Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (1): 113-123.DOI: 10.19852/j.cnki.jtcm.2023.01.010
• Original articles • Previous Articles Next Articles
Efficacy of suspended moxibustion stimulating Shenshu (BL23) and Guanyuan (CV4) on the amygdala-HPA axis in rats with kidney-Yang deficiency symptom pattern induced by hydrocortisone
MIN Youjiang1,2,3, YAO Haihua2( ), WANG Zhiqin3, LUO Kaitao4(
), WANG Zhiqin3, LUO Kaitao4( ), SUN Jie3, YUAN Zheng2, WU Huiqi3, CHENG Lihong3
), SUN Jie3, YUAN Zheng2, WU Huiqi3, CHENG Lihong3
- 1 School of Traditional Chinese Medicine, Nanchang Medical College, Nanchang 330053, China
2 Traditional Chinese Medicine Department, Shanghai Eighth People’s Hospital, Shanghai 200235, China
3 Acupuncture Department, Affiliated Hospital of Jiangxi University of Traditional Chinese Medicine, Nanchang 330006, Jiangxi, China
4 Acupuncture Department, Jiaxing Hospital of Traditional Chinese Medicine, Jiaxing 314001, China
-
Received:2021-12-21Accepted:2022-03-11Online:2023-02-15Published:2023-01-10 -
Contact:YAO Haihua,LUO Kaitao -
About author:LUO Kaitao, Acupuncture Department, Jiaxing Hospital of Traditional Chinese Medicine, Jiaxing 314001, China. lkt740813@163.com. Telephone:+8618217463558; +8613758395805
YAO Haihua, Outpatient Clinic Office, Shanghai Eighth People’s Hospital, Shanghai 200235, China. myj2002@126.com;
-
Supported by:Study on the Mechanism of Suspension Moxibustion with Moxa Stick in Treating Hydrocortisone Rats with Kidney-Yang Deficiency based on Hippocampus (amygdala)-HPA Axis(81660817)
Cite this article
MIN Youjiang, YAO Haihua, WANG Zhiqin, LUO Kaitao, SUN Jie, YUAN Zheng, WU Huiqi, CHENG Lihong. Efficacy of suspended moxibustion stimulating Shenshu (BL23) and Guanyuan (CV4) on the amygdala-HPA axis in rats with kidney-Yang deficiency symptom pattern induced by hydrocortisone[J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 113-123.
share this article
| Gene | Full name | Sequences (5-3′) | Product size (bp) |
|---|---|---|---|
| 11β-HSD1 | 11 beta- hydroxysteroid dehydrogenase type 1 | Sense primer: AAA ATA CCT CCT CCC CGT CC antisense primer: AGG CAG CGA GAC ACC ACC | 219 |
| MR | Mineralocorticoid receptor | Sense primer: AGA AGC TGG GGA AGT TAA AAG G antisense primer: TCG GAG CGA TGT ATG TGG TC | 102 |
| GR | Glucocorticoid receptor | Sense primer: CAT TAC CAC AGC TCA CCC CTA C antisense primer: GCA ATC ACT TGA CGC CCA C | 148 |
| CR | Corticotropin-releasing hormone | Sense primer: TGG CTC TGT CGC CCT GTC antisense primer: CAG CGG GAC TTC TGT TGA GG | 186 |
| ACTH | Adrenocorticotrophic hormone | Sense primer: CTC CTG CTT CAG ACC TCC ATA G antisense primer: GGC TGT TCA TCT CCG TTG C | 161 |
| GAPDH | Glyceraldehyde 3-phosphate dehydrogenase | Sense primer: GGA GTC TAC TGG CGT CTT CAC antisense primer: ATG AGC CCT TCC ACG ATG C | 237 |
Table 1 Primer sequences for real-time quantitative polymerase chain reaction
| Gene | Full name | Sequences (5-3′) | Product size (bp) |
|---|---|---|---|
| 11β-HSD1 | 11 beta- hydroxysteroid dehydrogenase type 1 | Sense primer: AAA ATA CCT CCT CCC CGT CC antisense primer: AGG CAG CGA GAC ACC ACC | 219 |
| MR | Mineralocorticoid receptor | Sense primer: AGA AGC TGG GGA AGT TAA AAG G antisense primer: TCG GAG CGA TGT ATG TGG TC | 102 |
| GR | Glucocorticoid receptor | Sense primer: CAT TAC CAC AGC TCA CCC CTA C antisense primer: GCA ATC ACT TGA CGC CCA C | 148 |
| CR | Corticotropin-releasing hormone | Sense primer: TGG CTC TGT CGC CCT GTC antisense primer: CAG CGG GAC TTC TGT TGA GG | 186 |
| ACTH | Adrenocorticotrophic hormone | Sense primer: CTC CTG CTT CAG ACC TCC ATA G antisense primer: GGC TGT TCA TCT CCG TTG C | 161 |
| GAPDH | Glyceraldehyde 3-phosphate dehydrogenase | Sense primer: GGA GTC TAC TGG CGT CTT CAC antisense primer: ATG AGC CCT TCC ACG ATG C | 237 |
| Gene | Full name | Sequences (5-3′) |
|---|---|---|
| 11β-HSD1 | 11 beta-hydroxysteroid dehydrogenase type 1 | CAGAUGCCAGGUUUGUGCUCAAGUAAAUCCAAAAUGGAUAGCCUUACUCA |
| CRH | Corticotropin releasing hormone | CAAUACAAAUAACGCUGUUUUGUUACUACAAAGAAACACACUUUGUGCA |
| MR | Mineralocorticoid receptor | GGAUGGAGAGGAUAGCAAUCCCGGCAGUCGCCCUACUGACGGUGGG |
| GR | Glucocorticoid receptor | UGCUUGUGGAGCCUUUCGAGAAAUCAAGGAGAAUCCUCUGCUGCUU |
Table 2 Primer sequences for Hybridization in situ
| Gene | Full name | Sequences (5-3′) |
|---|---|---|
| 11β-HSD1 | 11 beta-hydroxysteroid dehydrogenase type 1 | CAGAUGCCAGGUUUGUGCUCAAGUAAAUCCAAAAUGGAUAGCCUUACUCA |
| CRH | Corticotropin releasing hormone | CAAUACAAAUAACGCUGUUUUGUUACUACAAAGAAACACACUUUGUGCA |
| MR | Mineralocorticoid receptor | GGAUGGAGAGGAUAGCAAUCCCGGCAGUCGCCCUACUGACGGUGGG |
| GR | Glucocorticoid receptor | UGCUUGUGGAGCCUUUCGAGAAAUCAAGGAGAAUCCUCUGCUGCUU |
| Group | n | CORT | ACTH | CRH |
|---|---|---|---|---|
| Norm | 6 | 2149±214 | 641±47 | 106±15 |
| Model | 6 | 712±98a | 204±29a | 48±5a |
| CBX | 6 | 880±148a | 259±30a | 60±6ad |
| Moxi | 6 | 1578±204abc | 485±38abc | 83±7abc |
| Moxi+CBX | 6 | 1154±183ade | 339±40abcf | 69±8abe |
Table 3 Comparison of the level of CORT, ACTH and CRH in serum (ng/L, $\bar{x}\pm s$)
| Group | n | CORT | ACTH | CRH |
|---|---|---|---|---|
| Norm | 6 | 2149±214 | 641±47 | 106±15 |
| Model | 6 | 712±98a | 204±29a | 48±5a |
| CBX | 6 | 880±148a | 259±30a | 60±6ad |
| Moxi | 6 | 1578±204abc | 485±38abc | 83±7abc |
| Moxi+CBX | 6 | 1154±183ade | 339±40abcf | 69±8abe |
| Group | n | MR | GR | 11β-HSD1 | CRH |
|---|---|---|---|---|---|
| Norm | 6 | 6.63±2.60 | 2.13±0.88 | 1.61±0.75 | 1.35±0.40 |
| Model | 6 | 1.90±0.54a | 0.60±0.16a | 0.54±0.06a | 0.49±0.16a |
| CBX | 6 | 3.11±0.42a | 1.02±0.15a | 0.39±0.12a | 0.64±0.11a |
| Moxi | 6 | 3.98±0.76ab | 1.31±0.29ab | 0.93±0.19ad | 0.93±0.21abe |
| Moxi+CBX | 6 | 2.64±0.36a | 0.79±0.14ac | 0.74±0.12a | 0.79±0.16af |
Table 4 Comparison of the relative mRNA expressions of MR, GR, 11β-HSD1 and CRH in amygdala (/GAPDH) (‰, $\bar{x}\pm s$)
| Group | n | MR | GR | 11β-HSD1 | CRH |
|---|---|---|---|---|---|
| Norm | 6 | 6.63±2.60 | 2.13±0.88 | 1.61±0.75 | 1.35±0.40 |
| Model | 6 | 1.90±0.54a | 0.60±0.16a | 0.54±0.06a | 0.49±0.16a |
| CBX | 6 | 3.11±0.42a | 1.02±0.15a | 0.39±0.12a | 0.64±0.11a |
| Moxi | 6 | 3.98±0.76ab | 1.31±0.29ab | 0.93±0.19ad | 0.93±0.21abe |
| Moxi+CBX | 6 | 2.64±0.36a | 0.79±0.14ac | 0.74±0.12a | 0.79±0.16af |
| Group | n | 11β-HSD1 | CRH | ACTH |
|---|---|---|---|---|
| Norm | 6 | 2.43±0.73 | 3.31±1.11 | 4.48±0.31 |
| Model | 6 | 0.74±0.25a | 0.46±0.23a | 2.99±0.45a |
| CBX | 6 | 0.47±0.27a | 1.39±0.52ad | 3.24±0.36a |
| Moxi | 6 | 1.76±0.46bc | 2.66±0.77bc | 4.17±0.22bc |
| Moxi+CBX | 6 | 1.27±0.34ac | 1.93±0.40ab | 3.81±0.26ed |
Table 5 Comparison of the relative mRNA expressions of 11β-HSD1, CRH in hypothalamus and ACTH in pituitary (/GAPDH) (‰, $\bar{x}\pm s$)
| Group | n | 11β-HSD1 | CRH | ACTH |
|---|---|---|---|---|
| Norm | 6 | 2.43±0.73 | 3.31±1.11 | 4.48±0.31 |
| Model | 6 | 0.74±0.25a | 0.46±0.23a | 2.99±0.45a |
| CBX | 6 | 0.47±0.27a | 1.39±0.52ad | 3.24±0.36a |
| Moxi | 6 | 1.76±0.46bc | 2.66±0.77bc | 4.17±0.22bc |
| Moxi+CBX | 6 | 1.27±0.34ac | 1.93±0.40ab | 3.81±0.26ed |
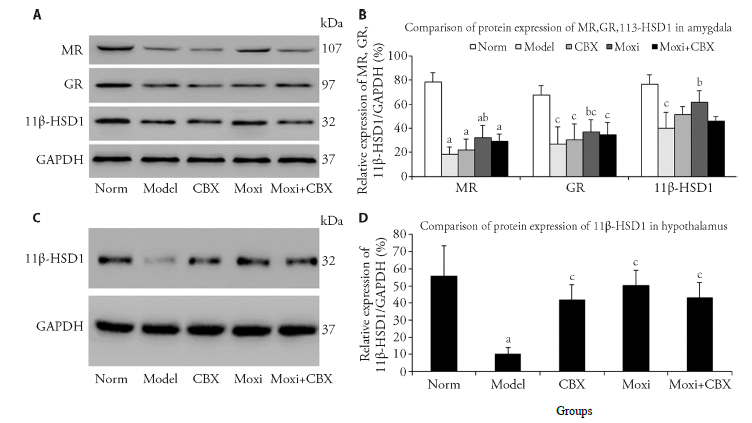
Figure 1 MR, GR and 11β-HSD1 activation in the amygdala and hypothalamus following treatment A: western blotting bands for MR, GR, 11β-HSD1 and GAPDH expression in the amygdala; B: compiled results in a bar graph for the ratio of MR, GR and 11β-HSD1 / GAPDH expression; C: western blotting bands for 11β-HSD1 and GAPDH expression in hypothalamus; D: compiled results in a bar graph for the ratio of 11β-HSD1 / GAPDH expression. aP < 0.01, cP < 0.05 versus norm; bP < 0.05, dP < 0.01, versus model. Data are shown as the mean ± standard error of the mean (1-way analysis of variance and Student-Newman-Keuls post hoc test, n = 4 rats/group). norm: normal control group; model: model group; CBX: carbenoxolone intraperitoneal injection group; moxi: moxibustion group; moxi + CBX: moxibustion plus carbenoxolone intraperitoneal injection group. MR: mineralocorticoid receptor; GR: glucocorticoid receptor; 11β-HSD1: 11 beta-hydroxysteroid dehydrogenase type 1; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.
| Group | n | MR | GR | 11β-HSD1 | CRH |
|---|---|---|---|---|---|
| Norm | 6 | 0.379±0.132 | 0.409±0.120 | 0.533±0.090 | 0.580±0.108 |
| Model | 6 | 0.036±0.010a | 0.025±0.019a | 0.175±0.092a | 0.073±0.057a |
| CBX | 6 | 0.077±0.026a | 0.074±0.072a | 0.110±0.066a | 0.119±0.080a |
| Moxi | 6 | 0.224±0.080abc | 0.258±0.095ef | 0.384±0.127bf | 0.337±0.181abc |
| Moxi+CBX | 6 | 0.112±0.040ad | 0.129±0.085a | 0.257±0.077af | 0.212±0.110ae |
Table 6 Comparison of the IOD values of MR, GR, 11β-HSD1 and CRH mRNA-positive cells in amygdala ($\bar{x}\pm s$)
| Group | n | MR | GR | 11β-HSD1 | CRH |
|---|---|---|---|---|---|
| Norm | 6 | 0.379±0.132 | 0.409±0.120 | 0.533±0.090 | 0.580±0.108 |
| Model | 6 | 0.036±0.010a | 0.025±0.019a | 0.175±0.092a | 0.073±0.057a |
| CBX | 6 | 0.077±0.026a | 0.074±0.072a | 0.110±0.066a | 0.119±0.080a |
| Moxi | 6 | 0.224±0.080abc | 0.258±0.095ef | 0.384±0.127bf | 0.337±0.181abc |
| Moxi+CBX | 6 | 0.112±0.040ad | 0.129±0.085a | 0.257±0.077af | 0.212±0.110ae |

Figure 2 mRNA expression of MR and GR in the amygdala following treatment (in situ hybridization, × 200) Bar = 50 μm. A: negative; B: expression of MR in norm; C: expression of MR in model; D: expression of MR in CBX; E: expression of MR in moxi; F: expression of MR in CBX + moxi; G: negative; H: expression of GR in norm; I: expression of GR in model; J: expression of GR in CBX; K: expression of GR in moxi; L: expression of GR in CBX + moxi. insitu hybridization-positive cells contained brown spots or particles, as indicated by the arrows. norm: normal control group; model: model group; CBX: carbenoxolone intraperitoneal injection group; moxi: moxibustion group; moxi + CBX: moxibustion plus carbenoxolone intraperitoneal injection group. MR: mineralocorticoid receptor; GR: glucocorticoid receptor.

Figure 3 mRNA expression of 11β-HSD1 and CRH in the amygdala following treatment (in situ hybridization, × 200) Bar = 50 μm. A: negative; B: expression of 11β-HSD1 in norm; C: expression of 11β-HSD1 in model; D: expression of 11β-HSD1 in CBX; E: expression of 11β-HSD1 in moxi; F: expression of 11β-HSD1 in CBX + moxi; G: negative; H: expression of CRH in norm; I: expression of CRH in model; J: expression of CRH in CBX; K: expression of CRH in moxi; L: expression of CRH in CBX + moxi. insitu hybridization-positive cells contained brown spots or particles, as indicated by the arrows. Norm: normal control group; model: model group; CBX: carbenoxolone intraperitoneal injection group; moxi: moxibustion group; moxi + CBX: moxibustion plus carbenoxolone intraperitoneal injection group. 11β-HSD1: 11 beta- hydroxysteroid dehydrogenase type 1; CRH: corticotropin-releasing hormone.
| Group | n | 11β-HSD1 | CRH |
|---|---|---|---|
| Norm | 6 | 0.520±0.098 | 0.478±0.098 |
| Model | 6 | 0.199±0.106a | 0.127±0.088a |
| CBX | 6 | 0.098±0.066a | 0.228±0.105c |
| Moxi | 6 | 0.405±0.107b | 0.392±0.072e |
| Moxi+CBX | 6 | 0.289±0.107cd | 0.327±0.044f |
Table 7 Comparison of the IOD values of 11β-HSD1 and CRH mRNA-positive cells in hypothalamus (± s)
| Group | n | 11β-HSD1 | CRH |
|---|---|---|---|
| Norm | 6 | 0.520±0.098 | 0.478±0.098 |
| Model | 6 | 0.199±0.106a | 0.127±0.088a |
| CBX | 6 | 0.098±0.066a | 0.228±0.105c |
| Moxi | 6 | 0.405±0.107b | 0.392±0.072e |
| Moxi+CBX | 6 | 0.289±0.107cd | 0.327±0.044f |

Figure 4 mRNA expression of 11β-HSD1 and CRH in the hypothalamus following treatment (in situ hybridization, × 200) Bar = 50 μm. A: negative; B: expression of 11β-HSD1 in norm; C: expression of 11β-HSD1 in model; D: expression of 11β-HSD1 in CBX; E: expression of 11β-HSD1 in moxi; F: expression of 11β-HSD1 in CBX + moxi; G: negative; H: expression of CRH in norm; I: expression of CRH in model; J: expression of CRH in CBX; K: expression of CRH in moxi; L: expression of CRH in CBX + moxi. insitu hybridization-positive cells contained brown spots or particles, as indicated by the arrows. Norm: normal control group; model: model group; CBX: carbenoxolone intraperitoneal injection group; moxi: moxibustion group; moxi + CBX: moxibustion plus carbenoxolone intraperitoneal injection group. 11β-HSD1: 11 beta- hydroxysteroid dehydrogenase type 1; CRH: corticotropin-releasing hormone.
| 1 | Xiao AJ, He L, Ouyang X, Liu JM, Chen MR. Comparison of the anti-apoptotic effects of 15- and 35-minute suspended moxibustion after focal cerebral ischemia/reperfusion injury. Neural Regen Res 2018; 13: 257-64. |
| 2 | Ye R, Zhang AX, Jiang RR, et al. Clinical study of moxibustion in the treatment of female sexual dysfunction with kidney-Yang deficiency. Si Zhen Guo Yi Guo Yao 2014; 25: 2700-2. |
| 3 | Zhu W. Moxibustion with ginger to treat 55 cases of low sperm motility of kidney-Yang deficiency. Beijing Zhong Yi 2000; 19: 48-9. |
| 4 | Wang Y, Xu JH, Hu ZH, et al. Observations on the therapeutic effect of Du meridian moxibustion on polycystic ovarian syndrome of spleen-kidney Yang deficiency type. Shanghai Zhen Jiu Za Zhi 2015; 34: 35-7. |
| 5 | Wen Y, Ren AL, Deng LW. Discussion on auricular acupuncture point combined with warm box moxibustion in the treatment of ovulation disorders with kidney-Yang deficiency. Hunan Zhong Yi Za Zhi 2013; 29: 72-3. |
| 6 | Li LA, Yang XJ, Du GM, Jin TM, Xu J. The central regulatory mechanism of HPA axis for stress response. Zhong Guo Shou Yi Za Zhi 2010; 46: 65-son7. |
| 7 |
Thompson BL, Erickson K, Schulkin J, Rosen JB. Corticosterone facilitates retention of contextually conditioned fear and increases CRH mRNA expression in the amygdala. Behav Brain Res 2004; 149: 209-13.
PMID |
| 8 | Makino S, Hasmoto K, Godd PW. Multiple feedback mechanisms activatingcorticotrophin-releasing hormone system in the brain during stress. Pharmocol Biochem Bebav 2002; 73: 147-52. |
| 9 |
Oitxl MS, Champagne DL, Veen RVD, Kloet ERD. Brain development under stress: hypotheses of glucocorticoid actions revisited. Neurosci Biobehav Rev 2010; 34: 853-66.
DOI PMID |
| 10 | He WB, Zhang JL, Chen NH. Analysis of relationship between brain and kidney of TCM based on negative feedback regulation in the hippocampus-HPA axis. Zhong Guo Zhong Yi Yao Za Zhi 2016; 31: 3426-8. |
| 11 |
De-Kloet ER, Vreugdenhil-Reugdenhil E, Oitzl MS, Joels M. Brain corticosteroid receptor balance in health and disease. Endocr Rev 1998; 19: 269-301.
DOI PMID |
| 12 | Harris HJ, Kotelvtse-Votelevtsev Y, Mullins JJ, Seckl JR, Holmes MC. Intracellular regeneration of glucocorticoids by 11β-hydroxysteroid (11β-HSD)-1 plays a key role in regulation hypothalamic-pituitary-adrenal axis: analysis of 11 beat-HSD-1 deficient mice. Endocrinology 2001; 142: 114-20. |
| 13 | Li XR, Zhong LY. 11β-HSD1 is involved in neuroendocrine regulation of the hippocampus and hypothalamic-pituitary-adrenal axis. J southeast Univ (MedSci Edi) 2009; 8: 358-60. |
| 14 | Mattsson C, Lai M, Noble J, et al. Obese Zucker rats have reduced mineralocorticoid receptor and 11 beta-hydroxysteroid dehy-drogenase type 1 expression in the hippocampus: implications for dysregulation of the hypothalamic-pituitary-adrenal axis in obesity. Endocrinology 2003;144: 2997-3003. |
| 15 | Zeng YB, Liu YM. Expression of connexin 32 in brain tissue of epileptic rats and the effect of carbenoxolone on it. J Clin Neurol 2013; 26: 115-7. |
| 16 | Yau JL, Noble J, Kenyon CJ, et al. Lack of tissue glucocorticoid reactivation in 11β-hydroxysteriod dehydrogenase type 1 knockout mice ameliorates age-related learning impairments. Proc Nat1 Acad Sci USA 2001; 98: 4716-21. |
| 17 | Shen ZY. Study on localization of kidney-Yang deficiency syndrome. Zhong Guo Zhong Xi Yi Jie He Za Zhi 1997; 17: 50-2. |
| 18 | Min YJ, Yao HH, Cheng LH. The effect of suspended moxa stick moxibustion on points Shenshu (BL23) and Guanyuan (CV4) on the pituitary-adrenal axis and the pituitary-thyroid axis in rats with kidney-Yang deficiency. Shanghai Zhen Jiu Za Zhi 2016; 35: 1469-52. |
| 19 | Min YJ, Yan ZG, Yang HY, Yang XM, Guo CX. Qualitative and quantitative experiment study on influential factors of acupuncture efficacy. Liaoning Zhong Yi Za Zhi 2008; 35: 1923-27. |
| 20 | Zhang Y, Xu SY, Liu MN, et al. Comparative studies on chemical contents and effect in kidney-Yang deficiency rats of salt-processed product and wine-processed product of cuscutae semen. Evid Based Complement Alternat Med 2019; 2019: 2049497. |
| 21 | Lin WZ, Wang P. Experimental acupuncture. China: Shanghai Science and Technology Press, 1999: 288-9. |
| 22 | Kuang AK. Effect of some pro-Yang drugs on depletion induced by large dose of corticosteroids. Zhong Hua Nei Ke Za Zhi 1963; 2: 113-6. |
| 23 | Chen YH, Ou-yang YQ, Sun Q, Chen J, Zou YH. A primary discussion of diagnosis index choosing in standardization study of animal model with kidney-Yang deficiency. Zhong Guo Zhong Yi Ji Chu Yi Xue Za Zhi 2003; 9: 26-30. |
| 24 | Shun LJ, Yao HH, Min YJ, Cheng LH. Effects of different acupuncture on the levels of serum CORT and ACTH and the gene expression of hypothalamic CRH in rats with deficiency of kidney-Yang induced by hydrocortisone: an orthogonal design. Zhong Hua Zhong Yi Yao Za Zhi 2016; 31: 2337-41. |
| 25 | Chen XY. Animal modeling of practical TCM syndromes. Beijing:Beijing Medical University, Peking Union Medical College Joint Press, 1993: 100-17. |
| 26 | Chen Q. Methodology of pharmacology research of Traditional Chinese Medicine. Beijing: the people's medical publishing house, 1993: 982-1001. |
| 27 | Wu PY, Yu ZY. Yougui wan in the treatment of 30 cases of spleen-kidney Yang deficiency syndrome caused by hormone withdrawal in patients with nephrotic syndrome. Jiangxi Zhong Yi Yao 2010; 41: 43-4. |
| 28 | Kou WW, Zhang MF. Clinical observation of Wenshentianjing pill in the treatment of 100 cases of hormone-dependent nephropathy with kidney-Yang deficiency. Zhong Guo Can Ji Yi Xue 2014; 22: 131-2. |
| 29 | AzumaK, Zhou Q, Niwa M, Kubo KY. Association between mastication, the hippocampus, and the HPA Axis: a com-prehensive review. Int J Mol Sci 2017; 18: 1687. |
| 30 |
Herman JP, Mcklveen JM, Ghosal S, et al. Regulation of the hypothalamic-pituitary-adrenocortical stress response. Compr Physiol 2016; 6: 603-21.
DOI PMID |
| 31 | Kolber BJ, Roberts MS, Howell MP, Wozniak DF, Sands MS, Muglia LJ. Central amygdala glucocorticoid receptor action promotes fear associated CRH activation and conditioning. Proc Natl Acad Sci 2008; 105: 12004-9. |
| 32 | Silvermang MN, Sternberg EM. Glucocorticoid regulation of infla-mmation and its functional correlates: from HPA axis to glucocorticoid receptor dysfunction. Ann N Y Acad Sci 2012; 12: 55-63. |
| 33 | Min YJ, Deng L, Hong ES. Orthogonal study on different acupuncture factors based on hypothalamic- pituitary-adrenal axis in rats with kindey-Yang deficiency. Shanghai Zhen Jiu Za Zhi 2016; 35: 355-9. |
| 34 |
Welberg LA, Seckl JR, Holmes MC. Inhibition of 11beta-hydroxysteroid dehydrogenase, the foeto-placental barrier to maternal glucocorticoids, permanently programs amygdala GR mRNA expression and anxiety-like behaviour in the offspring. Eur J Neurosci 2000; 12: 1047-54.
PMID |
| 35 | Ren DW, Pei JC. Randomized controlled study on effect of moxibustion of different acupoint groups on the hypothalamic-pituitary-adrenal axis in rat. Shi Yong Zhong Yi Nei Ke Xue Za Zhi 2014; 28: 80-1. |
| 36 | Wu Q (Ming dynasty). Yi Zong Jin Jian. Xinjiang: Xinjiang People's Medical Publishing House, 2015: 866. |
| 37 | Zhou JS, Jin ZG, Tao ZL. The segmental distribution of the primary sensory neuron of the acupoint Guanyuan (CV4) in the spinal ganglion. Shanghai Zhen Jiu Za Zhi 2001; 20: 40-1. |
| 38 | Zhao C, Shi Y, Cui YH, et al. Effect of suspended moxibustion on the pRb expression of peripheral blood in the kidney-Yang deficiency aged people. Huanqiu Zhong Yi 2012; 5: 350-3. |
| [1] | LI Chaoran, YANG Yan, FENG Chuwen, LI Heng, QU Yuanyuan, WANG Yulin, WANG Delong, WANG Qingyong, GUO Jing, SHI Tianyu, SUN Xiaowei, WANG Xue, HOU Yunlong, SUN Zhongren, YANG Tiansong. Integrated 'omics analysis for the gut microbiota response to moxibustion in a rat model of chronic fatigue syndrome [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1176-1189. |
| [2] | WANG Miao, ZHU Yan, ZHAO Hui, ZHAO Hongfang. Moxibustion enables protective effects on rheumatoid arthritis-induced myocardial injury via transforming growth factor beta1 signaling and metabolic reprogramming [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1190-1199. |
| [3] | LI Xingjie, LIU Qiqi, XIA Rui, LIU Jun, WANG Dan, SHI Jiao, KUANG Yuxing, DAI Yalan, HUANG Haoyu, TANG Wei, CHEN Shangjie. Moxibustion modulates working memory in patients with amnestic mild cognitive impairment: a functional magnetic resonance imaging study [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 801-808. |
| [4] | MA Fangfang, ZHANG Hewei, LI Bingxue, CHENG Peiyu, YU Mingwei, WANG Xiaomin. Acupuncture and moxibustion for malignant tumor patients with psychological symptoms of insomnia, anxiety and depression: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 441-456. |
| [5] | YAO Yao, ZHAO Zhenni, CHEN Fengqin, LENG Yufei, PANG Xiangtian, XU Xiao, SUN Zhiling. Effectiveness of moxibustion alone on lumbar disc herniation: a Meta-analysis of randomized controlled trials [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 14-26. |
| [6] | YANG Jun, XIONG Jun, XU Shaozhong, XIE Hongwu, XIANG Jie. Effect and cerebral mechanism of moxibustion at heat-sensitized Yaoyangguan (GV3) in patients with lumbar disc herniation and myofascial pain syndrome by resting-state functionality magnetic resonance imaging: protocol for an observational study [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 175-180. |
| [7] | FANG Jing, PAN Wen, WANG Xiangyun, LI Fengxing, ZHAO Ling, HUANG Zouqin, SHEN Xueyong. Efficacy of stimulating Mingmen (GV4) and Guanyuan (CV4) on kidney Yang deficiency in rat model: laser irradiation vs traditional moxibustion [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 972-979. |
| [8] | ZHOU Haiyan, ZHONG Yumei, GAO Xiuhua, WU Fei, JIA Min, YANG Xin. Efficacy of Moxa-burning heat stimulating Zusanli (ST36) and Shenshu (BL23) on expressions of macrophage migration inhibitory factor and macrophage apoptosis in rabbits with adjuvant-induced arthritis [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 980-987. |
| [9] | WANG Wei, LI Qingling, MA Qiang, XIA Ran, GAO Bing, WANG Yi, WANG Jing. Effects of moxibustion at bilateral Feishu (BL13) and Xinshu (BL15) combined with benazepril on myocardial cells apoptosis index and apoptosis-related proteins cytochrome c and apoptosis-inducing factor in rats with chronic heart failure [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 227-233. |
| [10] | MA Tingting, WU Jie, YANG Lijie, FENG Fen, YANG Huilin, ZHANG Jinhua, ZHONG Yanjin, NING Qing, HUANG Lirong, LIN Youbing, YAN Jue, CHEN Guiquan, HOU Tianshu, WANG Li, REN Yuanfang, TAN Jing. Ginger-indirect moxibustion plus acupuncture versus acupuncture alone for chronic fatigue syndrome: a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 242-249. |
| [11] | Feng HAO, Qiang WANG, Lei LIU, Libin WU, Ronglin CAI, Jiajia SANG, Jun HU, Jie WANG, Qing YU, Lu HE, Yingchao SHEN, Yiming MIAO, Ling HU, Zijian WU. Effect of moxibustion on autophagy and the inflammatory response of synovial cells in rheumatoid arthritis model rats [J]. Journal of Traditional Chinese Medicine, 2022, 42(1): 73-82. |
| [12] | PAN Lijia, MA Shuya, WEN Jing, ZHANG Xiaoqi, XING Haijiao, JIA Chunsheng. Direct contact moxibustion promotes apoptosis of gastric cancer cells in rats by regulating intestinal flora [J]. Journal of Traditional Chinese Medicine, 2021, 41(6): 943-952. |
| [13] | LIU Di, WU Yongli, LI Chun, WANG Minglei, MA Xiaoxiu, LIU Junwei, ZHANG Yanling, YANG Lei. Warming moxibustion attenuates inflammation and cartilage degradation in experimental rabbit knee osteoarthritis [J]. Journal of Traditional Chinese Medicine, 2021, 41(6): 959-967. |
| [14] | ZHAO Qingyi, SUN Yi, ZHOU Jing, GAO Yanling, MA Guizhi, HU Zhihai, WANG Yi, SHI Yin. Effectiveness of herb-partitioned moxibustion combined with electroacupuncture on polycystic ovary syndrome in patients with symptom pattern of kidney deficiency and phlegm-dampness [J]. Journal of Traditional Chinese Medicine, 2021, 41(6): 985-993. |
| [15] | WANG Zhaoqin, CUI Yunhua, HUANG Yan, WU Huangan, ZHAO Jimeng, HE Jinsen, LIU Huirong, ZHENG Handan, WU Luyi, HU Zhihai, LIU Yanan. Herb-partitioned moxibustion regulated the miRNA expression profile in the thyroid tissues of rats with experimental autoimmune thyroiditis [J]. Journal of Traditional Chinese Medicine, 2021, 41(5): 789-798. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

