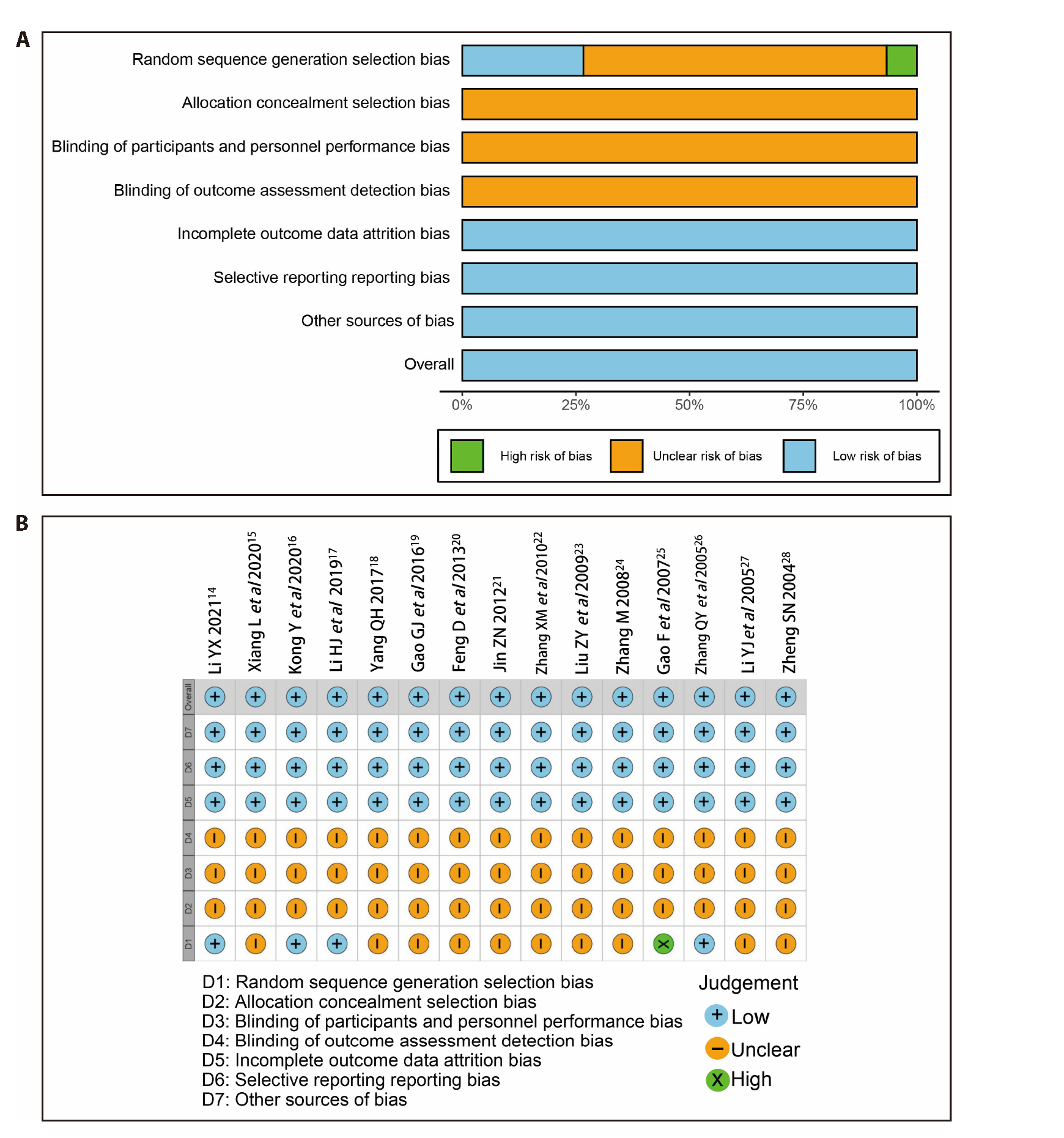
Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (5): 851-859.DOI: 10.19852/j.cnki.jtcm.20230713.001
• Meta Analysis • Previous Articles Next Articles
Effectiveness and safety of Pingxiao capsule (平消胶囊) as adjuvant therapy in treatment of breast cancer: a systematic review and Meta-analysis
YANG Yuqing1, CHEN Yuhuan1, LI Chunxiao2,3,4( ), LING Xiao2,3,4, WANG Panpan2,3,4, GUO Jing1, ZHANG Yingying1
), LING Xiao2,3,4, WANG Panpan2,3,4, GUO Jing1, ZHANG Yingying1
- 1 Henan University of Chinese Medicine, Zhengzhou 450046, China
2 Department of Pharmacy, the First Affiliated Hospital of Henan University of Chinese Medicine
3 Henan Province Engineering Research Center for Clinical Application, Evaluation and Transformation of Traditional Chinese Medicine
4 Henan Provincial Key Laboratory for Clinical Pharmacy of Traditional Chinese Medicine, Zhengzhou 450000, China
-
Received:2022-09-16Accepted:2022-12-22Online:2023-10-15Published:2023-07-13 -
Contact:LI Chunxiao, Department of Pharmacy, the First Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou 450000, China.lichunxiao@126.com . Telephone: +86-15890180908 -
Supported by:Henan Province Science and Technology Research Project: Construction of COS Optimization Method for Traditional Chinese Medicine Injection in Treatment of Acute Stroke Based on MMR(232102310476);Henan Province Top-notch Talent Training Project of Traditional Chinese Medicine: Construction of A Comprehensive Evaluation Method for Post-Marketing Clinical Safety of Traditional Chinese Medicine Injections Based on MMR(2022ZYBJ05);Henan Province Traditional Chinese Medicine Scientific Research Project: Optimization of Core Indicators for Comprehensive Evaluation of Clinical Safety of Traditional Chinese Medicine Injections Based on Analytic Hierarchy Process-Fuzzy Comprehensive Evaluation Method-Taking Shuganning As An Example(2022ZY1049)
Cite this article
YANG Yuqing, CHEN Yuhuan, LI Chunxiao, LING Xiao, WANG Panpan, GUO Jing, ZHANG Yingying. Effectiveness and safety of Pingxiao capsule (平消胶囊) as adjuvant therapy in treatment of breast cancer: a systematic review and Meta-analysis[J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 851-859.
share this article

Figure 1 Flowchart of the literature selection process CNKI: China National Knowledge Infrastructure Database; Wanfang: Wanfang database; VIP: China Science and Technology Journal Database; CBM: Chinese Biomedical Literature Database; EI: Engineering Index.
| Study | Sample size (E/C) | Age (years, E/C) | Intervention | Duration (weeks) | Outcome | |
|---|---|---|---|---|---|---|
| E | C | |||||
| Li YX 2021 | 40/40 | 50.81/50.72 | Pingxiao+capecitabine | Capecitabine | 6 | ①②⑥ |
| Xiang L et al 2020 | 36/40 | 54.5/53.6 | Pingxiao+capecitabine | Capecitabine | 6 | ①②⑥ |
| Kong Y et al 2020 | 59/59 | 52.7/51.9 | Pingxiao+CAF | CAF | 12-18 | ①②④⑥ |
| Li HJ et al 2019 | 49/49 | 51.08/52.13 | Pingxiao+capecitabine | Capecitabine | - | ①②④ |
| Yang QH 2017 | 42/42 | 54.87/55.42 | Pingxiao+FEC/CAF | FEC/CAF | 12-18 | ⑤ |
| Gao GJ et al 2016 | 73/73 | 54.71/52.46 | Pingxiao+FEC | FEC | - | ④⑥ |
| Feng D et al 2013 | 33/33 | 45.6/45.6 | Pingxiao+tamoxifen | Tamoxifen | ①②⑥ | |
| Jin ZN 2012 | 31/29 | 56/56 | Pingxiao+FEC/CAF | FEC/CAF | 12-18 | ④⑤ |
| Zhang XM et al 2010 | 44/41 | 67.2/66.8 | Pingxiao+letrozol | Letrozol | - | ①②③⑥ |
| Liu ZY et al 2009 | 28/26 | 54/54 | Pingxiao+FEC | FEC | - | ①③⑥ |
| Zhang M 2008 | 54/54 | 46.5/46.5 | Pingxiao+tamoxifen | Tamoxifen | 24 | ①② |
| Gao F et al 2007 | 50/46 | 48/43 | Pingxiao+CAF | CAF | 12 | ⑤⑥ |
| Zhang QY et al 2005 | 27/27 | 50/52 | Pingxiao+FEC+TE+P | FEC+TE+ P | 18 | ①②③⑥ |
| Li YJ et al 2005 | 30/30 | 43/43 | Pingxiao+CAF | CAF | 8 | ①②⑥ |
| Zheng SN 2004 | 45/42 | 47.8/47.5 | Pingxiao+CAF | CAF | 15 | ⑤⑥ |
Table 1 characteristics of included studies
| Study | Sample size (E/C) | Age (years, E/C) | Intervention | Duration (weeks) | Outcome | |
|---|---|---|---|---|---|---|
| E | C | |||||
| Li YX 2021 | 40/40 | 50.81/50.72 | Pingxiao+capecitabine | Capecitabine | 6 | ①②⑥ |
| Xiang L et al 2020 | 36/40 | 54.5/53.6 | Pingxiao+capecitabine | Capecitabine | 6 | ①②⑥ |
| Kong Y et al 2020 | 59/59 | 52.7/51.9 | Pingxiao+CAF | CAF | 12-18 | ①②④⑥ |
| Li HJ et al 2019 | 49/49 | 51.08/52.13 | Pingxiao+capecitabine | Capecitabine | - | ①②④ |
| Yang QH 2017 | 42/42 | 54.87/55.42 | Pingxiao+FEC/CAF | FEC/CAF | 12-18 | ⑤ |
| Gao GJ et al 2016 | 73/73 | 54.71/52.46 | Pingxiao+FEC | FEC | - | ④⑥ |
| Feng D et al 2013 | 33/33 | 45.6/45.6 | Pingxiao+tamoxifen | Tamoxifen | ①②⑥ | |
| Jin ZN 2012 | 31/29 | 56/56 | Pingxiao+FEC/CAF | FEC/CAF | 12-18 | ④⑤ |
| Zhang XM et al 2010 | 44/41 | 67.2/66.8 | Pingxiao+letrozol | Letrozol | - | ①②③⑥ |
| Liu ZY et al 2009 | 28/26 | 54/54 | Pingxiao+FEC | FEC | - | ①③⑥ |
| Zhang M 2008 | 54/54 | 46.5/46.5 | Pingxiao+tamoxifen | Tamoxifen | 24 | ①② |
| Gao F et al 2007 | 50/46 | 48/43 | Pingxiao+CAF | CAF | 12 | ⑤⑥ |
| Zhang QY et al 2005 | 27/27 | 50/52 | Pingxiao+FEC+TE+P | FEC+TE+ P | 18 | ①②③⑥ |
| Li YJ et al 2005 | 30/30 | 43/43 | Pingxiao+CAF | CAF | 8 | ①②⑥ |
| Zheng SN 2004 | 45/42 | 47.8/47.5 | Pingxiao+CAF | CAF | 15 | ⑤⑥ |

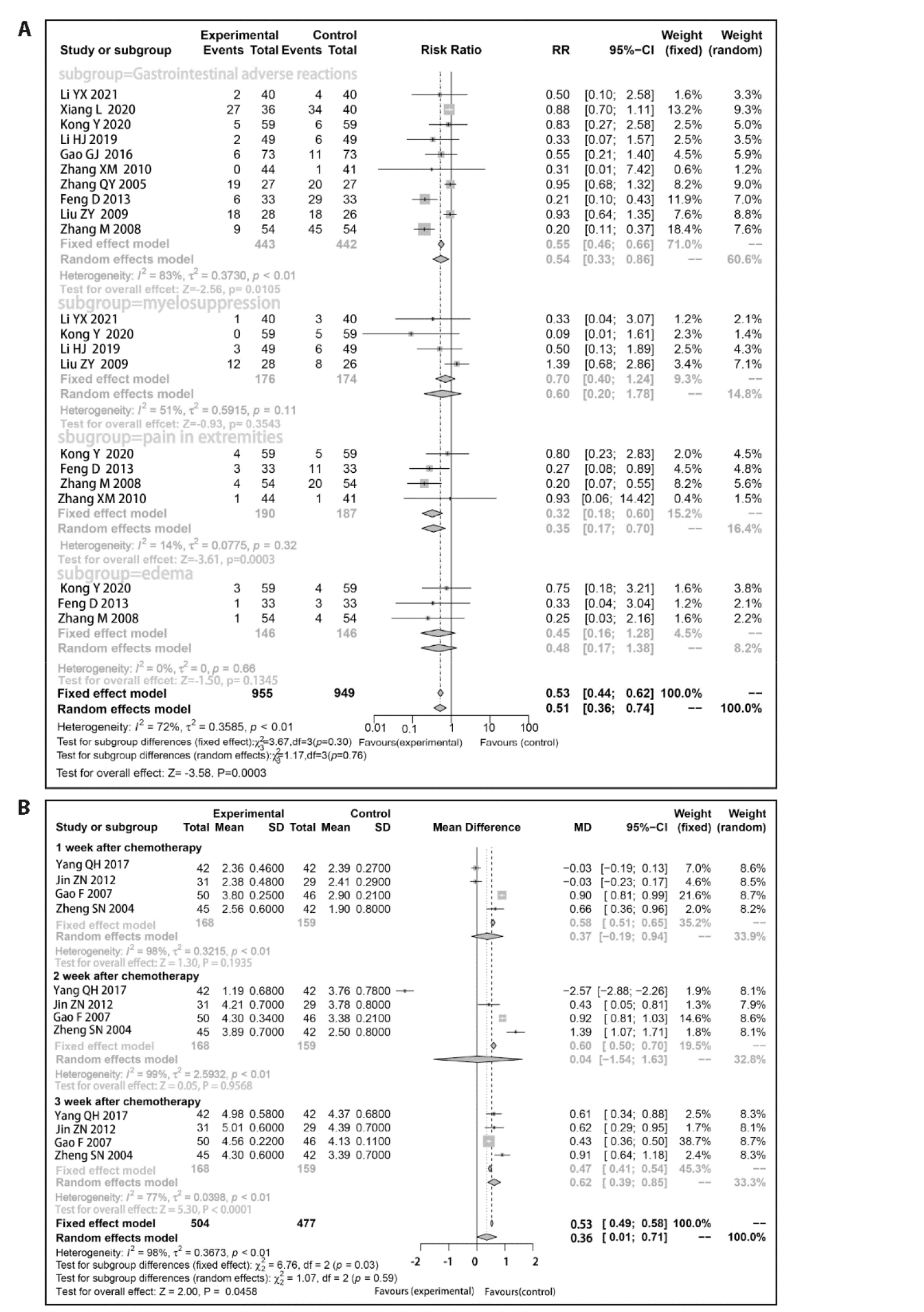
Figure 3 Forest plot of outcomes of the indicators A: forest plot of objective response rate; B: forest plot of disease control rate; C: forest plot of quality of life.
| Outcome | Number of studies | Sample size | SMD | 95% CI | I 2 value (%) | P value | |
|---|---|---|---|---|---|---|---|
| E | C | ||||||
| CD3+ | 3 | 139 | 137 | 1.42 | 0.76, 2.09 | 83% | < 0.001 |
| CD4+ | 4 | 212 | 210 | 1.18 | 0.70, 1.66 | 80% | < 0.001 |
Table 2 Meta-analysis of CD3+, CD4+
| Outcome | Number of studies | Sample size | SMD | 95% CI | I 2 value (%) | P value | |
|---|---|---|---|---|---|---|---|
| E | C | ||||||
| CD3+ | 3 | 139 | 137 | 1.42 | 0.76, 2.09 | 83% | < 0.001 |
| CD4+ | 4 | 212 | 210 | 1.18 | 0.70, 1.66 | 80% | < 0.001 |
| Study | CD8+ | CD4+/CD8+ | |||||
|---|---|---|---|---|---|---|---|
| SMD | 95% CI | P value | SMD | 95% CI | P value | ||
| Kong Y et al 2020 | -1.69 | -2.11, -1.27 | < 0.000 | 0.65 | 0.28, 1.02 | 0.0006 | |
| Gao GJ et al 2016 | 2.22 | 1.80, 2.63 | 0.000 | 2.14 | 1.64, 2.64 | < 0.000 | |
| Jin ZN 2012 | -1.68 | -2.27, -1.10 | < 0.000 | 0.82 | 0.29, 1.35 | 0.002 | |
Table 3 CD8+, CD4+/CD8+: Pingxiao capsule + chemotherapy vs Chemotherapy
| Study | CD8+ | CD4+/CD8+ | |||||
|---|---|---|---|---|---|---|---|
| SMD | 95% CI | P value | SMD | 95% CI | P value | ||
| Kong Y et al 2020 | -1.69 | -2.11, -1.27 | < 0.000 | 0.65 | 0.28, 1.02 | 0.0006 | |
| Gao GJ et al 2016 | 2.22 | 1.80, 2.63 | 0.000 | 2.14 | 1.64, 2.64 | < 0.000 | |
| Jin ZN 2012 | -1.68 | -2.27, -1.10 | < 0.000 | 0.82 | 0.29, 1.35 | 0.002 | |
| 1. |
Kolak A, Kamińska M, Sygit K, et al. Primary and secondary prevention of breast cancer. Ann Agric Environ Med 2017; 24: 549-53.
DOI PMID |
| 2. |
Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021; 71: 209-49.
DOI URL |
| 3. | Hao J, Chen WQ, Li N, et al. China guide line for the screening and early detection of female breast cancer (2021, Beijing). Zhong Guo Zhong Liu 2021; 30: 161-91. |
| 4. | Ferlay JEM, Lam F, Colombet M, et al. Global cancer observatory: cancer today. lyon, France: international agency for research on cancer, 2021-02-26, cited 2022-07-26. Available from URL: https://gco.iarc.fr/today. |
| 5. | Chinese anti-cancer association breast cancer professional committee. Chinese anti-cancer association breast cancer diagnosis and treatment guidelines and specifications (2021 Edition). Zhong Guo Ai Zheng Za Zhi 2021; 31: 954-1040. |
| 6. | Chen XD, Zhang FY, Chen Q, He ZM. Meta-analysis of the effect of capecitabine metronomic chemotherapy in advanced breast cancer. Shaoyang Xue Yuan Xue Bao (Natural Science Edition) 2021; 18: 32-8. |
| 7. | Zhou SS. Combinational efficacy of trastuzumab and capecitabine in patients with early HER2 positive breast cancer. Hunan Shi Fan Da Xue Xue Bao (Medical Sciences) 2021; 18: 127-30. |
| 8. | Liu QX, Xiang Q, Zhang Z, et al. Influence factors of chemotherapy toxicities in breast cancer patients. Zhong Guo Lin Chuang Yao Li Xue Za Zhi 2018; 34: 475-78. |
| 9. | Zhang KL, Si WT, Shao HM. Application of Traditional Chinese Medicine treatment in the adverse reactions of tumor chemotherapy. Zhong Guo Zhong Yi Ji Zheng 2015; 24: 1212-14. |
| 10. | Li X, Liu YF, Fan YY, Pei XH. Benefits of Chinese medicine intervention on the survival of breast cancer: a Meta-analysis. Zhong Yi Yao Lin Chuang ZA Zhi 2021; 33: 1922-26. |
| 11. | He XA, Gao NN, Ma X, Li L. Effects of Traditional Chinese Medicine on quality of life and toxicity of patients with chemoradiotherapy after breast cancer surgery. Zhong Liu Yao Xue 2016; 6: 349-55. |
| 12. | Zhu FX. Analysis of the effect of Pingxiao capsule on the treatment of patients with uterine fibroids and mammary gland hyperplasia. Dang Dai Yi Yao Lun Cong 2015; 13: 187-88. |
| 13. | Pu MJ, Du JZ, Wang XY, et al. Effect and mechanism of Pingxiao capsule containing-serum combined with paclitaxel on breast cancer cells. Guangxi Yi Ke Da Xue Xue Bao 2021; 38: 1717-22. |
| 14. | Li YX. Clinical observation of the combination of Pingxiao capsule and capecitabine in the treatment of advanced breast cancer. Zhong Hua Yang Sheng Bao Jian 2021; 39: 7-10. |
| 15. | Xiang L, Zhou JB, Qian HY, et al. The efficacy and safety of Pingxiao capsule combined with capecitabine in patients with advanced breast cancer. Zhong Guo Lin Chuag Yan Jiu 2020; 12: 81-3. |
| 16. | Kong Y, Ying JK, Yang C. Observation on the effect of CAF regimen combined with Pingxiao capsule in the treatment of breast cancer. Xian Dai Shi Yong Yi Xue 2020; 32: 492-93. |
| 17. | Li HJ, Chen GH, Li WL, et al. Clinical study on Pingxiao capsule combined with capecitabine in treatment of advanced breast cancer. Xian Dai Yao Wu Yu Lin Chuang 2019; 34: 2151-55. |
| 18. | Yang HQ. Study on Clinical efficacy of Pingxiao capsule in treating breast cancer. Gansu Ke Ji 2017; 33: 93-4. |
| 19. | Gao GJ, Yang WM. Pingxiao capsule alleviates the adverse reactions of postoperative chemotherapy for breast cancer and its effect on immune function. Yin Shi Bao Jian 2016; 3: 78. |
| 20. | Feng D, Han B, Liu F. Analysis of the effect of Pingxiao capsule combined with tamoxifen in the treatment of breast cancer in the later stage. Zhong Guo Bao Jian Ying Yang 2013; 23: 6824. |
| 21. | Jin ZN. Clinical study on efficacy of CAF chemotherapy combining with Pingxiao capsule on patients with breast cancer after operation. Liaoning Zhong Yi Za Zhi 2012; 39: 98-9. |
| 22. | Zhang XM, Gao Y, Li S, et al. Clinical observation of Pingxiao capsule combined with letrozole in postmenopausal patients with advanced breast cancer. Xian Dai Zhong Liu Yi Xue 2010; 18: 1749-50. |
| 23. | Liu ZY, Wu YJ. Clinical observation of Pingxiao capsule combined with chemotherapy in the treatment of advanced breast cancer. Zhong Guo Zhong Yi Yao Xian Dai Yuan Cheng Jiao Yu Za Zhi 2009; 7: 104-5. |
| 24. | Zhang M. The effect of Pingxiao capsule combined with tamoxifen in the treatment of breast cancer in late stage. Xian Dai Zhong Liu Yi Xue 2008; 10: 1785. |
| 25. | Gao F, Song R. Clinical effect of Pingxiao capsule in adjuvant effect of chemotherapy in 50 cases of breast cancer. Zhong Guo Zhong Liu Lin Chuang 2007; 34: 887-88. |
| 26. | Zhang QY, Zhao WH, Lai YJ, Kang XM. Effect on late-stage mammary cancer treated by endocrinotherapy or chemotherapy combined with Pingxiao capsule. Zhong Guo Zhong Xi Yi Jie He Za Zhi 2005; 25: 1074-76. |
| 27. | Li YJ, Li ZG, Wu T. Observation of curative effect of Pingxiao capsule combined with chemotherapy before breast cancer surgery. Xian Dai Zhong Liu Yi Xue 2005; 13: 260-62. |
| 28. | Zheng SN. Effect of Pingxiao capsule on synergistic and detoxification of postoperative chemotherapy for breast cancer. Xian Dai Zhong Liu Yi Xue 2004; 123: 248. |
| 29. | Zhang Y. Chinese medicine treatment of breast cancer. Guang Ming Zhong Yi 2009; 24: 178-81. |
| 30. | Zhang YJ, Han B, Huang HR, et al. Effect of Xiaozhong Sanjie decoction combined with Pingxiao capsule on inflammatory factors and immune factors in patients with non-lactation mastitis. Yi Xue Zong Shu 2020; 26: 1856-59. |
| 31. | Zhao ZP, Zhang YM, Guo LM, Cheng SX, Miao H. Clinical study of Pingxiao capsule combined with chemotherapy in the treatment of advanced cancer. Xian Dai Zhong Liu Yi Xue 1996; 3: 142-4. |
| 32. | Zhao XS, Jiang YX, Xu YD. Clinical study of Pingxiao capsule combined with gemcitabine and cisplatin in the treatment of advanced cholangiocarinoma. Zhong Hua Ji Ceng Yi Xue Za Zhi 2021; 28: 178-83. |
| 33. | Li HJ, Ge P, Jing RJ, et al. Clinical study on Pingxiao capsule combined with oxaliplatin in treatment of esophageal cancer. Xian Dai Yao Wu Yu Lin Chuang 2019; 34: 3421-25. |
| 34. |
He M, Tan B, Vasan K, et al. S1RT1 and AMPK pathways are essential for the proliferation and survival of primary effusion lymphoma cells. J Pathol 2017; 242: 309-21.
DOI URL |
| 35. |
Elmasry OS, Brown BL, Dobson PRM. AMPK activation of apoptotic markers in human breast cancer cell lines with different p53 backgrounds: MCF- 7, MDA-MB-231 and T47D cells. Asian Pac J Cancer Prev 2019; 20: 3763-70.
DOI URL |
| 36. | Wang J, Yue ZG, Dong MZ, et al. Study on anti-tumor effect and mechanism of Pingxiao capsule. Zhong Hua Zhong Xi Yi Jie He Za Zhi 2017; 32: 4658-63. |
| 37. | Zhu QG. Molecular biological mechanism of Pingxiao capsule against lung cancer. Nan Fang Yi Ke Da Xue Xue Bao 2008; 11: 2069-71. |
| 38. | Han XR, Yang JM, Luo YZ, et al. Chemopreventive effect of Pingxiao capsule on breast cancer in rats and its mechanism. Zhong Hua Ru Xian Bing Za Zhi (electronic edition) 2013; 7: 329-33. |
| 39. | Cai MC, Zhang HQ, Xie W, et al. Mechanism of Pingxiao capsule on colon cancer based on network pharmacology and molecular docking. Yao Xue Fu Wu Yu Yan Jiu 2021; 21: 321-26+64. |
| 40. | Hang CY, Chen JQ, Shen Y, et al. Study on the effect of Pingxiao capsule on multidrug resistant gene P-gp, TOPOⅡ and GST-π for breast cancer. Ling Nan Xian Dai Lin Chuang Wai Ke 2011; 11: 38-40. |
| [1] | QIN Xiaoyu, WANG Chunai, XUE Jianjun, ZHANG Jie, LU Xiaoting, DING Shengshuang, GE Long, WANG Minzhen. Efficacy of electroacupuncture on myocardial protection and postoperative rehabilitation in patients undergoing cardiac surgery with cardiopulmonary bypass: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 1-15. |
| [2] | DAI Xiaoling, ZHANG Anming, LIN Hui, SHI Bei, REN Yi, WEN Hongzhu, FEI Xiaoyan, LIN Jiang. Qingchang suppositry (清肠栓) induced remission in patients with mild-to-moderate ulcerative proctitis: a multicenter, prospective, randomized, parallel-controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 156-162. |
| [3] | LIU Tingting, LIU Tongou, LIU Mingfu. Effectiveness and safety of acupuncture in treatment of pregnancy-related symptoms: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 16-26. |
| [4] | WANG Yichen, WU Shiyi, WANG Zhengyan, CHANG Wenling, XIE Zhihao, TANG Xing, ZHAO Songmei, ZHOU Jing, CHEN Zehong, WANG Chao, YANG Chunxia. Efficacy of Zhumian Tang formula granules (助眠汤配方颗粒) combined with eszopiclone for the treatment of poor sleep quality: a multi-center, randomized controlled, superiority trial [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 163-171. |
| [5] | YANG Yi, YE Huijun, ZHENG Huiling, JIN Lihua. Clinical observation on 90 cases of primary dysmenorrhea treated by buccal acupuncture therapy: a randomized controlled study [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 172-181. |
| [6] | WANG Jiabao, ZHANG Lishuang, NIU Baihan, YU Yajun, YANG Fengwen, MIAO Lin, CHAI Lijuan, DING Xinya, SUN Yingjie, WANG Yujing, WANG Lin, ZHANG Han, WANG Yi, LI Lin. Efficacy and safety of Weichang’ an pill (胃肠安丸) combined with Western Medicine on gastrointestinal diseases: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1057-1067. |
| [7] | FAN Rong, HE Haoyu, TANG Tao, CUI Hanjin. Long-term effects of Qingfei Paidu decoction (清肺排毒汤) in patients with coronavirus disease 2019 acute pneumonia after treatment: a protocol for systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1068-1071. |
| [8] | DAI Zeqi, LIAO Xing, GUAN Yueyue, ZENG Zixiu, TANG Jun, HU Jing. Bloodletting puncture in the treatment of acute ischemic stroke: protocol for a mixed-method study of a multi-center randomized controlled trial and focus group [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1259-1267. |
| [9] | XU Yani, ZHANG Yutong, HE Weile, DAI Linglin, TANG Ding, WANG Jialing, ZHANG Xufen, CHEN Qin, CHEN Lifang, WANG Zhanglian, ZHAN Mingjie. Efficiency and safety of acupuncture for women with premature ovarian insufficiency: study protocol for a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1268-1274. |
| [10] | XU Xiangru, ZHOU Yi, CHEN Gang, LEI Ming, ZHANG Wen, WU Xinxin, PU Yuting, CHEN Caiyu, SUN Yuting, ZHOU Shuang, FANG Bangjiang. Clinical efficacy of Buzhong Yiqi decoction (补中益气汤) in the treatment of hospital-acquired pneumonia with multi-drug resistant bacteria: a prospective, randomized, multicenter controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1010-1018. |
| [11] | ZHAO Ming, LUO Yimiao, WANG Huichan, CAO Yu, MA Lina, PEI Hui, LI Hao. Guilingji capsule (龟龄集胶囊) for Alzheimer's disease: secondary analysis of a randomized non-inferiority controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1019-1025. |
| [12] | ZHANG Meizhen, HAO Xiaohui, TANG Yiting, CHEN Yupeng, HE Puyu, ZHAO Liming, PANG Bing, NI Qing. Efficacy and safety of Buyang Huanwu decoction (补阳还五汤) for diabetic peripheral neuropathy: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 841-850. |
| [13] | ZHAO HuiYan, JUN Purumea, LEE Chaewon, HAN Chang-Hyun. Acupoint catgut embedding for simple obesity in animal studies: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 860-867. |
| [14] | TANG Wenjuan, HU Yu, ZHU Man, DONG Mingzhi, LIU Tieming, SARWAR Ammar, ZHAN Yingzhuan, ZHANG Yanmin. Pingxiao capsule (平消胶囊) inhibits lung metastasis of triple-negative breast cancer and sensitizes breast cancer to radiotherapy [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 897-905. |
| [15] | SUN Wu, ZHAO Yuwei, LIAO Liang, ZHAO Zhonghui, CHEN Shiqi, YAN Xiaoling, WANG Xueyao, CHAO Guojun, ZHOU Jian. Effectiveness and safety of Xuebijing injection for patients with coronavirus disease 2019: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 631-639. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||