Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (1): 105-112.DOI: 10.19852/j.cnki.jtcm.2023.01.009
• Original articles • Previous Articles Next Articles
Huoxue Jiedu Huayu recipe (活血解毒化瘀方) alleviates contralateral renal fibrosis in unilateral ureteral obstruction rats by inhibiting the transformation of macrophages to myofibroblast
XIONG Yunzhao1, LIU Lingjin2, LIU Ziqian2, CHEN Gege2, HAO Juan2, GAO Xiaomeng2, QIANG Panpan2, WANG Zheng3( ), XU Qingyou3(
), XU Qingyou3( )
)
- 1 Graduate School, Hebei University of Chinese Medicine; Hebei Key Laboratory of Integrative Medicine on Liver-Kidney Patterns, Hebei University of Chinese Medicine; Department of Internal Medicine, Hebei University of Chinese Medicine, Shijiazhuang 050091, China
2 Graduate School, Hebei University of Chinese Medicine; Hebei Key Laboratory of Integrative Medicine on Liver-Kidney Patterns, Hebei University of Chinese Medicine, Shijiazhuang 050091, China
3 Graduate School, Hebei University of Chinese Medicine; Hebei Key Laboratory of Integrative Medicine on Liver-Kidney Patterns, Hebei University of Chinese Medicine; Department of Internal Medicine, Hebei University of Chinese Medicine, Shijiazhuang 050091, China
-
Received:2022-05-18Accepted:2022-09-14Online:2023-02-15Published:2023-01-10 -
Contact:WANG Zheng,XU Qingyou -
About author:Prof. WANG Zheng, Department of Internal Medicine, Hebei University of Chinese Medicine, Shijiazhuang 050091, China. 1017425887@qq.com. Telephone:+86-15350567700
Prof. XU Qingyou, Department of Internal Medicine, Hebei University of Chinese Medicine, Shijiazhuang 050091, China. qingyouxu@sohu.com;
-
Supported by:Effect of Macrophage-to-Myofibroblast Transition in Contralateral Kidney of Unilateral Ureteral Obstruction Rats Through the Aldosterone/ MR/SGK1 Pathway and Inhibition of Chinese Herbs(81873251);Aldosterone-induced Macrophage Polarization-lymphatic Endothelial Cell-like Trans-formation Involved in Renal Fibrosis and the Protective Effect of Yiqi Jiedu Huayu Herbs(82205006);Mechanism of Renal Fibrosis Induced by NF-kB/IL-8 Induced MMT in UUO Rats(BJK2022004)
Cite this article
XIONG Yunzhao, LIU Lingjin, LIU Ziqian, CHEN Gege, HAO Juan, GAO Xiaomeng, QIANG Panpan, WANG Zheng, XU Qingyou. Huoxue Jiedu Huayu recipe (活血解毒化瘀方) alleviates contralateral renal fibrosis in unilateral ureteral obstruction rats by inhibiting the transformation of macrophages to myofibroblast[J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 105-112.
share this article

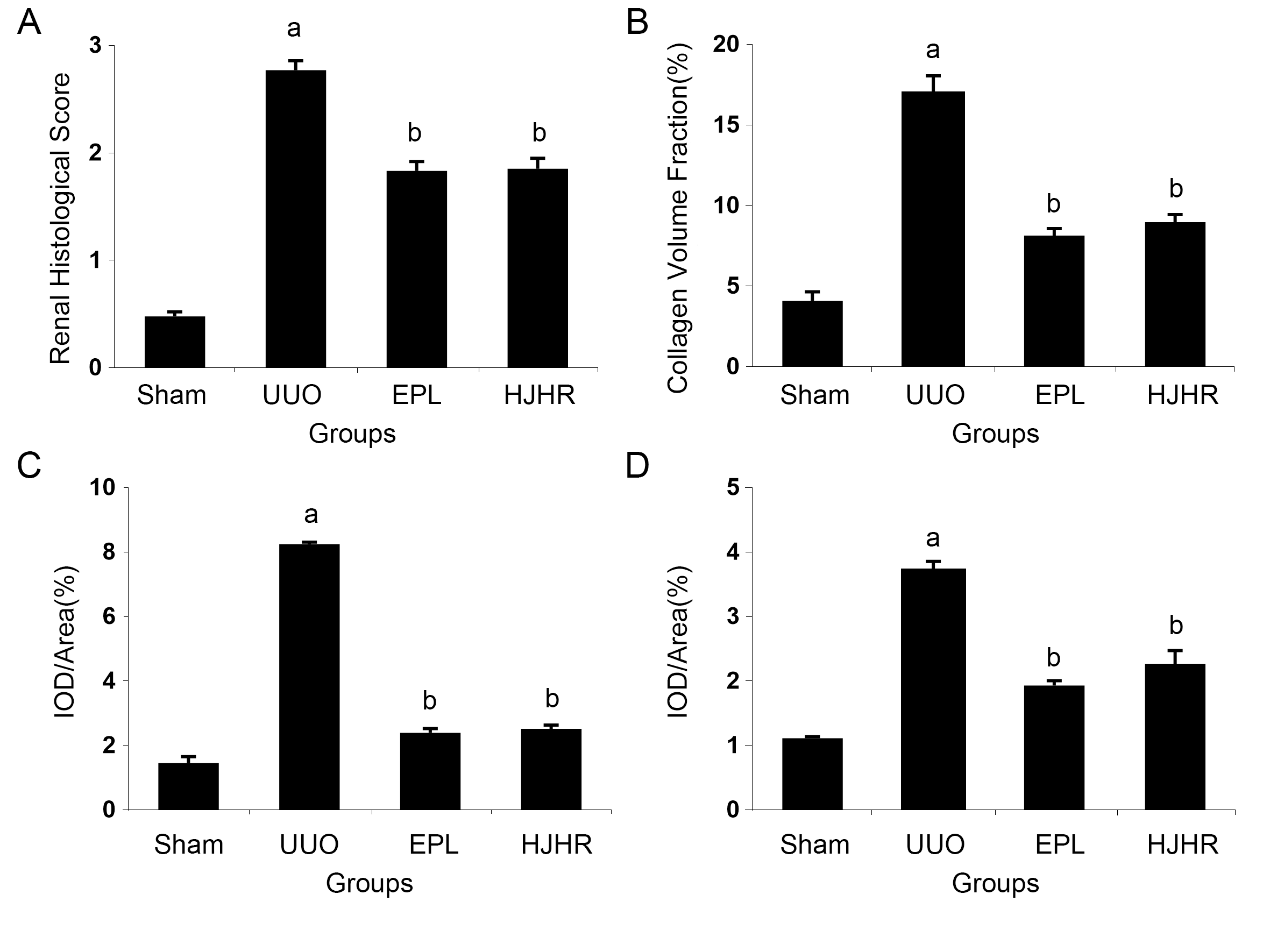
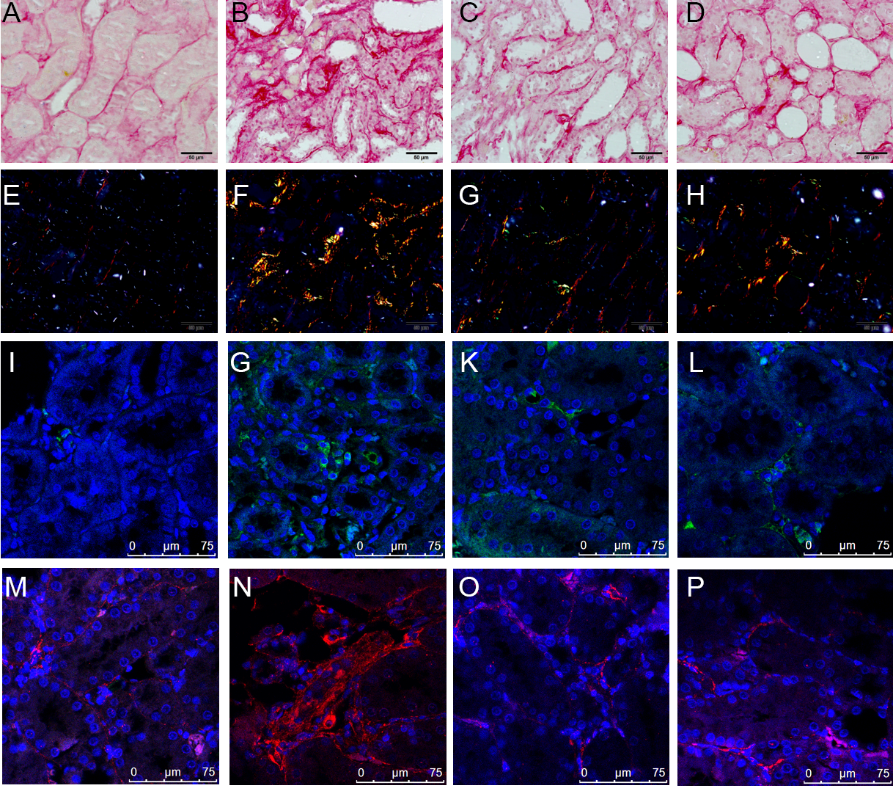
Figure 1 Effects of long-term UUO and treatment with Huoxue Jiedu Huayu recipe on renal histology and fibrosis of the contralateral kidneys A-D: kidney sections from all groups were stained with HE for morphological changes (× 40). A: sham group; B: UUO group; C: EPL group; D: HJHR group. E-H: kidney sections from all groups were stained with Masson’s trichrome for fibrosis (× 40). E: sham group; F: UUO group; G: EPL group; H: HJHR group. I-L: Kidney sections from all groups were stained with immunohistochemistry staining using antibodies against α-SMA to examine renal infiltration of myofibroblasts in the kidneys (× 40). I: sham group; J: UUO group; K: EPL group; L: HJHR group. M-P: kidney sections from all groups were stained with immunohistochemistry staining using antibodies against vimentin to examine renal infiltration of myofibroblasts in the kidneys (× 40). M: sham group; N: UUO group; O: EPL group; P: HJHR group. UUO: unilateral ureteral obstruction; HE: hematoxylin-eosin; EPL: eplerenone; HJHR: Huoxue Jiedu Huayu recipe; α-SMA: α-smooth muscle actin.

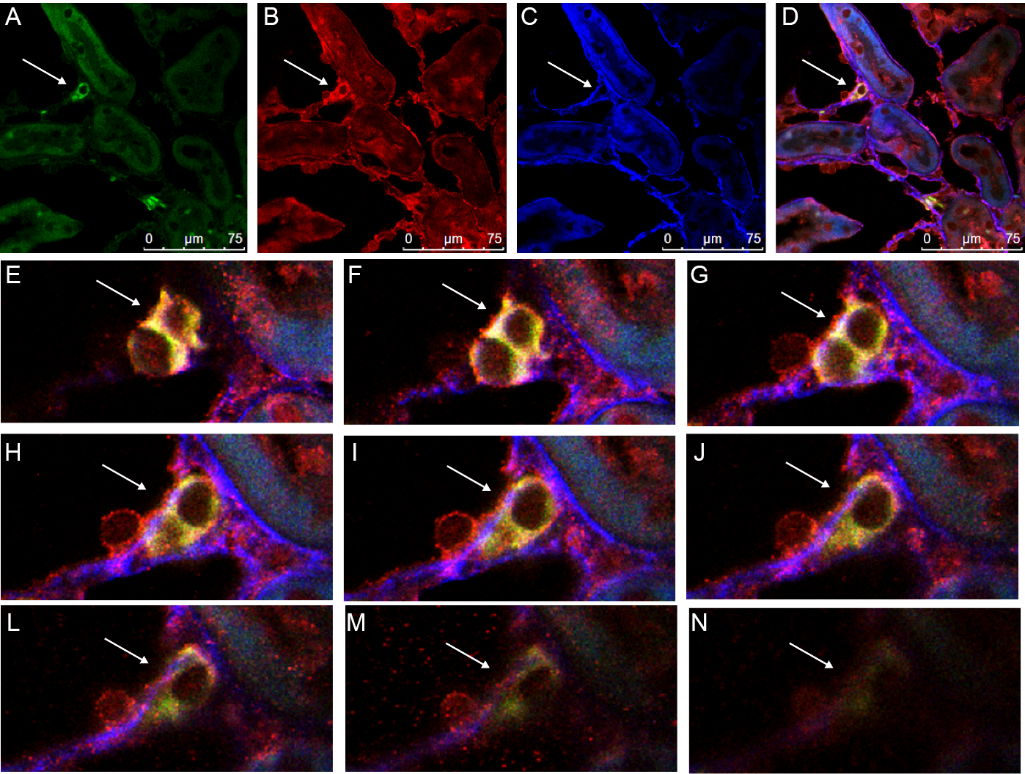
Figure 2 M2 macrophages are the major source of MMT in the contralateral kidneys of long-term UUO rats A-D: kidney sections from UUO group were stained with immunofluorescent co-staining of CD68 with the M2 macrophage marker CD206. A: nuclei were stained with DAPI in blue; B: macrophage marker CD68 were stained with FITC in green; C: M2 macrophage marker CD206 were stained with TRITC in red; D: immunofluorescent co-staining of CD68 (green) with the M2 macrophage marker CD206 (red). E-H: kidney sections from UUO group were stained with immunofluorescent co-staining of CD68 with the M1 macrophage marker iNOS. E: nuclei were stained with DAPI in blue; F: macrophage marker CD68 were stained with FITC in green; G: M2 macrophage marker iNOS were stained with TRITC in red; H: Immunofluorescent co-staining of CD68 (green) with the M1 macrophage marker iNOS (red). MMT: macrophage-to-myofibroblast transition; UUO: unilateral ureteral obstruction; DAPI: 4,6-diamino-2-phenyl indole; FITC: fluorescein isothiocyanate; TRITC: tetraethyl rhodamine isothiocyanate; iNOS: inducible nitric oxide synthase. All pictures are selected from the UUO group.
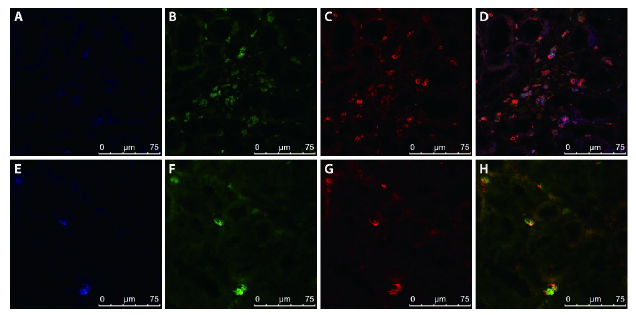
Figure 3 M2 macrophages are the major source of MMT in the contralateral kidneys of long-term UUO rats A-D: kidney sections from UUO group were stained with immunofluorescent multi-staining of CD68, the M2 macrophage marker CD206 and α-SMA. A: macrophage marker CD68 were stained with Alexa-405 in blue; B: M2 macrophage marker CD206 were stained with FITC in green; C: α-SMA were stained with TRITC in red; D: immunofluorescent multi -staining of CD68 (blue), CD206 (green) with the α-SMA (red). E-H: kidney sections from UUO group were stained with immunofluorescent multi-staining of CD68, the M1 macrophage marker iNOS and α-SMA. E: macrophage marker CD68 were stained with Alexa-405 in blue; F: M1 macrophage marker iNOS were stained with FITC in green; G: α-SMA were stained with TRITC in red; H: Immunofluorescent multi-staining of CD68 (blue), iNOS (green) with the α-SMA (red). MMT: macrophage-to-myofibroblast transition; UUO: unilateral ureteral obstruction; α-SMA: α-smooth muscle actin.; iNOS: inducible nitric oxide synthase; FITC: fluorescein isothiocyanate; TRITC: tetraethyl rhodamine isothiocyanate. All pictures are selected from the UUO group.
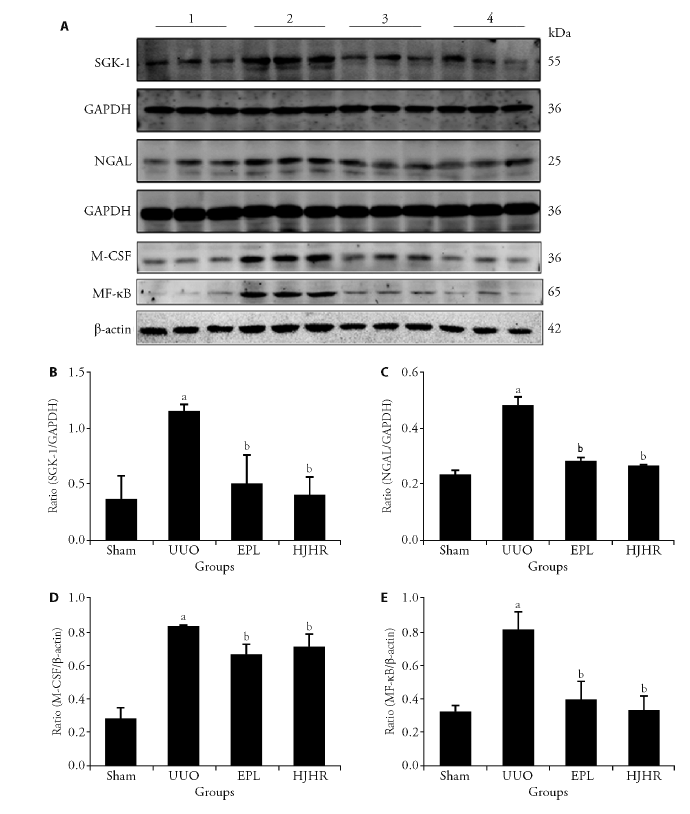
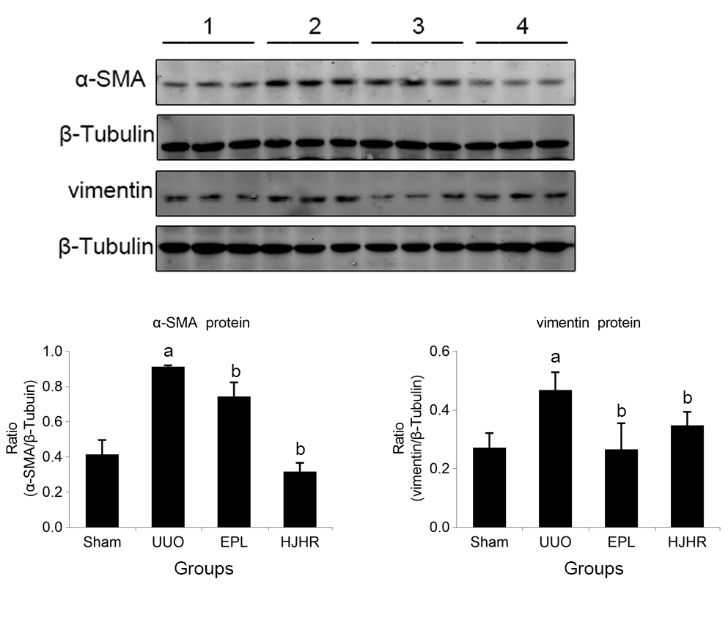
Figure 4 Effects of long-term UUO and treatment with Huoxue Jiedu Huayu recipe on the expression of MR downstream molecules A: Western blots using antibodies against SGK-1, NGAL, M-CSF and NF-κB to examine the activation of MR in the kidneys; B: protein ratio of SGK-1/GAPDH; C: protein ratio of NGAL/GAPDH; D: protein ratio of M-CSF/β-actin; E: protein ratio of NF-κB/β-actin. Western blots using antibodies against SGK-1, NGAL, M-CSF and NF-κB to examine the activation of MR in the kidneys 1: Sham group; 2: UUO group; 3: EPL group; 4: HJHR group. SGK-1: serine-glucocorticoid kinase-1; NGAL: neutrophil gelatinase-associated lipocalin; M-CSF: macrophage-colony-stimulating factor; NF-κB: nuclear factor-κB; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; MR: mineralocorticoid receptor; UUO: unilateral ureteral obstruction; EPL: eplerenone; HJHR: Huoxue Jiedu Huayu recipe. Images are representative of n = 12 rats in each group. Staining quantification data are presented as the mean ± stand error of mean, n = 6. aP < 0.05 vs the sham; bP < 0.05 vs the UUO.
| Group | n | SGK-1 mRNA | NF-κB mRNA | MCP-1 mRNA | TGF-β1 mRNA |
|---|---|---|---|---|---|
| Sham | 6 | 1.000±0.000 | 1.000±0.000 | 1.000±0.000 | 1.000±0.000 |
| UUO | 6 | 2.553±0.346a | 3.426±0.195a | 1.908±0.174a | 7.503±1.962a |
| EPL | 6 | 0.922±0.102b | 1.889±0.367b | 1.377±0.168b | 2.318±0.517b |
| HJHR | 6 | 0.964±0.322b | 1.964±0.475b | 1.445±0.071b | 5.262±0.653b |
Table 1 Expression of SGK-1, NF-κB, MCP-1 and TGF-β1 mRNA in kidney tissue of UUO rats
| Group | n | SGK-1 mRNA | NF-κB mRNA | MCP-1 mRNA | TGF-β1 mRNA |
|---|---|---|---|---|---|
| Sham | 6 | 1.000±0.000 | 1.000±0.000 | 1.000±0.000 | 1.000±0.000 |
| UUO | 6 | 2.553±0.346a | 3.426±0.195a | 1.908±0.174a | 7.503±1.962a |
| EPL | 6 | 0.922±0.102b | 1.889±0.367b | 1.377±0.168b | 2.318±0.517b |
| HJHR | 6 | 0.964±0.322b | 1.964±0.475b | 1.445±0.071b | 5.262±0.653b |
| 1 |
Chung EYM, Strippoli GFM. Aldosterone antagonists in addition to renin angiotensin system antagonists for preventing the progression of CKD: editorial summary of a cochrane review. Am J Kidney Dis 2021; 77: 810-12.
DOI PMID |
| 2 |
Yang J, Li J, Tan R, et al. Protocatechualdehyde attenuates obstructive nephropathy through inhibiting lncRNA9884 induced inflammation. Phytother Res 2021; 35: 1521-33.
DOI PMID |
| 3 | Schwenk MH, Hirsch JS, Bomback AS. Aldosterone blockade in CKD: emphasis on pharmacology. Adv Chronic Kidney Dis 2015; 22: 123-32. |
| 4 | Young MJ. Mechanisms of mineralocorticoid receptor-mediated cardiac fibrosis and vascular inflammation. Curr Opin Nephrol Hypertens 2008; 17: 174-80. |
| 5 |
Belden Z, Deiuliis JA, Dobre M, Rajagopalan S. The role of the mineralocorticoid receptor in inflammation: focus on kidney and vasculature. Am J Nephrol 2017; 46: 298-314.
DOI PMID |
| 6 | Lieber GB, Fernandez X, Mingo GG, et al. Mineralocorticoid receptor antagonists attenuate pulmonary inflammation and bleomycin-evoked fibrosis in rodent models. Eur J Pharmacol 2013; 718: 290-8. |
| 7 |
Joffe HV, Adler GK. Effect of aldosterone and mineralocorticoid receptor blockade on vascular inflammation. Heart Fail Rev 2005; 10: 31-7.
PMID |
| 8 |
Del Vecchio L, Procaccio M, Vigano S, Cusi D. Mechanisms of disease: the role of aldosterone in kidney damage and clinical benefits of its blockade. Nat Clin Pract Nephrol 2007; 3: 42-9.
PMID |
| 9 |
Eddy AA. The origin of scar-forming kidney myofibroblasts. Nat Med 2013; 19: 964-6.
DOI PMID |
| 10 |
LeBleu VS, Taduri G, O'Connell J, et al. Origin and function of myofibroblasts in kidney fibrosis. Nat Med 2013; 19: 1047-53.
DOI PMID |
| 11 | Nikolic-Paterson DJ, Wang S, Lan HY. Macrophages promote renal fibrosis through direct and indirect mechanisms. Kidney Int Suppl (2011) 2014; 4: 34-8. |
| 12 | Wang S, Meng XM, Ng YY, et al. TGF-beta/Smad3 signalling regulates the transition of bone marrow-derived macrophages into myofibroblasts during tissue fibrosis. Oncotarget 2016; 7: 8809-22. |
| 13 | Wang CH, Wang Z, Liang LJ, et al. The inhibitory effect of eplerenone on cell proliferation in the contralateral kidneys of rats with unilateral ureteral obstruction. Nephron 2017; 136: 328-38. |
| 14 | Xiong Y, Chang Y, Hao J, et al. Eplerenone attenuates fibrosis in the contralateral kidney of UUO rats by preventing macrophage-to-myofibroblast transition. Front Pharmacol 2021; 12: 620433. |
| 15 | Xu QY, Han L, Qin JG, et al. Zhao Yuyong's pathogenesis theory and clinical application of "kidney collateral stasis". Zhong Guo Zhong Yi Yao Za Zhi 2010; 25: 702-04. |
| 16 | Zhang Y, Hao J, Ma X, et al. Huoxue Jiedu Huayu recipe ameliorates mesangial cell pyroptosis in contralateral kidney of UUO Rats. Evid Based Complement Alternat Med 2020; 2020: 2530431. |
| 17 |
Ranjit S, Dvornikov A, Levi M, Furgeson S, Gratton E. Characterizing fibrosis in UUO mice model using multiparametric analysis of phasor distribution from FLIM images. Biomed Opt Express 2016; 7: 3519-30.
PMID |
| 18 | Fan HY, Yang MY, Qi D, et al. Salvianolic acid A as a multifunctional agent ameliorates doxorubicin-induced nephropathy in rats. Sci Rep 2015; 5: 12273. |
| 19 |
Li L, Shen Y, Ding Y, Liu Y, Su D, Liang X. Hrd1 participates in the regulation of collagen I synthesis in renal fibrosis. Mol Cell Biochem 2014; 386: 35-44.
DOI PMID |
| 20 |
Wynn TA, Barron L. Macrophages: master regulators of inflammation and fibrosis. Semin Liver Dis 2010; 30: 245-57.
DOI PMID |
| 21 | Martin-Fernandez B, Rubio-Navarro A, Cortegano I, et al. Aldosterone induces renal fibrosis and inflammatory M1-Macrophage subtype via mineralocorticoid receptor in rats. PLoS One 2016; 11: e0145946. |
| 22 | Wang YY, Jiang H, Pan J, et al. Macrophage-to-myofibroblast transition contributes to interstitial fibrosis in chronic renal allograft injury. J Am Soc Nephrol 2017; 28: 2053-67. |
| 23 | Sun JY, Li C, Shen ZX, et al. Mineralocorticoid receptor deficiency in macrophages inhibits neointimal hyperplasia and suppresses macrophage inflammation through SGK1-AP1/NF-kappa B pathways. Arterioscler Thromb Vasc Biol 2016; 36: 874-85. |
| 24 |
Chevalier RL. Obstructive nephropathy: lessons from cystic kidney disease. Nephron 2000; 84: 6-12.
PMID |
| 25 | Ma X, Chang Y, Xiong Y, Wang Z, Wang X, Xu Q. Eplerenone ameliorates cell pyroptosis in contralateral kidneys of rats with unilateral ureteral obstruction. Nephron 2019; 142: 233-42. |
| 26 | Navaneethan SD, Nigwekar SU, Sehgal AR, Strippoli GF. Aldosterone antagonists for preventing the progression of chronic kidney disease: a systematic review and Meta-analysis. Clin J Am Soc Nephrol 2009; 4: 542-51. |
| 27 |
Shrestha A, Che RC, Zhang AH. Role of aldosterone in renal fibrosis. Adv Exp Med Biol 2019; 1165: 325-46.
DOI PMID |
| 28 |
Feng Y, Wang Q, Wang Y, Yard B, Lang F. SGK1-mediated fibronectin formation in diabetic nephropathy. Cell Physiol Biochem 2005; 16: 237-44.
DOI PMID |
| 29 | Lang F, Voelkl J. Therapeutic potential of serum and glucocorticoid inducible kinase inhibition. Expert Opin Investig Drugs 2013; 22: 701-14. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||