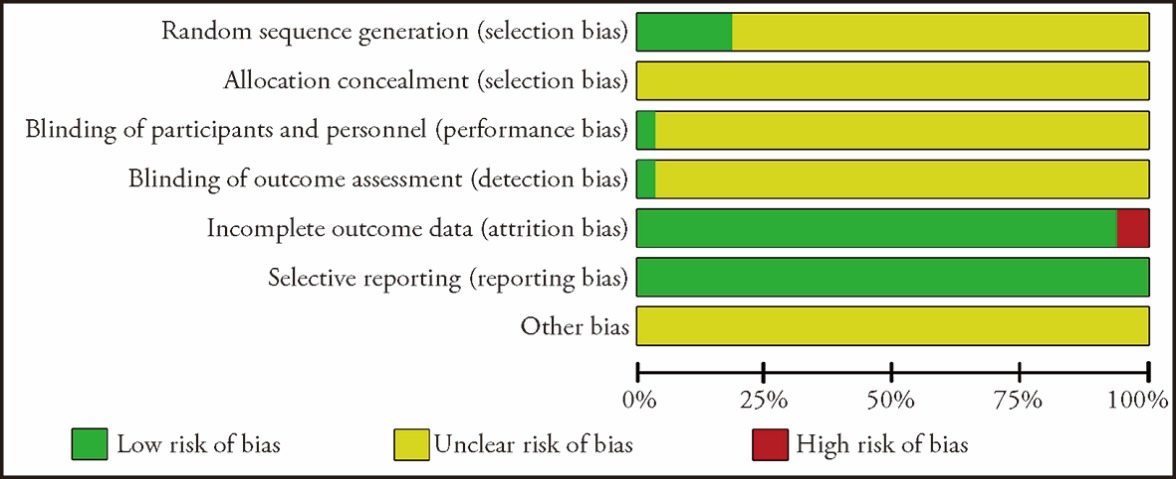
Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (6): 1057-1067.DOI: 10.19852/j.cnki.jtcm.20230814.003
• Meta-Analyses • Previous Articles Next Articles
Efficacy and safety of Weichang’ an pill (胃肠安丸) combined with Western Medicine on gastrointestinal diseases: a systematic review and Meta-analysis
WANG Jiabao1, ZHANG Lishuang2, NIU Baihan3, YU Yajun1, YANG Fengwen1, MIAO Lin1, CHAI Lijuan1, DING Xinya1, SUN Yingjie4, WANG Yujing4, WANG Lin4, ZHANG Han1, WANG Yi1( ), LI Lin1(
), LI Lin1( )
)
- 1 Institute of Traditional Chinese Medicine, Key Laboratory of Pharmacology of Traditional Chinese Medical Formulae, State Key Laboratory of Component-based Chinese Medicine, Tianjin University of Traditional Chinese Medicine, Tianjin 301617, China
2 Department of Cardiology, Tianjin Binhai New Area Hospital of Traditional Chinese Medicine, Tianjin 300451, China
3 Scientific Research Department, the First Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou 450000, China
4 Tianjin Zhongxin Pharmaceutical Group Co., Ltd., Lerentang Pharmaceutical Factory, Tianjin 413204, China
-
Received:2022-08-16Accepted:2022-11-23Online:2023-10-25Published:2023-08-14 -
Contact:WANG Yi, Key Laboratory of Pharmacology of Traditional Chinese Medical Formulae, Ministry of Education, Tianjin University of Traditional Chinese Medicine, Tianjin 301617, China. wangyi@tjutcm.edu.cn; LI Lin, Key Laboratory of Pharmacology of Traditional Chinese Medical Formulae, Ministry of Education, Tianjin University of Traditional Chinese Medicine, Tianjin 301617, China. lilintcm@126.com. Telephone: +86-17725301732 -
Supported by:Study on Compound Chinese Medicine(TD13-5053);Research on the Basic Biology of the Wind Drugs Effects of “Dissipate Wind to Dissipate Heat, Dispel Wind to Relieve Pain” via TLR4/NF-κB Signalling Pathway(2018KJ007);Innovation Team and Talents Cultivation Program of National Administration of Traditional Chinese Medicine(ZYYCXTD-C-202009);Research on the Therapeutic Effect and Mechanism of “Wind Dispeling” Herb Vitex Trifolia for Migraine Treatment via CGRP/ERK/NF-κB/NLRP3 Signaling Pathway(82004331);Research on the Effects of Erzhi pills via 11β-HSD1-GC/GR Pathway on Cognitive Function and Its Mechanism of Chronic Mild and Unpredictable Stress Depression Mice(81973792)
Cite this article
WANG Jiabao, ZHANG Lishuang, NIU Baihan, YU Yajun, YANG Fengwen, MIAO Lin, CHAI Lijuan, DING Xinya, SUN Yingjie, WANG Yujing, WANG Lin, ZHANG Han, WANG Yi, LI Lin. Efficacy and safety of Weichang’ an pill (胃肠安丸) combined with Western Medicine on gastrointestinal diseases: a systematic review and Meta-analysis[J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1057-1067.
share this article
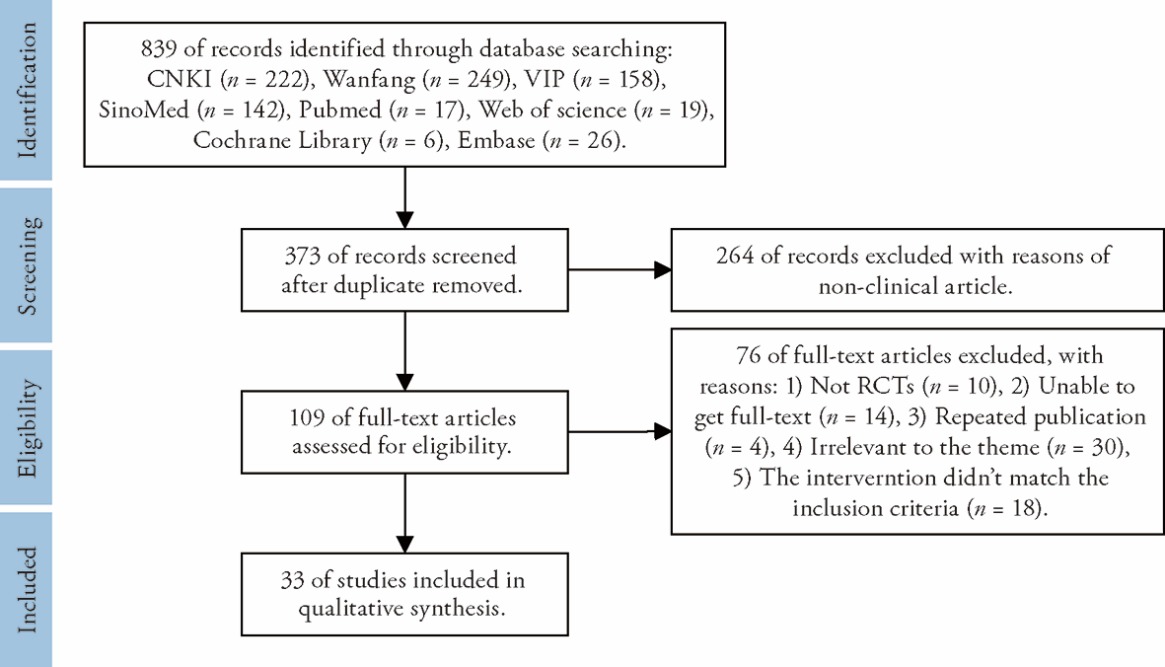
Figure 1 Flow diagram of the study selection CNKI: China National Knowledge Infrastructure Database; VIP: China Science and Technology Journal Database; RCT: randomized controlled trial.
| Disease type | Study | Sample size (T/C) | Age (T/C) | Course of Treatment | Outcome |
|---|---|---|---|---|---|
| AAGR-prevention | Li MF et al 2013 | 55/57 | 3-12 | 2-5 | (1) Comprehensive curative effect, (2) Symptomatic effect, (3) Clinical symptoms score |
| Lu JX et al 2013 | 40/40 | 4.52 (3-9)/4.63 (3-10) | 5 | (1) Gastrointestinal reaction rate, (2) Severity of diarrhea | |
| Wu XL et al 2014 | 100/100 | 1/3-3 | - | (1) Gastrointestinal reaction rate, (2) Duration of Diarrhea, (3) Severity of diarrhea | |
| AAGR-treatment | Ma JY et al 2017 | 51/51 | 3.11±1.75/3.15±1.68 | 7 | (1) Clinical efficacy |
| Pang ZF et al 2017 | 29/29 | 31.29±3.53/32.01±3.49 | 7 | (1) Clinical efficacy, (2) PGR, (3) MOT | |
| Yang LP et al 2016 | 58/50 | - | 6-7 | (1) Clinical efficacy, (2) Duration of diarrhea | |
| AD | Wei JJ et al 2018 | 31/29 | 37.42±11.81 /40.83±16.65 | - | (1) Clinical efficacy, (2) Clinical symptom score |
| FAP | He XY et al 2013 | 40/40 | 8.6 (3-13)/8.7 (3-14) | - | (1) Clinical efficacy |
| FD | Deng HY et al 2019 | 30/30 | 70.1±10.2/71.4±10.2 | 28 | (1) Clinical efficacy, (2) Clinical symptom score, (3) Relapse rate |
| Liu ZQ et al 2017 | 56/56 | 45±4.5/44.5±4.7 | 28 | (1) Clinical efficacy, (2) NO, (3) GAS, (4) MOT, (5) SP, (6) Gastric emptying rate, (7) Clinical symptom score, (8) Relapse rate | |
| Lin XH et al 2018 | 36/36 | 35.89±3.63/36.01±3.69 | 7 | (1) Clinical efficacy, (2) MOT, (3) SS, (4) Clinical symptom score | |
| Kong JF et al 2020 | 46/46 | 5.5±2.0/5.0±1.7 | 14 | (1) Clinical efficacy, (2) Clinical symptom score, | |
| Wei XG et al 2019 | 50/50 | 45.20±12.45 /47.12±10.98 | 14 | (1) Clinical efficacy, (2) MOT, (3) NO, (4) SP, (5) GAS, (6) Gastric emptying rate, (7) Clinical symptom score | |
| Zhou XR et al 2018 | 55/55 | 6.4±1.7/6.6±1.5 | 28 | (1) Clinical efficacy, (2) MOT, (3) NO, (4) SP, (5)5-HT, (6) Clinical symptom score, (7) Bradycardia rate, (8) Electrogastrogram | |
| Hp+ INF | Li Z et al 2015 | 49/49 | 8.0±1.9/7.5±1.3 | 14 | (1) Hp eradication rate, (2) Duration of abdominal pain, (3) Duration of vomiting, (4) Duration of inappetence |
| Xu JJ et al 2017 | 48/45 | 10.7±2.5/9.8±2.6 | 7 | (1) Clinical efficacy, (2) Clinical symptom efficacy | |
| IBS | Liu B et al 2017 | 63/65 | 31.38±4.02/30.59±4.47 | 28 | (1) Clinical efficacy, (2) SLC-90, (3) QOL (4) PSQI |
| Liu YD et al 2012 | 35/35 | 50.5±3.2/50.2±3.6 | 28 | (1) Clinical efficacy | |
| Ren HS et al 2014 | 36/36 | 18-60 | 28 | (1) Clinical efficacy | |
| Wang CM et al 2014 | 40/40 | 38.9 (15-55) | 14 | (1) Clinical efficacy | |
| Zhao J et al 2017 | 40/40 | 36.4±9.6/36.9±10.1 | 28 | (1) Clinical efficacy, (2) Clinical symptom score | |
| ML | Hao LC et al 2014 | 30/30 | 5.1 (2.5-8)/5.2 (2.8-9) | 7 | (1) Duration of abdominal pain, (2) Duration of vomiting, (3) Duration of lymph nodes disappeared |
| Niu Y et al 2014 | 30/30 | 6.8±0.5/7.2±0.7 | 14 | (1) Clinical efficacy, (2) Clinical symptom efficacy, (3) abdominal colour doppler ultrasound | |
| RVD | Dai WQ et al 2016 | 40/40 | 1.77±0.67/1.75±0.64 | 3 | (1) Clinical efficacy, (2) Duration of abdominal distension, (3) CK, (4) CK-MB, (5) Rotavirus turn to negativity |
| Fan XR et al 2016 | 60/60 | 15.3±3.2/14.9±3.4 | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of vomiting, (4) Duration of fever, (5) IL-10, (6) IL-2 | |
| Gao LY et al 2013 | 100/138 | 3.5±2.5/3.4±2.6 | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of vomiting, (4) Duration of fever | |
| Liu K et al 2016 | 101/105 | 1.8 (0.5-2.8)/2 (0.4-3) | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of vomiting, (4) Duration of fever, (5) Duration of dehydration | |
| Shang WG et al 2020 | 60/60 | 3±1.7/2.7±1.6 | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of vomiting, (4) Duration of fever, (5) CK, (6) CK-MB, (7) Rotavirus turn to negativity | |
| Wu XJ et al 2020 | 30/30 | 1.47±0.27/1.46±0.27 | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of vomiting, (4) Duration of fever | |
| Zhou YL et al 2014 | 80/80 | 1.68±0.34/1.64±0.32 | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of fever, (4) LDH, (5) CK, (6) CK-MB | |
| UC | Chen YY et al 2021 | 40/40 | 37.20±3.48/37.31±3.45 | 90 | (1) Clinical symptom score, (2) T lymphocyte subsets |
| Gao XM et al 2019 | 53/53 | 41.14±6.01/41.53±7.12 | - | (1) Clinical efficacy, (2) Clinical symptom score, (3) Times of diarrhea, (4) IL-1β, (5) IL-6, (6) TNF-α, (7) CRP | |
| Zhao JH et al 2020 | 26/25 | 30.2±3.58/30.58±3.4 | 90 | (1) Clinical efficacy, (2) IL-1β, (3) IL-6, (4) TNF-α, (5) CRP |
Table 1 Basic Characteristics of the included studies
| Disease type | Study | Sample size (T/C) | Age (T/C) | Course of Treatment | Outcome |
|---|---|---|---|---|---|
| AAGR-prevention | Li MF et al 2013 | 55/57 | 3-12 | 2-5 | (1) Comprehensive curative effect, (2) Symptomatic effect, (3) Clinical symptoms score |
| Lu JX et al 2013 | 40/40 | 4.52 (3-9)/4.63 (3-10) | 5 | (1) Gastrointestinal reaction rate, (2) Severity of diarrhea | |
| Wu XL et al 2014 | 100/100 | 1/3-3 | - | (1) Gastrointestinal reaction rate, (2) Duration of Diarrhea, (3) Severity of diarrhea | |
| AAGR-treatment | Ma JY et al 2017 | 51/51 | 3.11±1.75/3.15±1.68 | 7 | (1) Clinical efficacy |
| Pang ZF et al 2017 | 29/29 | 31.29±3.53/32.01±3.49 | 7 | (1) Clinical efficacy, (2) PGR, (3) MOT | |
| Yang LP et al 2016 | 58/50 | - | 6-7 | (1) Clinical efficacy, (2) Duration of diarrhea | |
| AD | Wei JJ et al 2018 | 31/29 | 37.42±11.81 /40.83±16.65 | - | (1) Clinical efficacy, (2) Clinical symptom score |
| FAP | He XY et al 2013 | 40/40 | 8.6 (3-13)/8.7 (3-14) | - | (1) Clinical efficacy |
| FD | Deng HY et al 2019 | 30/30 | 70.1±10.2/71.4±10.2 | 28 | (1) Clinical efficacy, (2) Clinical symptom score, (3) Relapse rate |
| Liu ZQ et al 2017 | 56/56 | 45±4.5/44.5±4.7 | 28 | (1) Clinical efficacy, (2) NO, (3) GAS, (4) MOT, (5) SP, (6) Gastric emptying rate, (7) Clinical symptom score, (8) Relapse rate | |
| Lin XH et al 2018 | 36/36 | 35.89±3.63/36.01±3.69 | 7 | (1) Clinical efficacy, (2) MOT, (3) SS, (4) Clinical symptom score | |
| Kong JF et al 2020 | 46/46 | 5.5±2.0/5.0±1.7 | 14 | (1) Clinical efficacy, (2) Clinical symptom score, | |
| Wei XG et al 2019 | 50/50 | 45.20±12.45 /47.12±10.98 | 14 | (1) Clinical efficacy, (2) MOT, (3) NO, (4) SP, (5) GAS, (6) Gastric emptying rate, (7) Clinical symptom score | |
| Zhou XR et al 2018 | 55/55 | 6.4±1.7/6.6±1.5 | 28 | (1) Clinical efficacy, (2) MOT, (3) NO, (4) SP, (5)5-HT, (6) Clinical symptom score, (7) Bradycardia rate, (8) Electrogastrogram | |
| Hp+ INF | Li Z et al 2015 | 49/49 | 8.0±1.9/7.5±1.3 | 14 | (1) Hp eradication rate, (2) Duration of abdominal pain, (3) Duration of vomiting, (4) Duration of inappetence |
| Xu JJ et al 2017 | 48/45 | 10.7±2.5/9.8±2.6 | 7 | (1) Clinical efficacy, (2) Clinical symptom efficacy | |
| IBS | Liu B et al 2017 | 63/65 | 31.38±4.02/30.59±4.47 | 28 | (1) Clinical efficacy, (2) SLC-90, (3) QOL (4) PSQI |
| Liu YD et al 2012 | 35/35 | 50.5±3.2/50.2±3.6 | 28 | (1) Clinical efficacy | |
| Ren HS et al 2014 | 36/36 | 18-60 | 28 | (1) Clinical efficacy | |
| Wang CM et al 2014 | 40/40 | 38.9 (15-55) | 14 | (1) Clinical efficacy | |
| Zhao J et al 2017 | 40/40 | 36.4±9.6/36.9±10.1 | 28 | (1) Clinical efficacy, (2) Clinical symptom score | |
| ML | Hao LC et al 2014 | 30/30 | 5.1 (2.5-8)/5.2 (2.8-9) | 7 | (1) Duration of abdominal pain, (2) Duration of vomiting, (3) Duration of lymph nodes disappeared |
| Niu Y et al 2014 | 30/30 | 6.8±0.5/7.2±0.7 | 14 | (1) Clinical efficacy, (2) Clinical symptom efficacy, (3) abdominal colour doppler ultrasound | |
| RVD | Dai WQ et al 2016 | 40/40 | 1.77±0.67/1.75±0.64 | 3 | (1) Clinical efficacy, (2) Duration of abdominal distension, (3) CK, (4) CK-MB, (5) Rotavirus turn to negativity |
| Fan XR et al 2016 | 60/60 | 15.3±3.2/14.9±3.4 | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of vomiting, (4) Duration of fever, (5) IL-10, (6) IL-2 | |
| Gao LY et al 2013 | 100/138 | 3.5±2.5/3.4±2.6 | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of vomiting, (4) Duration of fever | |
| Liu K et al 2016 | 101/105 | 1.8 (0.5-2.8)/2 (0.4-3) | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of vomiting, (4) Duration of fever, (5) Duration of dehydration | |
| Shang WG et al 2020 | 60/60 | 3±1.7/2.7±1.6 | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of vomiting, (4) Duration of fever, (5) CK, (6) CK-MB, (7) Rotavirus turn to negativity | |
| Wu XJ et al 2020 | 30/30 | 1.47±0.27/1.46±0.27 | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of vomiting, (4) Duration of fever | |
| Zhou YL et al 2014 | 80/80 | 1.68±0.34/1.64±0.32 | 3 | (1) Clinical efficacy, (2) Duration of diarrhea, (3) Duration of fever, (4) LDH, (5) CK, (6) CK-MB | |
| UC | Chen YY et al 2021 | 40/40 | 37.20±3.48/37.31±3.45 | 90 | (1) Clinical symptom score, (2) T lymphocyte subsets |
| Gao XM et al 2019 | 53/53 | 41.14±6.01/41.53±7.12 | - | (1) Clinical efficacy, (2) Clinical symptom score, (3) Times of diarrhea, (4) IL-1β, (5) IL-6, (6) TNF-α, (7) CRP | |
| Zhao JH et al 2020 | 26/25 | 30.2±3.58/30.58±3.4 | 90 | (1) Clinical efficacy, (2) IL-1β, (3) IL-6, (4) TNF-α, (5) CRP |
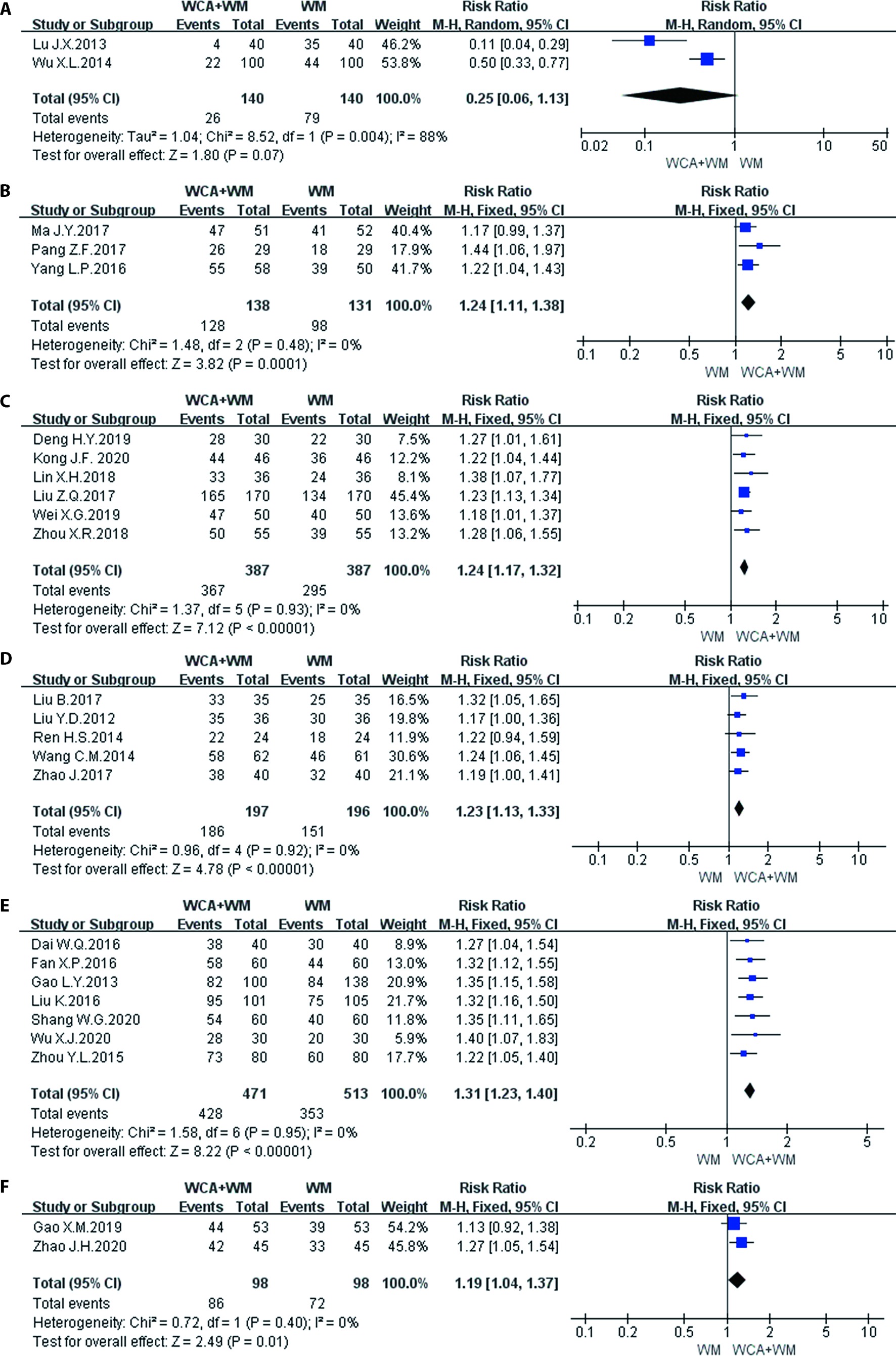
Figure 3 Forest map of efficacy on gastrointestinal diseases A: prevention of antibiotic-associated gastrointestinal reaction; B: treatment of antibiotic-associated gastrointestinal reaction; C: functional dyspepsia; D: irritable bowel syndrome; E: rotavirus diarrhea; F: ulcerative colitis.
| Included study | Dizziness or Headache | Diarrhea | Palpitation | Somnolence | Fatigue | Skin rash | Abdominal pain |
|---|---|---|---|---|---|---|---|
| Deng HY 2019 | 1:1 | 1:1 | 0:1 | - | - | - | - |
| Hao LC et al 2014 | - | - | - | - | - | 1:1 | - |
| Liu K 2016 | - | - | - | - | - | 2:2 | 4:5 |
| Liu YD et al 2012 | - | - | - | 2:1 | 1:1 | - | - |
| Li Z 2015 | - | 4:9 | - | - | - | - | - |
| Pang ZF 2017 | 1:2 | - | - | - | - | 1:1 | - |
| Ren HS 2014 | 2:0 | - | - | - | 0:1 | - | - |
| Total | 4:3 | 5:10 | 0:1 | 2:1 | 1:2 | 4:4 | 4:5 |
Table 2 Adverse reaction (E : C)
| Included study | Dizziness or Headache | Diarrhea | Palpitation | Somnolence | Fatigue | Skin rash | Abdominal pain |
|---|---|---|---|---|---|---|---|
| Deng HY 2019 | 1:1 | 1:1 | 0:1 | - | - | - | - |
| Hao LC et al 2014 | - | - | - | - | - | 1:1 | - |
| Liu K 2016 | - | - | - | - | - | 2:2 | 4:5 |
| Liu YD et al 2012 | - | - | - | 2:1 | 1:1 | - | - |
| Li Z 2015 | - | 4:9 | - | - | - | - | - |
| Pang ZF 2017 | 1:2 | - | - | - | - | 1:1 | - |
| Ren HS 2014 | 2:0 | - | - | - | 0:1 | - | - |
| Total | 4:3 | 5:10 | 0:1 | 2:1 | 1:2 | 4:4 | 4:5 |
| 1. |
Sperber AD, Bangdiwala SI, Drossman DA, et al. Worldwide prevalence and burden of functional gastrointestinal disorders, results of Rome foundation global study. Gastroenterology. 2021; 160: 99-114 e3.
DOI PMID |
| 2. | Drossman DA. Functional gastrointestinal disorders: history, pathophysiology, clinical features and Rome IV. Gastroenterology 2016; S0016- 5085 00223-7. |
| 3. | Tack J, Drossman DA. What's new in Rome IV? Neurogastroenterol Motil 2017; 29. |
| 4. |
Tamma PD, Avdic E, Li DX, Dzintars K, Cosgrove SE. Association of adverse events with antibiotic use in hospitalized patients. JAMA Intern Med 2017; 177: 1308-15.
DOI PMID |
| 5. | Shatila M, Thomas AS. Current and future perspectives in the diagnosis and management of helicobacter pylori infection. J Clin Med 2022; 1: 5086. |
| 6. | Xing DM, Zhu MJ, Liu CX, Wang H. Outcome measures in clinical trials of Traditional Chinese Medicine for stable angina pectoris. Acupunct Herb Med 2021; 1: 99-106. |
| 7. | Duya PA, Chen YX, Bai LD, et al. Nature products of Traditional Chinese Medicine provide new ideas in γδ T cell for tumor immunotherapy. Acupunct Herb Med 2022; 2: 78-83. |
| 8. | Yang QR, Jiang D, Meng K, Wang CJ, Chen R, Wang Y. Study on the molecular mechanism of Weichang'an pill in the treatment of irritable bowel syndrome based on network pharmacology. Zhong Yi Za Zhi 2020; 62: 1-7. |
| 9. |
Jia ST, Chai LJ, Zhang J, et al. Wei Chang An pill regulates gastrointestinal motility in a bidirectional manner. Pharm Biol 2021; 59: 1452-63.
DOI PMID |
| 10. |
Qu Z, Zhang JZ, Gao WY, Guo HM, Liu CX. Bidirectional effects of methanol extract of Wei-Chang-An pill on gastrointestinal transit and the spasmolytic activity on isolated rat jejunum. J Ethnopharmacol 2014; 155: 203-12.
DOI PMID |
| 11. | Zhang J. The research of anti-rotavirus activity of Weichang’an pill. Tianjin: Tianjin Medical University; 2011: 27. |
| 12. | Ling NS. Improving research on traditional Weichang’an pill to modern Chinese medicine. Tianjin: Tianjin University; 2007: 99-100. |
| 13. | Li MF, Hu SY, Li XM, et al. Multicenter study of Weichang'an pill in the prevention and treatment of gastrointestinal symptoms caused by azithromycin. Zhong Guo Zhong Xi Yi Jie He Er Ke Xue 2013; 5: 237-39. |
| 14. | Lu JX. Observation on Curative Effect of Weichang'an pill in preventing and treating 40 cases of azithromycin gastrointestinal reaction. Zhong Guo Yi Yao Zhi Nan 2013; 11: 484-5. |
| 15. | Wu XL, Zhang HY, Tian L, Liang K. Observation on the clinical effect of Weichang'an pill in preventing antibiotic-related diarrhea in children. Zhong Guo Bao Jian Ying Yang 2014: 3314-5. |
| 16. | Yang LP, Ma JY, Guo XN, Wang XH. Fifty-eight cases with pediatric antibody-related diarrhea treated with Weichang'an pill. Henan Zhong Yi 2016; 36: 871-3. |
| 17. | Pang ZF. Weichang'an pill in the treatment of gastrointestinal reactions induced by macrolides for 58 cases. Zhong Guo Zhong Yi Yao Xian Dai Yuan Cheng Jiao Yu 2017; 15: 90-2. |
| 18. | Ma JY, Yang LP. Effect of Weichang'an pill on antibiotic-related diarrhea in children with bronchopneumonia. Lin Chuang He Li Yong Yao 2017; 10: 69-70. |
| 19. | Zhou XR, Su RH, Xu GF, Yang LP, Jia LX, He AM. Effect of Weichang’an pill combined with domperidone on functional dyspepsia in children. Hebei Yi Ke Da Xue Xue Bao 2018; 39: 77-81. |
| 20. | Liu ZQ, Guo MW. The influence of integrated Chinese-western therapy on gastrointestinal dynamics and relapse in treatment of functional dyspepsia. Hubei Zhong Yi Yao Da Xue Xue Bao 2017; 19: 61-4. |
| 21. | Lin XH. Clinical observation on Weichang'an pills in the treatment of functional dyspepsia. Zhong Guo Zhong Yi Yao Xian Dai Yuan Cheng Jiao Yu 2018; 16: 98-9. |
| 22. | Deng HY. Clinical observation on integrated Chinese-Western therapy in treatment of senile functional dyspepsia. Hubei Zhong Yi Yao Da Xue Xue Bao 2019; 21: 74-6. |
| 23. | Kong JF. Clinical study of Weichang’an pill combined with mosapride tablets in the treatment of children with functional dyspepsia. Zhong Guo Xian Dai Yao Ying Yong 2020; 14: 119-21. |
| 24. | Wei XG. Clinical effect of Weichang’an pill combined with domperidone in the treatment of functional dyspepsia. Henan Yi Xue Yan Jiu 2019; 28: 2425-26. |
| 25. | Zhao J, Zhao Y. Clinical effect of Weichang’an pills combined with live bacteria preparations in the treatment of irritable bowel syndrome after infection. Neimenggu Zhong Yi Yao 2017; 36: 79. |
| 26. | Wang CM. The application and curative effect analysis of Weichang’an pill in diarrhea-type irritable bowel syndrome. Zhong Guo Shi Yong Yi Yao 2014; 9: 165-6. |
| 27. | Ren HS. Clinical observation of Weichang'an pill combined with bifidus triple viable bacteria and trimebutine in the treatment of diarrhea-type irritable bowel syndrome. Zhong Guo Shi Yong Yi Yao 2014; 8: 113-4. |
| 28. | Liu YD, Zhang BF, Zhang YR. The effect of triple therapy on irritable bowel syndrome in adults. Zhong Guo Lao Nian Xue Za Zhi 2012; 32: 3563-4. |
| 29. | Liu B, Liu L. Clinical curative effect of trimebutine maleate combined with Weichang'an pill in the treatment of irritable bowel syndrome. Dang Dai Yi Xue 2017; 23: 77-9. |
| 30. | Zhou YL, Shen ZH, Yano Y, Wang SY. The protective effect and clinical effect of Weichang’an pill on myocardial injury caused by rotavirus enteritis in children. Zhong Guo Zhong Xi Yi Jie He Xiao Hua Za Zhi 2015; 23: 898-9. |
| 31. | Wu XJ. Observation on the curative effect of Weichang'an pill combined with boulardii in the treatment of rotavirus enteritis in children. Mu Ying Shi Jie 2020: 143. |
| 32. | Shang WG, Xu J. Clinical effect of Weichang'an pill in the treatment of child patients with rotavirus enteritis and its influence on myocardial enzyme spectrum. Zhong Guo Yao Xue Jing Ji Xue 2020; 15: 74-7. |
| 33. | Liu K, Chen F, Lü Q, Wang Q, Fan DW. Clinical observation of Weichang'an pill in treating infantile rotavirus enteritis. Guo Ji Er Tong Za Zhi 2016; 43: 968-70. |
| 34. | Gao LY, Gao F. Clinical observation on the treatment of 100 cases of rotavirus enteritis in children with Weichang'an pill. Zhong Guo Bao Jian Ying Yang 2013: 462. |
| 35. | Fan XP, Zhang CY, Zhao QH, Li XH. Clinical effect of Weichang’an pill in the treatment of virus enteritis. Guo Ji Bing Du Xue Za Zhi 2016; 23: 10-2. |
| 36. | Dai WQ, Huang QF. Clinical observation of Weichang'an pill in treating rotavirus infection in children. Shi Jie Zui Xin Yi Xue Xin Xi Wen Zhai 2016; 16: 117-9. |
| 37. | Zhao JH. Effect of Weichang'an pill combined with mesalazine on serum inflammatory index level in patients with ulcerative colitis. Chengde Yi Xue Yuan Xue Bao 2020; 37: 487-90. |
| 38. | Gao XM, Zhang W, Lu WJ. Clinical study on Weichang'an pills combined with mesalazine in treatment of ulcerative colitis. Xian Dai Yao Wu Yu Lin Chuang 2019; 34: 3319-22. |
| 39. | Chen YY. Effect analysis of Weichang 'an pill combined with bifidobacteria triple viable capsule in the treatment of ulcerative colitis. Xian Dai Zhen Duan Yu Zhi Liao 2021; 32: 1867-9. |
| 40. | Li Z. Clinical observation of Weichang'an pills combined with quadruple therapy in treatment of helicobacter pylori infection in children. Xian Dai Yao Wu Yu Lin Chuang 2015; 30: 576-8. |
| 41. |
Chen Y, Chu F, Lin J, et al. The mechanisms of action of Weichang'an pill (WCAP) treat diarrhoea-predominant irritable bowel syndrome (IBS-D) using network pharmacology approach and in vivo studies. J Ethnopharmacol 2021; 275: 114119.
DOI URL |
| 42. | Zhang T, Su XL, Mao XY, et al. Effects of Weichang'an pill on Treg/Th17 immune balance in ulcerative colitis model mice. Zhong Yi Za Zhi 2020; 61: 1983-9. |
| 43. | Wei JJ, Yao F, Xu ZX, Chang H, Lu RY. Clinical observation of Weichang'an pills combined with montmorillonite powder in treatment of acute diarrhea of dyspeptic retention gastroenteric disease. Xian Dai Yao Wu Yu Lin Chuang 2018; 33: 1402-5. |
| 44. | He XY. Analysis of the therapeutic effect of Weichang'an on 40 children with functional abdominal pain. Zhong Guo Yi Yao Zhi Nan 2013; 11: 636-7. |
| 45. | Xu JJ, Xue N, Wei XX. Clinical study on Weichang'an pill in treating children's Hp infection-related gastritis. Zhong Guo Xian Dai Yao Wu Ying Yong 2017; 11:124-6. |
| 46. | Hao LC, Li XY, Dong Y, et al. Observation on the effect of Weichang'an pill in adjuvant treatment of mesenteric lymphadenitis in children. Zhong Guo Bao Jian Ying Yang 2014: 4560. |
| 47. | Niu Y, Ma XL. Clinical observation on treating the acute mesenteric lymphadenitis in children with the Weichang'an pill combined with antibiotic. Zhong Guo Wei Sheng Biao Zhun 2014; 5: 111-3. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||