Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (6): 965-971.DOI: 10.19852/j.cnki.jtcm.20220928.003
• Research Articles • Previous Articles Next Articles
Effect of electroacupuncture on inflammatory signal expression in local tissues of rats with chronic pelvic pain syndrome based on purinergic 2X7 receptor/NOD-like receptor pyrin domain-containing 3 signal pathway
XU Chang1, LI Na2, WU Xiaoling1, DAI Xingye1, YANG Zhiwen1, SUN Qianhui1, SHI Tianyu1, CHAI Yemao3, PANG Dandan5( ), CHENG Kai4(
), CHENG Kai4( )
)
- 1 Major in Acupuncture and Tuina, school of Acupuncture and Tuina, Beijing University of Chinese Medicine, Beijing 100029, China
2 Experimental science of acupuncture and moxibustion teaching and research section, school of Acupuncture and Tuina, Beijing University of Chinese Medicine, Beijing 100029, China
3 Complex laboratory, school of Acupuncture and Tuina, Beijing University of Chinese Medicine, Beijing 100029, China
4 Meridians and acupoints teaching and research section, school of Acupuncture and Tuina, Beijing University of Chinese Medicine, Beijing 100029, China
5 Academy of Chinese Medical Sciences, Beijing 100700, China
-
Received:2022-04-22Accepted:2022-07-25Online:2022-12-15Published:2022-09-28 -
Contact:PANG Dandan,CHENG Kai -
About author:PANG Dandan, Academy of Chinese Medical Sciences, Beijing 100700, China. Pangdan000@126.com,+86-19801282717
Prof. CHENG Kai, meridians and acupoints teaching and research section, school of Acupuncture and Tuina, Beijing University of Chinese Medicine, Beijing 100029, China. kevin@chengs.cn;
-
Supported by:RNA Sequencing Technology Screening the Mechanism of Acupuncture on Pain in Chronic Pelvic Pain Syndrome Rats(2020-JYB-XJSJJ-018)
Cite this article
XU Chang, LI Na, WU Xiaoling, DAI Xingye, YANG Zhiwen, SUN Qianhui, SHI Tianyu, CHAI Yemao, PANG Dandan, CHENG Kai. Effect of electroacupuncture on inflammatory signal expression in local tissues of rats with chronic pelvic pain syndrome based on purinergic 2X7 receptor/NOD-like receptor pyrin domain-containing 3 signal pathway[J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 965-971.
share this article
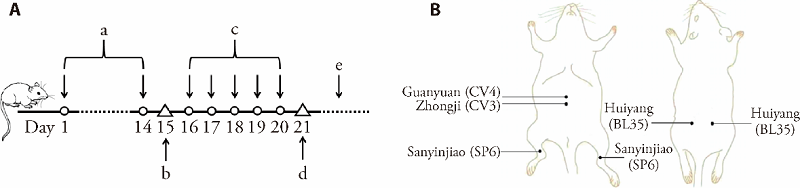
Figure 1 Experimental design and procedure A: thermal stimulation pain threshold was tested prior to and after 5 d consecutive treatment of EA. a: day 1 to day 14: CPPS model establishment; b: behavioral test before EA; c: day 16 to day 20: the 1st treatment course of EA; d: behavioral test after the 1st treatment course of EA; e: repeat the 1st course of EA for a total of 4 courses. B: schematic diagram showing the location of acupoints [Guanyuan (CV4), Zhongji (CV3), Sanyinjiao (SP6), Huiyang (BL35)] of the rat. EA: electroacupuncture; CPPS: chronic pelvic pain syndrome.
| Gene | Primer sequence(5’-3’) |
|---|---|
| P2X7R | Forward primer: AGGTGGCAGTTCAGGGAGGAATC Reverse primer: TGTATTTGGGTTGACAGCGATGGG |
| NLRP3 | Forward primer: GAGCTGGACCTCAGTGACAATGC Reverse primer: AGAACCAATGCGAGATCCTGACAAC |
| caspase-1 | Forward primer: AAACACCCACTCGTACACGTCTTG Reverse primer: AGGTCAACATCAGCTCCGACTCTC |
| IL-18 | Forward primer: GGCCTCTATTTGAAGATATGACTGATT Reverse primer: CCTCTAGGCTGGCTATCTTTATACATAC |
| GAPDH | Forward primer: ACCACAGTCCATGCCATCAC Reverse primer: TCCACCACCCTGTTGCTGTA |
Table 1 Primer sequence of detection genes
| Gene | Primer sequence(5’-3’) |
|---|---|
| P2X7R | Forward primer: AGGTGGCAGTTCAGGGAGGAATC Reverse primer: TGTATTTGGGTTGACAGCGATGGG |
| NLRP3 | Forward primer: GAGCTGGACCTCAGTGACAATGC Reverse primer: AGAACCAATGCGAGATCCTGACAAC |
| caspase-1 | Forward primer: AAACACCCACTCGTACACGTCTTG Reverse primer: AGGTCAACATCAGCTCCGACTCTC |
| IL-18 | Forward primer: GGCCTCTATTTGAAGATATGACTGATT Reverse primer: CCTCTAGGCTGGCTATCTTTATACATAC |
| GAPDH | Forward primer: ACCACAGTCCATGCCATCAC Reverse primer: TCCACCACCCTGTTGCTGTA |
| Group | n | Body weight (g) | Wet weight of prostate (mg) | PI |
|---|---|---|---|---|
| Control | 8 | 405.69±16.77 | 452.55±21.46 | 1.12±0.07 |
| Model | 8 | 361.1±13.81a | 703.11±40.48a | 1.95±0.17a |
| EA | 8 | 399.43±17.95b | 473.56±25.13b | 1.19±0.09b |
Table 2 Comparisons of body weight, wet weight of prostate and PI in rats ($\bar{x} \pm s$)
| Group | n | Body weight (g) | Wet weight of prostate (mg) | PI |
|---|---|---|---|---|
| Control | 8 | 405.69±16.77 | 452.55±21.46 | 1.12±0.07 |
| Model | 8 | 361.1±13.81a | 703.11±40.48a | 1.95±0.17a |
| EA | 8 | 399.43±17.95b | 473.56±25.13b | 1.19±0.09b |
| Group | n | Before acupuncture | 1st course | 2nd course | 3rd course | 4th course |
|---|---|---|---|---|---|---|
| Control | 10 | 11.2±0.8 | 11.0±0.9 | 10.9±0.8 | 10.8±1.1 | 10.6±0.5 |
| Model | 10 | 5.4±0.4a | 5.5±0.7a | 5.5±0.5a | 5.4±0.6a | 5.4±0.6a |
| EA | 10 | 5.6±0.5a | 6.0±0.5a | 6.7±0.6a | 8.0±0.7b | 10.0±0.7b |
Table 3 Thermal stimulation pain threshold of rats in each group at different time periods (s, $\bar{x} \pm s$)
| Group | n | Before acupuncture | 1st course | 2nd course | 3rd course | 4th course |
|---|---|---|---|---|---|---|
| Control | 10 | 11.2±0.8 | 11.0±0.9 | 10.9±0.8 | 10.8±1.1 | 10.6±0.5 |
| Model | 10 | 5.4±0.4a | 5.5±0.7a | 5.5±0.5a | 5.4±0.6a | 5.4±0.6a |
| EA | 10 | 5.6±0.5a | 6.0±0.5a | 6.7±0.6a | 8.0±0.7b | 10.0±0.7b |
| Group | n | TNF-α | IL-1β | PGE2 |
|---|---|---|---|---|
| Control | 8 | 114.4±6.7 | 42.3±9.8 | 144.1±25.7 |
| Model | 8 | 170.7±8.8a | 113.0±16.3a | 251.7±10.5a |
| EA | 8 | 118.7±9.4b | 52.6±13.3b | 153.6±27.8b |
Table 4 Expressions of TNF-α, IL-1β and PGE2 of rats in each group (ng/mL)
| Group | n | TNF-α | IL-1β | PGE2 |
|---|---|---|---|---|
| Control | 8 | 114.4±6.7 | 42.3±9.8 | 144.1±25.7 |
| Model | 8 | 170.7±8.8a | 113.0±16.3a | 251.7±10.5a |
| EA | 8 | 118.7±9.4b | 52.6±13.3b | 153.6±27.8b |
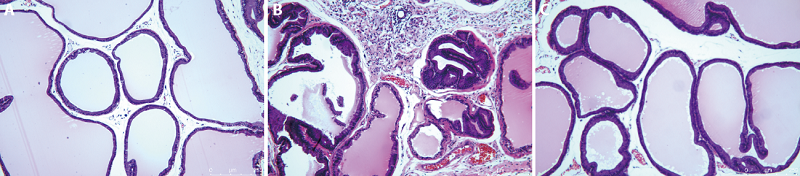
Figure 2 Comparison of prostatic histomorphology in rats A-C: HE staining of rat prostate tissue. A: histomorphology of prostate in the control group (×100); B: histomorphology of prostate in the model group (×100). Box represents atrophy of glandular cavity and decrease of secretion, arrow represents inflammatory cells infiltration; C: histomorphology of prostate in the EA group (×100). HE: hematoxylin-eosin; EA: electroacupuncture.
| Group | n | P2X7R mRNA | NLRP3 mRNA | Caspase-1 mRNA | IL-18 mRNA |
|---|---|---|---|---|---|
| Control | 6 | 1.20±0.68 | 1.12±0.53 | 1.10±0.61 | 1.04±0.35 |
| Model | 6 | 3.83±2.01a | 35.65±21.16b | 6.73±3.44b | 1.75±0.88a |
| EA | 6 | 1.73±1.02c | 1.54±0.39d | 2.05±1.53c | 1.09±0.25c |
Table 5 Comparison of the expression levels of P2X7R, NLRP3, Caspase-1 and IL-18 mRNA of each group ($\bar{x} \pm s$)
| Group | n | P2X7R mRNA | NLRP3 mRNA | Caspase-1 mRNA | IL-18 mRNA |
|---|---|---|---|---|---|
| Control | 6 | 1.20±0.68 | 1.12±0.53 | 1.10±0.61 | 1.04±0.35 |
| Model | 6 | 3.83±2.01a | 35.65±21.16b | 6.73±3.44b | 1.75±0.88a |
| EA | 6 | 1.73±1.02c | 1.54±0.39d | 2.05±1.53c | 1.09±0.25c |
| [1] |
Feng Y, Jiang S, Wang Z, et al. Oyster hydrolysate-zinc complex ameliorates carrageenan-induced rat prostatitis via an anti-inflammatory mechanism and reduced oxidative stress. J Funct Foods 2020; 72: 104066.
DOI URL |
| [2] | Smothers A, Young S, Constantine L. Management of chronic nonbacterial prostatitis and chronic pelvic pain syndrome in the adult male patient with comorbid conditions. J Christ Nurs 2020; 37: E21-6. |
| [3] | Wang W, Muhammad N, Baig M, et al. Experimental rodent models of chronic prostatitis and evaluation criteria. Biomed pharmacother 2018; 108C: 1894-901. |
| [4] |
Carter GT, Duong V, Ho S, et al. Side effects of commonly prescribed analgesic medications. Phys Med Rehabil Clin N Am 2014; 25: 457-70.
DOI PMID |
| [5] |
Natalia RN, Jyothi M, Jordan BK, Junjie M, Brandon KB. Cost-effectiveness of treatment options for neuropathic pain: a systematic review. Pharmaco Economics 2019; 37: 669-88.
DOI URL |
| [6] |
Watanabe S, Iwai T, Tanabe M. Intraplantar injection of sialidase reduces mechanical allodynia during inflammatory pain. J Pharmacol Sci 2017; 133: 49-52.
DOI PMID |
| [7] |
Xu ZF, Hong SH, Wang SJ, et al. Neuroendocrine-immune regulating mechanisms for the anti-inflammatory and analgesic actions of acupuncture. World J Tradit Chin Med 2020; 6: 384-92.
DOI URL |
| [8] |
Zhang K, Zeng X, Chen Y, et al. Therapeutic effects of Qian-Yu decoction and its three extracts on carrageenan-induced chronic prostatitis/chronic pelvic pain syndrome in rats. BMC Complement Altern Med 2017; 17: 75.
DOI URL |
| [9] |
Liu B, Long W, Wang RR. Electroacupuncture alleviates neuropathic pain by modulating Th2 infiltration and inhibiting microglial activation in the spinal cord of rats with spared nerve injury. World J Tradit Chin Med 2020; 6: 448-55.
DOI URL |
| [10] |
Radhakrishnan R, Nallu RS. Development and characterisation of a novel animal model of prostate inflammation-induced chronic pelvic pain. Inflammopharmacology 2009; 17: 23-8.
DOI PMID |
| [11] |
Huang AC, Liu M, Tsai T, et al. Low-frequency electroacupuncture at acupoints Guanyuan (CV4) and Zhongji (CV3) lengthen ejaculatory latency and improves sexual behavior in male rats. Chin J Physiol 2020; 63: 163-70.
DOI URL |
| [12] |
Wu Y, Jiang Y, Shao X, et al. Proteomics analysis of the amygdala in rats with CFA-induced pain aversion with electro-acupuncture stimulation. J Pain Res 2019; 12: 3067-78.
DOI PMID |
| [13] |
Liu Y, Liu LR, Wang XM. Electroacupuncture at points Baliao and Huiyang(BL35) for post-stroke detrusor overactivity. Neural Regen Res 2013; 8: 1663-72.
DOI URL |
| [14] |
Sun WP, Kou DQ, Yu ZJ, et al. A transcriptomic analysis of neuropathic pain in rat dorsal root ganglia following peripheral nerve injury. Neuromol Med 2020; 22: 250-63.
DOI PMID |
| [15] |
Fang JQ, Fang JF, Liang Y, Du JY. Electroacupuncture mediates extracellular signal-regulated kinase 1/2 pathways in the spinal cord of rats with inflammatory pain. BMC Complem Altern M 2014; 14: 285.
DOI URL |
| [16] |
Zhang W, Fang Y, Shi MF, et al. Optimal acupoint and session of acupuncture for patients with chronic prostatitis/chronic pelvic pain syndrome: a Meta-analysis. Transl Androl Urol 2021; 10: 143-53.
DOI PMID |
| [17] |
Zhang YQ, Ji GC, Wu GC, Zhao ZQ. Excitatory amino acid receptor antagonists and electroacupuncture synergetically inhibit carrageenan-induced behavioral hyperalgesia and spinal fos expression in rats. Pain 2002; 99: 525-35.
DOI URL |
| [18] |
Zheng HD, Wang ZQ, Li SS, et al. Effect of acupoints on acupuncture-moxibustion and its therapeutic mechanism. World J Tradit Chin Med 2020; 6:239-48.
DOI URL |
| [19] |
Xu ZF, Hong SH, Wang SJ, et al. Neuroendocrine-immune regulating mechanisms for the anti-inflammatory and analgesic actions of acupuncture. World J Tradit Chin Med 2020; 6:384-92.
DOI URL |
| [20] |
Lee SH, Lee BC. Use of acupuncture as a treatment method for chronic prostatitis/chronic pelvic pain syndromes. Curr Urol Rep 2011; 12: 288-96.
DOI URL |
| [21] | Yang WY. Clinical observation on electroacupuncture at Zhongliao and Huiyang points in the treatment of overactive bladder. Beijing: Beijing TCM University, 2016: 32. |
| [22] |
Shojaei-Zarghani S, Khosroushahi AY, Rafraf M. Oncopreventive effects of theanine and theobromine on dimethylhydrazine-induced colon cancer model. Biomed Pharmacother 2021; 134: 111140.
DOI PMID |
| [23] | Li T, Sun L, Huang ZQ. Research progress on the mechanism of acupuncture in the treatment of chronic prostatitis/pelvic pain syndrome. Jiangsu Zhong Yi Yao 2021; 53: 78-82. |
| [24] | Lin SS, Chen YZ, Zhou XY, Wang J. Research progress on anti-inflammatory mechanism of acupuncture therapy for autoimmune diseases based on inflammatory cytokines. Guangming Zhong Yi 2021; 36: 3372-5. |
| [25] | He QX, Li XD. Research progress on the relationship between cytokines and chronic prostatitis. Zhong Hua Nan Ke Xue Za Zhi 2011; 17: 939-42. |
| [26] | Wan HJ. Expression and significance of NLRP3 inflammasome and its downstream factor IL-1β/IL-18 in rat model of allergic rhinitis. Zhengzhou: Zhengzhou University, 2015: 2. |
| [27] | Zhao YD, Wang XF, Wang JW, Han DY, Zhao ZT. Effects of warming-promotion needling on rats with chronic non-bacteriaI prostatitis on TNF-a and IL-β. Zhong Hua Zhong Yi Yao Xue Kan 2016; 34: 129-31. |
| [28] |
Zhang J, Li X, Gao Y, et al. Effects of puerarin on the inflammatory role of burn-related procedural pain mediated by P2X7 receptors. Burns 2013; 39: 610-8.
DOI PMID |
| [29] |
Ismael S, Ahmed HA, Adris T, et al. The NLRP 3 inflammasome: a potential therapeutic target for traumatic brain injury. Neural Regen Res 2021; 16: 49-57.
DOI URL |
| [30] | Zhang Y. Role and significance of P2X7R - mediated IL-1β in chronic abacterial prostatitis model. Anhui: Anhui Medical University, 2019: 28-9. |
| [31] |
Garré JM, Silva HM, Lafaille JJ, Yang G. P2X7 receptor inhibition ameliorates dendritic spine pathology and social behavioral deficits in Rett syndrome mice. Nat Commun 2020; 11: 1784.
DOI PMID |
| [32] |
Friedle SA, Brautigam VM, Nikodemova M, Wright ML, Watters JJ. The P2X7-Egr pathway regulates nucleotide-dependent inflammatory gene expression in microglia. Glia 2011; 59: 1-13.
DOI PMID |
| [33] |
Wang W, Muhammad N, Baig MMFA, Abbas TM, Zhou X. Experimental rodent models of chronic prostatitis and evaluation criteria. Biomed Pharmacother 2018; 108: 1894-901.
DOI PMID |
| [34] |
Wagenlehner FME, Van Till JWO, Magri V, et al. National institutes of health chronic prostatitis symptom index (NIH-CPSI) symptom evaluation in multinational cohorts of patients with chronic prostatitis/chronic pelvic pain syndrome. Eur Urol 2013; 63: 953-9.
DOI PMID |
| [35] |
Yuan Z, Liu XL, Deng WT, et al. Correlation study of chronic nonbacterial prostatitis with the levels of COX-2 and PGE2 in prostatic secretion. Int Urol Nephrol 2014; 46: 1871-5.
DOI PMID |
| [36] | Fulgenzi A, Ticozzi P, Gabel CA, et al. Periodate oxidized ATP (oATP) reduces hyperalgesia in mice: involvement of P2X7 receptors and implications for therapy. Int J Immunopath Ph 2008; 21: 61-71. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

