Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (2): 427-438.DOI: 10.19852/j.cnki.jtcm.2026.02.015
• Original Articles • Previous Articles Next Articles
Effect of Huiyang Shengji unguent (回阳生肌膏) on the NOD-like receptor family pyrin domain containing protein 3/caspase1/ gasdermin D pathway and lymphatic angiogenesis in patients with diabetic foot ulcer
YU Fangning1, LIN Li1, TANG Xiao1, HE Xiujuan3, ZHANG Bo1, CHEN Yukun2, MA Yizhao2, LIU Zeyu1, YE Jinsheng4, XU Xuying1( )
)
- 1
Department of Vascular Surgery ,Beijing Traditional Chinese Medicine Hospital, Capital Medical University Beijing 100010, China
2Clinical Medical College ,Beijing University of Chinese Medicine Beijing 100020, China
3Research Laboratory for the Theory and Application of Wound Healing ,Beijing Institute of Traditional Chinese Medicine Beijing 100010, China
4Department of General Surgery of Beijing Traditional Chinese Medicine Hospital ,Capital Medical University Beijing 100010, China
-
Received:2025-01-11Accepted:2025-06-05Online:2026-04-15Published:2026-04-04 -
Contact:XU Xuying, Department of Vascular Surgery, Beijing Traditional Chinese Medicine Hospital, Capital Medical University, Beijing 100010, China. xxy7341@163.com; Telephone: +86-10-87906642 -
About author:YU Fangning and LIN Li are co-first authors and contributed equally to this work
-
Supported by:National Natural Science Foundation of China: Mechanism Study of Huiyang Shengji Ointment in Inducing Acute Inflammation in Chronic Diabetic Foot Ulcers and Promoting Wound Healing Based on the Signal Transducer and Activator of Transcription 3/Forkhead Box Protein M1 Pathway(Grant numbers 82174388)
Cite this article
YU Fangning, LIN Li, TANG Xiao, HE Xiujuan, ZHANG Bo, CHEN Yukun, MA Yizhao, LIU Zeyu, YE Jinsheng, XU Xuying. Effect of Huiyang Shengji unguent (回阳生肌膏) on the NOD-like receptor family pyrin domain containing protein 3/caspase1/ gasdermin D pathway and lymphatic angiogenesis in patients with diabetic foot ulcer[J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 427-438.
share this article
| Item | HYSJ group (n = 10) | Control group (n = 10) | P value | |
|---|---|---|---|---|
| Gender (n) | Male | 7 | 8 | 0.6 |
| Female | 3 | 2 | ||
| Age (years) | 67.0±10.5 | 70.4±10.7 | 0.5 | |
| Fasting blood glucose (mmol/L) | 6.2±0.8 | 6.3±1.1 | 0.9 | |
| Glycated hemoglobin (%) | 7.1±1.5 | 7.5±1.9 | 0.6 | |
| TP (g/L) | 65.4±9.3 | 68.7±7.0 | 0.4 | |
| ALB (g/L) | 35.8±7.1 | 35.4±4.2 | 0.9 | |
| ALT (U/L) | 18.5±12.4 | 28.4±41.3 | 0.5 | |
| UA (mmol/L) | 7.4±2.5 | 8.2±3.4 | 0.6 | |
| Scr (umol/L) | 67.1±27.1 | 84.8±30.2 | 0.2 | |
| Hgb (g/L) | 109.7±18.0 | 110.3±22.2 | 0.9 | |
Table 1 Comparison of baseline data in the two patient groups ($\stackrel{-}{x}\pm s$)
| Item | HYSJ group (n = 10) | Control group (n = 10) | P value | |
|---|---|---|---|---|
| Gender (n) | Male | 7 | 8 | 0.6 |
| Female | 3 | 2 | ||
| Age (years) | 67.0±10.5 | 70.4±10.7 | 0.5 | |
| Fasting blood glucose (mmol/L) | 6.2±0.8 | 6.3±1.1 | 0.9 | |
| Glycated hemoglobin (%) | 7.1±1.5 | 7.5±1.9 | 0.6 | |
| TP (g/L) | 65.4±9.3 | 68.7±7.0 | 0.4 | |
| ALB (g/L) | 35.8±7.1 | 35.4±4.2 | 0.9 | |
| ALT (U/L) | 18.5±12.4 | 28.4±41.3 | 0.5 | |
| UA (mmol/L) | 7.4±2.5 | 8.2±3.4 | 0.6 | |
| Scr (umol/L) | 67.1±27.1 | 84.8±30.2 | 0.2 | |
| Hgb (g/L) | 109.7±18.0 | 110.3±22.2 | 0.9 | |
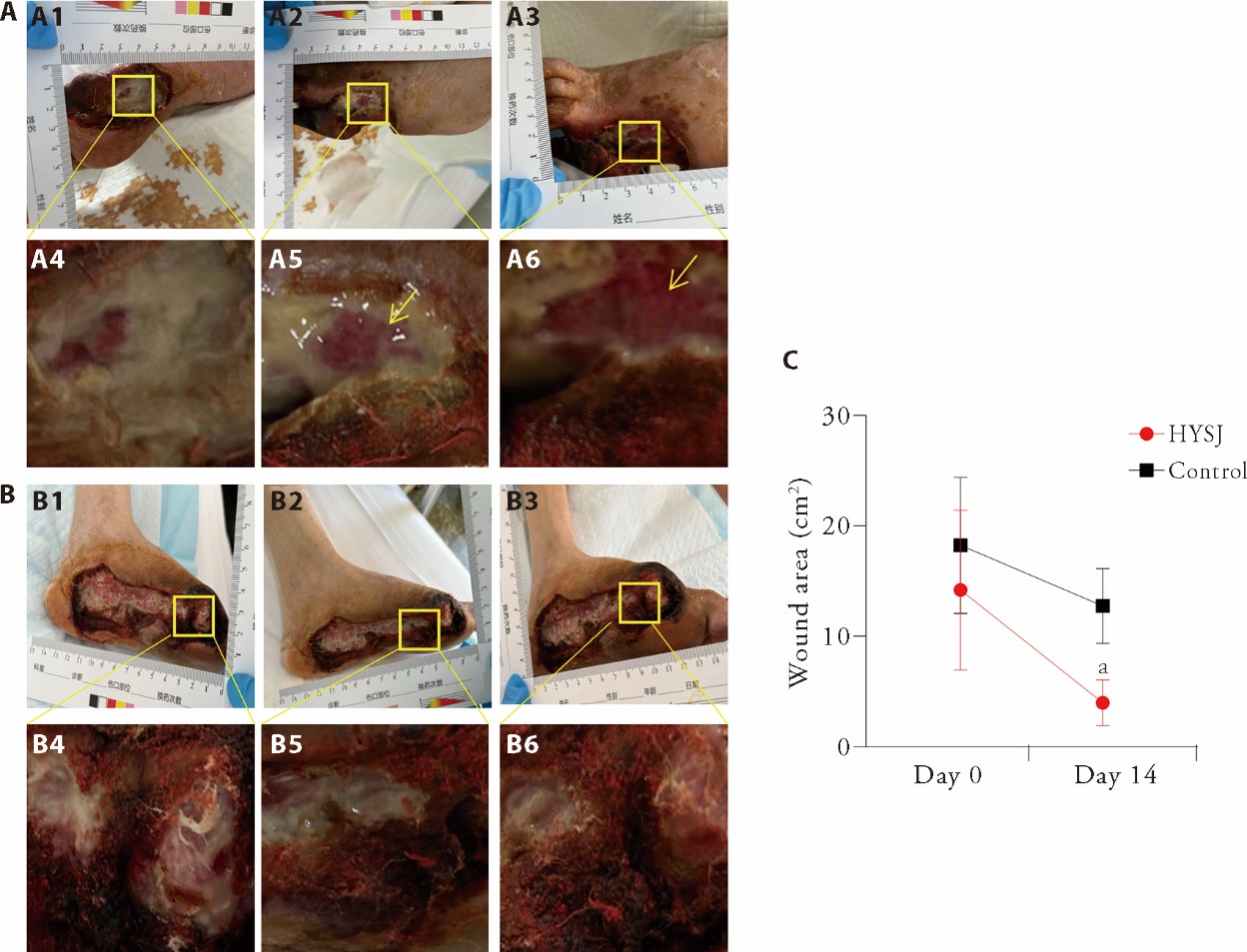
Figure 1 Photographs of the wounds in each group and the changes in wound area 14 d after the intervention A: photographs of typical wounds in the HYSJ group; B: photographs of typical wounds in the control group; C: the wound area before intervention and on the 14th day of intervention; A1, B1: the wound morphology before intervention; A2, B2: the wound morphology on the 7th day of intervention; A3, B3: the wound morphology on the 14th day of intervention; A4, B4: the local wound morphology before intervention; A5, B5: the local wound morphology on the 7th day of intervention; A6, B6: the local wound morphology on the 14th day of intervention. HYSJ group: treated with Huiyang Shengji unguent; control group: treated with bFGF. HYSJ: Huiyang Shengji unguent; bFGF: basic fibroblast growth factor. The independent sample t-test was used for between-group comparisons. Data were expressed as mean ± standard deviation (n = 10). Compared with control group, aP < 0.001.
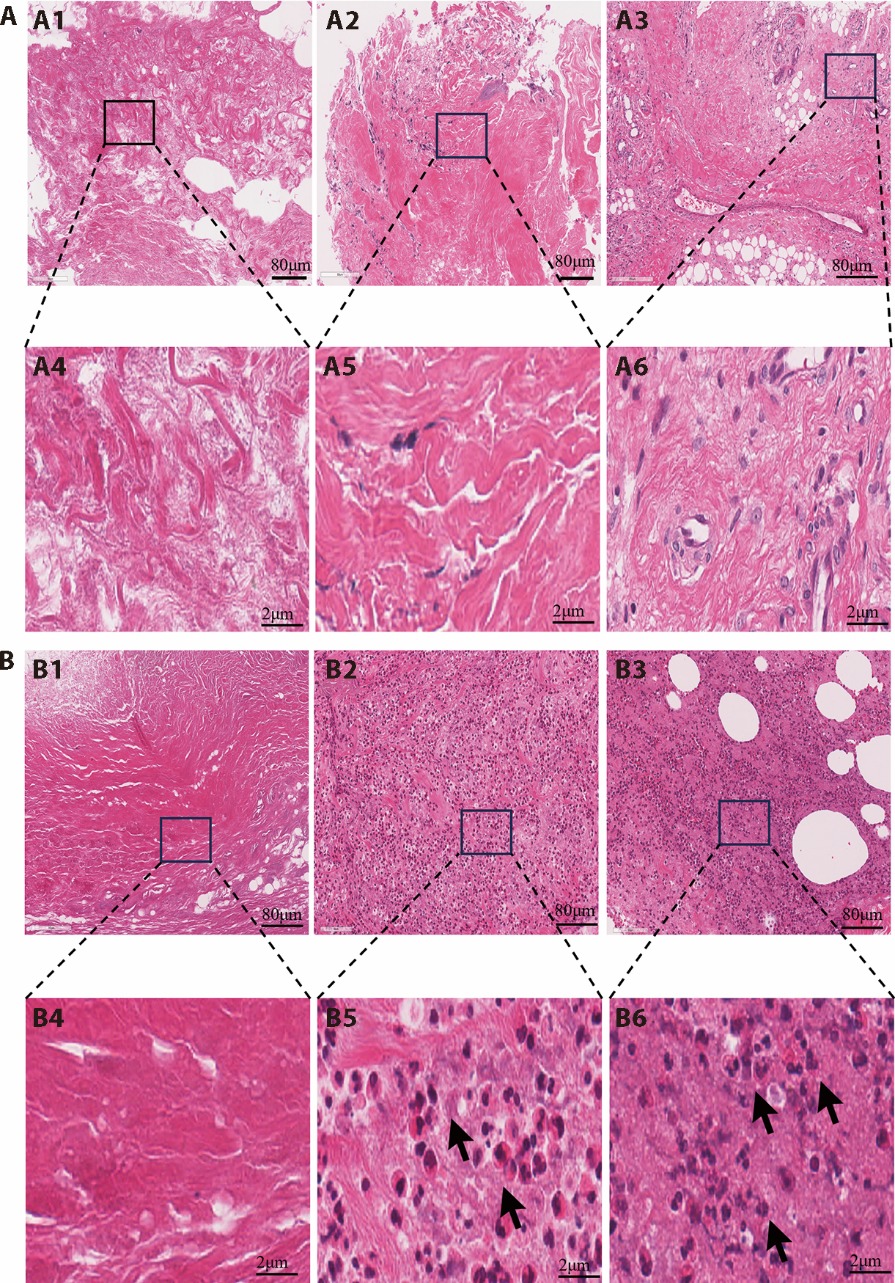
Figure 2 HE staining images of the wound tissues from each group at different intervention time A: HE staining of wound tissue in the control group on days 0, 7, and 14 of treatment (× 40); B: HE staining of wound tissue in the HYSJ group on days 0, 7, and 14 of treatment (× 40); A1, B1: image before treatment; A2, B2: image on day 7 of treatment (× 40); A3, B3: image on day 14 of treatment (× 40); A4, B4: detail image before treatment; A5, B5: detail image on day 7 of treatment; A6, B6: detail image on day 14 of treatment. The arrows indicate infiltrated neutrophils. Scale bar: 80 μm. HYSJ group: treated with Huiyang Shengji unguent; control group: treated with bFGF. HE: hematoxylin-eosin; HYSJ: Huiyang Shengji unguent; bFGF: basic fibroblast growth factor.
| Item | Control group (n = 10) | HYSJ group (n = 10) | |
|---|---|---|---|
| IL-1β | Day 0 | 2472.35 (379.63, 82825.95) | 2366.69 (1011.29, 15433.128) |
| Day 7 | 1864.33 (994.49, 2583.92) | 604.83 (367.05, 1169.90)a | |
| Day 14 | 2035.07 (875.39, 2933.65) | 548.94 (115.37, 692.52)ab | |
| IL-18 | Day 0 | 1073.13 (412.47, 4554.46) | 2036.31 (334.36, 8956.71) |
| Day 7 | 2400.49 (1384.87, 5397.56) | 388.13 (118.52, 883.67)a | |
| Day 14 | 2191.53 (375.93, 6433.65) | 143.71 (113.41, 690.34)ab | |
Table 2 Changes of IL-1β and IL-18 contents in wound secretion of two groups of patients [pg/mL, M (Q1, Q3)]
| Item | Control group (n = 10) | HYSJ group (n = 10) | |
|---|---|---|---|
| IL-1β | Day 0 | 2472.35 (379.63, 82825.95) | 2366.69 (1011.29, 15433.128) |
| Day 7 | 1864.33 (994.49, 2583.92) | 604.83 (367.05, 1169.90)a | |
| Day 14 | 2035.07 (875.39, 2933.65) | 548.94 (115.37, 692.52)ab | |
| IL-18 | Day 0 | 1073.13 (412.47, 4554.46) | 2036.31 (334.36, 8956.71) |
| Day 7 | 2400.49 (1384.87, 5397.56) | 388.13 (118.52, 883.67)a | |
| Day 14 | 2191.53 (375.93, 6433.65) | 143.71 (113.41, 690.34)ab | |
| Treatment time | Control group (n = 10) | HYSJ group (n = 10) |
|---|---|---|
| Day 0 | 0.39±0.17 | 0.21±0.31 |
| Day 7 | 0.15±0.10 | 0.45±0.18ab |
| Day 14 | 0.26±0.11 | 0.42±0.19b |
Table 3 Changes of VEGF-C content in wound exudates of two groups ($ \bar{x} \pm s$, ng/mL)
| Treatment time | Control group (n = 10) | HYSJ group (n = 10) |
|---|---|---|
| Day 0 | 0.39±0.17 | 0.21±0.31 |
| Day 7 | 0.15±0.10 | 0.45±0.18ab |
| Day 14 | 0.26±0.11 | 0.42±0.19b |

Figure 3 Analysis of IHC staining of lymphatic vessel formation-related proteins in wound tissues in each group at different intervention time A: IHC staining images of LYVE-1, PROX-1, and VEGFR-3 proteins in the wound tissue of the control group on days 0, 7, and 14 of treatment; B: IHC staining images of LYVE-1, PROX-1, and VEGFR-3 proteins in the wound tissue of the HYSJ group on days 0, 7, and 14 of treatment; C: quantitative IHC analysis of LYVE -1of wound tissue in the two groups; D: quantitative IHC analysis of PROX-1 of wound tissue in the two groups; E: quantitative IHC analysis of VEGFR-3 of wound tissue in the two groups; A1, B1: LYVE-1 protein before intervention (× 40); A2, B2: PROX-1 protein before intervention (× 40); A3, B3: VEGFR-3 protein before intervention (× 40); A4, B4: LYVE-1 on the 7th day of intervention (× 40); A5, B5: PROX-1 on the 7th day of intervention (× 40); A6, B6: VEGFR-3 on the 7th day of intervention (× 40); A7, B7: LYVE-1 on the 14th day of intervention (× 40); A8, B8: PROX-1 on the 14th day of intervention (× 40); A9, B9: VEGFR-3 on the 14th day of intervention (× 40). The arrows indicate positive staining points. Scale bar: 80 μm. HYSJ group: treated with Huiyang Shengji unguent; control group: treated with bFGF. IHC: immunohistochemical; LYVE-1: lymphatic vessel endothelial hyaluronan receptor 1; PROX-1: prospero homeobox protein 1; VEGFR-3: vascular endothelial growth factor receptor 3; HYSJ: Huiyang Shengji unguent; bFGF: basic fibroblast growth factor. Data were expressed as mean ± standard deviation (n = 6). The independent sample t-test was used for between-group comparisons. Compared with control group, aP < 0.001; bP < 0.01; cP < 0.05.
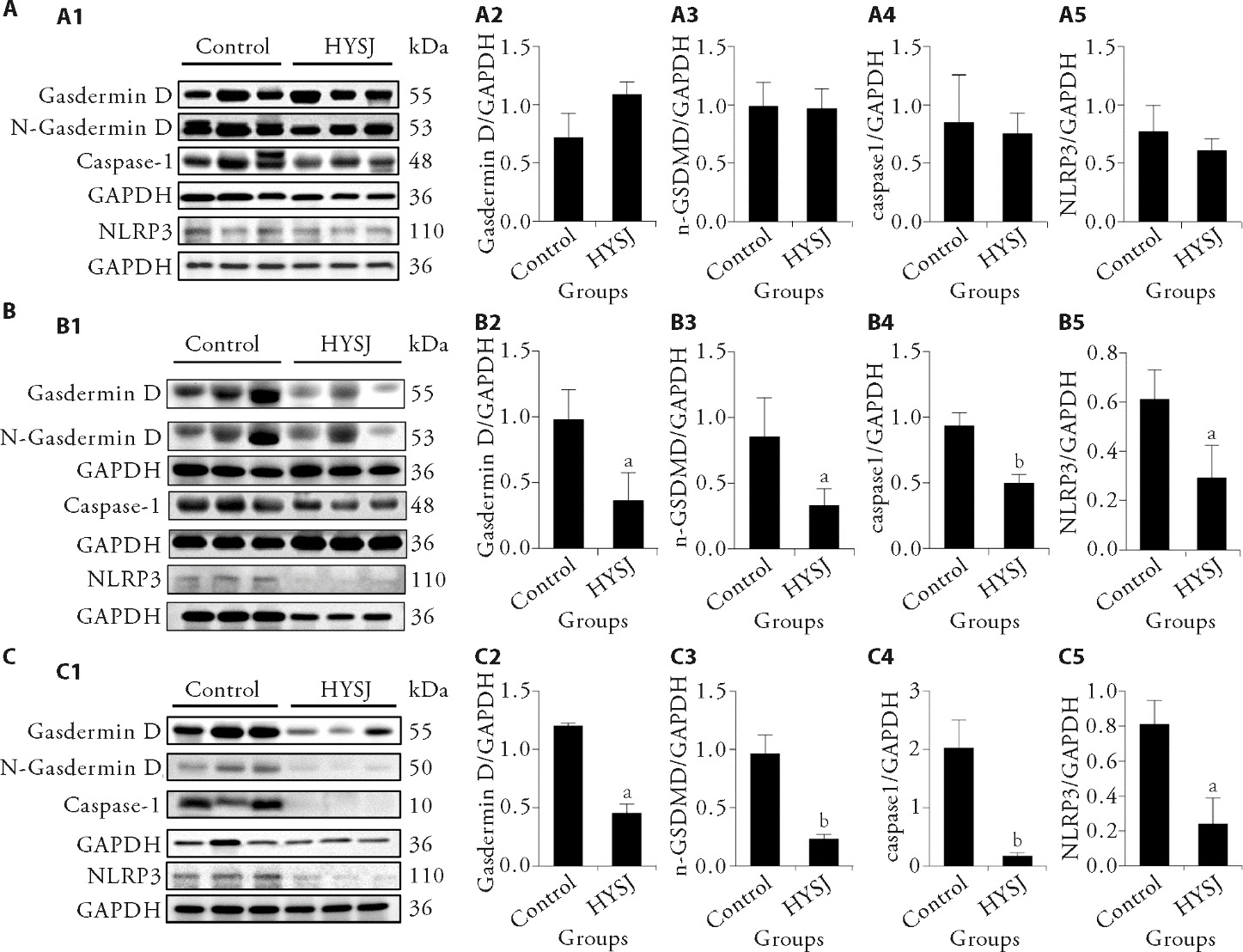
Figure 4 Western blot analysis of inflammatory-related proteins in the wound tissues in each group at different intervention time A: relative expression levels of the various proteins in wound tissues in the two patient groups before treatment; B: relative expression levels of the various proteins in wound tissues in the two patient groups of treatment for 7 d; C: relative expression levels of the various proteins in wound tissues in the two patient groups of treatment for 14 d; A1, B1, C1: the protein electrophoresis bands; A2, B2, C2: expression levels of Gasdermin D protein; A3, B3, C3: expression levels of N-Gasdermin D protein; A4, B4, C4: expression levels of caspase-1 protein; A5, B5, C5: expression levels of NLRP3 protein. HYSJ group: treated with Huiyang Shengji unguent; control group: treated with bFGF. NLRP3: NOD-like receptor family, pyrin domain containing protein 3; HYSJ: Huiyang Shengji unguent; bFGF: basic fibroblast growth factor. Data were expressed as mean ± standard deviation (n = 3). The independent sample t-test was used for between-group comparisons. Compared with control group, aP < 0.05, bP < 0.01.
| 1. | Lu B, Hu J, Wen J, et al. Determination of peripheral neuropathy prevalence and associated factors in Chinese subjects with diabetes and pre-diabetes - Shanghai diabetic neuropathy epidemiology and molecular genetics study (SH-DREAMS). PLoS One 2013; 8: e61053. |
| 2. |
Zhang P, Lu J, Jing Y, Tang S, Zhu D, Bi Y. Global epidemiology of diabetic foot ulceration: a systematic review and Meta-analysis. Ann Med 2017; 49: 106-16.
DOI URL |
| 3. |
Xiong Y, Tang R, Li X, Liu H. Effect of Neibu Huangqi Youhua formula (内补黄芪汤优化方) on postoperative wound healing, inflammatory factors and pain mediators of anal fistula. J Tradit Chin Med 2025; 45: 628-32.
DOI |
| 4. |
Meng J, Zhang H, Cao Y, et al. Zuyangping (足疡平) formula promotes skin wound healing in diabetic rats. J Tradit Chin Med 2024; 44: 1194-203.
DOI |
| 5. | Dong Y, Huang F, Wang Y, et al. Treatment of sore and ulcer with Yin syndrome of ecthyma by professor Wang Yuzhang’s method of reviving yang and promoting tissue regeneration. Jilin Zhong Yi Yao 2015; 35: 999-1002. |
| 6. | Dong Y, Huang F, Wang Y, et al. Report of typical cases of ulcers in Yin type of ecthyma treated with Wang Yuzhang’s method of reviving yang to generate muscle. Shi Jie Zhong Yi Yao 2015; 10: 1912-3. |
| 7. | Zhou M, Wu L, Tan C, Xu X. Study of Huiyang Shengji ointment on anti-functional damage of mouse bone marrow endothelial progenitor cells. Beijing Zhong Yi Yao Da Xue Xue Bao 2022; 45: 41-52. |
| 8. | Zhou M, Jia X, Xu X. Intervention effect of Huiyang Shengji ointment on human-derived microvascular endothelial cells. Huan Qiu Zhong Yi Yao 2020; 13: 784-90. |
| 9. | Wu L, Guo H, Yang Y, Xu X. Exploration on the mechanism of Huiyang Shengji ointment in the treatment of diabetic foot ulcer. Beijing Zhong Yi Yao 2021; 40: 956-62. |
| 10. | Cai H, Wang P, Zhang B, Dong X. Expression of the NEK7/NLRP3 inflammasome pathway in patients with diabetic lower extremity arterial disease. BMJ Open Diabetes Res Care 2020; 8: e001808. |
| 11. |
Huang W, Jiao J, Liu J, et al. MFG-E 8 accelerates wound healing in diabetes by regulating “NLRP3 inflammasome-neutrophil extracellular traps” axis. Cell Death Discov 2020; 6: 84.
DOI PMID |
| 12. |
Wang T, Zhao J, Zhang J, et al. Heparan sulfate inhibits inflammation and improves wound healing by downregulating the NLR family pyrin domain containing 3 (NLRP3) inflammasome in diabetic rats. J Diabetes 2018; 10: 556-63.
DOI PMID |
| 13. |
Zhao Y, Wang Q, Yan S, et al. Bletilla striata polysaccharide promotes diabetic wound healing through inhibition of the NLRP3 inflammasome. Front Pharmacol 2021; 12: 659215.
DOI URL |
| 14. |
Zhou J, Wei T, He Z. ADSCs enhance VEGFR3-mediated lymphangiogenesis via METTL3-mediated VEGF-C m6A modification to improve wound healing of diabetic foot ulcers. Mol Med 2021; 27: 146.
DOI PMID |
| 15. |
Ducoli L, Detmar M. Beyond PROX1: transcriptional, epigenetic, and noncoding RNA regulation of lymphatic identity and function. Dev Cell 2021; 56: 406-26.
DOI PMID |
| 16. | Chinese Medical Doctor Association Branch of Integrative Chinese and Western Medicine, Chinese Association of Integrated Traditional and Western Medicine Evidence-Based Medicine Professional Committee. Clinical research methods of TCM and integrated Traditional Chinese and Western Medicine. Zhong Guo Zhong Xi Yi Jie He Za Zhi 2015; 35: 901-32. |
| 17. | Diabetes Society of Chinese Medical Association, Infectious Diseases Branch of Chinese Medical Association, Tissue Repair and Regeneration Branch of Chinese Medical Association. Guidelines for the prevention and treatment of diabetic foot in China (2019 edition). Zhong Hua Tang Niao Bing Za Zhi 2019; 11: 92-108. |
| 18. | Meng J, Xu X, Wang G, Zhou M, Xie S. Effect of Huiyang Shengji Paste on wound macrophages in patients with spleen-kidney Yang deficiency syndrome of chronic skin ulcer. Zhong Yi Za Zhi 2019; 60: 391-5. |
| 19. |
Yadav JP, Singh AK, Grishina M, et al. Insights into the mechanisms of diabetic wounds: Pathophysiology, molecular targets, and treatment strategies through conventional and alternative therapies. Inflammopharmacology 2024; 32: 149-228.
DOI |
| 20. |
Luo T, Jia X, Feng WD, et al. Bergapten inhibits NLRP3 inflammasome activation and pyroptosis via promoting mitophagy. Acta Pharmacol Sin 2023; 44: 1867-78.
DOI |
| 21. |
Kataru RP, Jung K, Jang C, et al. Critical role of CD11b+ macrophages and VEGF in inflammatory lymphangiogenesis, antigen clearance, and inflammation resolution. Blood 2009; 113: 5650-9.
DOI PMID |
| 22. | Liu JJD. Liu Juanzi Gui Yi Fang. Beijing: China Publishing Group Digital Media, 2000: 4. |
| 23. | Wang G, Wang Y, Wang L, Xu X. Exploration of the mechanism of action of prescriptions represented by the “three methods of promoting tissue regeneration” on diabetic foot with chronic sore. Shi Jie Zhong Yi Yao 2020; 15: 2116-21. |
| 24. |
Zhang Y, Zhao X, Liu Y, Yang X. Sulforaphane and ophthalmic diseases. Food Sci Nutr 2024; 12: 5296-311.
DOI PMID |
| 25. |
Yu Y, Zhao H, Liu J, et al. Glucose-triggered NIR-responsive photothermal antibacterial gelatin/dextran hydrogel simultaneously targeting the high glucose and infection microenvironment in diabetic wound. Int J Biol Macromol 2025; 300: 140325.
DOI URL |
| 26. | Liu S, Wan R, Li Q, et al. Enhancing diabetic muscle repair through W-GA nanodots: a nanomedicinal approach to ameliorate myopathy in type 2 diabetes. Burns Trauma 2025; 13: tkae059. |
| 27. |
Kjaer M, Frederiksen AKS, Nissen NI, et al. Multinutrient supplementation increases collagen synthesis during early wound repair in a randomized controlled trial in patients with inguinal hernia. J Nutr 2020; 150: 792-9.
DOI PMID |
| 28. |
Smith TJ, Wilson M, Whitney C, et al. Supplemental protein and a multinutrient beverage speed wound healing after acute sleep restriction in healthy adults. J Nutr 2022; 152: 1560-73.
DOI URL |
| 29. | Jia YC, Qiu S, Xu J, Kang QL, Chai YM. Docosahexaenoic acid improves diabetic wound healing in a rat model by restoring impaired plasticity of macrophage progenitor cells. Plast Reconstr Surg 2020; 145: 942e-50e. |
| 30. | Kovacic JC, Mercader N, Torres M, Boehm M, Fuster V.Epithelial-to-mesenchymal and endothelial-to-mesenchymal transition: from cardiovascular development to disease. Circulation 2012; 125: 1795-808. |
| 31. |
Galiano RD, Tepper OM, Pelo CR, et al. Topical vascular endothelial growth factor accelerates diabetic wound healing through increased angiogenesis and by mobilizing and recruiting bone marrow-derived cells. Am J Pathol 2004; 164: 1935-47.
DOI PMID |
| 32. |
Oliver G. Lymphatic vasculature development. Nat Rev Immunol 2004; 4: 35-45.
DOI PMID |
| 33. |
Renò F, Sabbatini M. Breaking a vicious circle: Lymphangiogenesis as a new therapeutic target in wound healing. Biomedicines 2023; 11: 656.
DOI URL |
| 34. | Seoane PI, Lee B, Hoyle C, et al. The NLRP3-inflammasome as a sensor of organelle dysfunction. J Cell Biol 2020; 219: e202006194. |
| 35. |
Li W, Cao T, Luo C, et al. Crosstalk between ER stress, NLRP 3 inflammasome, and inflammation. Appl Microbiol Biotechnol 2020; 104: 6129-40.
DOI |
| 36. |
Mangan MSJ, Olhava EJ, Roush WR, Seidel HM, Glick GD, Latz E. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat Rev Drug Discov 2018; 17: 688.
DOI PMID |
| 37. |
Wang L, Hauenstein AV. The NLRP3 inflammasome: Mechanism of action, role in disease and therapies. Mol Aspects Med 2020; 76: 100889.
DOI URL |
| 38. |
Wani K, AlHarthi H, Alghamdi A, Sabico S, Al-Daghri NM. Role of NLRP3 inflammasome activation in obesity-mediated metabolic disorders. Int J Environ Res Public Health 2021; 18: 511.
DOI URL |
| 39. | Lin J, Chen M, Liu D, et al. Exogenous hydrogen sulfide protects human umbilical vein endothelial cells against high glucose-induced injury by inhibiting the necroptosis pathway. Int J Mol Med 2018; 41: 1477-86. |
| 40. |
Dieterich LC, Seidel CD, Detmar M. Lymphatic vessels: new targets for the treatment of inflammatory diseases. Angiogenesis 2014; 17: 359-71.
DOI PMID |
| [1] | XU Pingyuan, ZHU Heng, ZHOU Ruonan, WEI Yaping, ZHU Ziwei, XU Fangyuan, XIANG Yingying, CAO Yue, SHEN Lixuan, WANG Ziwei, XUE Yingying, YU Xizhong, FANG Penghua, SHANG Wenbin. Beneficial effects of Huanglian Jiedu decoction (黄连解毒汤 ) on metabolic syndrome: a prospective randomized controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 411-417. |
| [2] | LI Hongzheng, LIN Guosheng, PENG Yuxuan, Churov Alexey Viktorovich, YANG Wenwen, WANG Jie, LU Jieming, LIAO Feifei, YU Ruotong, WEI Yue, ZHAO Zhiru, LU Aimei, LI Peng, SHEN Aling, LONG Linzi, QU Hua, FU Changgeng. Candidate biomarker identification for blood stasis syndrome among coronary artery disease patients using the Olink proteomics platform [J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 490-500. |
| [3] | LI Dongqi, WANG Tongxing, WANG Zixuan, YAN Yihui, LI Jie, GU Jiaojiao, LI Cuiru, WANG Aili, SUN Lingling, MENG Yongjie, ZHANG Zeyu, HOU Yunlong, GAO Huailin. Improving glucose tolerance in obese rats: the role of Jinlida granules (津力达颗粒 ) in gut microbiota modulation [J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 62-72. |
| [4] | WEI Mumu, YAN Xiaoyu, ZHANG Yujing, ZHANG Xinxin, YAN Yongbin. Soufeng Yuchuan formula (搜风愈喘方) alleviates asthma airway inflammation and suppresses the progression of asthma by inhibiting ferroptosis in airway epithelial cells [J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 73-84. |
| [5] | ZOU Xiaoyun, WEI Minmin, JIA Shouning, QI Yongfu, LI Hailing, LIU Yan, SHI Xiujuan, LI Junru. Weimishu prescription (胃糜舒方) alleviates liver-stomach disharmony and chronic gastritis by reducing inflammation and regulating gastric stem cell differentiation [J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 95-109. |
| [6] | LEI Xiaochun, LIU Cuizhen, LIN Xiujuan, XIE Xiangyu, KE Wei, QIU Zhenwen, TANG Hongmei, HUANG Yushen, ZHANG Lijuan, HUANG Baoyuan, WAN Xin, LI Detang. Effect of improved Yupingfeng powder prescription (玉屏风散加味) on interleukin-33/suppression of tumorigenicity 2 pathway in mice with ovalbumins-induced allergic rhinitis [J]. Journal of Traditional Chinese Medicine, 2025, 45(6): 1215-1227. |
| [7] | LI Keyao, SHU Ye, CHANG Jing, TANG Jianping, ZHANG Litao, WEI Zhu. Psoriasis intervention by Huai’er (Trametes): unveiling novel targets via network pharmacology [J]. Journal of Traditional Chinese Medicine, 2025, 45(6): 1317-1329. |
| [8] | WANG Wei, LONG Qi, FU Ling, WU Haiqiao. Jianpi Yifei Tongluo recipe (健脾益肺通络方剂) attenuates inflammation by promoting the expression of interferon regulatory factor 4 in the rat model of chronic obstructive pulmonary disease [J]. Journal of Traditional Chinese Medicine, 2025, 45(5): 1048-1058. |
| [9] | LI Weijia, LU Jing, MA Chao, LIU Mengmeng, PEI Ke, CHEN Hongyan, LIN Zhe, LYU Guangfu. Hamayou (Oviductus Ranae) protein hydrolysate ameliorates depression by regulating the mitogen-activated protein kinase pathway [J]. Journal of Traditional Chinese Medicine, 2025, 45(3): 493-507. |
| [10] | MIN Yu, ZHENG Meifeng, SUN Ju, PENG Zetong, CAO Zhixian, HUANG Xiaohua. Systematic acupuncture explains acupuncture at Baihui (GV20) and Fengchi (GB20) targeting the inflammatory response to regulate migraine [J]. Journal of Traditional Chinese Medicine, 2025, 45(3): 610-617. |
| [11] | YUAN Jiayao, WU Suhui, MENG Yufan, LI Hanbing, LI Genlin, XU Jiangyan. Yishen Tongluo formula (益肾通络方) ameliorates kidney injury via modulating inflammation and apoptosis in streptozotocin-induced diabetic kidney disease mice [J]. Journal of Traditional Chinese Medicine, 2025, 45(2): 254-265. |
| [12] | HUANG Jiaen, LUO Qing, DONG Gengting, PENG Weiwen, HE Jianhong, DAI Weibo. Xiahuo Pingwei San (夏藿平胃散) attenuated intestinal inflammation in dextran sulfate sodium-induced ulcerative colitis mice through inhibiting the receptor for advanced glycation end-products signaling pathway [J]. Journal of Traditional Chinese Medicine, 2025, 45(2): 311-325. |
| [13] | TIAN Yuan, BU He, WANG Tieshan, YANG Dongliang, ZHANG Wei, LIU Tong, ZHANG Li, HUO Zejun. Efficacy of electro-acupuncture at “Weizhong” (BL40) on macrophage polarization in rats with injured lumbar multifidus [J]. Journal of Traditional Chinese Medicine, 2025, 45(2): 335-347. |
| [14] | AN Yuanyuan, LIU Wang, LI Yanjie, WANG Yanchun, REN Xiaobin, HE Hongbing. Effect and mechanism of Sanqi (Radix Notoginseng) in treating periodontitis [J]. Journal of Traditional Chinese Medicine, 2025, 45(1): 66-75. |
| [15] | HANG Wenlu, WANG Lin, BO Yun, ZUO Shurun, WANG Songquan, LI Haiquan, BU Chunlu, ZHAO Jie, ZHOU Xianmei. Bufei Huoxue capsule (补肺活血胶囊) alleviates silicosis by inhibiting the activation of nucleotide-like receptor containing pyrin domain 3 inflammasome and macrophages polarization based on network pharmacology [J]. Journal of Traditional Chinese Medicine, 2024, 44(6): 1236-1246. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

