Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (6): 1092-1102.DOI: 10.19852/j.cnki.jtcm.2023.06.004
• Research Articles • Previous Articles Next Articles
Hepatic transcriptome delineates the therapeutic effects of Sanren Tang (三仁汤) on high-fat diet-induced non-alcoholic fatty liver disease
YIN Yixiao1,2,3, TANG Hao1,2,3, FANG Yi1,2,3, LIU Wei1,2,3, WANG Jun1,2,3, HU Yiyang4,5,6, PENG Jinghua1,2,3( )
)
- 1 Institute of Liver diseases, Shuguang Hospital affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China
2 Key Laboratory of Liver and Kidney Diseases (Shanghai University of Traditional Chinese Medicine), Ministry of Education, Shanghai 201203, China
3 Shanghai Key Laboratory of Traditional Chinese Clinical Medicine, Shanghai 201203, China
4 Institute of Clinical Pharmacology, Shuguang Hospital affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, 201203, China
5 Key Laboratory of Liver and Kidney Diseases (Shanghai University of Traditional Chinese Medicine), Ministry of Education, Shanghai 201203, China
6 Shanghai Key Laboratory of Traditional Chinese Clinical Medicine, Shanghai 201203, China
-
Received:2022-09-11Accepted:2022-12-25Online:2023-10-25Published:2023-11-01 -
Contact:PENG Jinghua, Institute of Liver diseases, Shuguang Hospital affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, 201203, China; Key Laboratory of Liver and Kidney Diseases (Shanghai University of Traditional Chinese Medicine), Ministry of Education, Shanghai 201203, China; Shanghai Key Laboratory of Traditional Chinese Clinical Medicine, Shanghai 201203, China. pengjinghua2004@163.com. Telephone: +86-21-20256526 -
Supported by:Science and Technology Commission Shanghai Municipality: Exploring the Mechanism of Qushi Huayu Formula in Treating Non-alcoholic Fatty Liver Disease from the Perspective of Intestinal Barrier Function(17PJ1408900);Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine: “Si Ming” Scholar(SGXZ-201911)
Cite this article
YIN Yixiao, TANG Hao, FANG Yi, LIU Wei, WANG Jun, HU Yiyang, PENG Jinghua. Hepatic transcriptome delineates the therapeutic effects of Sanren Tang (三仁汤) on high-fat diet-induced non-alcoholic fatty liver disease[J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1092-1102.
share this article
| Group | n | Body weight (g) | Food intake (g/mouse per day) | ||||
|---|---|---|---|---|---|---|---|
| 0 week | 4th week | 8th week | 12th week | 16th week | |||
| Control | 10 | 22.94±0.71 | 24.94±1.67 | 27.22±2.22 | 29.04±2.59 | 30.10±2.97 | 2.18±0.12 |
| HFD | 10 | 23.02±0.66 | 27.65±1.99a | 32.90±3.39a | 36.91±4.56a | 41.25±4.08a | 2.07±0.14 |
| L-SRT | 10 | 22.90±0.64 | 27.69±1.71 | 33.27±2.98 | 36.69±4.85 | 36.39±5.08 | 2.03±0.11b |
| H-SRT | 10 | 23.11±0.64 | 27.93±2.19 | 31.63±3.07 | 34.71±4.08 | 36.31±3.70 | 2.00±0.18a |
| OCA | 10 | 23.11±0.57 | 27.99±1.28 | 33.03±2.09 | 36.69±4.27 | 37.80±4.22 | 1.96±0.14a |
Table 1 Body weight and food intake
| Group | n | Body weight (g) | Food intake (g/mouse per day) | ||||
|---|---|---|---|---|---|---|---|
| 0 week | 4th week | 8th week | 12th week | 16th week | |||
| Control | 10 | 22.94±0.71 | 24.94±1.67 | 27.22±2.22 | 29.04±2.59 | 30.10±2.97 | 2.18±0.12 |
| HFD | 10 | 23.02±0.66 | 27.65±1.99a | 32.90±3.39a | 36.91±4.56a | 41.25±4.08a | 2.07±0.14 |
| L-SRT | 10 | 22.90±0.64 | 27.69±1.71 | 33.27±2.98 | 36.69±4.85 | 36.39±5.08 | 2.03±0.11b |
| H-SRT | 10 | 23.11±0.64 | 27.93±2.19 | 31.63±3.07 | 34.71±4.08 | 36.31±3.70 | 2.00±0.18a |
| OCA | 10 | 23.11±0.57 | 27.99±1.28 | 33.03±2.09 | 36.69±4.27 | 37.80±4.22 | 1.96±0.14a |
| Group | n | TNF-α | IL-1β | IL-6 |
|---|---|---|---|---|
| Control | 10 | 1.00±0.27 | 1.00±0.13 | 1.00±0.25 |
| HFD | 10 | 1.78±0.53a | 1.41±0.04a | 1.32±0.29a |
| L-SRT | 10 | 1.19±0.56b | 0.84±0.27c | 0.93±0.38b |
| H-SRT | 10 | 1.33±0.35b | 1.38±0.03 | 1.43±0.38 |
| OCA | 10 | 1.67±0.41 | 0.79±0.35c | 0.63±0.17c |
Table 3 Relative mRNA expression of inflammatory factors in the liver tissue
| Group | n | TNF-α | IL-1β | IL-6 |
|---|---|---|---|---|
| Control | 10 | 1.00±0.27 | 1.00±0.13 | 1.00±0.25 |
| HFD | 10 | 1.78±0.53a | 1.41±0.04a | 1.32±0.29a |
| L-SRT | 10 | 1.19±0.56b | 0.84±0.27c | 0.93±0.38b |
| H-SRT | 10 | 1.33±0.35b | 1.38±0.03 | 1.43±0.38 |
| OCA | 10 | 1.67±0.41 | 0.79±0.35c | 0.63±0.17c |

Figure 1 Sanren Tang ameliorates hepatic histology in NAFLD mice A: haematoxylin-eosin staining of the liver sections (× 200magnification). B: oil red staining of the liver sections (200 × magnification). 1: control group, 2: high-fat diet (HFD) group, 3: low-dose Sanren Tang (L-SRT) group, 4: high-dose Sanren Tang (H-SRT) group, 5: obeticholic acid (OCA) group. Mice in the control group were fed a control diet (D12450B, 10% kcal from fat). The others were fed HFD (D12492, 60% kcal from fat) for 16 weeks. From the 13th to 16th week, the mice in the L-SRT, H-SRT and OCA groups were administered with L-SRT (crude drug 1.09 g/mL, 20 mL/kg body weight), H-SRT (crude drug 2.18 g/mL, 20 mL/kg body weight), and OCA (10 mg/kg), respectively. The others were administered with an equal volume of double-distilled water.
| Group | n | Fasting blood glucose (mg/dL) | Fasting insulin (ng/mL) | HOMA-IR | ALT (U/L) | Hepatic TG (mg/g tissue) | NAS |
|---|---|---|---|---|---|---|---|
| Control | 10 | 15.33±11.63 | 0.39±0.09 | 0.33±0.29 | 17.02±5.44 | 41.85±7.74 | 0 (0) |
| HFD | 10 | 52.96±17.29a | 1.36±0.47a | 4.01±2.26a | 66.99±42.17a | 61.22±13.17a | 7 (2)a |
| L-SRT | 10 | 56.15±13.16 | 0.69±0.25b | 2.03±0.87c | 24.93±26.46c | 40.17±16.27b | 1 (1.75)c |
| H-SRT | 10 | 57.38±10.14 | 0.62±0.17b | 1.84±0.47d | 9.95±7.19c | 35.51±15.73b | 1 (1.5)c |
| OCA | 10 | 49.69±19.34 | 1.23±0.64 | 2.69±1.58 | 24.69±19.01 | 41.81±10.88b | 1 (1)b |
Table 2 Laboratory and pathological parameters of the mice
| Group | n | Fasting blood glucose (mg/dL) | Fasting insulin (ng/mL) | HOMA-IR | ALT (U/L) | Hepatic TG (mg/g tissue) | NAS |
|---|---|---|---|---|---|---|---|
| Control | 10 | 15.33±11.63 | 0.39±0.09 | 0.33±0.29 | 17.02±5.44 | 41.85±7.74 | 0 (0) |
| HFD | 10 | 52.96±17.29a | 1.36±0.47a | 4.01±2.26a | 66.99±42.17a | 61.22±13.17a | 7 (2)a |
| L-SRT | 10 | 56.15±13.16 | 0.69±0.25b | 2.03±0.87c | 24.93±26.46c | 40.17±16.27b | 1 (1.75)c |
| H-SRT | 10 | 57.38±10.14 | 0.62±0.17b | 1.84±0.47d | 9.95±7.19c | 35.51±15.73b | 1 (1.5)c |
| OCA | 10 | 49.69±19.34 | 1.23±0.64 | 2.69±1.58 | 24.69±19.01 | 41.81±10.88b | 1 (1)b |

Figure 2 Hepatic transcriptome profile and hepatic genes reversed by SRT in HFD-induced NAFLD A: PCA of transcriptome data; B: clustering analysis of sample-to-sample distances; C: number of DEGs; D: DEG intersection; E: heatmap of the top 20 genes up-or down-regulated by HFD and reversed by H-SRT; F: GO analysis of total genes regulated by HFD and reversed by H-SRT; G: KEGG pathway enrichment analysis of total genes regulated by HFD and reversed by H-SRT. PCA: principal component analysis; DEGs: differentially expressed genes; ECM: extracellular matrix; HFD: high-fat diet; NAFLD: non-alcoholic fatty liver disease; H-SRT: high-dose of Sanren Tang; GO: gene ontology; KEGG: Kyoto Encyclopedia of Genes and Genomes. Samples from the control, HFD, and H-SRT groups (4 samples per group) were randomly chosen for RNA-seq. Mice in the control group were fed a control diet (D12450B, 10% kcal from fat). The others were fed HFD (D12492, 60% kcal from fat) for 16 weeks. From the 13th to 16th week, the mice in the L-SRT, H-SRT and OCA groups were administered with L-SRT (crude drug 1.09 g/mL, 20 mL/kg body weight), H-SRT (crude drug 2.18 g/mL, 20 mL/kg body weight), and OCA (10 mg/kg), respectively. The others were administered with an equal volume of double-distilled water.
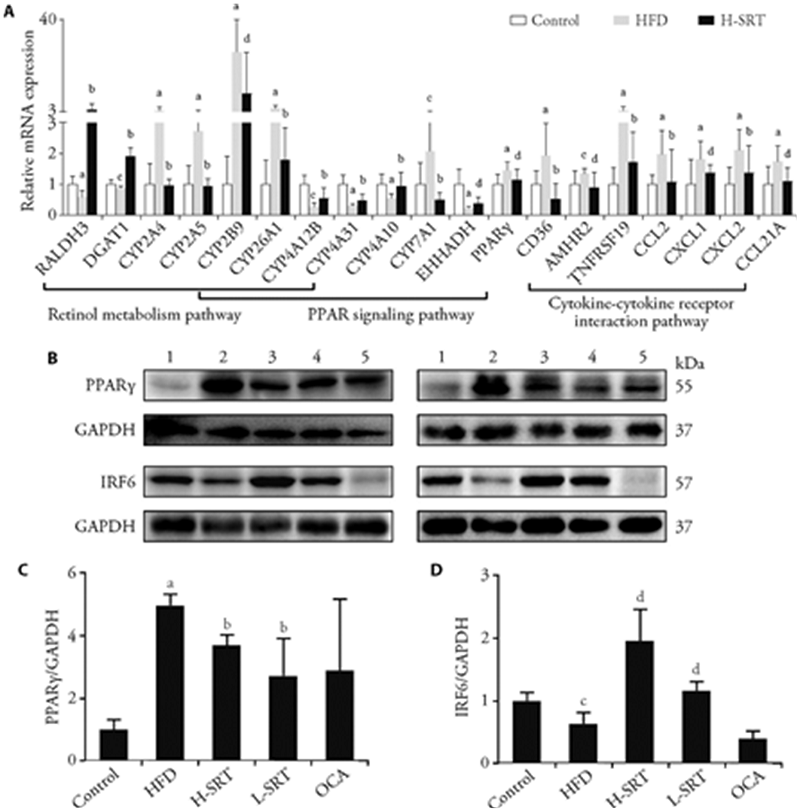
Figure 3 Verification of the genes enriched in the top three KEGG pathways A: relative hepatic mRNA expression levels of genes enriched in the top three KEGG pathways; B: representative images of Western-blotting of PPARγ and IRF 6 in liver tissue; C: the densities analysis of Western-blotting and of PPARγ; D: the densities analysis of Western-blotting band of IRF6. 1: control; 2: HFD; 3: H-SRT; 4: L-SRT; 5: OCA. KEGG: Kyoto Encyclopedia of Genes and Genomes; PPARγ: proliferator-activated receptor γ; IRF: interferon regulatory factor; HFD: high-fat diet; L-SRT: low-dose Sanren Tang; H-SRT: high-dose Sanren Tang; OCA: obeticholic acid; GAPDH: glyceraldehyde 3-phosphate dehydrogenase. Ten samples per group were used for detection of mRNA expression. The data of Western-blotting were obtained from three samples per group chosen randomly. aP < 0.01, vs control, bP < 0.01, vs HFD, cP < 0.05, vs control, dP < 0.05, vs HFD. The original uncut image of the Western-blot can be found in supplementary Figures7 and 8. Mice in the control group were fed a control diet (D12450B, 10% kcal from fat). The others were fed HFD (D12492, 60% kcal from fat) for 16 weeks. From the 13th to 16th week, the mice in the L-SRT, H-SRT and OCA groups were administered with L-SRT (crude drug 1.09 g/mL, 20 mL/kg body weight), H-SRT (crude drug 2.18 g/mL, 20 mL/kg body weight), and OCA (10 mg/kg), respectively. The others were administered with an equal volume of double-distilled water.
| 1. | Francque SM, Marchesini G, Kautz A, et al. Non-alcoholic fatty liver disease: apatient guideline. JHEP Rep 2021; 3: 100322. |
| 2. | Zhang S, Li J. Expert consensus on TCM diagnosis and treatment of nonalcoholic fatty liver disease (2017). Lin Chuang GanDan Bing Za Zhi 2017; 33: 2270-4. |
| 3. | Liu P. Clinical observation of Jiangzhi Sanren decoction in the treatment of nonalcoholic steatohepatitis with damp-heat intrinsic type. Shenyang: Liaoning Uniersity of Traditional Chinese Medicine, 2019: 1-37. |
| 4. | The National Administration of Traditional Chinese Medicine and the National Health Commission of People's Republic of China. Notification of the National Administration of Traditional Chinese Medicine and the National Health Commission of People's Republic of China on Printing and distributing the Management Standards of Chinese Medicine decocting rooms in medical institutions. Gazette of the National Health Commission of People's Republic of China 2009; 6: 29-31. |
| 5. |
Reagan-Shaw S, Nihal M, Ahmad N. Dose translation from animal to human studies revisited. FASEB J 2008; 22: 659-61.
DOI PMID |
| 6. |
Leng J, Huang F, Hai Y, et al. Amelioration of non-alcoholic steatohepatitis by Qushi Huayu decoction is associated with inhibition of the intestinal mitogen-activated protein kinase pathway. Phytomedicine 2020; 66: 153135.
DOI URL |
| 7. | Rodrigues PM, Afonso MB, Simão AL, et al. miR-21 ablation and obeticholic acid ameliorate nonalcoholic steatohepatitis in mice. Cell Death Dis 2017; 8: e2748. |
| 8. |
Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology 2005; 41: 1313-21.
DOI PMID |
| 9. |
Vogeser M, Konig D, Frey I, Predel HG, Parhofer KG, Berg A. Fasting serum insulin and the homeostasis model of insulin resistance (HOMA-IR) in the monitoring of lifestyle interventions in obese persons. Clin Biochem 2007; 40: 964-8.
DOI PMID |
| 10. |
Peng JH, Leng J, Tian HJ, et al. Geniposide and chlorogenic acid combination ameliorates non-alcoholic steatohepatitis involving the protection on the gut barrier function in mouse induced by high-fat diet. Front Pharmacol 2018; 9: 1399.
DOI URL |
| 11. |
Bolger AM, Lohse M, Usadel B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 2014; 30: 2114-20.
DOI PMID |
| 12. |
Kim D, Langmead B, Salzberg SL. HISAT: a fast spliced aligner with low memory requirements. Nat Methods 2015; 12: 357-60.
DOI PMID |
| 13. |
Trapnell C, Williams BA, Pertea G, et al. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 2010; 28: 511-5.
DOI PMID |
| 14. |
Anders S, Pyl PT, Huber W. HTSeq--a Python framework to work with high-throughput sequencing data. Bioinformatics 2015; 31: 166-9.
DOI PMID |
| 15. |
Tong J, Han CJ, Zhang JZ, et al. Hepatic interferon regulatory factor 6 alleviates liver steatosis and metabolic disorder by transcriptionally suppressing peroxisome proliferator-activated receptor gamma in mice. Hepatology 2019; 69: 2471-88.
DOI URL |
| 16. |
Blomhoff R, Green MH, Green JB, Berg T, Norum KR. Vitamin A metabolism: new perspectives on absorption, transport, and storage. Physiol Rev 1991; 71: 951-90.
PMID |
| 17. |
Senoo H, Mezaki Y, Fujiwara M. The stellate cell system (vitamin A-storing cell system). Anat Sci Int 2017; 92: 387-455.
DOI PMID |
| 18. |
Coelho JM, Cansancao K, Perez RM, et al. Association between serum and dietary antioxidant micronutrients and advanced liver fibrosis in non-alcoholic fatty liver disease: an observational study. PeerJ 2020; 8: e9838.
DOI URL |
| 19. |
Jeon D, Son M, Shim J. Dynamics of serum retinol and alpha-tocopherol levels according to non-alcoholic fatty liver disease status. Nutrients 2021; 13: 1720.
DOI URL |
| 20. |
Ashla AA, Hoshikawa Y, Tsuchiya H, et al. Genetic analysis of expression profile involved in retinoid metabolism in non-alcoholic fatty liver disease. Hepatol Res 2010; 40: 594-604.
DOI PMID |
| 21. |
Nakano M, Kelly EJ, Wiek C, Hanenberg H, Rettie AE. CYP4V2 in Bietti's crystalline dystrophy: ocular localization, metabolism of omega-3-polyunsaturated fatty acids, and functional deficit of the p.H331P variant. Mol Pharmacol 2012; 82: 679-86.
DOI URL |
| 22. |
Jarrar YB, Jarrar Q, Abed A, Abu-Shalhoob M. Effects of nonsteroidal anti-inflammatory drugs on the expression of arachidonic acid-metabolizing Cyp450 genes in mouse hearts, kidneys and livers. Prostaglandins Other Lipid Mediat 2019; 141: 14-21.
DOI URL |
| 23. |
Ito O, Nakamura Y, Tan L, et al. Expression of cytochrome P-450 4 enzymes in the kidney and liver: regulation by PPAR and species-difference between rat and human. Mol Cell Biochem 2006; 284: 141-8.
DOI PMID |
| 24. | Roh YS, Seki E. Chemokines and Chemokine Receptors in the Development of NAFLD. Adv Exp Med Biol 2018; 1061: 45-53. |
| 25. |
Mullen RD, Ontiveros AE, Moses MM, Behringer RR. AMH and AMHR2 mutations: aspectrum of reproductive phenotypes across vertebrate species. Dev Biol 2019; 455: 1-9.
DOI URL |
| 26. | Deng C, Lin YX, Qi XK, et al. TNFRSF19 Inhibits TGFbeta signaling through Interaction with TGFbeta receptor type I to promote tumorigenesis. Cancer Res 2018; 78: 3469-83. |
| 27. |
Gerard C, Rollins BJ. Chemokines and disease. Nat Immunol 2001; 2: 108-15.
DOI PMID |
| 28. |
Obstfeld AE, Sugaru E, Thearle M, et al. C-C chemokine receptor 2 (CCR2) regulates the hepatic recruitment of myeloid cells that promote obesity-induced hepatic steatosis. Diabetes 2010; 59: 916-25.
DOI PMID |
| 29. |
Ali AA, Fouda A, Abdelaziz ES, Abdelkawy K, Ahmed MH. The promising role of CCL2 as a noninvasive marker for nonalcoholic steatohepatitis diagnosis in Egyptian populations. Eur J Gastroenterol Hepatol 2021; 33: e954-60.
DOI URL |
| 30. | Bahcecioglu IH, Yalniz M, Ataseven H, et al. Levels of serum hyaluronic acid, TNF-alpha and IL-8 in patients with nonalcoholic steatohepatitis. Hepatogastroenterology 2005; 52: 1549-53. |
| 31. |
Yang L, Miura K, Zhang B, et al. TRIF differentially regulates hepatic steatosis and inflammation/fibrosis in mice. Cell Mol Gastroenterol Hepatol 2017; 3: 469-83.
DOI PMID |
| 32. |
Han L, Shen WJ, Bittner S, Kraemer FB, Azhar S. PPARs: regulators of metabolism and as therapeutic targets in cardiovascular disease. Part I: PPAR-alpha. Future Cardiol 2017; 13: 259-78.
DOI PMID |
| 33. |
Han L, Shen WJ, Bittner S, Kraemer FB, Azhar S. PPARs: regulators of metabolism and as therapeutic targets in cardiovascular disease. Part II: PPAR-beta/delta and PPAR-gamma. Future Cardiol 2017; 13: 279-96.
DOI PMID |
| 34. |
Matsusue K, Haluzik M, Lambert G, et al. Liver-specific disruption of PPARgamma in leptin-deficient mice improves fatty liver but aggravates diabetic phenotypes. J Clin Invest 2003; 111: 737-47.
PMID |
| 35. |
Houten SM, Denis S, Argmann CA, et al. Peroxisomal L-bifunctional enzyme (Ehhadh) is essential for the production of medium-chain dicarboxylic acids. J Lipid Res 2012; 53: 1296-303.
DOI PMID |
| 36. | Abdollahi H, Zamanian Azodi M, Hatami B. Protein interaction mapping interpretation of none alcoholic fatty liver disease model of rats after fat diet feeding. Gastroenterol Hepatol Bed Bench 2017; 10: S146-53. |
| 37. |
Schwarz M, Russell DW, Dietschy JM, Turley SD. Marked reduction in bile acid synthesis in cholesterol 7alpha-hydroxylase-deficient mice does not lead to diminished tissue cholesterol turnover or to hypercholesterolemia. J Lipid Res 1998; 39: 1833-43.
PMID |
| 38. |
Duan Y, Chen Y, Hu W, et al. Peroxisome Proliferator-activated receptor gamma activation by ligands and dephosphorylation induces proprotein convertase subtilisin kexin type 9 and low density lipoprotein receptor expression. J Biol Chem 2012; 287: 23667-77.
DOI URL |
| 39. |
Inoue M, Ohtake T, Motomura W, et al. Increased expression of PPARgamma in high fat diet-induced liver steatosis in mice. Biochem Biophys Res Commun 2005; 336: 215-22.
DOI URL |
| 40. |
Benton CR, Koonen DP, Calles-Escandon J, et al. Differential effects of contraction and PPAR agonists on the expression of fatty acid transporters in rat skeletal muscle. J Physiol 2006; 573: 199-210.
DOI URL |
| 41. |
Gao M, Ma Y, Alsaggar M, Liu D. Dual outcomes of rosiglitazone treatment on fatty liver. AAPS J 2016; 18: 1023-31.
DOI PMID |
| 42. |
Rada P, Gonzalez-Rodriguez A, Garcia-Monzon C, Valverde AM. Understanding lipotoxicity in NAFLD pathogenesis: is CD36a key driver? Cell Death Dis 2020; 11: 802.
DOI |
| 43. |
Miquilena-Colina ME, Lima-Cabello E, Sanchez-Campos S, et al. Hepatic fatty acid translocase CD36upregulation is associated with insulin resistance, hyperinsulinaemia and increased steatosis in non-alcoholic steatohepatitis and chronic hepatitis C. Gut 2011; 60: 1394-402.
DOI PMID |
| 44. |
Kawanishi N, Mizokami T, Yada K, Suzuki K. Exercise training suppresses scavenger receptor CD36 expression in kupffer cells of nonalcoholic steatohepatitis model mice. Physiol Rep 2018; 6: e13902.
DOI URL |
| 45. |
Koonen DP, Jacobs RL, Febbraio M, et al. Increased hepatic CD36 expression contributes to dyslipidemia associated with diet-induced obesity. Diabetes 2007; 56: 2863-71.
DOI PMID |
| 46. |
Wilson CG, Tran JL, Erion DM, Vera NB, Febbraio M, Weiss EJ. Hepatocyte-specific disruption of CD36 attenuates fatty liver and improves insulin sensitivity in HFD-fed mice. Endocrinology 2016; 157: 570-85.
DOI PMID |
| 47. | Kuo NC, Huang SY, Yang CY, Shen HH, Lee YM. Involvement of HO-1 and autophagy in the protective effect of magnolol in hepatic steatosis-induced NLRP3 inflammasome activation in vivo and in vitro. Antioxidants (Basel) 2020; 9. |
| 48. | Lee YS, Choi SS, Yonezawa T, et al. Honokiol, magnolol, and a combination of both compounds improve glucose metabolism in high-fat diet-induced obese mice. Food Science and Biotechnology 2015; 24: 8. |
| 49. |
Xiao Z, Ji Q, Fu YD, et al. Amygdalin ameliorates liver fibrosis through inhibiting activation of TGF-beta/smad signaling. Chin J Integr Med 2023; 29: 316-24.
DOI |
| 50. |
Elsaed WM. Amygdalin (Vitamin B17) pretreatment attenuates experimentally induced acute autoimmune hepatitis through reduction of CD4+ cell infiltration. Ann Anat 2019; 224: 124-32.
DOI PMID |
| 51. |
Tang F, Fan K, Wang K, Bian C. Amygdalin attenuates acute liver injury induced by D-galactosamine and lipopolysaccharide by regulating the NLRP3, NF-kappaB and Nrf2/NQO1 signalling pathways. Biomed Pharmacother 2019; 111: 527-36.
DOI URL |
| 52. | Sun J, Wang Z, Chen L, Sun G. Hypolipidemic effects and preliminary mechanism of chrysanthemum flavonoids, its main components luteolin and luteoloside in hyperlipidemia rats. Antioxidants (Basel) 2021; 10: 1309. |
| 53. |
Jain S, Jacobson KA. Purinergic signaling in liver pathophysiology. Front Endocrinol (Lausanne) 2021; 12: 718429.
DOI URL |
| [1] | YANG Shaojun, MA Yanhua, BAI Zhouxia, YU Ye, FANG Buwu, ZHANG Li, WANG Li. Intervention effect of Cigu Xiaozhi prescription (慈菇消脂方) on ceramide lipoapoptosis in non-alcoholic fatty liver disease [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 63-69. |
| [2] | YANG Jiayao, TAO Dongqing, MA Wei, LIU Song, LIAO Yan, SHU Lei, ZHANG Shu, LI Chenyu, DU Nianlong. Protective effects and mechanisms of Lizhong decoction (理中汤) against non-alcoholic fatty liver disease in a rat model [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 773-780. |
| [3] | ZHANG Chengfei, QIN Lingling, WANG Haiyan, SUN Boju, ZHAO Dan, ZHANG Qiue, ZHONG Fengying, WU Lili, LIU Tonghua. Efficacy of aqueous extract of flower of Edgeworthia gardneri (Wall.) Meisn on glucose and lipid metabolism in KK/Upj-Ay/J mice [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 187-193. |
| [4] | MA Yanhua, HAN Tao, YANG Shaojun, YU Chengzu, SHI Xia, WU Yan. Efficacy of Cigu Xiaozhi pill(慈菇消脂丸) on non-alcoholic steatohepatitis-associated lipoapoptosis through stress-activated c-Jun N-terminal kinase signalling pathway [J]. Journal of Traditional Chinese Medicine, 2021, 41(1): 79-88. |
| [5] | Wang Jianxing, Yu Xiaohan, Jiang Yan, Wang Yan, Li Ying, Han Shuying. Effects of a fermented buckwheat flower and leaf extract on the blood glucose and lipid profile of type 2 diabetic db/db mice [J]. Journal of Traditional Chinese Medicine, 2020, 40(2): 197-203. |
| [6] | Xiao Huanming, Jiang Junmin, Xie Yubao, Shi Meijie, Tian Guangjun, Zhao Pengtao, Zhang Chaozhen, Chen Huijun, Xu Chanyuan, Chi Xiaoling. Colon hydrotherapy plus Traditional Chinese Medicine to treat non-alcoholic fatty liver disease: a pilot study [J]. Journal of Traditional Chinese Medicine, 2017, 37(03): 393-396. |
| [7] | Wang Yan, Wang Xiaozhong, Zeng Binfang, Guo Feng, He Yongquan, He Jiaying, Liu Bin. Association between peroxisome proliferator-activated receptor γ gene polymorphism and susceptibility to northwest dryness syndrome [J]. Journal of Traditional Chinese Medicine, 2017, 37(02): 201-206. |
| [8] | Liang Huiqing, Yang Jiaen, Tang Jinmo, Wu Chuncheng, Li Hongshan, Chen Shaodong. Optimization of dosage ratio of chlorogenic acid and gardenia glycosides in the treatment of rats with fatty liver disease induced by high-fat feed [J]. Journal of Traditional Chinese Medicine, 2016, 36(05): 683-688. |
| [9] | Shaodong Chen, Haihong Zhou, Manting Lin, Ran Mi, Long Li. Decoction vs extracts-mixed solution: effect of Yiqihuoxue formula on non-alcoholic fatty liver disease in rats [J]. Journal of Traditional Chinese Medicine, 2013, 33(04): 513-517. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

