Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (1): 51-61.DOI: 10.19852/j.cnki.jtcm.2026.01.005
• Original Articles • Previous Articles Next Articles
Ex vivo to in vivo extrapolation of primary absorbed compounds as multifunctional proxies of Zhiqiao (Fructus Aurantii Submaturus)-Houpo (Cortex Magnoliae Officinalis) herb pair
ZHOU Runze, XU Chendong, QIAN Haotian, HUANG Xi( )
)
- Institute of Comorbid Depression, Nanjing University of Chinese Medicine, Nanjing 210023, China
-
Received:2025-01-15Accepted:2025-05-09Online:2026-02-15Published:2026-01-28 -
Contact:Prof. HUANG Xi, Institute of Comorbid Depression, Nanjing University of Chinese Medicine, Nanjing 210023, China.290606@njucm.edu.cn ;Telephone: +86-13951761464 -
Supported by:Jiangsu Province’s Colleges and Universities(Integration of Chinese);National Natural Science Foundation of China [Methanism on Molecular and Hippocampus-Thalamus Neurocircuitry of Rapid Prokinetic and Antidepressant by Shugan Following Acute Stress(81973589);The Relationship Between Stress, Ghrelin Signaling, Liver-Soothing and Antidepressant Prokinetic Mechanisms(81573797)
Cite this article
ZHOU Runze, XU Chendong, QIAN Haotian, HUANG Xi. Ex vivo to in vivo extrapolation of primary absorbed compounds as multifunctional proxies of Zhiqiao (Fructus Aurantii Submaturus)-Houpo (Cortex Magnoliae Officinalis) herb pair[J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 51-61.
share this article
| Item | Control (n = 3) | Model (n = 3) | FPSDS (n = 3) | MH (n = 3) | Narirutin (n = 3) | Naringin (n = 3) | Hesperidin (n = 3) | Neohesperidin (n = 3) | Nobiletin (n = 3) | Honokiol (n = 3) | Magnolol (n = 3) | 8ACs (n = 3) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NO (μmol/L) Mean±SD | 507.67 ±20.13 | 21461.67 ±795.36 | 18577.33 ±69.72 | 14497.33 ±247.04 | 16201.67 ±292.17 | 16127.33 ±183.92 | 17053.67 ±763.52 | 12958.67 ±126.00 | 13246.67 ±301.02 | 17663.33 ±703.40 | 16503 ±997.77 | 16698.67 ±663.99 |
| Contribution (%) | 241.45 | 182.36 | 184.94 | 152.83 | 294.8 | 284.81 | 131.69 | 171.92 | 165.13 | |||
| eNOS (μmol/L) Mean±SD | 4.66 ±0.17 | 1.53 ±0.06 | 3.43 ±0.08 | 1.85 ±0.12 | 4.71 ±0.07 | 2.29 ±0.07 | 2.41 ±0.03 | 0.69 ±0.04 | 5.04 ±0.05 | 2.65 ±0.11 | 2.77 ±0.07 | 2.95 ±0.04 |
| Contribution (%) | 53.94 | 137.32 | 66.76 | 70.26 | 20.12 | 146.94 | 77.26 | 80.76 | 86.01 | |||
| ROS Mean±SD | 37.03 ±2.33 | 19.02 ±0.84 | 17.36 ±0.48 | 23.03 ±0.73 | 31.76 ±1.65 | 17.72 ±3.75 | 18.37 ±0.24 | 34.68 ±2.34 | 34.76 ±2.20 | 17.33 ±0.31 | 28.03 ±0.67 | 37.96 ±0.87 |
| Contribution (%) | 132.66 | 182.95 | 102.07 | 105.82 | 199.77 | 200.23 | 99.83 | 161.46 | 218.66 |
Table 1 Ranking of the contribution of 8 ACs alone or mixed to the efficacy of FPSDS in H2O2 treated endothelial cells
| Item | Control (n = 3) | Model (n = 3) | FPSDS (n = 3) | MH (n = 3) | Narirutin (n = 3) | Naringin (n = 3) | Hesperidin (n = 3) | Neohesperidin (n = 3) | Nobiletin (n = 3) | Honokiol (n = 3) | Magnolol (n = 3) | 8ACs (n = 3) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NO (μmol/L) Mean±SD | 507.67 ±20.13 | 21461.67 ±795.36 | 18577.33 ±69.72 | 14497.33 ±247.04 | 16201.67 ±292.17 | 16127.33 ±183.92 | 17053.67 ±763.52 | 12958.67 ±126.00 | 13246.67 ±301.02 | 17663.33 ±703.40 | 16503 ±997.77 | 16698.67 ±663.99 |
| Contribution (%) | 241.45 | 182.36 | 184.94 | 152.83 | 294.8 | 284.81 | 131.69 | 171.92 | 165.13 | |||
| eNOS (μmol/L) Mean±SD | 4.66 ±0.17 | 1.53 ±0.06 | 3.43 ±0.08 | 1.85 ±0.12 | 4.71 ±0.07 | 2.29 ±0.07 | 2.41 ±0.03 | 0.69 ±0.04 | 5.04 ±0.05 | 2.65 ±0.11 | 2.77 ±0.07 | 2.95 ±0.04 |
| Contribution (%) | 53.94 | 137.32 | 66.76 | 70.26 | 20.12 | 146.94 | 77.26 | 80.76 | 86.01 | |||
| ROS Mean±SD | 37.03 ±2.33 | 19.02 ±0.84 | 17.36 ±0.48 | 23.03 ±0.73 | 31.76 ±1.65 | 17.72 ±3.75 | 18.37 ±0.24 | 34.68 ±2.34 | 34.76 ±2.20 | 17.33 ±0.31 | 28.03 ±0.67 | 37.96 ±0.87 |
| Contribution (%) | 132.66 | 182.95 | 102.07 | 105.82 | 199.77 | 200.23 | 99.83 | 161.46 | 218.66 |

Figure 1 Effect of FM-derived ACs on anti-depression and prokinetic after acute stress A: effects of FM, 2ACs, and 3ACs on AFS rats in the FST test; B: effects of FM, 2ACs, and 3ACs AFS rats in the OFT test; C: effects of FM, 2ACs, and 3ACs on AFS rats in the GE test; D: effects of FM, 2ACs, and 3ACs on AFS rats in the IT test. Control group: no AFS modeling treatment; Model group: rats were forced to swim for 15 min; FM group: FM (20 g/kg) was administered by gavage 24 h after model establishment; 2ACs group: 2ACs was administered by gavage 24 h after model establishment; 3ACs group: 3ACs was administered by gavage 24 h after model establishment. FM: Zhiqiao (Fructus Aurantii Submaturus) and Houpo (Cortex Magnoliae Officinalis); AFS: acuted forced swimming; 2ACs: nobiletin (2.15 mg/kg) + magnolol (1.70 mg/kg); 3ACs group: nobiletin (2.15 mg/kg) + magnolol (1.70 mg/kg) + MH (11.86 mg/kg). FST: forced swimming test; OFT: open field test; GE: gastric emptying; IT: intestinal transit. One-way analysis of variance was used to compare more than two groups, followed by the least significant difference test to detect differences between groups. Data are shown as mean ± standard deviation (n = 6). Comparison with the Control group, aP < 0.05; comparison with the Model group, bP < 0.05.

Figure 2 Effects of FM-derived ACs on biochemical molecules in plasma after acute stress A: 5-HT; B: DA; C: Ghrelin; D: CRH; E: ACTH; F: MDA; G: ROS; H: IL-1β; I: TNF-α; J: IL-6. Control group: no AFS modeling treatment; Model group: rats were forced to swim for 15 min; FM group: FM (20 g/kg) was administered by gavage 24 h after model establishment; 2ACs group: 2ACs was administered by gavage 24 h after model establishment; 3ACs group: 3ACs was administered by gavage 24 h after model establishment. FM:Zhiqiao (Fructus Aurantii Submaturus) and Houpo (Cortex Magnoliae Officinalis); 2ACs: nobiletin (2.15 mg/kg) + magnolol (1.70 mg/kg); 3ACs group: nobiletin (2.15 mg/kg) + magnolol (1.70 mg/kg) + MH (11.86 mg/kg). 5-HT: 5-hydroxytryptamine; DA: dopamine; CRH: corticotropin-releasing hormone; ACTH: adrenocorticotropic hormone; MDA: malondialdehyde; ROS: reactive oxygen species; IL-1β: interleukin-1β; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; MH: meranzin hydrate. One-way analysis of variance was used to compare more than two groups, followed by the least significant difference test to detect differences between groups. Data are shown as mean ± standard deviation (n = 8). Comparison with the control group, aP < 0.05; comparison with the model group, bP < 0.05.
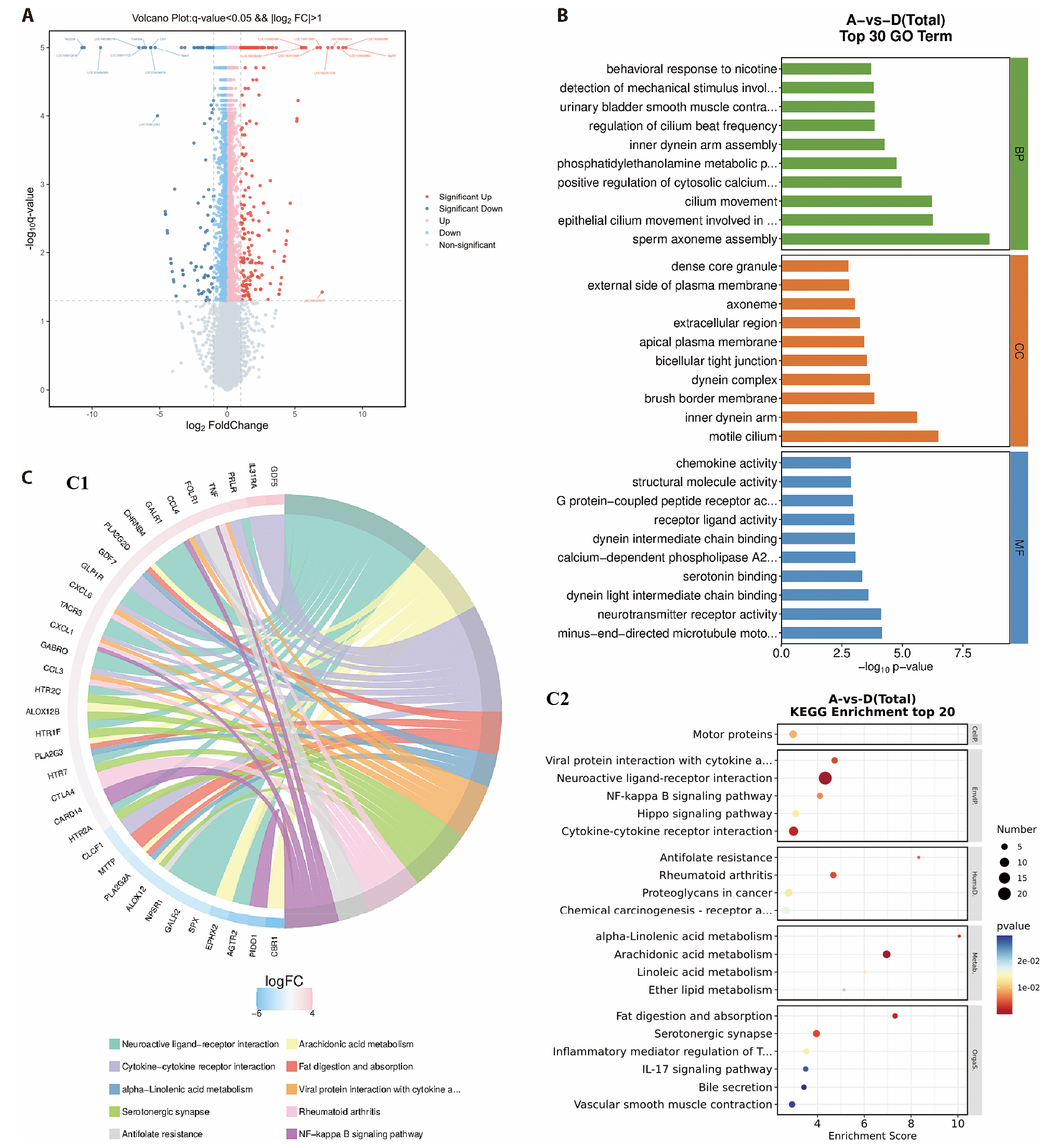
Figure 3 Therapeutic effect of FRA on DG in acute stress rats at the transcriptome level A: volcano diagram for the DEGs detected in DG after oral administration of FRA in the AFS rats (n?=?3 biologically independent animal samples in the model group; n?=?3 biologically independent animal samples in oral administration of FRA group; B: GO bar chart for DEGs enrichment after FRA treatment; C: KEGG enrichment analysis of differential genes; C1: KEGG chord enrichment analysis of differential genes; C2: KEGG bubble chart enrichment analysis of DEGs; GO enrichment and corresponding KEGG pathway enrichment analysis showing DEGs in the FRA group compared with the model group. Histogram: top 30 significantly enriched biological processes, cellular components, and molecular function. The horizontal axis represents ?log10 P of the pathway, and the vertical axis represents the GO terms. Chord diagram: top ten significantly enriched KEGG pathways, the right semicircle represents the names of four KEGG pathways the left semicircle represents DEGs in KEGG pathways. The color map represents the fold-change of genes (log2 FC), and the colored bands connect a gene to a specific GO term. Bubble diagram: the top 20 significantly enriched KEGG terms. The horizontal axis represents the enrichment score, the vertical axis represents the description, and bubble sizes represent enriched gene counts. CellP: cellular processes; HumaD: human diseases; Metab: metabolism; OrgaS: organismal systems; EnvIP: environmental information processing; DEG: different expression genes; FRA: Zhiqiao (Fructus Aurantii Submaturus); GO: gene ontology; KEGG: Kyoto encyclopedia of genes and genomes; FC: fold change.

Figure 4 GSEA of KEGG enrichment pathway of DEGs in FRA administration and model groups. A: neuroactive ligand-receptor interaction; B: IL-17 signaling pathway; C: cGMP-PKG signaling pathway; D: glutamatergic synapse. GESA: Gene Set Enrichment Analysis. KEGG: Kyoto encyclopedia of genes and genomes; DEGs: different expression genes; FRA: Zhiqiao (Fructus Aurantii Submaturus); IL-17: interleukin-17; cGMP-PKG: cyclic guanosine monophosphate-protein kinase G.
| 1. |
Otte C, Gold SM, Penninx BW, et al. Major depressive disorder. Nat Rev Dis Primers 2016; 2: 16065.
DOI PMID |
| 2. |
Malhi GS, Mann JJ. Depression. Lancet 2018; 392: 2299.
DOI PMID |
| 3. |
Hack LM, Tozzi L, Zenteno S, et al. A cognitive biotype of depression and symptoms, behavior measures, neural circuits, and differential treatment outcomes: a prespecified secondary analysis of a randomized clinical trial. JAMA Netw Open 2023; 6: e2318411.
DOI URL |
| 4. |
Drossman DA, Hasler WL. Rome IV-functional GI disorders: disorders of gut-brain interaction. Gastroenterology 2016; 150: 1257.
DOI PMID |
| 5. | Pinto-Sanchez MI, Hall GB, Ghajar K, et al. probiotic bifidobacterium longum NCC3001 reduces depression scores and alters brain activity: a pilot study in patients with irritable bowel syndrome. Gastroenterology 2017; 153: 448. |
| 6. |
Yang C, Lin X, Wang X, et al. The schizophrenia and gut microbiota: a bibliometric and visual analysis. Front Psychiatry 2022; 13: 1022472.
DOI URL |
| 7. |
Berwian IM, Walter H, Seifritz E, Huys QJ. Predicting relapse after antidepressant withdrawal-a systematic review. Psychol Med 2017; 47: 426.
DOI PMID |
| 8. | Pan SY, Zhou SF, Gao SH, et al. New perspectives on how to discover drugs from herbal medicines: CAM's outstanding contribution to modern therapeutics. Evid Based Complement Alternat Med 2013; 2013: 627375. |
| 9. |
Ekor M. The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol 2014; 4: 177.
DOI PMID |
| 10. |
Bent S. Herbal medicine in the United States: review of efficacy, safety, and regulation: grand rounds at University of California, San Francisco Medical Center. J Gen Intern Med 2008; 23: 854.
DOI PMID |
| 11. |
Shi S, Yan H, Chen Y, et al. Pharmacokinetic study of precisely representative antidepressant, prokinetic, anti-inflammatory and anti-oxidative compounds from Fructus aurantii and Magnolia Bark. Chem Biol Interact 2020; 315: 108851.
DOI URL |
| 12. | Sun SM (Tang dynasty). Qian Jin Yi Fang. Shanxi: Shanxi Science and Technology Publishing House, 2010: 806. |
| 13. |
Huang Y, Xu M, Li J, et al. Ex vivo to in vivo extrapolation of syringic acid and ferulic acid as grape juice proxies for endothelium-dependent vasodilation: redefining vasoprotective resveratrol of the French paradox. Food Chem 2021; 363: 130323.
DOI URL |
| 14. | Anacker C, Hen R. Adult hippocampal neurogenesis and cognitive flexibility-linking memory and mood. Nat Rev Neurosci 2017; 18: 335. |
| 15. |
He H, He H, Mo L, et al. Priming of microglia with dysfunctional gut microbiota impairs hippocampal neurogenesis and fosters stress vulnerability of mice. Brain Behav Immun 2024; 115: 280.
DOI URL |
| 16. |
Porsolt RD, Le Pichon M, Jalfre M. Depression: a new animal model sensitive to antidepressant treatments. Nature 1977; 266: 730.
DOI |
| 17. |
Poole DP, Pelayo JC, Cattaruzza F, et al. Transient receptor potential ankyrin 1 is expressed by inhibitory motoneurons of the mouse intestine. Gastroenterology 2011; 141: 565.
DOI PMID |
| 18. |
Wu Z, Chen L, Guo W, et al. Oral mitochondrial transplantation using nanomotors to treat ischaemic heart disease. Nat Nanotechnol 2024; 19: 1375.
DOI |
| 19. |
Magalhães R, Novais A, Barrière DA, et al. A resting-state functional MR imaging and spectroscopy study of the dorsal hippocampus in the chronic unpredictable stress rat model. J Neurosci 2019; 39: 3640.
DOI PMID |
| 20. |
Hisey E, Purkey A, Gao Y, et al. A ventromedial prefrontal-to-lateral entorhinal cortex pathway modulates the gain of behavioral responding during threat. Biol Psychiatry 2023; 94: 239.
DOI URL |
| 21. |
Slattery DA, Cryan JF. Using the rat forced swim test to assess antidepressant-like activity in rodents. Nat Protoc 2012; 7: 1009.
DOI PMID |
| 22. |
Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci 2012; 13: 701.
DOI PMID |
| 23. |
Krishnan V, Nestler EJ. The molecular neurobiology of depression. Nature 2008; 455: 894.
DOI |
| 24. |
Papadopoulos AS, Cleare AJ. Hypothalamic-pituitary-adrenal axis dysfunction in chronic fatigue syndrome. Nat Rev Endocrinol 2011; 8: 22.
DOI PMID |
| 25. |
Gold PW. The organization of the stress system and its dysregulation in depressive illness. Mol Psychiatry 2015; 20: 32.
DOI |
| 26. |
Yang J, Xia X, Du M, et al. Highly effective nobiletin-MPN in yeast microcapsules for targeted modulation of oxidative stress, NLRP 3 inflammasome activation, and immune responses in ulcerative colitis. J Agric Food Chem 2024; 72: 13054.
DOI URL |
| 27. |
Tao L, Zhang Q, Liu L, et al. Magnolol preserves the integrity of the intestinal epithelial barrier and mitigates intestinal injury through activation of PPAR gamma in COPD rat. J Ethnopharmacol 2025; 338: 119068.
DOI URL |
| 28. |
Zhang X, Han L, Liu J, et al. Pharmacokinetic study of 7 compounds following oral administration of Fructus Aurantii to depressive rats. Front Pharmacol 2018; 9: 131.
DOI URL |
| 29. |
Zhang YJ, Huang W, Huang X, et al. Fructus Aurantii induced antidepressant effect via its monoaminergic mechanism and prokinetic action in rat. Phytomedicine 2012; 19: 1101.
DOI URL |
| 30. |
Villemure C, Bushnell MC. Mood influences supraspinal pain processing separately from attention. J Neurosci 2009; 29: 705.
DOI PMID |
| 31. |
Ziegler K, Folkard R, Gonzalez AJ, et al. Primary somatosensory cortex bidirectionally modulates sensory gain and nociceptive behavior in a layer-specific manner. Nat Commun 2023; 14: 2999.
DOI PMID |
| 32. |
Nagai M, Kishi K, Kato S. Insular cortex and neuropsychiatric disorders: a review of recent literature. Eur Psychiatry 2007; 22: 387.
DOI URL |
| 33. |
Alexopoulos GS. Depression in the elderly. Lancet 2005; 365: 1961.
DOI PMID |
| 34. |
Teixeira CM, Rosen ZB, Suri D, et al. Hippocampal 5-HT input regulates memory formation and schaffer collateral excitation. Neuron 2018; 98: 992.
DOI PMID |
| 35. |
HU YY, LIU XG, ZHAO P, et al. Integration of serum pharmacochemistry with network pharmacology to reveal the potential mechanism of Yangqing Chenfei formula (养清尘肺方) for the treatment of silicosis. J Tradit Chin Med 2024; 44: 784-93.
DOI |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

