| T-SOD | 1.13±0.20 | 0.36±0.10a | 0.78±0.15b |
| MDA | 2.99±0.48 | 5.41±1.25a | 2.47±0.50b |
Table 5 Effect of Wedelolactone on oxidative stress markers in T2DM rats ($ \bar{x} \pm s$)
| Item | Control group (n = 10) | T2DM group (n = 10) | T2DM+WED group (n = 10) |
|---|---|---|---|
| GSH | 8.88±0.56 | 4.94±0.35a | 7.35±0.47b |
| T-SOD | 1.13±0.20 | 0.36±0.10a | 0.78±0.15b |
| MDA | 2.99±0.48 | 5.41±1.25a | 2.47±0.50b |
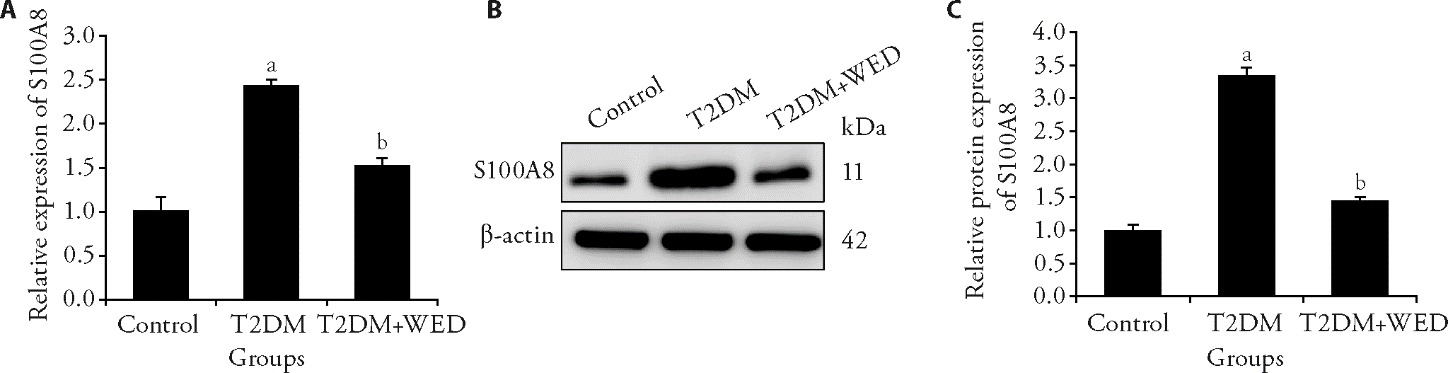
Figure 3 Wedelolactone modulates S100A8 expression in liver tissues of T2DM rats A: relative mRNA expression of S100A8; B: representative protein band of S100A8; C: quantification of S100A8 protein expression. Control group: rats were fed a standard diet and received daily oral gavage of physiological saline from weeks 8 to 12; T2DM group: modeled rats continued on the HFD and received daily oral gavage of physiological saline during weeks 8-12; T2DM + WED group: modeled rats received daily oral gavage of Wedelolactone (50 mg/kg) from weeks 8 to 12, while remaining on the HFD. T2DM: type 2 diabetes mellitus; HFD: high-fat diet; WED: Wedelolactone. Differences in means between groups were examined using one-way analysis of variance. Data were present as mean ± standard deviation (n = 10). aP < 0.01 compared to the Control group; bP < 0.01 compared to the T2DM group.
| 1. | Zhao YY, Xing HC, Wang XM, et al. Management of diabetes mellitus in patients with chronic liver diseases. J Diabetes Res 2019; 2019: 6430486. |
| 2. |
Saeedi P, Petersohn I, Salpea P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res Clin Pract 2019; 157: 107843.
DOI URL |
| 3. | Zhu FY, Dong ZW, Zhu WY, Liu ZQ. Advances in anti-diabetes drugs and their synthesis. Fa Jiao Ke Ji Tong Xun 2016; 45: 175-81. |
| 4. |
Rehman K, Akash MSH. Mechanism of generation of oxidative stress and pathophysiology of type 2 diabetes mellitus: how are they interlinked? J Cell Biochem 2017; 118: 3577-85.
DOI PMID |
| 5. | Banik S, Ghosh A. The association of oxidative stress biomarkers with type 2 diabetes mellitus: a systematic review and Meta-analysis. Health Sci Rep 2021; 4: e389. |
| 6. |
Gjorgjieva M, Mithieux G, Rajas F. Hepatic stress associated with pathologies characterized by disturbed glucose production. Cell Stress 2019; 3: 86-99.
DOI PMID |
| 7. |
Klisic A, Isakovic A, Kocic G, et al. Relationship between oxidative stress, inflammation and dyslipidemia with fatty liver index in patients with type 2 diabetes mellitus. Exp Clin Endocrinol Diabetes 2018; 126: 371-8.
DOI URL |
| 8. | Bigagli E, Lodovici M. Circulating oxidative stress biomarkers in clinical studies on type 2 diabetes and its complications. Oxid Med Cell Longev 2019; 2019: 5953685. |
| 9. |
Ye HW, Wang RX, Wei JJ, Wang Y, Zhang XF, Wang LH. Bioinformatics analysis identifies potential ferroptosis key gene in type 2 diabetic islet dysfunction. Front Endocrinol (Lausanne) 2022; 13: 904312.
DOI URL |
| 10. |
Peng SH, Liu L, Xie ZY, et al. Chinese herbal medicine for type 2 diabetes mellitus with nonalcoholic fatty liver disease: a systematic review and Meta-analysis. Front Pharmacol 2022; 13: 863839.
DOI URL |
| 11. | Wang Y, Dai ZL, Wang Q, et al. Clinical application of Traditional Chinese Medicine therapy for type 2 diabetes mellitus: an evidence map. Evid Based Complement Alternat Med 2022; 2022: 2755332. |
| 12. |
Bhatti JS, Sehrawat A, Mishra J, et al. Oxidative stress in the pathophysiology of type 2 diabetes and related complications: current therapeutics strategies and future perspectives. Free Radic Biol Med 2022; 184: 114-34.
DOI URL |
| 13. |
Nowotny K, Jung T, Hohn A, Weber D, Grune T. Advanced glycation end products and oxidative stress in type 2 diabetes mellitus. Biomolecules 2015; 5: 194-222.
DOI PMID |
| 14. |
Bugianesi E, McCullough AJ, Marchesini G. Insulin resistance: a metabolic pathway to chronic liver disease. Hepatology 2005; 42: 987-1000.
DOI PMID |
| 15. |
Aleksunes LM, Manautou JE. Emerging role of Nrf 2 in protecting against hepatic and gastrointestinal disease. Toxicol Pathol 2007; 35: 459-73.
DOI PMID |
| 16. | Hurtado-Carneiro V, Dongil P, Perez-Garcia A, Alvarez E, Sanz C. Preventing oxidative stress in the liver: an opportunity for GLP-1 and/or PASK. Antioxidants (Basel) 2021; 10: 2028. |
| 17. |
Chupradit S, Bokov D, Zamanian MY, Heidari M, Hakimizadeh E. Hepatoprotective and therapeutic effects of resveratrol: a focus on anti-inflammatory and antioxidative activities. Fundam Clin Pharmacol 2022; 36: 468-85.
DOI URL |
| 18. |
Mukai E, Fujimoto S, Inagaki N. Role of reactive oxygen species in glucose metabolism disorder in diabetic pancreatic beta-cells. Biomolecules 2022; 12: 1228.
DOI URL |
| 19. |
Mohamed J, Nazratun Nafizah AH, Zariyantey AH, Budin SB. Mechanisms of diabetes-iInduced liver damage: the role of oxidative stress and inflammation. Sultan Qaboos Univ Med J. 2016; 16: e132-41.
DOI URL |
| 20. |
Haythorne E, Lloyd M, Walsby-Tickle J, et al. Altered glycolysis triggers impaired mitochondrial metabolism and mTORC 1 activation in diabetic beta-cells. Nat Commun 2022; 13: 6754.
DOI |
| 21. |
Bogan JS. Ubiquitin-like processing of TUG proteins as a mechanism to regulate glucose uptake and energy metabolism in fat and muscle. Front Endocrinol (Lausanne) 2022; 13: 1019405.
DOI URL |
| 22. |
Verma G, Bowen A, Gheibi S, et al. Ribosomal biogenesis regulator DIMT1 controls beta-cell protein synthesis, mitochondrial function, and insulin secretion. J Biol Chem 2022; 298: 101692.
DOI URL |
| 23. |
Boodhoo K, Vlok M, Tabb DL, Myburgh KH, van de Vyver M. Dysregulated healing responses in diabetic wounds occur in the early stages postinjury. J Mol Endocrinol 2021; 66: 141-55.
DOI PMID |
| 24. | Tonacci A, Quattrocchi P, Gangemi S. IL33/ST 2 axis in diabetic kidney disease: a literature review. Medicina (Kaunas) 2019; 55: 50. |
| 25. |
Lu JL, Liang Y, Zhao JJ, Meng HY, Zhang XJ. Interleukin-33 prevents the development of autoimmune diabetes in NOD mice. Int Immunopharmacol 2019; 70: 9-15.
DOI PMID |
| 26. |
Lin J, Lan Y, Xiang DC, et al. IL-33 promotes pancreatic beta-cell survival and insulin secretion under diabetogenic conditions through PPARgamma. Eur J Pharmacol 2023; 959: 176059.
DOI URL |
| 27. |
Yi XM, Lian H, Li S. Signaling and functions of interleukin-33 in immune regulation and diseases. Cell Insight 2022; 1: 100042.
DOI URL |
| 28. |
De Jesus A, Keyhani-Nejad F, Pusec CM, et al. Hexokinase 1 cellular localization regulates the metabolic fate of glucose. Mol Cell 2022; 82: 1261-77 e9.
DOI PMID |
| 29. |
Rodrigues RM, He Y, Hwang S, et al. E-selectin-dependent inflammation and lipolysis in adipose tissue exacerbate steatosis-to-NASH progression via S100A8/9. Cell Mol Gastroenterol Hepatol 2022; 13: 151-71.
DOI URL |
| 30. |
Fang XY, Miao RY, Wei JH, Wu HR, Tian JX. Advances in multi-omics study of biomarkers of glycolipid metabolism disorder. Comput Struct Biotechnol J 2022; 20: 5935-51.
DOI URL |
| 31. | Zheng KI, Liu WY, Pan XY, et al. Combined and sequential non-invasive approach to diagnosing non-alcoholic steatohepatitis in patients with non-alcoholic fatty liver disease and persistently normal alanine aminotransferase levels. BMJ Open Diabetes Res Care 2020; 8: e001174. |
| 32. |
Daryabor G, Atashzar MR, Kabelitz D, Meri S, Kalantar K. The effects of type 2 diabetes mellitus on organ metabolism and the immune system. Front Immunol 2020; 11: 1582.
DOI PMID |
| 33. |
Yan S, Lu W, Zhou J, et al. Aqueous extract of Scrophularia ningpoensis improves insulin sensitivity through AMPK-mediated inhibition of the NLRP3 inflammasome. Phytomedicine 2022; 104: 154308.
DOI URL |
| 34. |
Yuan HL, Ouyang S, Yang RN, et al. Osthole alleviated diabetic neuropathic pain mediated by the P2X(4) receptor in dorsal root ganglia. Brain Res Bull 2018; 142: 289-96.
DOI URL |
| 35. |
Lee D, Lee DH, Choi S, Lee JS, Jang DS, Kang KS. Identification and isolation of active compounds from astragalus membranaceus that improve insulin secretion by regulating pancreatic beta-cell metabolism. Biomolecules 2019 ; 9: 618.
DOI URL |
| 36. |
Wang P, Liu Y, Zhang T, et al. Effects of root extract of morinda officinalis in mice with high-fat-diet/streptozotocin-induced diabetes and C2C12 myoblast differentiation. ACS Omega 2021; 6: 26959-68.
DOI URL |
| [1] | ZHENG Yanfeng, ZHANG Xinjiang, ZHANG Xiaomeng, LI Xiangji, XIN Chen, KONG Jingwei, WANG Xin, SUN Lan, RONG Peijing. Transcutaneous auricular vagus nerve stimulation improves emotional and cognitive functions in post-traumatic stress disorder rats through anti-inflammation, neuroprotection, and modulation of the hippocampal nuclear factor erythroid 2-related factor 2-heme oxygenase-1-glutathione peroxidase 4 pathway [J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 326-338. |
| [2] | NI Shuang, LIU Xiaofei, GUO Xiaoyan, GU Zuxi, WU Panqing, CONG Chao, LI Shengnan, GAO Xianwei, XU Lianwei. Mechanism of Tiaogeng decoction (调更汤) in a cognitive dysfunction mouse model [J]. Journal of Traditional Chinese Medicine, 2025, 45(5): 987-997. |
| [3] | LIU Xiaoyao, LI Jialin, WANG Weiling, FAN Qiongyin, SU Zeqi, HE Cheng, WANG Chunguo, GAO Jian, WANG Ting. Pharmacological effect and possible mechanism of Mudan Huaban recipe (牡丹化斑方) on melasma in mice induced by ultraviolet B and progesterone [J]. Journal of Traditional Chinese Medicine, 2025, 45(3): 518-527. |
| [4] | XU Bojun, TAO Tian, ZHAO Liangbin, ZHENG Hui, ZHAN huakui, GUO Julan. Bushen Tongluo recipe (补肾通络方) improves oxidative stress homeostasis, inhibits transforming growth factor/Notch signaling pathway, and regulates the lncRNA maternally expressed gene 3/miR-145 axis to delay diabetic kidney disease [J]. Journal of Traditional Chinese Medicine, 2025, 45(3): 561-570. |
| [5] | FENG Chuwen, LI Chaoran, Yang Yan, QU Yuanyuan, SUN Zhongren, SUN Weibo, LIU Tingting, LI Shulin, Yang Tiansong. Identifying potential biomarkers in the hippocampus of chronic fatigue syndrome rats treated with moxibustion at Zusanli (ST36): a proteomics study [J]. Journal of Traditional Chinese Medicine, 2025, 45(3): 571-585. |
| [6] | ZHENG Peng, MENG Ying, LIU Meijun, YU Di, LIU Huiying, WANG Fuchun, XU Xiaohong. Electroacupuncture inhibits hippocampal oxidative stress and autophagy in sleep-deprived rats through the protein kinase B and mechanistic target of rapamycin signaling pathway [J]. Journal of Traditional Chinese Medicine, 2024, 44(5): 974-980. |
| [7] | Zubaria Tul Ain, Iram Fatima, Sana Naseer, Sobia Kanwal, Tariq Mahmood. Assessment of phytochemicals, antioxidant, anti-hemolytic, anti-inflammatory and anti-cancer potential of flowers, leaves and stem extracts of Rosa arvensis [J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 804-812. |
| [8] | JIN Xiao, WU Bingxin, LIN Miaoyang, ZHONG Biying, LIN Luoqi, XU Danping. Clinical efficacy and gene chip expression analysis of Shenzhu Guanxin recipe granules (参术冠心方颗粒) in patients with intermediate coronary lesions [J]. Journal of Traditional Chinese Medicine, 2024, 44(3): 545-553. |
| [9] | HUANG Hongmei, YANG Maojun, LI Ting, WANG Dandan, LI Ying, TANG Xiaochi, YUAN Lu, GU Shi, XU Yong. Neferine inhibits the progression of diabetic nephropathy by modulating the miR-17-5p/nuclear factor E2-related factor 2 axis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 44-53. |
| [10] | ZHANG Xiaoying, WANG Ruixuan, WANG Yiqing, XU Fanxing, YAN Tingxu, WU Bo, ZHANG Ming, JIA Ying. Spinosin protects Neuro-2a/APP695 cells from oxidative stress damage by inactivating p38 [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 868-875. |
| [11] | LIU Bingbing, LI Jieru, SI Jianchao, CHEN Qi, YANG Shengchang, JI Ensheng. Ginsenoside Rb1 alleviates chronic intermittent hypoxia-induced diabetic cardiomyopathy in db/db mice by regulating the adenosine monophosphate-activated protein kinase/Nrf2/heme oxygenase-1 signaling pathway [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 906-914. |
| [12] | ZHOU Hua, LI Hui, WANG Haihua. Potential protective effects of the water-soluble Chinese propolis on experimental ulcerative colitis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 925-933. |
| [13] | HAN Rui, CHANG Junzhao, LIU Qianqian, LIU Haitao, LI Junwei. Efficacy of electroacupunture at Zusanli (ST36) on jumping-injured muscle based on transcriptome sequencing and genes analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 322-328. |
| [14] | WANG Yide, GAO Zhen. Yinyanghuo (Herba Epimedii Brevicornus) and its components for chronic obstructive pulmonary disease: preclinical evidence and possible mechanisms [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 386-396. |
| [15] | HENG Xianpei, LI Liang, YANG Liuqin, WANG Zhita. Efficacy of Dangua Fang (丹瓜方) on endothelial cells damaged by oxidative stress [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 900-907. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

