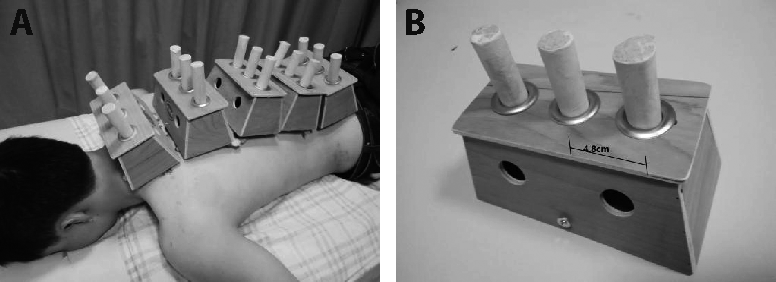
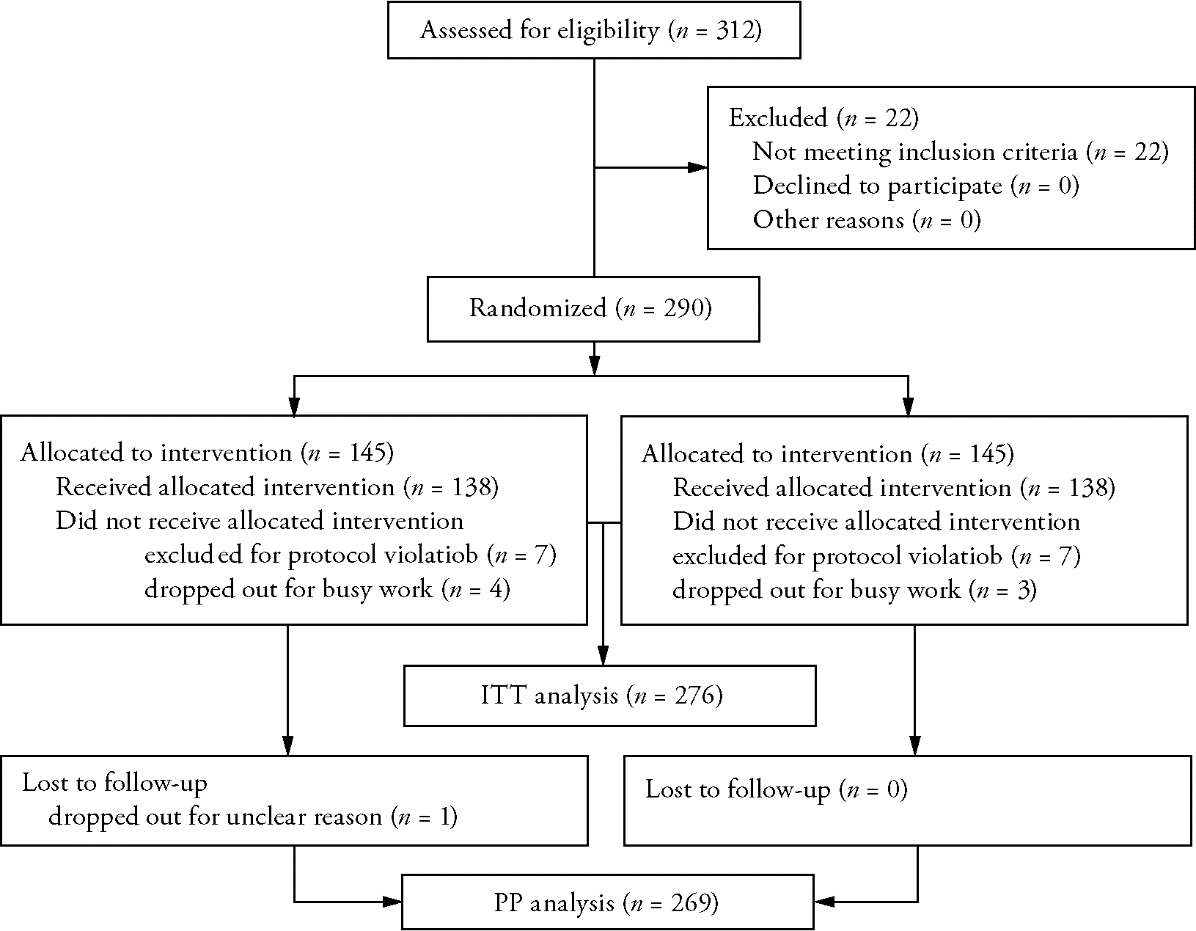
Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (2): 242-249.DOI: 10.19852/j.cnki.jtcm.20211214.003
• Research Articles • Previous Articles Next Articles
Ginger-indirect moxibustion plus acupuncture versus acupuncture alone for chronic fatigue syndrome: a randomized controlled trial
MA Tingting1, WU Jie1( ), YANG Lijie1, FENG Fen1, YANG Huilin2, ZHANG Jinhua2, ZHONG Yanjin2, NING Qing2, HUANG Lirong2, LIN Youbing2, YAN Jue3, CHEN Guiquan3, HOU Tianshu4, WANG Li4, REN Yuanfang2, TAN Jing5
), YANG Lijie1, FENG Fen1, YANG Huilin2, ZHANG Jinhua2, ZHONG Yanjin2, NING Qing2, HUANG Lirong2, LIN Youbing2, YAN Jue3, CHEN Guiquan3, HOU Tianshu4, WANG Li4, REN Yuanfang2, TAN Jing5
- 1 Center of Preventive Medicine, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610075, China
2 College of Acupuncture and Tuina, Chengdu University of Traditional Chinese Medicine, Chengdu 610075, China
3 Department of acupuncture, Center of Preventive Medicine, Affiliated TCM Hospital of Southwest Medical University, Luzhou 646600, China
4 Center of Preventive Medicine, Chengdu Integrated TCM and Western Medicine Hospital, Chengdu 610021, China
5 Center of Chinese Evidence-Based Medicine, Sichuan University West China Hospital, Chengdu 610041, China
-
Received:2021-05-16Accepted:2021-08-22Online:2021-12-14Published:2021-12-14 -
Contact:WU Jie -
About author:WU Jie, Center of Preventive Medicine, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610075, China. drwujie@163.com, Telephone: +86-028-87783128
-
Supported by:Science and Technology Support Program of Sichuan Province Science and Technology Agency: Clinical Researches of Acupuncture for “Unhealthy Status” Based on the TCM Theory of “All Diseases Originate from Qi’s Problem”(2014SZ0152)
Cite this article
MA Tingting, WU Jie, YANG Lijie, FENG Fen, YANG Huilin, ZHANG Jinhua, ZHONG Yanjin, NING Qing, HUANG Lirong, LIN Youbing, YAN Jue, CHEN Guiquan, HOU Tianshu, WANG Li, REN Yuanfang, TAN Jing. Ginger-indirect moxibustion plus acupuncture versus acupuncture alone for chronic fatigue syndrome: a randomized controlled trial[J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 242-249.
share this article
| Item | Group A (n=138) | Group B (n=138) | |
|---|---|---|---|
| Age (years) [median (QL-QU)] | 27.5 (24.5-41.4) | 27.3 (24.5-38.2) | |
| 18-29.99 | 78 (56.52) | 82 (59.42) | |
| 30-39.99 | 22 (15.94) | 25 (18.12) | |
| 40-55.49 | 38 (27.54) | 31 (22.46) | |
| Female [n (%)] | 109 (78.99) | 113 (81.88) | |
| BMI (kg/m2) [mean (SD)] | 21.44 (2.55) | 21.20 (2.54) | |
| Duration of disease (month) [Median (QL-QU)] | 18 (10-30) | 18 (12-30) | |
| Expectant degree for effect [n (%)] | Low | 27 (19.57) | 20 (14.49) |
| Moderate | 68 (49.28) | 77 (55.80) | |
| High | 43 (31.16) | 41 (29.71) | |
| FSS at baseline [mean (SD)] | 39.69 (9.17) | 39.45 (9.13) | |
| SPHERE at baseline [mean (SD)] | 17.54 (4.79) | 17.46 (5.21) | |
| SDS at baseline [mean (SD)] | 48.21 (10.39) | 50.17 (10.99) | |
| HAMA at baseline [mean (SD)] | 12.86 (6.36) | 13.67 (8.22) |
Table 1 Characteristics of patients at baseline
| Item | Group A (n=138) | Group B (n=138) | |
|---|---|---|---|
| Age (years) [median (QL-QU)] | 27.5 (24.5-41.4) | 27.3 (24.5-38.2) | |
| 18-29.99 | 78 (56.52) | 82 (59.42) | |
| 30-39.99 | 22 (15.94) | 25 (18.12) | |
| 40-55.49 | 38 (27.54) | 31 (22.46) | |
| Female [n (%)] | 109 (78.99) | 113 (81.88) | |
| BMI (kg/m2) [mean (SD)] | 21.44 (2.55) | 21.20 (2.54) | |
| Duration of disease (month) [Median (QL-QU)] | 18 (10-30) | 18 (12-30) | |
| Expectant degree for effect [n (%)] | Low | 27 (19.57) | 20 (14.49) |
| Moderate | 68 (49.28) | 77 (55.80) | |
| High | 43 (31.16) | 41 (29.71) | |
| FSS at baseline [mean (SD)] | 39.69 (9.17) | 39.45 (9.13) | |
| SPHERE at baseline [mean (SD)] | 17.54 (4.79) | 17.46 (5.21) | |
| SDS at baseline [mean (SD)] | 48.21 (10.39) | 50.17 (10.99) | |
| HAMA at baseline [mean (SD)] | 12.86 (6.36) | 13.67 (8.22) |
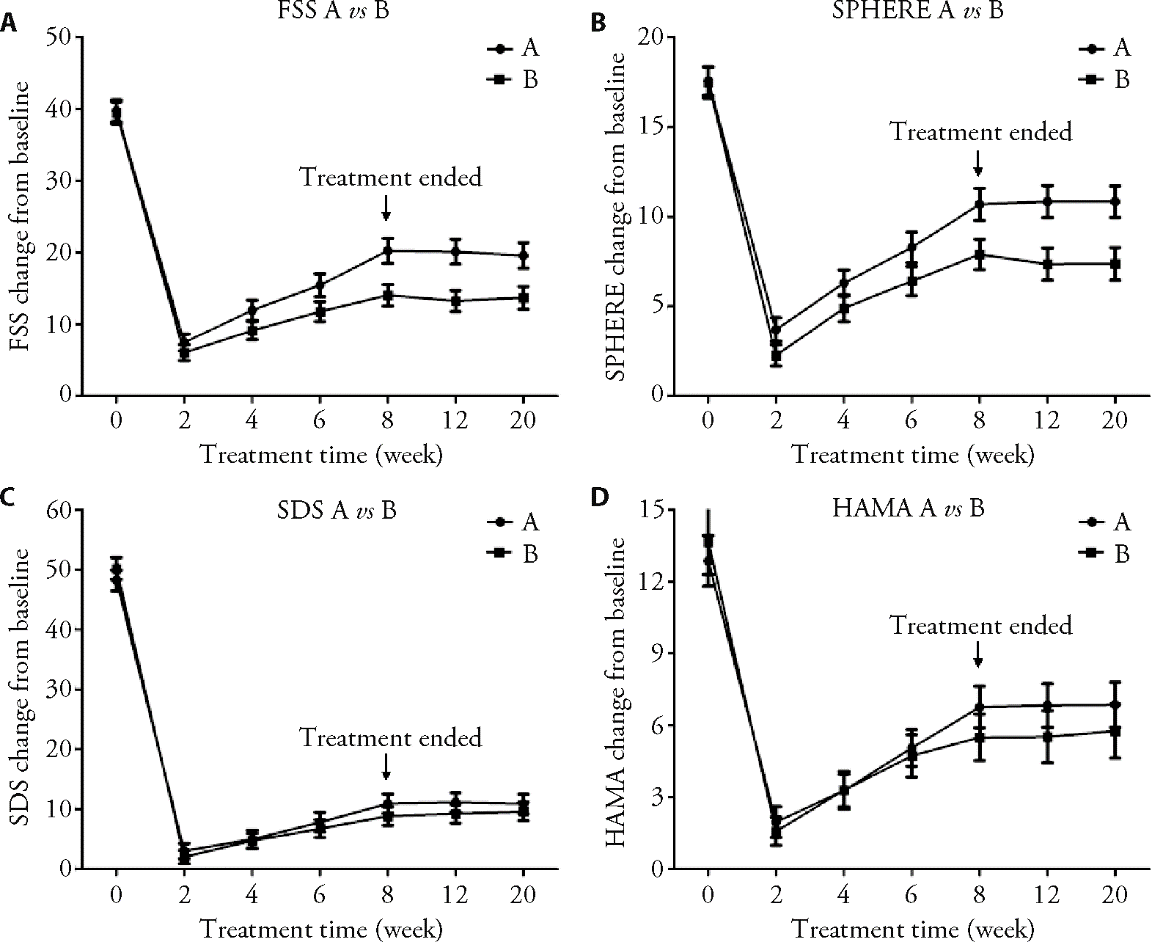
Figure 3 Outcome change during treatment and follow-up Data were shown by mean and 95% confidence interval of difference from baseline, by setting the starting point as the absolute value of specific outcome at baseline. A: Fatigue Severity Scale (FSS) score at baseline and changes after 2, 4, 6 and 8 weeks' treatment and during the follow-up period in A and B groups. Group A demonstrated superiority after 4 weeks treatment, and its superiority sustained until week 20; B: Somatic and Psychological Health Report (SPHERE) at baseline and changes after 2, 4, 6 and 8 weeks' treatment and during the follow-up period in A and B groups. Group A demonstrated superiority after 2 weeks treatment, and its superiority sustained until week 20; C: Self-rating Depression Scale (SDS) at baseline and changes after 2, 4, 6 and 8 weeks' treatment and during the follow-up period in A and B groups. There was no statistical difference between A and B groups; D: Hamilton Anxiety (HAMA) scale at baseline and changes after 2, 4, 6 and 8 weeks' treatment and during the follow-up period in A and B groups. There was no statistical difference between A and B groups).
| Time | FSS | SPHERE | SDS | HAMA | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Group A (n =138) | Group B (n = 138) | MD (95% CI) | Group A (n = 138) | Group B (n = 138) | MD (95% CI) | Group A (n = 138) | Group B (n = 138) | MD (95% CI) | Group A (n = 138) | Group B (n = 138) | MD (95%CI) | ||||||||
| Week 2 | 7.47 (6.88) | 6.06 (6.62) | 1.41 (–0.18, 3.01) | 3.70 (3.90) | 2.27 (3.52) | 1.43 (0.56, 2.31) | 2.99 (7.57) | 2.01 (6.82) | 0.97 (–0.73, 2.67) | 1.96 (3.81) | 1.57 (3.54) | 0.38 (﹣0.48, 1.25) | |||||||
| Week 4 | 11.94 (8.42) | 9.12 (7.52) | 2.82 (0.94, 4.70) | 6.30 (4.37) | 4.89 (4.48) | 1.41 (0.36, 2.45) | 4.95 (9.13) | 4.69 (7.78) | 0.26 (–1.74, 2.26) | 3.28 (4.11) | 3.29 (4.72) | –0.01 (–1.06, 1.03) | |||||||
| Week 6 | 15.44 (9.61) | 11.76 (8.30) | 3.68 (1.56, 5.80) | 8.28 (5.09) | 6.41 (4.91) | 1.87 (0.69, 3.05) | 7.81 (9.77) | 6.74 (8.80) | 1.07 (–1.12, 3.27) | 5.05 (4.61) | 4.72 (5.29) | 0.33 (–0.85, 1.50) | |||||||
| Week 8 (Treat end) | 20.22 (10.46) | 14.03 (8.99) | 6.20 (3.89, 8.50) | 10.67 (5.36) | 7.89 (5.03) | 2.78 (1.56, 4.01) | 10.90 (9.68) | 8.83 (9.17) | 2.07 (–0.15, 4.30) | 6.76 (5.22) | 5.49 (5.84) | 1.27 (–0.04, 2.58) | |||||||
| Week 12 (Follow–up) | 20.10 (10.31) | 13.25 (8.85) | 6.86 (4.59, 9.12) | 10.83 (5.35) | 7.36 (5.31) | 3.48 (2.22, 4.74) | 11.14 (9.39) | 9.22 (9.29) | 1.92 (–0.28, 4.12) | 6.83 (5.46) | 5.52 (6.50) | 1.31 (–0.10, 2.73) | |||||||
| Week 20 (Follow–up) | 19.56 (10.64) | 13.68 (9.48) | 5.88 (3.50, 8.25) | 10.83 (5.26) | 7.37 (5.43) | 3.46 (2.20, 4.72) | 10.86 (9.75) | 9.59 (9.12) | 1.27 (–0.96, 3.49) | 6.86 (5.64) | 5.76 (6.76) | 1.09 (–0.37, 2.56) | |||||||
Table 2 Outcome difference between groups A and B across various times
| Time | FSS | SPHERE | SDS | HAMA | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Group A (n =138) | Group B (n = 138) | MD (95% CI) | Group A (n = 138) | Group B (n = 138) | MD (95% CI) | Group A (n = 138) | Group B (n = 138) | MD (95% CI) | Group A (n = 138) | Group B (n = 138) | MD (95%CI) | ||||||||
| Week 2 | 7.47 (6.88) | 6.06 (6.62) | 1.41 (–0.18, 3.01) | 3.70 (3.90) | 2.27 (3.52) | 1.43 (0.56, 2.31) | 2.99 (7.57) | 2.01 (6.82) | 0.97 (–0.73, 2.67) | 1.96 (3.81) | 1.57 (3.54) | 0.38 (﹣0.48, 1.25) | |||||||
| Week 4 | 11.94 (8.42) | 9.12 (7.52) | 2.82 (0.94, 4.70) | 6.30 (4.37) | 4.89 (4.48) | 1.41 (0.36, 2.45) | 4.95 (9.13) | 4.69 (7.78) | 0.26 (–1.74, 2.26) | 3.28 (4.11) | 3.29 (4.72) | –0.01 (–1.06, 1.03) | |||||||
| Week 6 | 15.44 (9.61) | 11.76 (8.30) | 3.68 (1.56, 5.80) | 8.28 (5.09) | 6.41 (4.91) | 1.87 (0.69, 3.05) | 7.81 (9.77) | 6.74 (8.80) | 1.07 (–1.12, 3.27) | 5.05 (4.61) | 4.72 (5.29) | 0.33 (–0.85, 1.50) | |||||||
| Week 8 (Treat end) | 20.22 (10.46) | 14.03 (8.99) | 6.20 (3.89, 8.50) | 10.67 (5.36) | 7.89 (5.03) | 2.78 (1.56, 4.01) | 10.90 (9.68) | 8.83 (9.17) | 2.07 (–0.15, 4.30) | 6.76 (5.22) | 5.49 (5.84) | 1.27 (–0.04, 2.58) | |||||||
| Week 12 (Follow–up) | 20.10 (10.31) | 13.25 (8.85) | 6.86 (4.59, 9.12) | 10.83 (5.35) | 7.36 (5.31) | 3.48 (2.22, 4.74) | 11.14 (9.39) | 9.22 (9.29) | 1.92 (–0.28, 4.12) | 6.83 (5.46) | 5.52 (6.50) | 1.31 (–0.10, 2.73) | |||||||
| Week 20 (Follow–up) | 19.56 (10.64) | 13.68 (9.48) | 5.88 (3.50, 8.25) | 10.83 (5.26) | 7.37 (5.43) | 3.46 (2.20, 4.72) | 10.86 (9.75) | 9.59 (9.12) | 1.27 (–0.96, 3.49) | 6.86 (5.64) | 5.76 (6.76) | 1.09 (–0.37, 2.56) | |||||||
| 1. |
Reeves WC, Lloyd A, Vernon SD, et al. Identification of ambiguities in the 1994 chronic fatigue syndrome research case definition and recommendations for resolution. BMC Health Serv Res 2003; 3: 25-33.
DOI URL |
| 2. |
Capelli E, Zola R, Lorusso L, Venturini L, Sardi F, Ricevuti G. Chronic fatigue syndrome/myalgic encephalomyelitis: an update. Int J Immunopathol Pharmacol 2010; 23: 981-9.
PMID |
| 3. | Ng SM, Yiu YM. Acupuncture for chronic fatigue syndrome: a randomized, sham-controlled trial with single-blinded design. Altern Ther Health Med 2013; 19: 21-6. |
| 4. | Oka T, Tanahashi T, Chijiwa T, Lkhagvasuren B, Sudo N, Oka K. Isometric yoga improves the fatigue and pain of patients with chronic fatigue syndrome who are resistant to conventional therapy: a randomized, controlled trial. Bio Psycho Soc Med 2014; 8: 27. |
| 5. | Price JR, Mitchell E, Tidy E, et al. Cognitive behaviour therapy for chronic fatigue syndrome in adults (review). Cochrane Database Syst Rev 2008; 2008: 1-67. |
| 6. |
White PD, Goldsmith KA, Johnson AL, et al. Comparison of adaptive pacing therapy, cognitive behaviour therapy, graded exercise therapy, and specialist medical care for chronic fatigue syndrome (PACE): a randomised trial. Lancet 2011; 377: 823-36.
DOI PMID |
| 7. |
Van Cauwenbergh D, De Kooning M, Ickmans K, Nijs J. How to exercise people with chronic fatigue syndrome: evidence-based practice guidelines. Eur J Clin Invest 2012; 42: 1136-44.
DOI PMID |
| 8. |
Wang T, Zhang Q, Xue X, Yeung A. A systematic review of acupuncture and moxibustion treatment for chronic fatigue syndrome in China. Am J Chin Med 2008; 36: 1-24.
DOI URL |
| 9. |
Wang YY, Li XX, Liu JP, Luo H, Ma LX, Alraek T. Traditional Chinese Medicine for chronic fatigue syndrome: a systematic review of randomized clinical trials. Complement Ther Med 2014; 22: 826-33.
DOI URL |
| 10. | Tan HJ, Luo TT, Qu WZ, Wen MX, Yang L, Wu X. Acupoint selection rules of acupuncture for chronic fatigue syndrome based on data mining technology. Zhong Guo Zhong Yi Ji Chu Yi Xue Za Zhi 2017; 23: 242-4+98. |
| 11. |
Wang T, Xu C, Pan K, Xiong H. Acupuncture and moxibustion for chronic fatigue syndrome in Traditional Chinese Medicine: a systematic review and Meta-analysis. BMC Complement Altern Med 2017; 17: 163.
DOI URL |
| 12. |
Lerdal A, Wahl A, Rustoen T, Hanestad BR, Moum T. Fatigue in the general population: a translation and test of the psychometric properties of the Norwegian version of the fatigue severity scale. Scand J Public Health 2005; 33: 123-30.
DOI URL |
| 13. | Hickie IB, Davenport TA, Hadzi-Pavlovic D, et al. Development of a simple screening tool for common mental disorders in general practice. Med J Aust 2001; 175(Suppl):S10-7. |
| 14. | Zhang RF. A randomized controlled study on treating chronic fatigue syndrome by acupuncturing acupoints on meridians. Chengdu: Chengdu University of Traditional Chinese Medicine, 2012: 21-41. |
| 15. |
Wang W, Russell A, Yan Y. Traditional Chinese Medicine and new concepts of predictive, preventive and personalized medicine in diagnosis and treatment of suboptimal health. EPMA J 2014; 5: 4.
DOI PMID |
| 16. | National Collaborating Centre for Primary Care (UK), Chronic fatigue syndrome/myalgic encephalomyelitis (or encephalopathy): diagnosis and management of chronic fatigue syndrome/myalgic encephalomyelitis (or encephalopathy) in adults and children (Royal College of General Practitioners, London, 2007). |
| 17. | Yancey JR, Thomas SM. Chronic fatigue syndrome: diagnosis and treatment. Am Fam Physician 2012; 86: 741-6. |
| 18. |
Kim JE, Seo BK, Choi JB, et al. Acupuncture for chronic fatigue syndrome and idiopathic chronic fatigue: a multicenter, nonblinded, randomized controlled trial. Trials 2015; 16: 314.
DOI URL |
| 19. | Amorim D, Amado J, Brito I, et al. Acupuncture and electroacupuncture for anxiety disorders: a systematic review of the clinical research. Complement Ther Clinl Pract 2018; 31: 31-7. |
| 20. |
Bosch P, van den Noort M, Staudte H, Lim S. Schizophrenia and depression: a systematic review of the effectiveness and the working mechanisms behind acupuncture. Explore (NY) 2015; 11: 281-91.
DOI URL |
| 21. |
Kim HG, Yoo SR, Park HJ, Son CG. Indirect moxibustion (CV4 and CV8) ameliorates chronic fatigue: a randomized, double-blind, controlled study. J Altern Complement Med 2013; 19: 134-40.
DOI URL |
| 22. |
Shu Q, Wang H, Litscher D, et al. Acupuncture and moxibustion have different effects on fatigue by regulating the autonomic nervous system: a pilot controlled clinical trial. Sci Rep 2016; 6: 37846.
DOI URL |
| 23. |
S Pettersson, IE Lundberg, MH Liang, et al. Determination of the minimal clinically important difference for seven measures of fatigue in Swedish patients with systemic lupus erythematosus. Scand J Rheumatol 2015; 44: 1-5.
DOI URL |
| [1] | QIN Xiaoyu, WANG Chunai, XUE Jianjun, ZHANG Jie, LU Xiaoting, DING Shengshuang, GE Long, WANG Minzhen. Efficacy of electroacupuncture on myocardial protection and postoperative rehabilitation in patients undergoing cardiac surgery with cardiopulmonary bypass: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 1-15. |
| [2] | DAI Xiaoling, ZHANG Anming, LIN Hui, SHI Bei, REN Yi, WEN Hongzhu, FEI Xiaoyan, LIN Jiang. Qingchang suppositry (清肠栓) induced remission in patients with mild-to-moderate ulcerative proctitis: a multicenter, prospective, randomized, parallel-controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 156-162. |
| [3] | WANG Yichen, WU Shiyi, WANG Zhengyan, CHANG Wenling, XIE Zhihao, TANG Xing, ZHAO Songmei, ZHOU Jing, CHEN Zehong, WANG Chao, YANG Chunxia. Efficacy of Zhumian Tang formula granules (助眠汤配方颗粒) combined with eszopiclone for the treatment of poor sleep quality: a multi-center, randomized controlled, superiority trial [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 163-171. |
| [4] | YANG Yi, YE Huijun, ZHENG Huiling, JIN Lihua. Clinical observation on 90 cases of primary dysmenorrhea treated by buccal acupuncture therapy: a randomized controlled study [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 172-181. |
| [5] | DAI Zeqi, LIAO Xing, GUAN Yueyue, ZENG Zixiu, TANG Jun, HU Jing. Bloodletting puncture in the treatment of acute ischemic stroke: protocol for a mixed-method study of a multi-center randomized controlled trial and focus group [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1259-1267. |
| [6] | XU Yani, ZHANG Yutong, HE Weile, DAI Linglin, TANG Ding, WANG Jialing, ZHANG Xufen, CHEN Qin, CHEN Lifang, WANG Zhanglian, ZHAN Mingjie. Efficiency and safety of acupuncture for women with premature ovarian insufficiency: study protocol for a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1268-1274. |
| [7] | XU Xiangru, ZHOU Yi, CHEN Gang, LEI Ming, ZHANG Wen, WU Xinxin, PU Yuting, CHEN Caiyu, SUN Yuting, ZHOU Shuang, FANG Bangjiang. Clinical efficacy of Buzhong Yiqi decoction (补中益气汤) in the treatment of hospital-acquired pneumonia with multi-drug resistant bacteria: a prospective, randomized, multicenter controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1010-1018. |
| [8] | ZHAO Ming, LUO Yimiao, WANG Huichan, CAO Yu, MA Lina, PEI Hui, LI Hao. Guilingji capsule (龟龄集胶囊) for Alzheimer's disease: secondary analysis of a randomized non-inferiority controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1019-1025. |
| [9] | ZHANG Meizhen, HAO Xiaohui, TANG Yiting, CHEN Yupeng, HE Puyu, ZHAO Liming, PANG Bing, NI Qing. Efficacy and safety of Buyang Huanwu decoction (补阳还五汤) for diabetic peripheral neuropathy: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 841-850. |
| [10] | YANG Yuqing, CHEN Yuhuan, LI Chunxiao, LING Xiao, WANG Panpan, GUO Jing, ZHANG Yingying. Effectiveness and safety of Pingxiao capsule (平消胶囊) as adjuvant therapy in treatment of breast cancer: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 851-859. |
| [11] | SUN Wu, ZHAO Yuwei, LIAO Liang, ZHAO Zhonghui, CHEN Shiqi, YAN Xiaoling, WANG Xueyao, CHAO Guojun, ZHOU Jian. Effectiveness and safety of Xuebijing injection for patients with coronavirus disease 2019: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 631-639. |
| [12] | WANG Chao, WU Qiong, LI Ping, WANG Zhigang, LOU Xusheng, LI Yuanyuan, ZHANG Lin. Effect of Traditional Chinese Medicine combined with Western Medicine on blood lipid levels and inflammatory factors in patients with angina pectoris in coronary heart disease identified as intermingled phlegm and blood stasis syndrome: a network Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 640-649. |
| [13] | ZHANG Xinghe, LI Qifu, YI Rong, XING Chonghui, JIN Yuhao, MENG Jiangqiong, FENG Jialei, ZHAO Siwen, LIANG Fanrong, GUO Taipin. Effect of catgut embedding at acupoints versus non-acupoints in abdominal obesity: a randomized clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 780-786. |
| [14] | LUO Xin, XIE Jing, HUANG Li, GAN Wenfan, CHEN Ming. Efficacy and safety of activating blood circulation and removing blood stasis of Traditional Chinese Medicine for managing renal fibrosis in patients with chronic kidney disease: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 429-440. |
| [15] | ZHANG Yuehong, SHAO Xianzhi, ZHAO Qianlong, ZHAN Hualong, ZHANG Jianhua, DU Sisi, CHEN Jing, LIU Yingfang, ZHOU Haiwang, CHEN Xinsheng, HONG Ying, LIAN Fengmei, TONG Xiaolin, BA Yuanming. Effectiveness of Xiangsha Liujun pills (香砂六君丸) on decreased digestive function in convalescent patients of coronavirus disease 2019: a randomized, double blind, placebo controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 552-558. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||