Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (1): 62-72.DOI: 10.19852/j.cnki.jtcm.2026.01.006
• Original Articles • Previous Articles Next Articles
Improving glucose tolerance in obese rats: the role of Jinlida granules (津力达颗粒 ) in gut microbiota modulation
LI Dongqi1, WANG Tongxing2,3, WANG Zixuan4, YAN Yihui1, LI Jie1, GU Jiaojiao1, LI Cuiru1, WANG Aili5, SUN Lingling6, MENG Yongjie1, ZHANG Zeyu1, HOU Yunlong2,3( ), GAO Huailin7(
), GAO Huailin7( )
)
- 1 Graduate School, Hebei University of Chinese Medicine, Shijiazhuang 050090, China
2 National Key Laboratory for Innovation and Transformation of Luobing Theory, Shijiazhuang 050035, China
3 Hebei Yiling Pharmaceutical Research Institute, Key Laboratory of State Administration of TCM (Cardio-Cerebral Vessel Collateral Diseases), Shijiazhuang 050035, China
4 Graduate School, Hebei Medical University, Shijiazhuang 050017, China
5 Department of Medical Oncology, Hebei Yiling Hospital, Shijiazhuang 050091, China
6 Graduate School, Henan University of Traditional Chinese Medicine, Zhengzhou 450046, China
7 Graduate School, Hebei University of Chinese Medicine, Shijiazhuang 050090, China; Endocrinology Department, Hebei Yiling Hospital, Shijiazhuang 050091, China
-
Received:2024-08-22Accepted:2025-05-28Online:2026-02-15Published:2026-01-28 -
Contact:HOU Yunlong, National Key Laboratory for Innovation and Transformation of Luobing Theory, Shijiazhuang 050035, China; Hebei Yiling Pharmaceutical Research Institute, Key Laboratory of State Administration of TCM (Cardio-Cerebral Vessel Collateral Diseases), Shijiazhuang 050035, China,houyunlonghrb@hotmail.com ; GAO Huailin, Graduate School, Hebei University of Chinese Medicine, Shijiazhuang 050090, China; Endocrinology Department, Hebei Yiling Hospital, Shijiazhuang 050091, China,gaohuailin@126.com ;Telephone: +86-18503210603 -
About author:LI Dongqi and WANG Tongxing are co-first authors and contributed equally to this work -
Supported by:National Key Research and Development Program 'Modernization Research of Traditional Chinese Medicine': Cardiovascular Event Chain (Metabolic Syndrome, Atherosclerosis, Myocardial Infarction, Arrhythmia, Heart Failure)(2017YFC700500);Key R&D Program of Hebei: Traditional Chinese Medicine Innovation Project: Clinical Research on the Treatment of Diabetes Foot with Collateral Drugs and the Mechanism of Its Influence on Collateral Vessel Reconstruction(223777155D);Scientific Research Project of Hebei Provincial Administration of Traditional Chinese Medicine: Clinical Study on Jinlida Granules in Treating Intestinal Dysfunction of diabetes and Its Effect on Short Chain Fatty Acids(2023179);Scientific Research Project of Hebei Provincial Administration of Traditional Chinese Medicine: Clinical Study on Tongluo Therapy for Diabetes Foot and Its Influence on Microcirculation(2018200)
Cite this article
LI Dongqi, WANG Tongxing, WANG Zixuan, YAN Yihui, LI Jie, GU Jiaojiao, LI Cuiru, WANG Aili, SUN Lingling, MENG Yongjie, ZHANG Zeyu, HOU Yunlong, GAO Huailin. Improving glucose tolerance in obese rats: the role of Jinlida granules (津力达颗粒 ) in gut microbiota modulation[J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 62-72.
share this article
| Group | n | 0 week | 4 weeks | 8 weeks | 12 weeks | 16 weeks | 20 weeks | 24 weeks |
|---|---|---|---|---|---|---|---|---|
| Control | 5 | 751±100 | 782±136 | 816±143 | 810±143 | 845±135 | 844±127 | 876±129 |
| HFD | 5 | 738±61 | 821±65 | 875±129 | 876±92 | 940±66 | 970±70 | 1032±80c |
| L-JLD | 7 | 727±93 | 751±104 | 774±95a | 791±102 | 813±99a | 861±105 | 898±115 |
| H-JLD | 7 | 702±68 | 742±81 | 747±75a | 763±85a | 790±93a | 799±108b | 827±128b |
| Sitagliptin | 7 | 738±52 | 790±63 | 815±86 | 840±92 | 888±106 | 907±110 | 950±124 |
Table 1 JLD reduced body weight gain (g, $\bar{x} \pm s$)
| Group | n | 0 week | 4 weeks | 8 weeks | 12 weeks | 16 weeks | 20 weeks | 24 weeks |
|---|---|---|---|---|---|---|---|---|
| Control | 5 | 751±100 | 782±136 | 816±143 | 810±143 | 845±135 | 844±127 | 876±129 |
| HFD | 5 | 738±61 | 821±65 | 875±129 | 876±92 | 940±66 | 970±70 | 1032±80c |
| L-JLD | 7 | 727±93 | 751±104 | 774±95a | 791±102 | 813±99a | 861±105 | 898±115 |
| H-JLD | 7 | 702±68 | 742±81 | 747±75a | 763±85a | 790±93a | 799±108b | 827±128b |
| Sitagliptin | 7 | 738±52 | 790±63 | 815±86 | 840±92 | 888±106 | 907±110 | 950±124 |
| Group | n | 0 min | 30 min | 60 min | 120 min |
|---|---|---|---|---|---|
| Control | 5 | 6.38±0.63 | 9.42±1.62 | 9.50±1.11 | 8.52±1.19 |
| HFD | 5 | 6.20±0.79 | 8.22±1.21 | 9.70±1.37 | 9.90±1.33c |
| L-JLD | 7 | 6.06±0.94 | 7.67±1.16a | 8.14±0.70a | 8.46±1.13a |
| H-JLD | 7 | 5.86±0.27 | 7.36±0.83b | 8.66±0.81 | 7.87±0.95b |
| Sitagliptin | 7 | 6.00±0.37 | 7.09±0.97b | 7.69±1.23b | 7.93±0.60b |
Table 2 JLD improved glucose tolerance (mmol/L, $\bar{x} \pm s$)
| Group | n | 0 min | 30 min | 60 min | 120 min |
|---|---|---|---|---|---|
| Control | 5 | 6.38±0.63 | 9.42±1.62 | 9.50±1.11 | 8.52±1.19 |
| HFD | 5 | 6.20±0.79 | 8.22±1.21 | 9.70±1.37 | 9.90±1.33c |
| L-JLD | 7 | 6.06±0.94 | 7.67±1.16a | 8.14±0.70a | 8.46±1.13a |
| H-JLD | 7 | 5.86±0.27 | 7.36±0.83b | 8.66±0.81 | 7.87±0.95b |
| Sitagliptin | 7 | 6.00±0.37 | 7.09±0.97b | 7.69±1.23b | 7.93±0.60b |

Figure 1 JLD improved the histopathology of colon tissues and ameliorated inflammation in colon tissues A: representative HE staining images of colon tissues (× 5 and × 20, scale bar = 500 μm). Intestinal epithelial cells are indicated with black arrows and the spacing of colonic glands is indicated with red arrows; B: Representative Masson staining of colon sections (× 10, scale bar = 250 μm); C: representative immunohistochemical staining for F4/80 in colon sections (× 20 and × 60, scale bar = 100 μm). A1, A6,B1, C1, C6: Control group; A2, A7, B2, C2, C7: HFD group; A3, A8, B3, C3, C8: L-JLD group; A4, A9, B4, C4, C9: H-JLD group; A5, A10, B5, C5, C10: Sitagliptin group. Control: fed with normal food + the same volume of 5% CMC was given by gavage for 25 weeks; HFD: fed with HFD + the same volume of 5% CMC was given by gavage for 25 weeks; L-JLD: fed with HFD + received an intragastric administration of JLD at a dosage of 1.5 g·kg?1·d?1 for 25 weeks ; H-JLD: fed with HFD + received an intragastric administration of JLD at a dosage of 3 g·kg?1·d?1 for 25 weeks ; Sitagliptin: fed with HFD + received an intragastric administration of sitagliptin at a dosage of 10 mg·kg?1·d?1 for 25 weeks. HFD: high-fat diet; L-JLD: low dose Jinlida; H-JLD: high dose Jinlida; HE: hematoxylin-eosin; CMC: carboxymethyl cellulose.
| Group | n | Collagen fibre | F4/80 |
|---|---|---|---|
| Control | 4 | 8.85±2.20 | 0.917±0.144 |
| HFD | 4 | 14.26±2.54a | 1.186±0.117c |
| L-JLD | 4 | 9.34±1.94b | 0.775±0.164b |
| H-JLD | 4 | 9.03±1.42b | 0.774±0.050b |
| Sitagliptin | 4 | 9.67±1.05b | 0.843±0.105b |
Table 3 The proportion of collagen fibre area and F4/80 positive area in the colon tissue of rats in each group (%,$\bar{x} \pm s$)
| Group | n | Collagen fibre | F4/80 |
|---|---|---|---|
| Control | 4 | 8.85±2.20 | 0.917±0.144 |
| HFD | 4 | 14.26±2.54a | 1.186±0.117c |
| L-JLD | 4 | 9.34±1.94b | 0.775±0.164b |
| H-JLD | 4 | 9.03±1.42b | 0.774±0.050b |
| Sitagliptin | 4 | 9.67±1.05b | 0.843±0.105b |
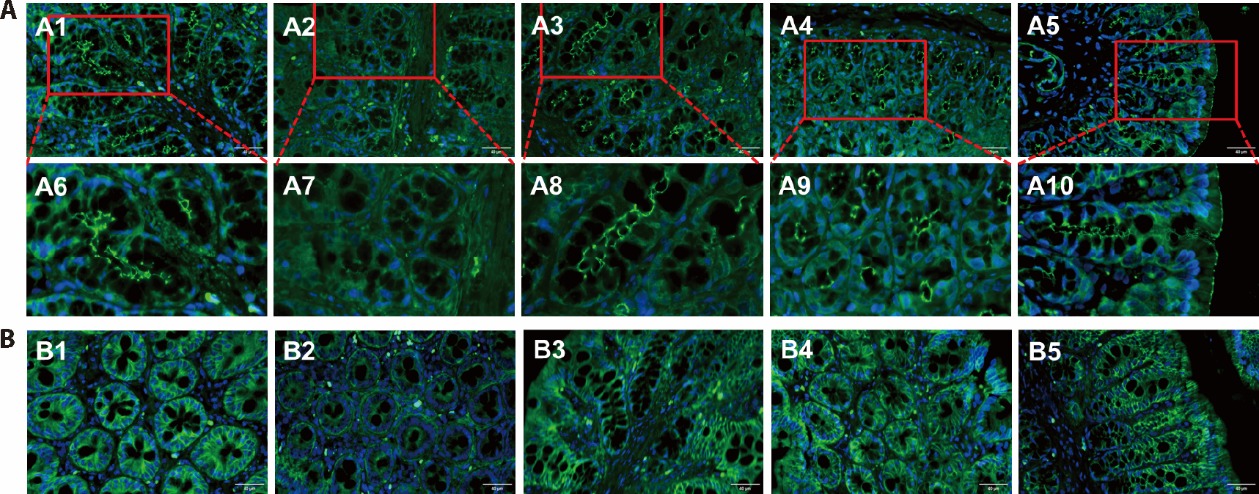
Figure 2 JLD protected the intestinal barrier in HFD-induced obese rats A: immunofluorescence of colonic ZO-1 (× 40 and × 80, scale bar = 40 μm); B: immunofluorescence of colonic Claudin-1 (× 40, scale bar = 40 μm). A1, B1: Control group; A2, B2: HFD group; A3, B3: L-JLD group; A4, B4: H-JLD group; A5, B5: Sitagliptin group. Control: fed with normal food + the same volume of 5% CMC was given by gavage for 25 weeks; HFD: fed with HFD + the same volume of 5% CMC was given by gavage for 25 weeks; L-JLD: fed with HFD + received an intragastric administration of JLD at a dosage of 1.5 g·kg?1·d?1 for 25 weeks; H-JLD: fed with HFD + received an intragastric administration of JLD at a dosage of 3 g·kg?1·d?1 for 25 weeks; Sitagliptin: fed with HFD + received an intragastric administration of sitagliptin at a dosage of 10 mg·kg?1·d?1 for 25 weeks. JLD: Jinlida granules; HFD: high-fat diet; ZO-1: zonula occludens-1; CMC: carboxymethyl cellulose; L-JLD: low dose Jinlida; H-JLD: high dose Jinlida.
| Group | n | ZO-1 | Claudin-1 |
|---|---|---|---|
| Control | 4 | 101.1±5.0 | 106.1±11.3 |
| HFD | 4 | 71.0±6.7a | 52.8±9.0a |
| L-JLD | 4 | 87.4±9.1b | 87.7±9.3b |
| H-JLD | 4 | 87.7±6.1b | 108.0±7.4c |
| Sitagliptin | 4 | 91.5±1.0b | 85.9±9.6b |
Table 4 Average fluorescence intensity of ZO-1 and Claudin-1 protein expression in colon tissue of rats in each group ($\bar{x} \pm s$)
| Group | n | ZO-1 | Claudin-1 |
|---|---|---|---|
| Control | 4 | 101.1±5.0 | 106.1±11.3 |
| HFD | 4 | 71.0±6.7a | 52.8±9.0a |
| L-JLD | 4 | 87.4±9.1b | 87.7±9.3b |
| H-JLD | 4 | 87.7±6.1b | 108.0±7.4c |
| Sitagliptin | 4 | 91.5±1.0b | 85.9±9.6b |

Figure 3 Analysis of the colon tissue transcriptome from the H-LJD and HFD groups A: volcano plot showing the DEGs in the H-JLD/HFD comparison; B: heatmap for the H-JLD/HFD comparison. C: top 20 KEGG pathways for the H-JLD/HFD comparison. The genes behind each bar represent the DEGs involved in the pathway. The red and green genes represent upregulated and downregulated DEGs in the H-JLD/HFD comparison, respectively. HFD: fed with HFD + the same volume of 5% CMC was given by gavage for 25 weeks; H-JLD: fed with HFD + received an intragastric administration of JLD at a dose of 3 g·kg?1·d?1 for 25 weeks; DEGs: differentially expressed genes; HFD: high-fat diet; H-JLD: high dose Jinlida. KEGG: kyoto encyclopedia of genes and genomes; CMC: carboxymethyl cellulose; FDR: false discovery rate; FC: fold change. DEGs were selected with the following conditional thresholds: q value (FDR) < 0.05 and absolute FC ≥ 2. n = 5 for each group.

Figure 4 JLD altered the diversity and composition of the gut microbiota in HFD-induced obese rats A: alpha-diversity measured by the Simpson index; B: NMDS plot based on the Bray-Curtis index; C: phylum-level distribution of the gut microbiota; D: relative abundances of Bacteroides species; E: genus-level distribution of the gut microbiota; F: relative abundances of Turicibacter; G: relative abundances of Faecalibaculum; H: relative abundances of Coriobacteriaceae; I: relative abundances of Lactobacillus reuteri; J: LDA at the genus and species levels. Control: fed with normal food + the same volume of 5% CMC was given by gavage for 25 weeks; HFD: fed with HFD + the same volume of 5% CMC was given by gavage for 25 weeks; H-JLD: fed with HFD + received an intragastric administration of JLD at a dose of 3 g·kg?1·d?1 for 25 weeks. JLD: Jinlida granules; HFD: high-fat diet; NMDS: nonmetric multidimensional scaling; CMC: carboxymethyl cellulose; LDA: linear discriminant analysis. Statistical analyses were measured using the least significant difference test and Tamhane's T2 test for pairwise comparisons between groups. Compared with control group, aP < 0.001, cP < 0.05; compared with HFD group, bP < 0.001, dP < 0.05. Control and HFD groups, n = 5; H-JLD group, n = 6.
| 1. |
Masood B, Moorthy M. Causes of obesity: a review. Clin Med (Lond) 2023; 23: 284-91.
DOI PMID |
| 2. |
Saltiel AR, Olefsky JM. Inflammatory mechanisms linking obesity and metabolic disease. J Clin Invest 2017; 127: 1-4.
DOI PMID |
| 3. |
Blüher M. Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol 2019; 15: 288-98.
DOI PMID |
| 4. |
Echouffo-Tcheugui JB, Perreault L, Ji L, Dagogo-Jack S. Diagnosis and management of prediabetes: a review. JAMA 2023; 329: 1206-16.
DOI PMID |
| 5. |
La Sala L, Pontiroli AE. Prevention of diabetes and cardiovascular disease in obesity. Int J Mol Sci 2020; 21: 8178.
DOI URL |
| 6. |
El Meouchy P, Wahoud M, Allam S, Chedid R, Karam W, Karam S. Hypertension related to obesity: pathogenesis, characteristics and factors for control. Int J Mol Sci 2022; 23: 12305.
DOI URL |
| 7. |
Ahmed B, Sultana R, Greene MW. Adipose tissue and insulin resistance in obese. Biomed Pharmacother 2021; 137: 111315.
DOI PMID |
| 8. |
Recena Aydos L, Aparecida do Amaral L, Serafim de Souza R, Jacobowski AC, Freitas Dos Santos E, Rodrigues Macedo ML. Nonalcoholic fatty liver disease induced by high-fat diet in C57bl/6 models. Nutrients 2019; 11: 3067.
DOI URL |
| 9. |
Fu Y, Wang Y, Gao H, et al. Associations among dietary omega-3 polyunsaturated fatty acids, the gut microbiota, and intestinal immunity. Mediators Inflamm 2021; 2021: 8879227.
DOI URL |
| 10. |
de Wit N, Derrien M, Bosch-Vermeulen H, et al. Saturated fat stimulates obesity and hepatic steatosis and affects gut microbiota composition by an enhanced overflow of dietary fat to the distal intestine. Am J Physiol Gastrointest Liver Physiol 2012; 303: G589-99.
DOI URL |
| 11. |
Gonçalves RV, Santos JDB, Silva NS, et al. Trans-fatty acids aggravate anabolic steroid-induced metabolic disturbances and differential gene expression in muscle, pancreas and adipose tissue. Life Sci 2019; 232: 116603.
DOI URL |
| 12. |
Malesza IJ, Malesza M, Walkowiak J, et al. High-fat, western-style diet, systemic inflammation, and gut microbiota: a narrative review. Cells 2021; 10: 3164.
DOI URL |
| 13. |
Bach Knudsen KE, Lærke HN, Hedemann MS, et al. Impact of diet-modulated butyrate production on intestinal barrier function and inflammation. Nutrients 2018; 10: 1499.
DOI URL |
| 14. |
Bahlouli W, Breton J, Lelouard M, et al. Stress-induced intestinal barrier dysfunction is exacerbated during diet-induced obesity. J Nutr Biochem 2020; 81: 108382.
DOI URL |
| 15. |
Dang Y, Ma C, Chen K, et al. The effects of a high-fat diet on inflammatory bowel disease. Biomolecules 2023; 13: 905.
DOI URL |
| 16. |
Jia W, Weng J, Zhu D, et al. Standards of medical care for type 2 diabetes in China 2019. Diabetes Metab Res Rev 2019; 35: e3158.
DOI URL |
| 17. |
Liu Y, Song A, Zang S, et al. Jinlida reduces insulin resistance and ameliorates liver oxidative stress in high-fat fed rats. J Ethnopharmacol 2015; 162: 244-52.
DOI PMID |
| 18. |
Zhou HR, Wang TX, Hao YY, et al. Jinlida granules reduce obesity in db/db mice by activating beige adipocytes. Biomed Res Int 2022; 2022: 4483009.
DOI URL |
| 19. |
Zhang H, Hao Y, Wei C, et al. Chinese medicine Jinlida granules improve high-fat-diet induced metabolic disorders using activation of brown adipose tissue in mice. Biomed Pharmacother 2019; 114: 108781.
DOI |
| 20. | Pan J, Xu Y, Chen S, et al. The effectiveness of Traditional Chinese Medicine Jinlida granules on glycemic variability in newly diagnosed type 2 diabetes: a double-blinded, randomized trial. J Diabetes Res 2021; 2021: 6303063. |
| 21. | Shi YL, Liu WJ, Zhang XF, et al. Effect of Chinese herbal medicine Jinlida granule in treatment of patients with impaired glucose tolerance. Chin Med J (Engl) 2016; 129: 2281-6. |
| 22. |
Tian J, Lian F, Yang L, Tong X. Evaluation of the Chinese herbal medicine Jinlida in type 2 diabetes patients based on stratification: results of subgroup analysis from a 12-week trial. J Diabetes 2018; 10: 112-20.
DOI PMID |
| 23. |
Hao YY, Cui WW, Gao HL, et al. Jinlida granules ameliorate the high-fat-diet induced liver injury in mice by antagonising hepatocytes pyroptosis. Pharm Biol 2022; 60: 274-81.
DOI URL |
| 24. |
Vuksan V, Xu ZZ, Jovanovski E, et al. Efficacy and safety of American ginseng (Panax quinquefolius L.) extract on glycemic control and cardiovascular risk factors in individuals with type 2 diabetes: a double-blind, randomized, cross-over clinical trial. Eur J Nutr 2019; 58: 1237-45.
DOI PMID |
| 25. |
Luo J, Chen Z, Guo Q, Chai Y, Bao Y. Effects of saponins isolated from Polygonatum sibiricum on H2O2-induced oxidative damage in RIN-m5F cells and its protective effect on pancreas. Food Chem Toxicol 2023; 175: 113724.
DOI URL |
| 26. |
Xu J, Wang Y, Xu DS, Ruan KF, Feng Y, Wang S. Hypoglycemic effects of MDG-1, a polysaccharide derived from ophiopogon japonicas, in the ob/ob mouse model of type 2 diabetes mellitus. Int J Biol Macromol 2011; 49: 657-62.
DOI PMID |
| 27. |
Han B, Sun H, Yang X, et al. Ultrasound-assisted enzymatic extraction of Corni Fructus alpha-glucosidase inhibitors improves insulin resistance in HepG2 cells. Food Funct 2021; 12: 9808-19.
DOI PMID |
| 28. |
Miao H, Zhao YH, Vaziri ND, et al. Lipidomics biomarkers of diet-induced hyperlipidemia and its treatment with poria cocos. J Agric Food Chem 2016; 64: 969-79.
DOI URL |
| 29. |
Cui L, Liu M, Chang X, Sun K. The inhibiting effect of the Coptis chinensis polysaccharide on the type II diabetic mice. Biomed Pharmacother 2016; 81: 111-9.
DOI PMID |
| 30. |
Feng M, Liu F, Xing J, Zhong Y, Zhou X. Anemarrhena saponins attenuate insulin resistance in rats with high-fat diet-induced obesity by the IRS-1/PI3K/AKT pathway. J Ethnopharmacol 2021; 277: 114251.
DOI URL |
| 31. |
Jia Q, Zhu R, Tian Y, et al. Salvia miltiorrhiza in diabetes: a review of its pharmacology, phytochemistry, and safety. Phytomedicine 2019; 58: 152871.
DOI URL |
| 32. |
Yang L, Chen J, Lu H, et al. Pueraria lobata for diabetes mellitus: past, present and future. Am J Chin Med 2019; 47: 1419-44.
DOI URL |
| 33. |
Zhang Y, Jin D, An X, Duan L, Duan Y, Lian F. Lychee seed as a potential hypoglycemic agent, and exploration of its underlying mechanisms. Front Pharmacol 2021; 12: 737803.
DOI URL |
| 34. |
Gu W, Wang Y, Zeng L, et al. Polysaccharides from Polygonatum kingianum improve glucose and lipid metabolism in rats fed a high fat diet. Biomed Pharmacother 2020; 125: 109910.
DOI PMID |
| 35. |
Wang JH, Bose S, Kim HG, Han KS, Kim H. Fermented Rhizoma atractylodis macrocephalae alleviates high fat diet-induced obesity in association with regulation of intestinal permeability and microbiota in rats. Sci Rep 2015; 5: 8391.
DOI |
| 36. |
Yan D, Fan P, Sun W, et al. Anemarrhena asphodeloides modulates gut microbiota and restores pancreatic function in diabetic rats. Biomed Pharmacother 2021; 133: 110954.
DOI PMID |
| 37. |
Kim D, Langmead B, Salzberg SL. HISAT: a fast spliced aligner with low memory requirements. Nat Methods 2015; 12: 357-60.
DOI PMID |
| 38. |
Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 2014; 15: 550.
DOI URL |
| 39. |
The Gene Ontology resource: enriching a gold mine. Nucleic Acids Res 2021; 49: D325-34.
DOI PMID |
| 40. |
Kanehisa M, Furumichi M, Sato Y, Ishiguro-Watanabe M, Tanabe M. KEGG: integrating viruses and cellular organisms. Nucleic Acids Res 2021; 49: D545-51.
DOI PMID |
| 41. |
Subramanian A, Tamayo P, Mootha VK, et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A 2005; 102: 15545-50.
DOI URL |
| 42. |
Kechin A, Boyarskikh U, Kel A, Filipenko M. cutPrimers: a new tool for accurate cutting of primers from reads of targeted next generation sequencing. J Comput Biol 2017; 24: 1138-43.
DOI PMID |
| 43. |
Langmead B, Salzberg SL. Fast gapped-read alignment with Bowtie 2. Nat Methods 2012; 9: 357-9.
DOI PMID |
| 44. |
Buchfink B, Xie C, Huson DH. Fast and sensitive protein alignment using DIAMOND. Nat Methods 2015; 12: 59-60.
DOI PMID |
| 45. |
Wu F, Shao Q, Hu M, et al. Wu-mei-wan ameliorates chronic colitis-associated intestinal fibrosis through inhibiting fibroblast activation. J Ethnopharmacol 2020; 252: 112580.
DOI URL |
| 46. |
Lee JS, Tato CM, Joyce-Shaikh B, et al. Interleukin-23-independent IL-17 production regulates intestinal epithelial permeability. Immunity 2015; 43: 727-38.
DOI PMID |
| 47. |
Tong Y, Gao H, Qi Q, et al. High fat diet, gut microbiome and gastrointestinal cancer. Theranostics 2021; 11: 5889-910.
DOI PMID |
| 48. |
Beam A, Clinger E, Hao L. Effect of diet and dietary components on the composition of the gut microbiota. Nutrients 2021; 13: 2795.
DOI URL |
| 49. |
Wang Y, Fei Y, Liu L, et al. Polygonatum odoratum polysaccharides modulate gut microbiota and mitigate experimentally induced obesity in rats. Int J Mol Sci 2018; 19: 3587.
DOI URL |
| 50. |
Hosomi K, Saito M, Park J, et al. Oral administration of blautia wexlerae ameliorates obesity and type 2 diabetes via metabolic remodeling of the gut microbiota. Nat Commun 2022; 13: 4477.
DOI PMID |
| 51. |
Jin Z, Fang Z, Pei Z, et al. A low molecular weight brown algae laminaria japonica glycan modulation of gut microbiota and body weight in mice. Food Funct 2021; 12: 12606-20.
DOI URL |
| 52. |
Louis S, Tappu RM, Damms-Machado A, Huson DH, Bischoff SC. Characterization of the gut microbial community of obese patients following a weight-loss intervention using whole metagenome shotgun sequencing. PLoS One 2016; 11: e0149564.
DOI URL |
| [1] | YUAN Menghua, SHAO Jianbin, YANG Yawei, ZHAO Yan, HU Xingang. Combined acupuncture and herb treatment improves intestinal flora inabdominally obese subjects based on 16s rRNA sequencing: a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 205-210. |
| [2] | ZHANG Xiaosi, ZHANG Shuangyuan, CHEN Hanqing, LIN Zhengdao, XIE Chune, LI Junxiang, LI Xiaohong. Hewei Jiangni recipe (和胃降逆方) improved the quality of life in patients with cold-heat mixed nonerosive reflux disease: a randomized, double-blinded study [J]. Journal of Traditional Chinese Medicine, 2025, 45(5): 1106-1118. |
| [3] | CHOI You Yeon, JIN Seong chul, KIM Mi Hye, BAEK Hee Kyung, KIM Dong Hyun, OH Sung Hyuk, YANG Woong Mo. Exploring the therapeutic potential of Morus alba Linne extract in targeting localized adiposity [J]. Journal of Traditional Chinese Medicine, 2025, 45(5): 970-978. |
| [4] | YE Wujie, YANG Yawei, ZHANG Da, TANG Ling, CUI Minying, FU Bin, ZHANG Meng, HU Xingang, ZHAO Yan. Effectiveness of combining Qingyanyin formulated granules (轻燕饮配方颗粒) with press needles in treating abdominal obesity: a multicenter randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2025, 45(1): 107-114. |
| [5] | YANG Chunyan, LUO Jia, PENG Weijie, DAI Weibo. Huaiyu pill (槐榆片) alleviates inflammatory bowel disease in mice via blocking toll like receptor 4/ myeloid differentiation primary response gene 88/ nuclear factor kappa B subunit 1 pathway [J]. Journal of Traditional Chinese Medicine, 2024, 44(5): 916-925. |
| [6] | ZHU Linghui, SUN Ziwei, GUAN Yuanyuan, LIU Meiyi, ZHENG Yi, YU Ruoxi, WANG Qi, LI Lingru. Differences in vascular endothelial function and serum proteome between obese people with phlegm-dampness constitution and balanced constitution [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 188-196. |
| [7] | QIN Xihui, PANG Jianli, XIONG Guan, FENG Jie. Bo′s abdominal acupuncture improves disordered metabolism in obese type 2 diabetic rats through regulating fibroblast growth factor 21 and its related adipokines [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1200-1208. |
| [8] | ZHAO HuiYan, JUN Purumea, LEE Chaewon, HAN Chang-Hyun. Acupoint catgut embedding for simple obesity in animal studies: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 860-867. |
| [9] | JIANG Li, FU Qiang, WANG Shidong, ZHAO Jinxi, CHEN Yu, LI Jiayue, XIAO Yonghua, HUANG Weijun, SUN Ruixi, XIAO Yao, SHEN Aijia, WANG Junheng, LIU Jiangteng, FU Xiaozhe, LI Yuanyuan, ZHAO Yu, XUE Taiqi. Effects of Shenlian formula (参连方) on microbiota and inflammatory cytokines in adults with type 2 diabetes: a double-blind randomized clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 760-769. |
| [10] | ZHANG Xinghe, LI Qifu, YI Rong, XING Chonghui, JIN Yuhao, MENG Jiangqiong, FENG Jialei, ZHAO Siwen, LIANG Fanrong, GUO Taipin. Effect of catgut embedding at acupoints versus non-acupoints in abdominal obesity: a randomized clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 780-786. |
| [11] | SUN Mengzhu, ZHANG Yujie, SONG Yafang, GUO Jing, WANG Yuhang, XIN Chen, GU Dongmei, SUN Jianhua, PEI Lixia. Electroacupuncture alleviates water avoidance stress-induced irritable bowel syndrome in mice by improving intestinal barrier functions and suppressing the expression of inflammatory cytokines [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 494-500. |
| [12] | CHEN Wenlin, LIANG Fang, ZHANG Yuncheng, LV Jinzhen, YANG Daguo. Effects of Zhenggan Huayu decoction (正肝化瘀方) combined with entecavir on gut microbiota in patients with chronic hepatitis B fibrosis [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 559-567. |
| [13] | WEI Jiali, LAI Lily, LIN Zhechao, LIU Jianping, HAN Mei. Acupoint catgut embedding versus acupuncture for simple obesity: a systematic review and Meta-analysis of randomized controlled trials [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 839-847. |
| [14] | YE Wujie, XING Jingyu, YU Zekai, HU Xingang, ZHAO Yan. Systematic review and Meta-analysis of acupuncture and acupoint catgut embedding for the treatment of abdominal obesity [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 848-857. |
| [15] | YANG Liping, YU Xinglin, ZHANG Chao, CHEN Pu, DUAN Xiaohua. Yajieshaba prevents lipopolysaccharide-induced intestinal barrier injury via anti-inflammatory and anti-apoptosis [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 707-714. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

