Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (1): 95-109.DOI: 10.19852/j.cnki.jtcm.2026.01.009
• Original Articles • Previous Articles Next Articles
Weimishu prescription (胃糜舒方) alleviates liver-stomach disharmony and chronic gastritis by reducing inflammation and regulating gastric stem cell differentiation
ZOU Xiaoyun1,2, WEI Minmin3, JIA Shouning4, QI Yongfu4, LI Hailing5, LIU Yan1, SHI Xiujuan1, LI Junru6( )
)
- 1 Medical colledge, Qinghai University, Xining 810016, China
2 Department of Hepatopathy, Qinghai Provincial Hospital of Traditional Chinese Medicine, Xining 810000, China
3 Department of Cardiology, Qinghai Provincial Hospital of Traditional Chinese Medicine, Xining 810000, China
4 Department of Reserch Institute of Traditional Chinese Medicine, Qinghai Provincial Hospital of Traditional Chinese Medicine, Xining 810000, China
5 Department of Pharmacy, Qinghai Provincial Hospital of Traditional Chinese Medicine, Xining 810000, China
6 Department of Spleen and Stomach Diseases, Qinghai Provincial Hospital of Traditional Chinese Medicine, Xining 810000, China
-
Received:2025-04-14Accepted:2025-11-20Online:2026-02-15Published:2026-01-28 -
Contact:Pro. LI Junru, Department of Spleen and Stomach Diseases, Qinghai Provincial Hospital of Traditional Chinese Medicine, Xining 810000, China.mlqinghai201314@sina.com ;Telephone: +86-13519732841 -
About author:Pro. LI Junru, Department of Spleen and Stomach Diseases, Qinghai Provincial Hospital of Traditional Chinese Medicine, Xining 810000, China. mlqinghai201314@sina.com;Telephone: +86-13519732841
-
Supported by:2023 Traditional Chinese Medicine Innovation Capability Enhancement Project from the National Administration of Traditional Chinese Medicine(no number);2024 Qinghai Province “Kunlun Talents ? High-End Innovative and Entrepreneurial Talents” Top Notch Talent Project(QHKLYC-GDCXCY-2024-152);Study the Mechanism of Weimishu Prescription in the Treatment of Chronic Gastritis with Liver-Stomach Disharmony
Cite this article
ZOU Xiaoyun, WEI Minmin, JIA Shouning, QI Yongfu, LI Hailing, LIU Yan, SHI Xiujuan, LI Junru. Weimishu prescription (胃糜舒方) alleviates liver-stomach disharmony and chronic gastritis by reducing inflammation and regulating gastric stem cell differentiation[J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 95-109.
share this article
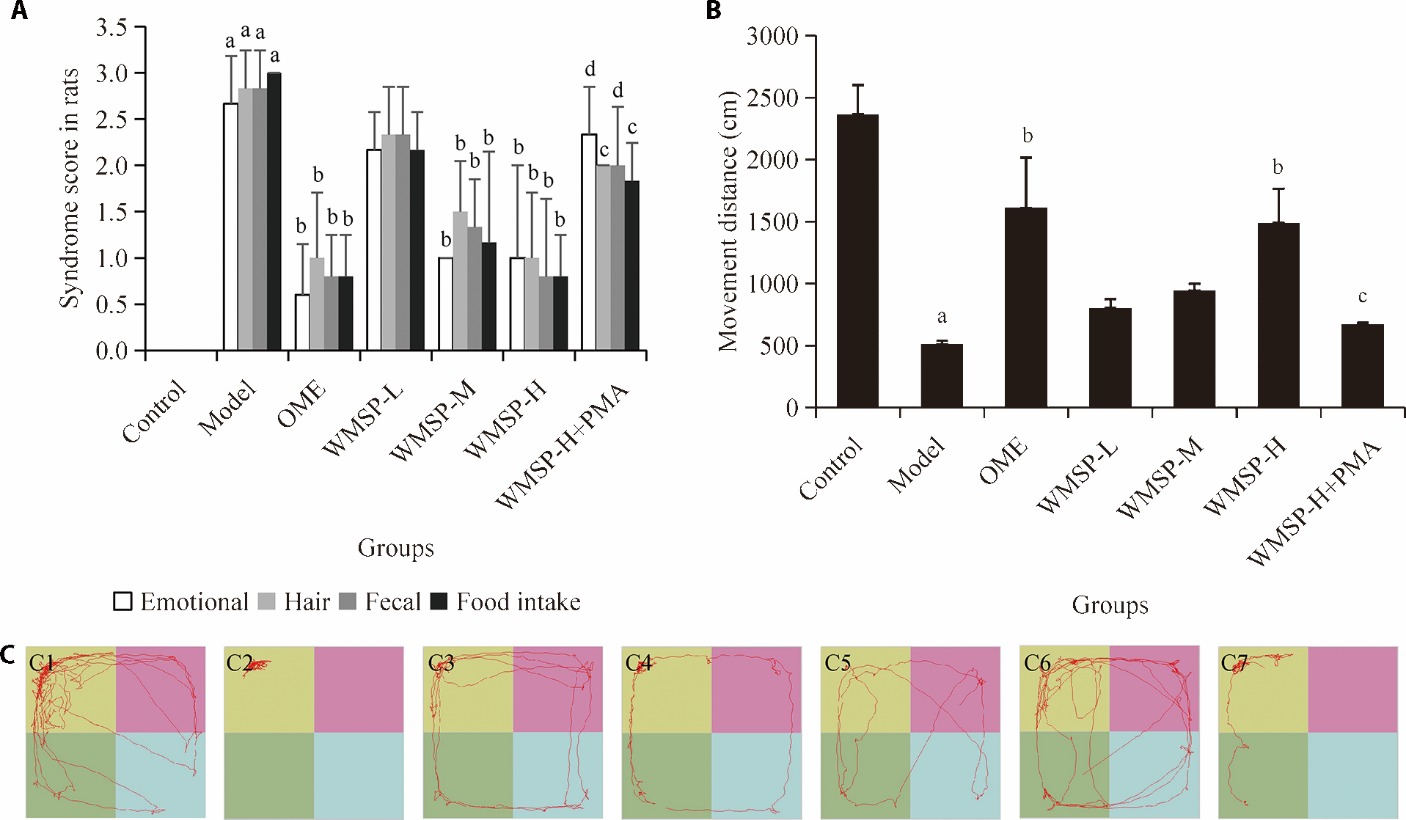
Figure 1 WMSP improved the liver-stomach disharmony syndrome in CG-LSD rats A: Traditional Chinese Medicine syndrome scores of CG-LSD rats based on emotional condition, hair condition, stool condition, and food intake (n = 6); B: moving distance of rats in OFT (n = 3); C: representative images of OFT; C1: control group; C2: model group; C3: OME group; C4: WMSP-L group; C5: WMSP-M group; C6: WMSP-H group; C7: WMSP-H + PMA group. Control group: normal rats without CG-LSD model or treatment; Model group: rats with CG-LSD, without treatment; OME group: CG-LSD rats treated with positive drug omeprazole (20 mg/kg); WMSP-L group: CG-LSD rats treated with low-dose WMSP (8.7 g·kg?1·d?1); WMSP-M group: CG-LSD rats treated with middle-dose WMSP (17.3 g·kg?1·d?1); WMSP-H group: CG-LSD rats treated with high-dose WMSP (34.6 g·kg?1·d?1); WMSP-H+PMA group: CG-LSD rats treated with high-dose WMSP (34.6 g·kg?1·d?1) and NF-κB agonist PMA (20 ng·kg?1·d?1). CG-LSD: chronic gastritis with liver-stomach disharmony; OFT: open field test; OME: omeprazole; WMSP: Weimishu prescription; PMA: phorbol 12-myristate 13-acetate; ANOVA: analysis of variance. Statistical analysis was performed using one-way ANOVA followed by post hoc testing. Data were presented as the mean ± standard deviation. aP < 0.01 vs the control group; bP < 0.01 vs the model group; cP < 0.05, dP < 0.01 vs the WMSP-H group.
| Week | Control (n = 6) | Model (n = 6) | OME (n = 5) | WMSP-L (n = 6) | WMSP-M (n = 6) | WMSP-H (n = 5) | WMSP-H+PMA (n = 6) |
|---|---|---|---|---|---|---|---|
| 0 | 225±3 | 222±3 | 225±6 | 225±5 | 220±4 | 224±4 | 222±5 |
| 1 | 307±8 | 303±12 | 297±16 | 295±9 | 295±12 | 302±7 | 296±12 |
| 2 | 379±23 | 359±18 | 356±18 | 357±19 | 357±28 | 360±18 | 358±22 |
| 3 | 451±12 | 403±17a | 404±15 | 405±24 | 409±33 | 407±21 | 403±16 |
| 4 | 502±18 | 451±17b | 454±16 | 460±24 | 451±49 | 460±26 | 443±31 |
| 5 | 529±18 | 474±19b | 480±12 | 478±31 | 480±48 | 479±17 | 476±22 |
| 6 | 554±16 | 490±29b | 492±29 | 496±31 | 494±45 | 494±19 | 491±19 |
| 7 | 605±23 | 507±18b | 505±23 | 509±38 | 511±39 | 509±17 | 509±26 |
| 8 | 624±28 | 516±20b | 558±36 | 542±27 | 552±41 | 562±13 | 536±22 |
| 9 | 632±44 | 524±17b | 586±40c | 554±29 | 565±44 | 580±25 | 548±35 |
| 10 | 646±33 | 535±14b | 603±29c | 576±23 | 584±44d | 596±20d | 563±43 |
| 11 | 655±32 | 529±31b | 625±26c | 589±24c | 602±45c | 614±17c | 572±44 |
Table 1 Body weight of rats (g, $\bar{x} \pm s$)
| Week | Control (n = 6) | Model (n = 6) | OME (n = 5) | WMSP-L (n = 6) | WMSP-M (n = 6) | WMSP-H (n = 5) | WMSP-H+PMA (n = 6) |
|---|---|---|---|---|---|---|---|
| 0 | 225±3 | 222±3 | 225±6 | 225±5 | 220±4 | 224±4 | 222±5 |
| 1 | 307±8 | 303±12 | 297±16 | 295±9 | 295±12 | 302±7 | 296±12 |
| 2 | 379±23 | 359±18 | 356±18 | 357±19 | 357±28 | 360±18 | 358±22 |
| 3 | 451±12 | 403±17a | 404±15 | 405±24 | 409±33 | 407±21 | 403±16 |
| 4 | 502±18 | 451±17b | 454±16 | 460±24 | 451±49 | 460±26 | 443±31 |
| 5 | 529±18 | 474±19b | 480±12 | 478±31 | 480±48 | 479±17 | 476±22 |
| 6 | 554±16 | 490±29b | 492±29 | 496±31 | 494±45 | 494±19 | 491±19 |
| 7 | 605±23 | 507±18b | 505±23 | 509±38 | 511±39 | 509±17 | 509±26 |
| 8 | 624±28 | 516±20b | 558±36 | 542±27 | 552±41 | 562±13 | 536±22 |
| 9 | 632±44 | 524±17b | 586±40c | 554±29 | 565±44 | 580±25 | 548±35 |
| 10 | 646±33 | 535±14b | 603±29c | 576±23 | 584±44d | 596±20d | 563±43 |
| 11 | 655±32 | 529±31b | 625±26c | 589±24c | 602±45c | 614±17c | 572±44 |

Figure 2 WMSP improves gastric mucosa injury in CG-LSD rats A1-A7: representative images of HE staining of gastric mucosal tissues (magnification = × 100), scare bar = 200 μm; A8-A14: representative images of HE staining of gastric mucosal tissues (magnification = × 400), scare bar = 50 μm. A1, A8: control group; A2, A9: model group; A3, A10: OME group; A4, A11: WMSP-L group; A5, A12: WMSP-M group; A6, A13: WMSP-H group; A7, A14: WMSP-H + PMA group; B: representative images of TUNEL staining. Scare bar = 20 μm. B1-B7: DAPI stained gastric mucosal epithelial cells in the control, model, OME, WMSP-L, WMSP-M, WMSP-H, and WMSP-H + PMA groups; B8-B14: TUNEL stained gastric mucosal epithelial cells in the control, model, OME, WMSP-L, WMSP-M, WMSP-H, and WMSP-H + PMA groups; B15-B21: merge of DAPI and TUNEL stained gastric mucosal epithelial cells in the control, model, OME, WMSP-L, WMSP-M, WMSP-H, and WMSP-H + PMA groups; C: histopathological scores of HE staining, characterized by mucosal cell atrophy, necrosis, and decline; inflammatory cell infiltration; hemorrhage; and fibrous tissue proliferation; D: the apoptosis rate of gastric mucosal epithelial cells. Control group: normal rats without CG-LSD model or treatment; Model group: rats with CG-LSD, without treatment; OME group: CG-LSD rats treated with positive drug omeprazole (20 mg/kg); WMSP-L group: CG-LSD rats treated with low-dose WMSP (8.7 g·kg?1·d?1); WMSP-M group: CG-LSD rats treated with middle-dose WMSP (17.3 g·kg?1·d?1); WMSP-H group: CG-LSD rats treated with high-dose WMSP (34.6 g·kg?1·d?1); WMSP-H + PMA group: CG-LSD rats treated with high-dose WMSP (34.6 g·kg?1·d?1) and NF-κB agonist PMA (20 ng·kg?1·d?1). CG-LSD: chronic gastritis with liver-stomach disharmony; OME: omeprazole; WMSP: Weimishu prescription; PMA: phorbol 12-myristate 13-acetate; ANOVA: analysis of variance. Statistical analysis was performed using one-way ANOVA followed by post hoc testing. Data were presented as the mean ± standard deviation (n = 3). aP < 0.01 vs the control group; bP < 0.01, cP < 0.05, vs the model group; dP < 0.01 vs the WMSP-H group.

Figure 3 WMSP promoted the survival and differentiation of gastric stem cells (GSCs) in CG-LSD rats A: representative images of flow cytometry. A1: control group; A2: model group; A3: OME group; A4: WMSP-L group; A5: WMSP-M group; A6: WMSP-H group; A7: WMSP-H + PMA group; B: the percentage of Lgr5+Ki67+ cells in gastric tissue detected by flow cytometry; C: representative images of immunofluorescence staining, scare bar = 50 μm; C1-C7: DAPI stained GSCs in the control, model, OME, WMSP-L, WMSP-M, WMSP-H, and WMSP-H + PMA groups; C8-C14: Lgr5 stained GSCs in the control, model, OME, WMSP-L, WMSP-M, WMSP-H, and WMSP-H + PMA groups; C15-C21: merge of DAPI and Lgr5 stained GSCs in the control, model, OME, WMSP-L, WMSP-M, WMSP-H, and WMSP-H + PMA groups; D: immunofluorescence staining results indicating the number of Lgr5-positive cells; E: the expression levels of the main cell marker (PGC), endocrine cell marker (SST), gastric gland mucous cell marker (MUC6), parietal cell marker (H+-K+ATP4B), and epithelial cell marker (E-cadherin) proteins detected by Western blot. Control group: normal rats without CG-LSD model or treatment; Model group: rats with CG-LSD, without treatment; OME group: CG-LSD rats treated with positive drug omeprazole (20 mg/kg); WMSP-L group: CG-LSD rats treated with low-dose WMSP (8.7 g·kg?1·d?1); WMSP-M group: CG-LSD rats treated with middle-dose WMSP (17.3 g·kg?1·d?1); WMSP-H group: CG-LSD rats treated with high-dose WMSP (34.6 g·kg?1·d?1); WMSP-H + PMA group: CG-LSD rats treated with high-dose WMSP (34.6 g·kg?1·d?1) and NF-κB agonist PMA (20 ng·kg?1·d?1). CG-LSD: chronic gastritis with liver-stomach disharmony; OME: omeprazole; WMSP: Weimishu prescription; PMA: phorbol 12-myristate 13-acetate; Lgr5: leucine-rich repeat-containing G protein-coupled receptor 5; GSCs: gastric stem cells; MUC6: mucin-6; PGC: pepsinogen C; SST: somatostatin; H+-K+ATP4B: hydrogen potassium ATPase beta; ANOVA: analysis of variance. Statistical analysis was performed using one-way ANOVA followed by post hoc testing. Data are presented as the mean ± standard deviation (n = 3). aP < 0.01 vs the control group; bP < 0.01 vs the model group; cP < 0.01 vs the WMSP-H group.
| Group | n | Mist1 | SOX2 | SOX9 | CD44 |
|---|---|---|---|---|---|
| Control | 3 | 1.033±0.305 | 1.017±0.230 | 1.03±0.31 | 1.013±0.222 |
| Model | 3 | 0.243±0.032a | 0.203±0.025a | 0.22±0.01a | 0.147±0.015a |
| OME | 3 | 0.813±0.227b | 0.837±0.206b | 0.90±0.25b | 0.850±0.182b |
| WMSP-L | 3 | 0.517±0.137 | 0.437±0.117 | 0.48±0.15 | 0.343±0.130 |
| WMSP-M | 3 | 0.630±0.135c | 0.533±0.143c | 0.58±0.07c | 0.557±0.146b |
| WMSP-H | 3 | 0.723±0.112b | 0.703±0.188b | 0.71±0.26b | 0.703±0.222b |
| WMSP-H+PMA | 3 | 0.393±0.091d | 0.313±0.050e | 0.35±0.08d | 0.253±0.045e |
Table 2 GSCs-related markers (Mist1, Sox2, SOX9, and CD44) mRNA expression (2-△△CT) detected using RT-qPCR ($\bar{x} \pm s$)
| Group | n | Mist1 | SOX2 | SOX9 | CD44 |
|---|---|---|---|---|---|
| Control | 3 | 1.033±0.305 | 1.017±0.230 | 1.03±0.31 | 1.013±0.222 |
| Model | 3 | 0.243±0.032a | 0.203±0.025a | 0.22±0.01a | 0.147±0.015a |
| OME | 3 | 0.813±0.227b | 0.837±0.206b | 0.90±0.25b | 0.850±0.182b |
| WMSP-L | 3 | 0.517±0.137 | 0.437±0.117 | 0.48±0.15 | 0.343±0.130 |
| WMSP-M | 3 | 0.630±0.135c | 0.533±0.143c | 0.58±0.07c | 0.557±0.146b |
| WMSP-H | 3 | 0.723±0.112b | 0.703±0.188b | 0.71±0.26b | 0.703±0.222b |
| WMSP-H+PMA | 3 | 0.393±0.091d | 0.313±0.050e | 0.35±0.08d | 0.253±0.045e |
| Group | n | MUC6 | PGC | SST | ATP4B | E-cadherin |
|---|---|---|---|---|---|---|
| Control | 3 | 1.000±0.048 | 1.000±0.046 | 1.000±0.054 | 1.006±0.117 | 1.001±0.058 |
| Model | 3 | 0.137±0.061a | 0.329±0.015a | 0.086±0.036a | 0.114±0.013a | 0.155±0.017a |
| OME | 3 | 0.775±0.060b | 0.711±0.060b | 0.836±0.088b | 0.850±0.066b | 0.772±0.028b |
| WMSP-L | 3 | 0.528±0.080b | 0.565±0.057b | 0.496±0.049b | 0.291±0.032b | 0.425±0.034b |
| WMSP-M | 3 | 0.637±0.114b | 0.615±0.063b | 0.663±0.049b | 0.400±0.081b | 0.537±0.021b |
| WMSP-H | 3 | 0.814±0.062b | 0.695±0.018b | 0.832±0.084b | 0.735±0.065b | 0.785±0.066b |
| WMSP-H+PMA | 3 | 0.336±0.053c | 0.412±0.029c | 0.151±0.024c | 0.146±0.033c | 0.277±0.039c |
Table 3 Western blot assay for quantitative analysis of PGC, SST, MUC6, H + -K + ATP4B, and E-cadherin proteins ($\bar{x} \pm s$)
| Group | n | MUC6 | PGC | SST | ATP4B | E-cadherin |
|---|---|---|---|---|---|---|
| Control | 3 | 1.000±0.048 | 1.000±0.046 | 1.000±0.054 | 1.006±0.117 | 1.001±0.058 |
| Model | 3 | 0.137±0.061a | 0.329±0.015a | 0.086±0.036a | 0.114±0.013a | 0.155±0.017a |
| OME | 3 | 0.775±0.060b | 0.711±0.060b | 0.836±0.088b | 0.850±0.066b | 0.772±0.028b |
| WMSP-L | 3 | 0.528±0.080b | 0.565±0.057b | 0.496±0.049b | 0.291±0.032b | 0.425±0.034b |
| WMSP-M | 3 | 0.637±0.114b | 0.615±0.063b | 0.663±0.049b | 0.400±0.081b | 0.537±0.021b |
| WMSP-H | 3 | 0.814±0.062b | 0.695±0.018b | 0.832±0.084b | 0.735±0.065b | 0.785±0.066b |
| WMSP-H+PMA | 3 | 0.336±0.053c | 0.412±0.029c | 0.151±0.024c | 0.146±0.033c | 0.277±0.039c |
| Group | n | Serum | Gastric mucosa | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| IL-1β | IL-6 | IL-17 | TNF-α | IL-1β | IL-6 | IL-17 | TNF-α | ||||
| Control | 3 | 3.4±0.7 | 19.4±3.8 | 5.2±0.3 | 28.9±3.1 | 2.0±0.6 | 14.8±3.7 | 2.9±0.4 | 16.3±2.2 | ||
| Model | 3 | 8.6±0.7a | 35.4±4.1a | 8.1±0.5a | 59.0±4.3a | 6.3±0.6a | 36.0±4.6a | 6.7±0.4a | 40.9±2.7a | ||
| OME | 3 | 5.9±1.0b | 25.9±2.9b | 6.5±0.8b | 37.8±3.5b | 3.8±0.8b | 20.6±3.6b | 4.3±0.7b | 19.4±5.7b | ||
| WMSP-L | 3 | 7.2±0.5c | 29.7±2.5c | 7.1±0.7c | 50.5±5.3c | 5.1±0.7c | 29.5±1.6c | 5.4±0.6c | 32.1±4.9c | ||
| WMSP-M | 3 | 6.0±1.0b | 26.5±2.5b | 6.7±0.5c | 44.9±4.8b | 4.3±0.6b | 23.6±2.2b | 4.7±0.9c | 26.1±5.3b | ||
| WMSP-H | 3 | 5.3±0.7b | 23.6±2.7b | 5.7±0.7b | 41.0±4.6b | 3.8±0.5b | 16.5±1.4b | 4.1±0.7b | 20.4±3.6b | ||
| WMSP-H+PMA | 3 | 7.8±0.7d | 31.3±3.1d | 7.3±0.4d | 53.9±3.7d | 5.8±0.7d | 33.3±3.0d | 6.1±0.6d | 35.2±2.7d | ||
Table 4 Levels of inflammatory factors in the serum and gastric mucosal tissues of rats (pg/mL, $\bar{x} \pm s$)
| Group | n | Serum | Gastric mucosa | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| IL-1β | IL-6 | IL-17 | TNF-α | IL-1β | IL-6 | IL-17 | TNF-α | ||||
| Control | 3 | 3.4±0.7 | 19.4±3.8 | 5.2±0.3 | 28.9±3.1 | 2.0±0.6 | 14.8±3.7 | 2.9±0.4 | 16.3±2.2 | ||
| Model | 3 | 8.6±0.7a | 35.4±4.1a | 8.1±0.5a | 59.0±4.3a | 6.3±0.6a | 36.0±4.6a | 6.7±0.4a | 40.9±2.7a | ||
| OME | 3 | 5.9±1.0b | 25.9±2.9b | 6.5±0.8b | 37.8±3.5b | 3.8±0.8b | 20.6±3.6b | 4.3±0.7b | 19.4±5.7b | ||
| WMSP-L | 3 | 7.2±0.5c | 29.7±2.5c | 7.1±0.7c | 50.5±5.3c | 5.1±0.7c | 29.5±1.6c | 5.4±0.6c | 32.1±4.9c | ||
| WMSP-M | 3 | 6.0±1.0b | 26.5±2.5b | 6.7±0.5c | 44.9±4.8b | 4.3±0.6b | 23.6±2.2b | 4.7±0.9c | 26.1±5.3b | ||
| WMSP-H | 3 | 5.3±0.7b | 23.6±2.7b | 5.7±0.7b | 41.0±4.6b | 3.8±0.5b | 16.5±1.4b | 4.1±0.7b | 20.4±3.6b | ||
| WMSP-H+PMA | 3 | 7.8±0.7d | 31.3±3.1d | 7.3±0.4d | 53.9±3.7d | 5.8±0.7d | 33.3±3.0d | 6.1±0.6d | 35.2±2.7d | ||

Figure 4 WMSP regulated the NF-κB/β-catenin pathway in CG-LSD rats A: Western blotting used to detect the expression of IκBα, p-IκBα, Smurf2, and β-catenin in gastric mucosal tissues; B: quantitative data of NF-κB p65-positive cells in the nucleus; C: immunofluorescence staining used to detect the nuclear localization of NF-κB p65. Scare bar = 50 μm. C1-C7: DAPI stained GSCs in the control, model, OME, WMSP-L, WMSP-M, WMSP-H, and WMSP-H + PMA groups; C8-C14: Lgr5 stained GSCs in the control, model, OME, WMSP-L, WMSP-M, WMSP-H, and WMSP-H + PMA groups; C15-C21: merge of DAPI and Lgr5 stained GSCs in the control, model, OME, WMSP-L, WMSP-M, WMSP-H, and WMSP-H + PMA groups; D: RT-qPCR performed to measure the expression levels of c-MYC in gastric mucosal tissues; E: RT-qPCR performed to measure the expression levels of AXIN2 in gastric mucosal tissues. Control group: normal rats without CG-LSD model or treatment; Model group: rats with CG-LSD, without treatment; OME group: CG-LSD rats treated with positive drug omeprazole (20 mg/kg); WMSP-L group: CG-LSD rats treated with low-dose WMSP (8.7 g·kg?1·d?1); WMSP-M group: CG-LSD rats treated with middle-dose WMSP (17.3 g·kg?1·d?1); WMSP-H group: CG-LSD rats treated with high-dose WMSP (34.6 g·kg?1·d?1); WMSP-H + PMA group: CG-LSD rats treated with high-dose WMSP (34.6 g·kg?1·d?1) and NF-κB agonist PMA (20 ng·kg?1·d?1). CG-LSD: chronic gastritis with liver-stomach disharmony; OME: omeprazole; WMSP: Weimishu prescription; PMA: phorbol 12-myristate 13-acetate; SMURF2: Smad-specific E3 ubiquitin ligase 2; IkBα: I-kappa-B-alpha; c-MYC: myelocytomatosis oncogene; AXIN2: axis inhibition protein 2; RT-qPCR: real-time quantitative polymerase chain reaction; ANOVA: analysis of variance. Statistical analysis was performed using one-way ANOVA followed by post hoc testing. Data are presented as the mean ± standard deviation (n = 3). aP < 0.01 vs the control group; bP < 0.01, cP < 0.05 vs the model group; dP < 0.01 vs the WMSP-H group.
| Group | n | SMURF2 | IκBα | p-IκBα | β-catenin |
|---|---|---|---|---|---|
| Control | 3 | 1.01±0.13 | 1.00±0.12 | 1.00±0.04 | 1.01±0.12 |
| Model | 3 | 2.71±0.13a | 1.15±0.11a | 6.08±0.29a | 0.23±0.06a |
| OME | 3 | 1.53±0.04b | 1.07±0.04 | 3.12±0.20b | 0.81±0.05b |
| WMSP-L | 3 | 2.02±0.09b | 1.07±0.04 | 4.84±0.12b | 0.58±0.04b |
| WMSP-M | 3 | 1.83±0.14b | 1.08±0.07 | 4.33±0.31b | 0.70±0.04b |
| WMSP-H | 3 | 1.51±0.09b | 1.05±0.14 | 3.23±0.15b | 0.86±0.07b |
| WMSP-H+PMA | 3 | 2.15±0.09c | 1.08±0.06 | 5.31±0.06c | 0.49±0.13c |
Table 5 Quantitative analysis of the NF-κB/β-catenin pathway proteins via Western blotting
| Group | n | SMURF2 | IκBα | p-IκBα | β-catenin |
|---|---|---|---|---|---|
| Control | 3 | 1.01±0.13 | 1.00±0.12 | 1.00±0.04 | 1.01±0.12 |
| Model | 3 | 2.71±0.13a | 1.15±0.11a | 6.08±0.29a | 0.23±0.06a |
| OME | 3 | 1.53±0.04b | 1.07±0.04 | 3.12±0.20b | 0.81±0.05b |
| WMSP-L | 3 | 2.02±0.09b | 1.07±0.04 | 4.84±0.12b | 0.58±0.04b |
| WMSP-M | 3 | 1.83±0.14b | 1.08±0.07 | 4.33±0.31b | 0.70±0.04b |
| WMSP-H | 3 | 1.51±0.09b | 1.05±0.14 | 3.23±0.15b | 0.86±0.07b |
| WMSP-H+PMA | 3 | 2.15±0.09c | 1.08±0.06 | 5.31±0.06c | 0.49±0.13c |
| 1. |
Wang B, Xiao L, Chen P, et al. Uncovering the role of Traditional Chinese Medicine in immune-metabolic balance of gastritis from the perspective of cold and hot: Jin Hong Tablets as a case study. Chin Med 2024; 19: 134.
DOI |
| 2. |
Liu Y, Huang T, Wang L, et al. Traditional Chinese Medicine in the treatment of chronic atrophic gastritis, precancerous lesions and gastric cancer. J Ethnopharmacol 2025; 337: 118812.
DOI URL |
| 3. |
Yang YY, Li KM, Xu GF, et al. Clinical manifestation, lifestyle, and treatment patterns of chronic erosive gastritis: a multicenter real-world study in China. World J Gastroenterol 2024; 30: 1108-20.
DOI URL |
| 4. |
Shi Y, Wei N, Wang K, et al. Deep learning-assisted diagnosis of chronic atrophic gastritis in endoscopy. Front Oncol 2023; 13: 1122247.
DOI URL |
| 5. |
Xu W, Jiang T, Shen K, et al. GADD45B regulates the carcinogenesis process of chronic atrophic gastritis and the metabolic pathways of gastric cancer. Front Endocrinol (Lausanne) 2023; 14: 1224832.
DOI URL |
| 6. |
Chinese Society of Gastroenterology, Cancer Collaboration Group of Chinese Society of Gastroenterology, Chinese Medical Association. Guidelines for diagnosis and treatment of chronic gastritis in China (2022, Shanghai). J Dig Dis 2023; 24: 150-80.
DOI URL |
| 7. |
Aziz F, Chakraborty A, Liu K, et al. Gastric tumorigenesis induced by combining Helicobacter pylori infection and chronic alcohol through IL-10 inhibition. Carcinogenesis 2022; 43: 126-39.
DOI URL |
| 8. | Wang L, Lian YJ, Dong JS, et al. Traditional Chinese Medicine for chronic atrophic gastritis: efficacy, mechanisms and targets. World J Gastroenterol 2025; 31: 102053. |
| 9. |
Kuang W, Xu J, Xu F, et al. Current study of pathogenetic mechanisms and therapeutics of chronic atrophic gastritis: a comprehensive review. Front Cell Dev Biol 2024; 12: 1513426.
DOI URL |
| 10. | Wang B (Tang dynasty). Huang Di Nei Jing Su Wen. Beijing: People's Medical Publishing House, 2013: 8. |
| 11. | Luo HY, Shen JW, Li JR, Jin DW, Guan YX, Li YS. Summary of clinical experience of Chief Physician Li Junru in the treatment of chronic atrophic gastritis in plateau areas. Zhong Guo Yi Yao Dao Bao 2024; 21: 143-7. |
| 12. |
Chen L, Wei S, He Y, et al. Treatment of chronic gastritis with Traditional Chinese Medicine: pharmacological activities and mechanisms. Pharmaceuticals (Basel) 2023; 16: 1308.
DOI URL |
| 13. |
Yuxi G, Ze LI, Nan C, et al. High-throughput sequencing analysis of differential microRNA expression in the process of blocking the progression of chronic atrophic gastritis to gastric cancer by Xianglian Huazhuo formula. J Tradit Chin Med 2024; 44: 703-12.
DOI |
| 14. | Yuan C, Xiaosi Z, Junxiang LI, et al. Hewei Jiangni granule alleviates visceral hypersensitivity of non-erosive reflux diseasestromal interaction molecule 1/transient receptor potential vanilloid subfamily member 1 pathway. J Tradit Chin Med 2025; 45: 1-12. |
| 15. |
Mengting L, Tao LI, Fuhao C, et al. Weichang' an pill alleviates functional dyspepsia through modulating brain-gut peptides and gut microbiota. J Tradit Chin Med 2024; 44: 1177-86.
DOI |
| 16. | Xu WW, Li JR. Brief analysis of Li Junru's experience in diagnosis and treatment of chronic erosive gastritis. Shi Jie Zui Xin Yi Xue Xin Xi Wen Zhai 2018; 18: 176-81. |
| 17. | Xu WW, Li JR, Cui XS. 30 Cases of chronic erosive gastritis with liver depression and Qi stagnation treated with modified Weimishu prescription. Zhong Guo Min Jian Liao Fa 2018; 26: 32-33. |
| 18. | Li JR, Xu WW, Zou XY, et al. Clinical study on treatment with Yiqi Huayu Shengji decoction for chronic erosive gastritis patients with Qi deficiency and blood stasis syndrome in Qinghai area. Zhong Yi Za Zhi 2018; 59: 2117-19+47. |
| 19. | Guan YX, Luo HY, Li JR. Effects of Weimishu prescription on gastric function and inflammatory response in chronic erosive gastritis with hepato-gastric disharmony patients. Zhong Guo Min Jian Liao Fa 2024; 32: 78-81. |
| 20. |
Liu M, Liu Q, Zou Q, et al. The composition and roles of gastric stem cells in epithelial homeostasis, regeneration, and tumorigenesis. Cell Oncol (Dordr) 2023; 46: 867-83.
DOI PMID |
| 21. |
Lee SH, Won Y, Gibbs D, et al. Amphiregulin switches progenitor cell fate for lineage commitment during gastric mucosal regeneration. Gastroenterology 2024; 167: 469-84.
DOI URL |
| 22. | He L, Zhang X, Zhang S, et al. H. Pylori-facilitated TERT/Wnt/β-Catenin triggers spasmolytic polypeptide-expressing metaplasia and oxyntic atrophy. Adv Sci (Weinh) 2025; 12: e2401227. |
| 23. |
McGowan KP, Delgado E, Hibdon ES, Samuelson LC. Differential sensitivity to Wnt signaling gradients in human gastric organoids derived from corpus and antrum. Am J Physiol Gastrointest Liver Physiol 2023; 325: G158-g73.
DOI URL |
| 24. |
Alvina FB, Chen TC, Lim HYG, Barker N. Gastric epithelial stem cells in development, homeostasis and regeneration. Development 2023; 150: dev201494.
DOI URL |
| 25. | Li K, Ma X, Li Z, et al. A Natural peptide from a Traditional Chinese Medicine has the potential to treat chronic atrophic gastritis by activating gastric stem cells. Adv Sci (Weinh) 2024; 11: e2304326. |
| 26. |
Takada H, Sasagawa Y, Yoshimura M, et al. Single-cell transcriptomics uncovers EGFR signaling-mediated gastric progenitor cell differentiation in stomach homeostasis. Nat Commun 2023; 14: 3750.
DOI PMID |
| 27. |
Wang YR, Yan XE, Ding MY, et al. Research on the signaling pathway and the related mechanism of Traditional Chinese Medicine intervention in chronic gastritis of the "inflammation-cancer transformation". Front Pharmacol 2024; 15: 1338471.
DOI URL |
| 28. |
Liu J, Zhuang Y, Wu J, et al. IKKβ mediates homeostatic function in inflammation via competitively phosphorylating AMPK and IκBα. Acta Pharm Sin B 2022; 12: 651-64.
DOI URL |
| 29. |
Xie Y, Zheng L, Chen W, Zeng Y, Yao K, Zhou T. Potential signal pathways and therapeutic effects of mesenchymal stem cell on oxidative stress in diseases. Curr Pharm Des 2024; 31: 83-94.
DOI URL |
| 30. |
Yu Q, Shi H, Ding Z, Wang Z, Yao H, Lin R. The E3 ubiquitin ligase TRIM31 attenuates NLRP3 inflammasome activation in Helicobacter pylori-associated gastritis by regulating ROS and autophagy. Cell Commun Signal 2023; 21: 1.
DOI PMID |
| 31. | State Pharmacopoeia Committee. Chinese Pharmacopoeia. Beijing: China Medical Science and Technology Press, 2025: 1-415. |
| 32. |
Liu F, Nong X, Qu W, Li X. Pharmacokinetics and tissue distribution of 12 major active components in normal and chronic gastritis rats after oral administration of Weikangling capsules. J Ethnopharmacol 2023; 316: 116722.
DOI URL |
| 33. |
Wang XX, Liu XJ, Wang YJ, et al. Chaihu Shugan powder inhibits interstitial cells of cajal mitophagy through USP 30 in the treatment of functional dyspepsia. J Ethnopharmacol 2024; 323: 117695.
DOI URL |
| 34. |
Hou LW, Fang JL, Zhang JL, et al. Auricular vagus nerve stimulation ameliorates functional dyspepsia with depressive-like behavior and inhibits the hypothalamus-pituitary-adrenal axis in a rat model. Dig Dis Sci 2022; 67: 4719-31.
DOI |
| 35. |
Qi L, Chang Y, Jintong Y, et al. Association of miR-499 rs3746444, miR-149 rs2292832 polymorphisms and their expression levels with helicobacter pylori-related gastric diseases and Traditional Chinese Medicine syndromes. J Tradit Chin Med 2024; 44: 1024-34.
DOI |
| 36. |
Liu J, Zhang G, Wang Y, et al. Screening and verification of hemostatic effective components group of Panax Notoginseng based on spectrum-effect relationships. J Ethnopharmacol 2024; 321: 117539.
DOI URL |
| 37. |
Zhang YY, Chen ZJ, Chen LP, et al. Astragali radix (Huangqi): a time-honored nourishing herbal medicine. Chin Med 2024; 19: 119.
DOI |
| 38. |
Chen R, Wang X, Xu H, Zhao R, Hu Q. Comparative study on volatile oils among Bupleuri Radix species and habitats: Yields, chemical characterization and antipyretic activities. Chem Biodivers 2022; 19: e202200549.
DOI URL |
| 39. |
Zhu Y, Lai Y. Pharmacological properties and derivatives of saikosaponins-a review of recent studies. J Pharm Pharmacol 2023; 75: 898-909.
DOI PMID |
| 40. |
Li YG, Wang XY, Chen HF, Yuan JB, Meng Y, Yang WL. Comparison of the chemical constituents of raw Fructus Aurantii and Fructus Aurantii stir-baked with bran, and the biological effects of auraptene. J Ethnopharmacol 2021; 269: 113721.
DOI URL |
| 41. |
Nascakova Z, He J, Papa G, Francas B, Azizi F, Müller A. Helicobacter pylori induces the expression of Lgr5 and stem cell properties in gastric target cells. Life Sci Alliance 2024; 7: e202402783.
DOI URL |
| 42. |
Chen F, Han Y, Yue J, et al. Oxidative stress and Kras mutation in Mist1+ cells act in a double-hit manner to drive gastric tumorigenesis. Cell Rep 2025; 44: 116014.
DOI URL |
| 43. |
Dong J, Wu X, Zhou X, et al. Spatially resolved expression landscape and gene-regulatory network of human gastric corpus epithelium. Protein Cell 2023; 14: 433-47.
DOI |
| 44. |
Huang KK, Ma H, Chong RHH, et al. Spatiotemporal genomic profiling of intestinal metaplasia reveals clonal dynamics of gastric cancer progression. Cancer Cell 2023; 41: 2019-37.e8.
DOI PMID |
| 45. |
Chen Q, Weng K, Lin M, et al. SOX9 modulates the transformation of gastric stem cells through biased symmetric cell division. Gastroenterology 2023; 164: 1119-36.e12.
DOI URL |
| 46. |
Huang W, Wen F, Gu P, et al. The inhibitory effect and mechanism of Yi-qi-hua-yu-jie-du decoction on the drug resistance of gastric cancer stem cells based on ABC transporters. Chin Med 2022; 17: 93.
DOI |
| 47. |
Chen Z, Gao Y, Zhang P, et al. Identification of gastric cancer stem cells with CD44 and Lgr 5 double labelling and their initial roles on gastric cancer malignancy and chemotherapy resistance. Cell Biol Toxicol 2024; 41: 12.
DOI |
| 48. |
Qin Y, Geng JX, Huang B. Clinical value of serum pepsinogen in the diagnosis and treatment of gastric diseases. World J Gastrointest Oncol 2023; 15: 1174-81.
DOI URL |
| 49. | Papantoniou K, Aggeletopoulou I, Pastras P, Triantos C. The role of somatostatin in the gastrointestinal tract. Biology (Basel) 2025; 14: 558. |
| 50. |
Arai J, Hayakawa Y, Tateno H, et al. Impaired glycosylation of gastric mucins drives gastric tumorigenesis and serves as a novel therapeutic target. Gastroenterology 2024; 167: 505-21.e19.
DOI PMID |
| 51. |
Della Bella C, Antico A, Panozzo MP, et al. Gastric Th17 cells specific for H+/K+-ATPase and serum IL-17 signature in gastric autoimmunity. Front Immunol 2022; 13: 952674.
DOI URL |
| 52. |
Tang Y, Yang G, Zhang J, et al. E-cadherin is required for the homeostasis of Lgr5+ gastric antral stem cells. Int J Biol Sci 2019; 15: 34-43.
DOI URL |
| 53. |
Lialios P, Alimperti S. Role of E-cadherin in epithelial barrier dysfunction: implications for bacterial infection, inflammation, and disease pathogenesis. Front Cell Infect Microbiol 2025; 15: 1506636.
DOI URL |
| 54. |
Wen X, Qin J, Zhang X, et al. MEK-mediated CHPF2 phosphorylation promotes colorectal cancer cell proliferation and metastasis by activating NF-κB signaling. Cancer Lett 2024; 584: 216644.
DOI URL |
| 55. |
Bordin F, Terriaca G, Apostolico A, et al. SMURF1 and SMURF 2 directly target GLI1 for ubiquitination and proteasome-dependent degradation. Cell Death Discov 2024; 10: 498.
DOI PMID |
| 56. |
Guo L, Li K, Ma Y, et al. MicroRNA-322-5p targeting Smurf2 regulates the TGF-β/Smad pathway to protect cardiac function and inhibit myocardial infarction. Hum Cell 2024; 37: 972-85.
DOI |
| [1] | LIANG Zihao, GAO Jie, SONG Jinyun, ZHENG Qin, ZHAO Hongyu. Network pharmacological and experimental validation of the mechanism of Chaihu Guizhi Ganjiang decoction (柴胡桂枝干姜汤) regulating T helper cell 17/regulatory T cell balance to improve autoimmune hepatitis [J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 110-118. |
| [2] | SUN Yan, ZHOU Haozheng, FENG Jianhui, LIN Zikai, HE Jie, WANG Zhenhua, GUO Yuting, WEN Shaohong, LI Gang. Baitouweng Tang (白头翁汤) alleviates dextran sulfate sodium-induced ulcerative colitis in mice: a network pharmacology combined with experimental study [J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 119-126. |
| [3] | GAO Yinli, ZHANG Linhua, ZHANG Yaqiang, ZHANG Liuhe, ZHI Nan, LIAN Haihong. Pinggan Yuyin Qingre formula (平肝育阴清热方) ameliorates meibomian gland dysfunction through the interleukin-17/nuclear factor kappa B pathway based on network pharmacology and experimental validation [J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 172-182. |
| [4] | LI Junwei, LIU Xinyue, MOU Xuan, XIE Hongliang, GAO Yating, ZHANG Aolin, HE Tao, YEUNG Tszching, TANG Chao, WU Xiaoke, FAN Xiaohui, WANG Chichiu, LI Lu. Therapeutic effect and anti-inflammatory mechanism of modified Shoutai pills (改良寿胎丸) against lipopolysaccharide-induced miscarriage in mice [J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 22-33. |
| [5] | LI Dongqi, WANG Tongxing, WANG Zixuan, YAN Yihui, LI Jie, GU Jiaojiao, LI Cuiru, WANG Aili, SUN Lingling, MENG Yongjie, ZHANG Zeyu, HOU Yunlong, GAO Huailin. Improving glucose tolerance in obese rats: the role of Jinlida granules (津力达颗粒 ) in gut microbiota modulation [J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 62-72. |
| [6] | WEI Mumu, YAN Xiaoyu, ZHANG Yujing, ZHANG Xinxin, YAN Yongbin. Soufeng Yuchuan formula (搜风愈喘方) alleviates asthma airway inflammation and suppresses the progression of asthma by inhibiting ferroptosis in airway epithelial cells [J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 73-84. |
| [7] | LEI Xiaochun, LIU Cuizhen, LIN Xiujuan, XIE Xiangyu, KE Wei, QIU Zhenwen, TANG Hongmei, HUANG Yushen, ZHANG Lijuan, HUANG Baoyuan, WAN Xin, LI Detang. Effect of improved Yupingfeng powder prescription (玉屏风散加味) on interleukin-33/suppression of tumorigenicity 2 pathway in mice with ovalbumins-induced allergic rhinitis [J]. Journal of Traditional Chinese Medicine, 2025, 45(6): 1215-1227. |
| [8] | LI Keyao, SHU Ye, CHANG Jing, TANG Jianping, ZHANG Litao, WEI Zhu. Psoriasis intervention by Huai’er (Trametes): unveiling novel targets via network pharmacology [J]. Journal of Traditional Chinese Medicine, 2025, 45(6): 1317-1329. |
| [9] | CHENG Ziqi, DONG Xin, Temuribagen , XU Caimeng, HU Shaonan, CHEN Qianwen, WANG Yuewu, WANG Haibo, HE Xiaoyu, XUE Dan, XUE Peifeng. Exploration of the mechanism of the Mongolian medicine Tonglaga-5 (通拉嘎-5) for the treatment of n-methyl-n′-nitro-n-nitrosoguanidine-induced chronic atrophic gastritis based on network pharmacology and metabolomics [J]. Journal of Traditional Chinese Medicine, 2025, 45(6): 1366-1375. |
| [10] | WANG Wei, LONG Qi, FU Ling, WU Haiqiao. Jianpi Yifei Tongluo recipe (健脾益肺通络方剂) attenuates inflammation by promoting the expression of interferon regulatory factor 4 in the rat model of chronic obstructive pulmonary disease [J]. Journal of Traditional Chinese Medicine, 2025, 45(5): 1048-1058. |
| [11] | LI Weijia, LU Jing, MA Chao, LIU Mengmeng, PEI Ke, CHEN Hongyan, LIN Zhe, LYU Guangfu. Hamayou (Oviductus Ranae) protein hydrolysate ameliorates depression by regulating the mitogen-activated protein kinase pathway [J]. Journal of Traditional Chinese Medicine, 2025, 45(3): 493-507. |
| [12] | DIAO Ruohan, DUAN Xingwu, LI Lingling, QU Tiange, FENG Huishang, CHEN Guangshan. Intervention and mechanism of Xiaoyin Anshen Yin (消银安神饮) in treatment of psoriasis combined with sleep disorders [J]. Journal of Traditional Chinese Medicine, 2025, 45(3): 552-560. |
| [13] | MIN Yu, ZHENG Meifeng, SUN Ju, PENG Zetong, CAO Zhixian, HUANG Xiaohua. Systematic acupuncture explains acupuncture at Baihui (GV20) and Fengchi (GB20) targeting the inflammatory response to regulate migraine [J]. Journal of Traditional Chinese Medicine, 2025, 45(3): 610-617. |
| [14] | YUAN Jiayao, WU Suhui, MENG Yufan, LI Hanbing, LI Genlin, XU Jiangyan. Yishen Tongluo formula (益肾通络方) ameliorates kidney injury via modulating inflammation and apoptosis in streptozotocin-induced diabetic kidney disease mice [J]. Journal of Traditional Chinese Medicine, 2025, 45(2): 254-265. |
| [15] | HUANG Haiyang, ZHU Shumin, ZHONG Shaowen, LIU Ying, HOU Shaozhen, GAO Jie, OU Jianzhao, DONG Mingguo, NING Weimin. Fuzheng Xuanfei Huashi prescription (扶正宣肺化湿方) suppresses inflammation in lipopolysaccharide-induced lung injury in mice via toll-like recptor 4/nuclear transcription factor κB and cyclooxygenase-2/prostaglandin E2 pathway [J]. Journal of Traditional Chinese Medicine, 2025, 45(2): 272-280. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

