Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (3): 559-567.DOI: 10.19852/j.cnki.jtcm.20230208.002
Previous Articles Next Articles
Effects of Zhenggan Huayu decoction (正肝化瘀方) combined with entecavir on gut microbiota in patients with chronic hepatitis B fibrosis
CHEN Wenlin1( ), LIANG Fang1, ZHANG Yuncheng1, LV Jinzhen1, YANG Daguo2
), LIANG Fang1, ZHANG Yuncheng1, LV Jinzhen1, YANG Daguo2
- 1 Department of Liver Diseases, Shenzhen Hospital, Beijing University of Chinese Medicine, Shenzhen 518172, China
2 Department of Liver Diseases, Shenzhen Third People's Hospital, Guangdong 518112, China
-
Accepted:2022-05-05Online:2023-06-15Published:2023-02-08 -
Contact:Dr. CHEN Wenlin, Department of Liver Diseases, Shenzhen Hospital, Beijing University of Chinese Medicine, Shenzhen, Guangdong 518172, China. chenguiren69@163.com. Telephone: +86-15013727635 -
Supported by:Shenzhen Science and Technology Plan Project: Study on the Mechanism of Zhenggan Huayu Decoction Regulating Intestinal Flora in Liver Fibrosis based on LPS-TRL4 Signal Pathway(JCYJ20180302150216419)
Cite this article
CHEN Wenlin, LIANG Fang, ZHANG Yuncheng, LV Jinzhen, YANG Daguo. Effects of Zhenggan Huayu decoction (正肝化瘀方) combined with entecavir on gut microbiota in patients with chronic hepatitis B fibrosis[J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 559-567.
share this article
| Before treatment (0 week) | After treatment (24 weeks) | ||||||
|---|---|---|---|---|---|---|---|
| Item | ETV (n = 21) | TCM (n = 38) | P value | ETV (n = 21) | TCM (n = 38) | P value | |
| Sex (F/M) | 3/17 | 3/35 | 0.405 | 0.176 | 0.086 | 0.405 | |
| Age | 40.45±7.35 | 41.70±9.29 | 0.456 | 40.45±7.35 | 41.70±9.29 | 0.456 | |
| ALT | 86.31±105.55 | 241.84±808.09 | 0.697 | 25.32±9.48 | 26.93±12.22 | 0.563 | |
| AST | 80.46±108.09 | 68.51±52.44 | 0.715 | 27.24±4.97 | 31.34±15.09 | 0.953 | |
| GGT | 63.01±48.67 | 92.53±138.73 | 0.841 | 36.38±24.81 | 40.73±31.62 | 0.759 | |
| ALP | 98.60±36.55 | 106.80±58.69 | 0.804 | 80.57±32.67 | 89.26±51.46 | 0.991 | |
| TBA | 13.59±13.09 | 24.39±38.46 | 0.532 | 11.33±17.36 | 13.60±28.28 | 0.654 | |
| TP | 73.59±6.55 | 72.82±5.75 | 0.637 | 71.79±8.05 | 74.48±5.25 | 0.328 | |
| TBIL | 22.89±13.56 | 26.70±23.48 | 0.953 | 19.06±6.41 | 18.63±6.64 | 0.715 | |
| DBIL | 6.01±5.98 | 8.59±12.24 | 0.741 | 3.45±1.14 | 3.97±2.80 | 0.878 | |
| IBIL | 17.44±7.68 | 18.10±11.97 | 0.663 | 15.61±5.44 | 14.23±5.08 | 0.268 | |
| CHE | 7485.12±2208.47 | 7555.21±2626.55 | 0.805 | 7815.45±1555.8 | 8081.00±2335.77 | 0.596 | |
| TG | 1.28±0.71 | 1.37±0.69 | 0.340 | 1.16±0.41 | 1.56±1.21 | 0.396 | |
| TC | 4.71±1.11 | 4.47±0.89 | 0.509 | 4.88±1.13 | 4.68±0.88 | 0.841 | |
| HDL-C | 1.39±0.34 | 1.22±0.37 | 0.283 | 1.43±0.27 | 1.28±0.22 | 0.087 | |
| LDL-C | 3.13±0.84 | 3.02±0.71 | 0.588 | 3.33±0.83 | 3.27±0.80 | 1.000 | |
| UREA | 4.87±1.15 | 4.69±0.91 | 0.540 | 29.08±107.35 | 4.86±0.78 | 0.334 | |
| CR | 72.57±13.14 | 70.72±8.75 | 0.741 | 75.46±9.86 | 73.99±11.98 | 0.841 | |
| UA | 365.34±102.41 | 342.92±79.17 | 0.760 | 369.56±110.51 | 361.81±65.52 | 0.769 | |
| HA | 182.92±237.93 | 202.16±272.55 | 0.800 | 87.81±88.53 | 60.15±41.98 | 0.266 | |
| PIIINP | 12.13±10.30 | 11.51±9.62 | 0.888 | 8.92±5.80 | 6.63±2.14 | 0.490 | |
| IV-C | 100.76±88.14 | 104.07±143.10 | 0.422 | 56.73±32.23 | 45.04±27.97 | 0.254 | |
| LN | 2604.00±10286.62 | 119.10±67.09 | 0.746 | 113.61±75.35 | 93.10±38.79 | 0.526 | |
| RL | 122.30±10.74 | 123.58±12.82 | 0.962 | 121.55±9.55 | 122.88±8.65 | 0.509 | |
| DPV | 10.85±1.24 | 10.50±0.76 | 0.617 | 10.60±1.28 | 10.50±0.65 | 0.440 | |
| TS | 41.15±5.72 | 37.38±6.42 | 0.041 | 36.55±5.01 | 35.50±5.33 | 0.594 | |
Table 1 Characteristics and clinical indices of participants
| Before treatment (0 week) | After treatment (24 weeks) | ||||||
|---|---|---|---|---|---|---|---|
| Item | ETV (n = 21) | TCM (n = 38) | P value | ETV (n = 21) | TCM (n = 38) | P value | |
| Sex (F/M) | 3/17 | 3/35 | 0.405 | 0.176 | 0.086 | 0.405 | |
| Age | 40.45±7.35 | 41.70±9.29 | 0.456 | 40.45±7.35 | 41.70±9.29 | 0.456 | |
| ALT | 86.31±105.55 | 241.84±808.09 | 0.697 | 25.32±9.48 | 26.93±12.22 | 0.563 | |
| AST | 80.46±108.09 | 68.51±52.44 | 0.715 | 27.24±4.97 | 31.34±15.09 | 0.953 | |
| GGT | 63.01±48.67 | 92.53±138.73 | 0.841 | 36.38±24.81 | 40.73±31.62 | 0.759 | |
| ALP | 98.60±36.55 | 106.80±58.69 | 0.804 | 80.57±32.67 | 89.26±51.46 | 0.991 | |
| TBA | 13.59±13.09 | 24.39±38.46 | 0.532 | 11.33±17.36 | 13.60±28.28 | 0.654 | |
| TP | 73.59±6.55 | 72.82±5.75 | 0.637 | 71.79±8.05 | 74.48±5.25 | 0.328 | |
| TBIL | 22.89±13.56 | 26.70±23.48 | 0.953 | 19.06±6.41 | 18.63±6.64 | 0.715 | |
| DBIL | 6.01±5.98 | 8.59±12.24 | 0.741 | 3.45±1.14 | 3.97±2.80 | 0.878 | |
| IBIL | 17.44±7.68 | 18.10±11.97 | 0.663 | 15.61±5.44 | 14.23±5.08 | 0.268 | |
| CHE | 7485.12±2208.47 | 7555.21±2626.55 | 0.805 | 7815.45±1555.8 | 8081.00±2335.77 | 0.596 | |
| TG | 1.28±0.71 | 1.37±0.69 | 0.340 | 1.16±0.41 | 1.56±1.21 | 0.396 | |
| TC | 4.71±1.11 | 4.47±0.89 | 0.509 | 4.88±1.13 | 4.68±0.88 | 0.841 | |
| HDL-C | 1.39±0.34 | 1.22±0.37 | 0.283 | 1.43±0.27 | 1.28±0.22 | 0.087 | |
| LDL-C | 3.13±0.84 | 3.02±0.71 | 0.588 | 3.33±0.83 | 3.27±0.80 | 1.000 | |
| UREA | 4.87±1.15 | 4.69±0.91 | 0.540 | 29.08±107.35 | 4.86±0.78 | 0.334 | |
| CR | 72.57±13.14 | 70.72±8.75 | 0.741 | 75.46±9.86 | 73.99±11.98 | 0.841 | |
| UA | 365.34±102.41 | 342.92±79.17 | 0.760 | 369.56±110.51 | 361.81±65.52 | 0.769 | |
| HA | 182.92±237.93 | 202.16±272.55 | 0.800 | 87.81±88.53 | 60.15±41.98 | 0.266 | |
| PIIINP | 12.13±10.30 | 11.51±9.62 | 0.888 | 8.92±5.80 | 6.63±2.14 | 0.490 | |
| IV-C | 100.76±88.14 | 104.07±143.10 | 0.422 | 56.73±32.23 | 45.04±27.97 | 0.254 | |
| LN | 2604.00±10286.62 | 119.10±67.09 | 0.746 | 113.61±75.35 | 93.10±38.79 | 0.526 | |
| RL | 122.30±10.74 | 123.58±12.82 | 0.962 | 121.55±9.55 | 122.88±8.65 | 0.509 | |
| DPV | 10.85±1.24 | 10.50±0.76 | 0.617 | 10.60±1.28 | 10.50±0.65 | 0.440 | |
| TS | 41.15±5.72 | 37.38±6.42 | 0.041 | 36.55±5.01 | 35.50±5.33 | 0.594 | |
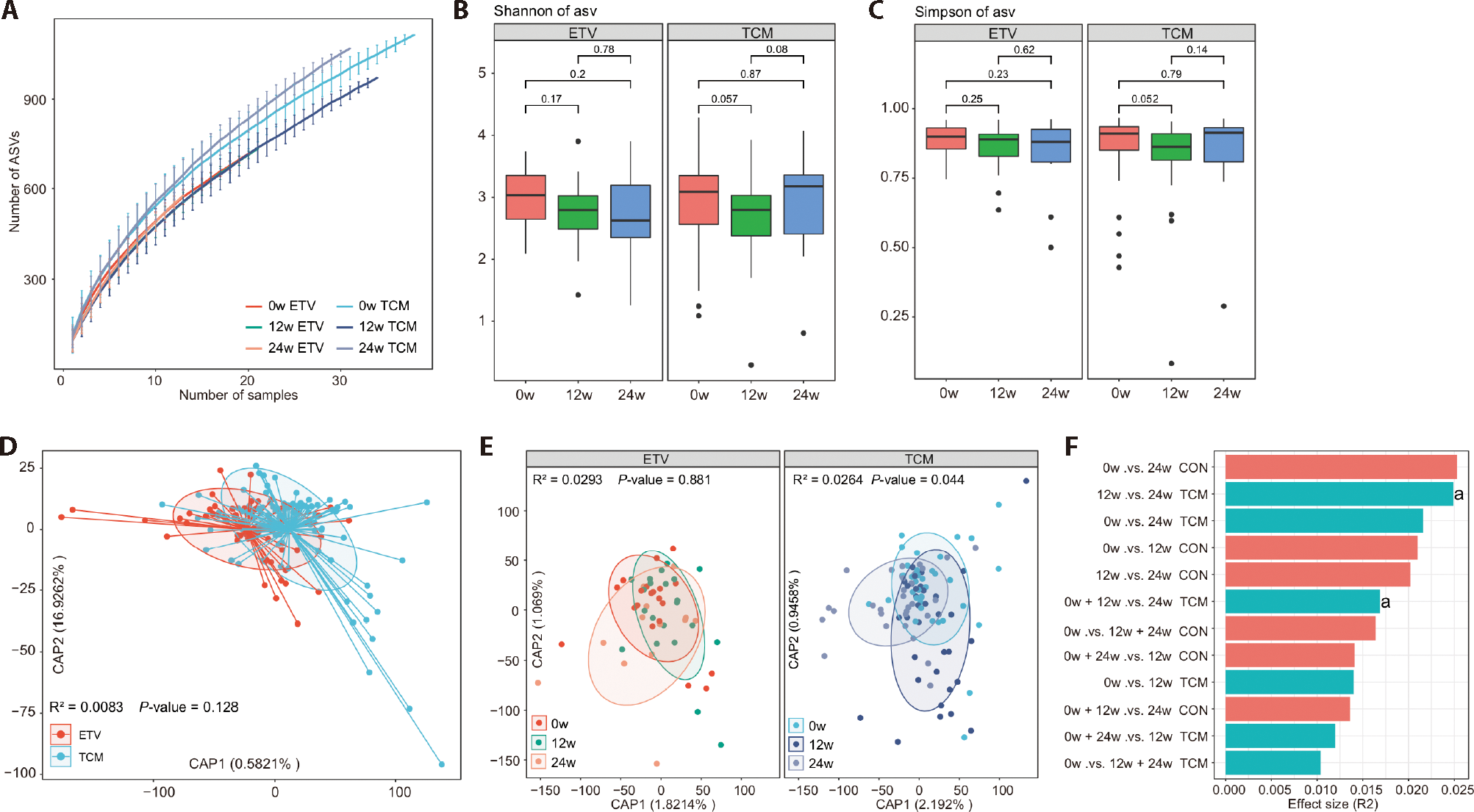
Figure 1 Effects of ETV and TCM on the gut microbiota of patients A: rarefaction curve analysis of ASVs in each group of samples. The number of identified ASVs in each group was calculated based on a randomly selected specific number of samples with 100 replacements. Quartiles numbers are plotted by a line segment; B, C: differences in gut microbiota diversity between ETV and TCM groups at different treatment periods. The significance level in the Wilcoxon signed-rank test was marked as a numerical P-value; D: the dbRDA analysis based on microbiota composition showed separations between groups. The R2 and P values generated by PERMANOVA analysis are shown in the lower-left quadrant of the plot; E: dbRDA analysis showed separations at different treatment periods within ETV and TCM groups, and R2 as well as P values generated by PERMANOVA analysis are shown at the top of the image; F: PERMANOVA analysis showed differences between treatment periods. Patients in the TCM group were treated with ZGHY 1 potion/d (oral administration in two doses) and ETV 0.5 mg/d, and the ETV group were treated with ETV 0.5 mg/d. All patients were treated for a total of 24 weeks, fecal samples were collected and analyzed in the 12 and 24 weeks, respectively. ETV: entecavir; TCM: Traditional Chinese Medicine (FZHY+ETV group); CON: control (ETV group); ZGHY: Zhenggan Huayu decoction; ASVs: amplicon sequence variants; dbRDA: distance-based redundancy analysis; PERMANOVA: permutational multivariate analysis of variance. Significant differences in TCM group: 12 w vs 24 w, aP<0.05; 0+12 w vs 24 w, aP<0.05. n = 38.

Figure 2 Microbiota composition in patients’ gut A: the composition of phyla levels in different groups. Only the top 10 members in relative abundance are shown; B: species differences at phylum levels between groups; C: the composition of family levels in different groups. Only the top 10 members in relative abundance are shown; D: species differences at family levels between groups. Patients in the TCM group were treated with ZGHY 1 potion/d (oral administration in two doses) and ETV 0.5 mg/d, and the ETV group were treated with ETV 0.5 mg/d. All patients were treated for a total of 24 weeks, fecal samples were collected and analyzed in the 12 and 24 weeks, respectively. ETV: entecavir; ZGHY: Zhenggan Huayu decoction; TCM: Traditional Chinese Medicine (FZHY + ETV group); CON: control (ETV group).

Figure 3 Heat maps show correlations between feature genera and clinical indicators Spearman correlation test showed that: * q<0.05; + q<0.01. A correlation coefficient>0.6 was considered as a positive correlation, and<-0.6 as a negative correlation. ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; CHE: cholinesterase; CR: creatinine; DBIL: direct bilirubin; GGT: γ-glutamyl transferase; HA: hyaluronic acid; HDL-C: high-density lipoprotein cholesterol; IBIL: indirect bilirubin; IV-C: Type IV collagen; LDL-C: low-density lipoprotein cholesterol; LN: laminin; PIIINP: type III procollagen peptide; TBA: total bile acid; TBIL: total bilirubin; TC: total cholesterol; TG: triglyceride; TP: total protein; UA: uric acid; UREA: urea; RL: right lobe of liver oblique diameter; DPV: portal vein diameter; TS: spleen thickness.
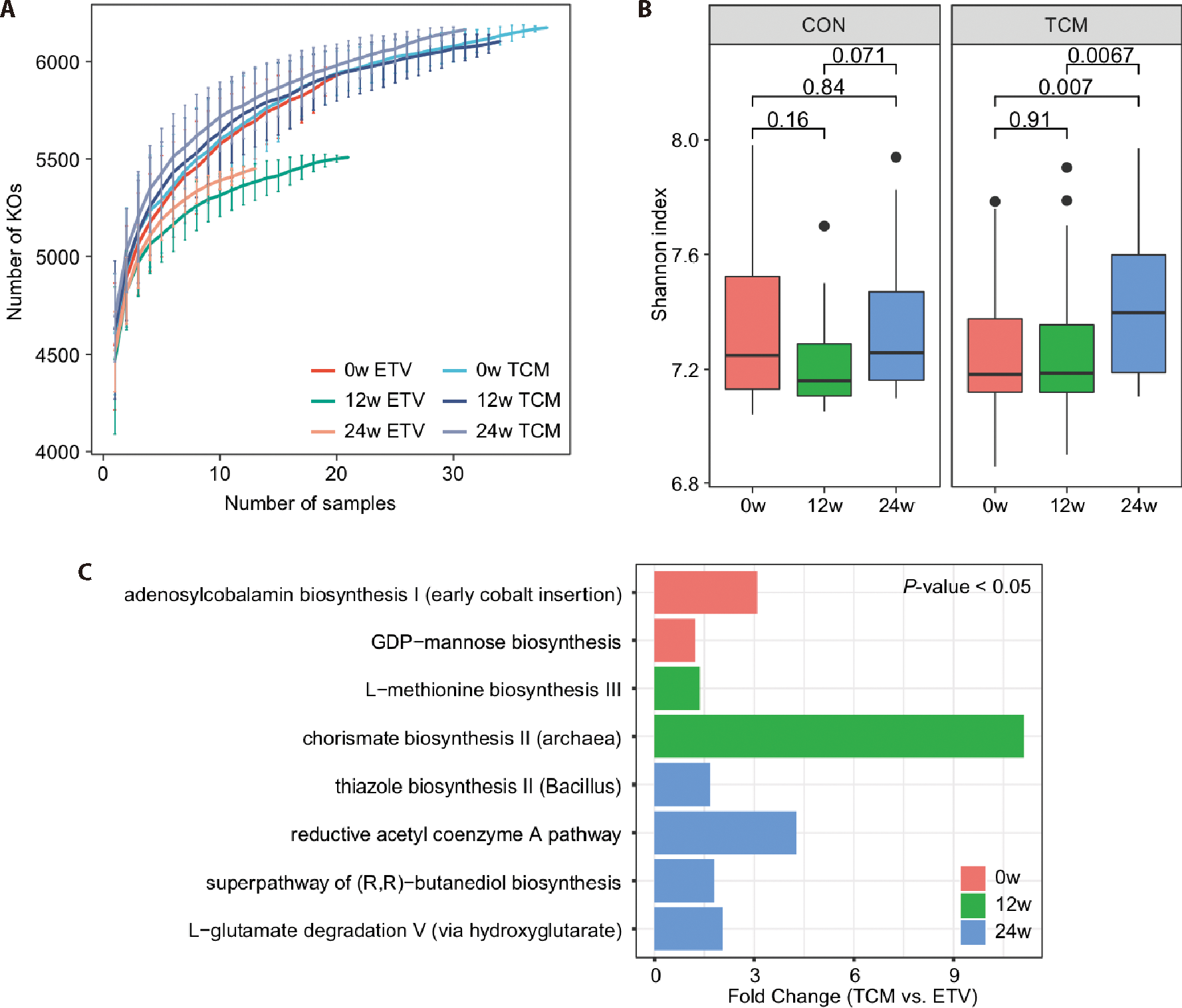
Figure 4 Effects of ETV and TCM on gut microbiota functions A: rarefaction curve analysis of KOs in each group of samples. The number of annotated KO genes in each group was calculated based on a randomly selected specific number of samples with 100 replacements. Quartile numbers were plotted by a line segment. B: boxplot showing the Shannon index of gut microbiota function for patients in ETV and TCM groups at different treatment periods, revealing microbiota function diversity in patients’ gut. C: fold change of gut microbiota function between patients in TCM and ETV groups. Only functional items with significant differences (P<0.05) are shown, TCM group: n = 38, ETV group, n = 21. Patients in the TCM group were treated with ZGHY 1 potion/d (oral administration in two doses) and ETV 0.5 mg/d, and the ETV group were treated with ETV 0.5 mg/d. All patients were treated for a total of 24 weeks, fecal samples were collected and analyzed in the 12 and 24 weeks, respectively. ETV: entecavir; TCM: Traditional Chinese Medicine (FZHY + ETV group); CON: control (ETV group); KO: Kyoto Encyclopedia of Genes and Genomes Orthology; ZGHY: Zhenggan Huayu decoction.
| [1] | Nguyen MH, Wong G, Gane E, Kao JH, Dusheiko G. Hepatitis B virus: advances in prevention, diagnosis, and therapy. Clin Microbiol Rev 2020; 33: e00046-19. |
| [2] |
Suhail M, Abdel-Hafiz H, Ali A, Fatima K, et al. Potential mechanisms of hepatitis B virus induced liver injury. World J Gastroenterol 2014; 20: 12462-72.
DOI URL |
| [3] |
Tang LSY, Covert E, Wilson E, Kottilil S. Chronic hepatitis B Infection: a review. JAMA 2018; 319: 1802-13.
DOI PMID |
| [4] |
Seto WK, Lo YR, Pawlotsky JM, Yuen MF. Chronic hepatitis B virus infection. Lancet 2018; 392: 2313-24.
DOI URL |
| [5] |
Sarin SK, Kumar M, Lau GK, et al. Asian-Pacific clinical practice guidelines on the management of hepatitis B: a 2015 update. Hepatol Int 2016; 10: 1-98.
DOI PMID |
| [6] |
Zhou WC, Zhang QB, Qiao L. Pathogenesis of liver cirrhosis. World J Gastroenterol 2014; 20: 7312-24.
DOI URL |
| [7] |
Calvaruso V, Craxì A. Fibrosis in chronic viral hepatitis. Best Pract Res Clin Gastroenterol 2011; 25: 219-30.
DOI PMID |
| [8] | Wang T, Jin W, Huang Q, et al. Clinical efficacy and safety of eight Traditional Chinese Medicine combined with entecavir in the treatment of chronic hepatitis B liver fibrosis in adults: a network Meta-analysis. Evid Based Complement Alternat Med: eCAM 2020; 2020: 7603410. |
| [9] |
Choi WM, Yip TC, Lim YS, Wong GL, Kim WR. Methodological challenges of performing Meta-analyses to compare the risk of hepatocellular carcinoma between chronic hepatitis B treatments. J Hepatol 2022; 76: 186-94.
DOI URL |
| [10] |
Lange CM, Bojunga J, Hofmann WP, et al. Severe lactic acidosis during treatment of chronic hepatitis B with entecavir in patients with impaired liver function. Hepatology 2009; 50: 2001-6.
DOI PMID |
| [11] |
Zhang L, Wang G, Hou W, Li P, Dulin A, Bonkovsky HL. Contemporary clinical research of Traditional Chinese Medicines for chronic hepatitis B in China: an analytical review. Hepatology 2010; 51: 690-8.
DOI PMID |
| [12] | Hu J, Teng J, Wang W, et al. Clinical efficacy and safety of Traditional Chinese Medicine Xiaoyao San in insomnia combined with anxiety. Medicine 2021; 100: e27608. |
| [13] |
Zhang DQ, Mu YP, Xu Y, Chen JM, Liu P, Liu W. Research progress in Chinese Medicine preparations for promoting blood circulation and removing blood stasis for cirrhotic patients with portal vein thrombosis following splenectomy. Chin J Integr Med 2020; 28: 855-863.
DOI |
| [14] | Huang C, Shen D, Sun S, et al. Effect of Fufang Biejia Ruangan Tablet on lowering biochemical and virological parameters of hepatic fibrosis in patients with chronic hepatitis B: protocol for a systematic review and Meta-analysis of randomized controlled trials and cohort studies. Medicine 2019; 98: e15297. |
| [15] |
Wang T, Zhou X, Liu H, et al. Fuzheng Huayu capsule as an adjuvant treatment for HBV-related cirrhosis: a systematic review and Meta-analysis. Phytother Res 2018; 32: 757-68.
DOI PMID |
| [16] |
Li X, Zhou D, Chi X, et al. Entecavir combining Chinese herbal medicine for HBeAg-positive chronic hepatitis B patients: a randomized, controlled trial. Hepatol Int 2020; 14:985-96.
DOI PMID |
| [17] |
Li X, Zhang L, Qiu M, et al. Chinese herbal medicine combined with entecavir to reduce the off-therapy recurrence risk in HBeAg-positive chronic hepatitis B patients: a multicener, double-blind, randomized controlled trial in China. Trials 2020; 21: 708.
DOI PMID |
| [18] |
Jones RM, Neish AS. Gut microbiota in intestinal and liver disease. Annu Rev Pathol 2021; 16: 251-75.
DOI PMID |
| [19] |
Wang X, Chen L, Wang H, Cai W, Xie Q. Modulation of bile acid profile by gut microbiota in chronic hepatitis B. J Cell Mol Med 2020; 24: 2573-81.
DOI PMID |
| [20] |
Bajaj JS. Alcohol, liver disease and the gut microbiota. Nat Rev Gastroenterol Hepatol 2019; 16: 235-46.
DOI PMID |
| [21] |
Fan Y, Pedersen O. Gut microbiota in human metabolic health and disease. Nat Rev Microbiol 2021; 19: 55-71.
DOI |
| [22] | Yang R, Xu Y, Dai Z, Lin X, Wang H. The immunologic role of gut microbiota in patients with chronic HBV infection. J Immunol Res 2018; 2018: 2361963. |
| [23] |
Zeng Y, Chen S, Fu Y, et al. Gut microbiota dysbiosis in patients with hepatitis B virus-induced chronic liver disease covering chronic hepatitis, liver cirrhosis and hepatocellular carcinoma. J Viral Hepat 2020; 27: 143-55.
DOI URL |
| [24] |
Lv LJ, Li SH, Li SC, et al. Early-onset preeclampsia is associated with gut microbial alterations in antepartum and postpartum women. Front Cell Infect Microbiol 2019; 9: 224.
DOI URL |
| [25] |
Bolyen E, Rideout JR, Dillon MR, et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 2019; 37: 852-7.
DOI PMID |
| [26] | Quast C, Pruesse E, Yilmaz P, et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 2013; 41: D590-6. |
| [27] |
Douglas GM, Maffei VJ, Zaneveld JR, et al. PICRUSt2 for prediction of metagenome functions. Nat Biotechnol 2020; 38: 685-8.
DOI PMID |
| [28] |
Ren Z, Li A, Jiang J, et al. Gut microbiome analysis as a tool towards targeted non-invasive biomarkers for early hepatocellular carcinoma. Gut 2019; 68: 1014-23.
DOI PMID |
| [29] | Li X, Wu S, Du Y, Yang L, Li Y, Hong B. Entecavir therapy reverses gut microbiota dysbiosis induced by hepatitis B virus infection in a mouse model. Int J Antimicrob Agents 2020; 56: 106000. |
| [30] |
Compare D, Coccoli P, Rocco A, et al. Gut--liver axis: the impact of gut microbiota on non alcoholic fatty liver disease. Nutr Metab Cardiovasc Dis 2012; 22: 471-6.
DOI PMID |
| [31] |
Wan MLY, El-Nezami H. Targeting gut microbiota in hepatocellular carcinoma: probiotics as a novel therapy. Hepatobiliary Surg Nutr 2018; 7: 11-20.
DOI URL |
| [32] |
Gevers D, Kugathasan S, Denson LA, et al. The treatment-naive microbiome in new-onset Crohn's disease. Cell Host Microbe 2014; 15: 382-92.
DOI PMID |
| [33] |
Bajaj JS, Heuman DM, Hylemon PB, et al. Altered profile of human gut microbiome is associated with cirrhosis and its complications. J Hepatol 2014; 60: 940-7.
DOI PMID |
| [34] |
Shek D, Chen D, Read SA, Ahlenstiel G. Examining the gut-liver axis in liver cancer using organoid models. Cancer Lett 2021; 510: 48-58.
DOI PMID |
| [35] |
Roberti MP, Yonekura S, Duong CPM, et al. Chemotherapy-induced ileal crypt apoptosis and the ileal microbiome shape immunosurveillance and prognosis of proximal colon cancer. Nat Med 2020; 26: 919-31.
DOI PMID |
| [36] | Kaakoush NO. Insights into the role of erysipelotrichaceae in the human host. Front Cell Infect Microbiol 2015; 5: 84. |
| [37] |
Lurie Y, Webb M, Cytter-Kuint R, Shteingart S, Lederkremer GZ. Non-invasive diagnosis of liver fibrosis and cirrhosis. World J Gastroenterol 2015; 21: 11567-83.
DOI URL |
| [38] | Qin F, Wu H, Li X, Han J. Correlation between changes in gut flora and serum inflammatory factors in children with noninfectious diarrhea. J Int Med Res 2020; 48: 300060519896154. |
| [39] |
Zhu L, Baker SS, Gill C, et al. Characterization of gut microbiomes in nonalcoholic steatohepatitis (NASH) patients: a connection between endogenous alcohol and NASH. Hepatology 2013; 57: 601-9.
DOI PMID |
| [40] |
Maccioni L, Gao B, Leclercq S, et al. Intestinal permeability, microbial translocation, changes in duodenal and fecal microbiota, and their associations with alcoholic liver disease progression in humans. Gut microbes 2020; 12: 1782157.
DOI URL |
| [41] |
Wang B, Zhang SQ, Dong JL, et al. Ambient temperature structures the gut microbiota of zebrafish to impact the response to radioactive pollution. Environ Pollut 2021; 293: 118539.
DOI URL |
| [42] |
Martin-Gallausiaux C, Marinelli L, Blottière HM, Larraufie P, Lapaque N. SCFA: mechanisms and functional importance in the gut. Proc Nutr Soc 2021; 80: 37-49.
DOI URL |
| [43] |
Louis P, Flint HJ. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol Lett 2009; 294: 1-8.
DOI PMID |
| [44] |
Suez J, Zmora N, Zilberman-Schapira G, et al. Post-antibiotic gut mucosal microbiome reconstitution is impaired by probiotics and improved by autologous FMT. Cell 2018; 174: 1406-23. e16.
DOI PMID |
| [1] | JIANG Li, FU Qiang, WANG Shidong, ZHAO Jinxi, CHEN Yu, LI Jiayue, XIAO Yonghua, HUANG Weijun, SUN Ruixi, XIAO Yao, SHEN Aijia, WANG Junheng, LIU Jiangteng, FU Xiaozhe, LI Yuanyuan, ZHAO Yu, XUE Taiqi. Effects of Shenlian formula (参连方) on microbiota and inflammatory cytokines in adults with type 2 diabetes: a double-blind randomized clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 760-769. |
| [2] | LUO Xin, XIE Jing, HUANG Li, GAN Wenfan, CHEN Ming. Efficacy and safety of activating blood circulation and removing blood stasis of Traditional Chinese Medicine for managing renal fibrosis in patients with chronic kidney disease: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 429-440. |
| [3] | ZHAO Ye, WANG Xian, GU Ling, LI Zihang, ZHU Jingtian, WANG Wenkai, ZHANG Liang, XUE Mei. Efficacy of Danggui Buxue decoction (当归补血汤) on diabetic nephropathy-induced renal fibrosis in rats and possible mechanism [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 507-513. |
| [4] | XIONG Yunzhao, LIU Lingjin, LIU Ziqian, CHEN Gege, HAO Juan, GAO Xiaomeng, QIANG Panpan, WANG Zheng, XU Qingyou. Huoxue Jiedu Huayu recipe (活血解毒化瘀方) alleviates contralateral renal fibrosis in unilateral ureteral obstruction rats by inhibiting the transformation of macrophages to myofibroblast [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 105-112. |
| [5] | XU Baogui, ZHENG Jiawen, TIAN Xiaoxiao, YUAN Falei, LIU Zhongliang, YANG Zuisu, DING Xianjun. Antihepatofibrotic effect of Guizhifuling pill (桂枝茯苓丸) on carbon tetrachloride-induced liver fibrosis in mice [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 715-722. |
| [6] | ZHAO Jie, WANG Li, CAO Ai-li, WANG Yun-man, CHI Yang-feng, WANG Yi, WANG Hao, PENG Wen. Huangqi decoction (黄芪汤) attenuates renal interstitial fibrosis via transforming growth factor-β1/mitogen-activated protein kinase signaling pathways in 5/6 nephrectomy mice [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 723-731. |
| [7] | HU Xijiao, LI Shuoxi, YANG Dongxia, GU Na, LIU Jinzhe, WANG Yawen, LIU Li, SUN Yiming. Modified Gexiazhuyu decoction (膈下逐瘀汤加减方) alleviates chronic salpingitis via p38 signaling pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 213-220. |
| [8] | Jiaru LIN, Li WANG, Bo CHEN, Santao OU, Jianhua QIN, Junming FAN. Shenweifang-containing serum inhibits transforming growth factor-β1-induced myofibroblast differentiation in normal rat kidney interstitial fibroblast cells [J]. Journal of Traditional Chinese Medicine, 2022, 42(1): 39-48. |
| [9] | ZHONG Jian, FANG Guiyu, WANG Zixia, CHEN Ping, LU Dengyong, SHI Xiaodong. Yishen Huoxue decoction(益肾活血方) attenuates unilateral ureteric obstruction-induced renal fibrosis and hypoxia-induced reactive oxygen species generation via adenosine monophosphate-activated protein kinase/peroxisome proliferator-activated receptor coa [J]. Journal of Traditional Chinese Medicine, 2021, 41(6): 875-882. |
| [10] | LU Feng, GENG Jiabao, ZHANG Jiawei, DONG Yu. Effect of entecavir plus Ganshuang granule(肝爽颗粒)on fibrosisand cirrhosis in patients with chronic hepatitis B [J]. Journal of Traditional Chinese Medicine, 2021, 41(4): 624-629. |
| [11] | Guo Sijia, Song Yalin, Feng Jihong, Liu Shuang, Li Yuechuan, Liu Min, Wei Luqing, Zhang Xian, Xie Hui, Sun Zengtao. Effects of Qizhukangxian granules on idiopathic pulmonary fibrosis: a randomized, double blind, placebo-controlled and multicenter clinical pilot trial [J]. Journal of Traditional Chinese Medicine, 2020, 40(4): 674-682. |
| [12] | Chen Hui, Liu Xiao, Su Yinxu, Chang Ke, Wu Haotian, Su Guangbao, Gong Jiening. Notch signaling pathway mediates the immunomodulatory mechanism of Yangfei Huoxue decoction alleviating bleomycin-induced pulmonary fibrosis in rats [J]. Journal of Traditional Chinese Medicine, 2020, 40(2): 204-211. |
| [13] | Wang Jiepeng, Fang Chaoyi, Wang Shaoxian, Fang Fang, Chu Xinqiao, Liu Na, Lu Chenxi, Wang Shuo, Li Wei. Danggui Buxue Tang ameliorates bleomycin-induced pulmonary fibrosis in rats through inhibiting transforming growth factor-β1/Smad3/plasminogen activator inhibitor-1 signaling pathway [J]. Journal of Traditional Chinese Medicine, 2020, 40(2): 236-244. |
| [14] | Pei Lixin, Shu Shengnan, Wang Xuanying, Ji Baoyu. Effect of chrysanthemum extract on myocardial fibrosis in rats with renovascular hypertension [J]. Journal of Traditional Chinese Medicine, 2019, 39(04): 542-549. |
| [15] | Bai Yunping, Zhao Peng, Wu Mingming, Li Jiansheng. A pharmacological approach to study the active compounds in Jinshui Huanxian formula in treatment of pulmonary fibrosis [J]. Journal of Traditional Chinese Medicine, 2019, 39(03): 364-379. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

