Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (1): 156-162.DOI: 10.19852/j.cnki.jtcm.20231121.004
• Original articles • Previous Articles Next Articles
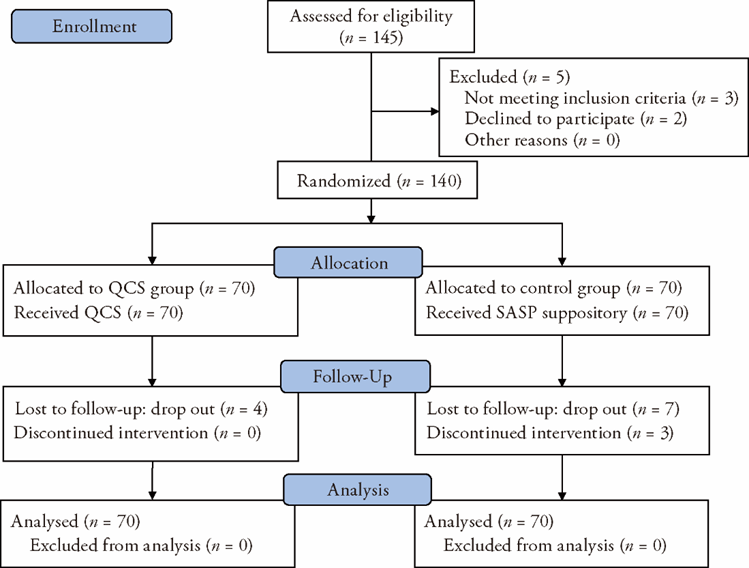
Qingchang suppositry (清肠栓) induced remission in patients with mild-to-moderate ulcerative proctitis: a multicenter, prospective, randomized, parallel-controlled clinical trial
DAI Xiaoling1, ZHANG Anming2, LIN Hui3, SHI Bei1, REN Yi2, WEN Hongzhu1, FEI Xiaoyan1, LIN Jiang1( )
)
- 1 Department of Gastroenterology, Longhua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai 200032, China
2 Department of Gastroenterology, Shanghai Putuo Traditional Chinese Medicine hospital, Shanghai 200063, China
3 Department of Protocology, Shanghai Tianshan Traditional Chinese Medicine hospital, Shanghai 200050, China
-
Received:2022-12-11Accepted:2023-03-23Online:2024-02-15Published:2023-11-21 -
Contact:LIN jiang, Department of Gastroenterology, Longhua hospital affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai 200032, China. linjiang@longhua.net. Telephone: +86-64385700-3425 -
Supported by:Shanghai Municipal Administration of Traditional Chinese Medicine: Leadship Training Program of Inheritance and Innovation Team of Shanghai School of Traditional Chinese Medicine(2021WXGLP-007)
Cite this article
DAI Xiaoling, ZHANG Anming, LIN Hui, SHI Bei, REN Yi, WEN Hongzhu, FEI Xiaoyan, LIN Jiang. Qingchang suppositry (清肠栓) induced remission in patients with mild-to-moderate ulcerative proctitis: a multicenter, prospective, randomized, parallel-controlled clinical trial[J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 156-162.
share this article
| Item | Study group (n = 70) | Control group (n = 70) | P value |
|---|---|---|---|
| Male/female (n) | 34/36 | 42/28 | 0.235 |
| Body mass index (kg/m2) | 22.08±2.61 | 22.62±2.97 | 0.264 |
| Age (years) | 43.10±10.95 | 43.32±13.33 | 0.539 |
| Disease level (mild/moderate, n) | 21/49 | 29/41 | 0.158 |
| Duration of disease (years) | 1.5 (0.08, 15) | 1.0 (0.08, 20) | 0.161 |
| Mayo score | 7 (3, 10) | 6 (3, 10) | 0.243 |
| Geboes score | 5 (2, 9) | 5 (2, 8) | 0.878 |
| ESR (mm/h) | 10 (2, 54) | 7.5 (1, 65) | 0.119 |
| CRP (mg/L) | 0.8 (0.5, 39) | 0.5 (1, 24) | 0.138 |
| Abdominal pain score (scores, n) | 0.606 | ||
| 0 | 32 | 36 | |
| 2 | 19 | 16 | |
| 4 | 4 | 14 | |
| 6 | 5 | 6 | |
| Diarrhea subscore (scores, n) | 0.052 | ||
| 0 | 2 | 2 | |
| 2 | 23 | 20 | |
| 4 | 27 | 24 | |
| 6 | 8 | 22 | |
| Purulent bloody stool score (scores, n) | 0.871 | ||
| 0 | 3 | 2 | |
| 2 | 19 | 22 | |
| 4 | 26 | 27 | |
| 6 | 22 | 19 |
Table 1 Patients demographics and baseline clinical characteristics
| Item | Study group (n = 70) | Control group (n = 70) | P value |
|---|---|---|---|
| Male/female (n) | 34/36 | 42/28 | 0.235 |
| Body mass index (kg/m2) | 22.08±2.61 | 22.62±2.97 | 0.264 |
| Age (years) | 43.10±10.95 | 43.32±13.33 | 0.539 |
| Disease level (mild/moderate, n) | 21/49 | 29/41 | 0.158 |
| Duration of disease (years) | 1.5 (0.08, 15) | 1.0 (0.08, 20) | 0.161 |
| Mayo score | 7 (3, 10) | 6 (3, 10) | 0.243 |
| Geboes score | 5 (2, 9) | 5 (2, 8) | 0.878 |
| ESR (mm/h) | 10 (2, 54) | 7.5 (1, 65) | 0.119 |
| CRP (mg/L) | 0.8 (0.5, 39) | 0.5 (1, 24) | 0.138 |
| Abdominal pain score (scores, n) | 0.606 | ||
| 0 | 32 | 36 | |
| 2 | 19 | 16 | |
| 4 | 4 | 14 | |
| 6 | 5 | 6 | |
| Diarrhea subscore (scores, n) | 0.052 | ||
| 0 | 2 | 2 | |
| 2 | 23 | 20 | |
| 4 | 27 | 24 | |
| 6 | 8 | 22 | |
| Purulent bloody stool score (scores, n) | 0.871 | ||
| 0 | 3 | 2 | |
| 2 | 19 | 22 | |
| 4 | 26 | 27 | |
| 6 | 22 | 19 |
| Symptom | Group | 0 week (n) | 12 weeks (n) | Remission rate (%) | P value |
|---|---|---|---|---|---|
| Abdominal pain | Study | 38 | 12 | 68.42 | 0.303 |
| Control | 36 | 14 | 61.11 | ||
| Diarrhea | Study | 68 | 15 | 77.94 | 1.000 |
| Control | 66 | 15 | 77.27 | ||
| Purulent bloody stool | Study | 67 | 11 | 83.58 | 1.000 |
| Control | 68 | 10 | 85.29 |
Table 2 Comparison of the remission rates of the main symptoms at week 12 between the two groups
| Symptom | Group | 0 week (n) | 12 weeks (n) | Remission rate (%) | P value |
|---|---|---|---|---|---|
| Abdominal pain | Study | 38 | 12 | 68.42 | 0.303 |
| Control | 36 | 14 | 61.11 | ||
| Diarrhea | Study | 68 | 15 | 77.94 | 1.000 |
| Control | 66 | 15 | 77.27 | ||
| Purulent bloody stool | Study | 67 | 11 | 83.58 | 1.000 |
| Control | 68 | 10 | 85.29 |
| Symptom | Group | 0 week | 12 weeks | Remission rate (%) | P value |
|---|---|---|---|---|---|
| Tenesmus | Study | 54 | 15 | 76.47 | 0.009 |
| Control | 56 | 42 | 25.00 | ||
| Anal burning | Study | 51 | 13 | 74.51 | 0.003 |
| Control | 54 | 38 | 29.63 | ||
| Fetid oris | Study | 18 | 15 | 16.67 | 1.000 |
| Control | 21 | 15 | 28.57 | ||
| Dark urine | Study | 19 | 14 | 26.31 | 0.425 |
| Control | 17 | 13 | 23.53 |
Table 3 Comparison of the remission rates of the TCM symptoms at week 12 between the two groups
| Symptom | Group | 0 week | 12 weeks | Remission rate (%) | P value |
|---|---|---|---|---|---|
| Tenesmus | Study | 54 | 15 | 76.47 | 0.009 |
| Control | 56 | 42 | 25.00 | ||
| Anal burning | Study | 51 | 13 | 74.51 | 0.003 |
| Control | 54 | 38 | 29.63 | ||
| Fetid oris | Study | 18 | 15 | 16.67 | 1.000 |
| Control | 21 | 15 | 28.57 | ||
| Dark urine | Study | 19 | 14 | 26.31 | 0.425 |
| Control | 17 | 13 | 23.53 |
| Group | n | ESR | CRP | ||
|---|---|---|---|---|---|
| 0 week | 12 weeks | 0 week | 12 weeks | ||
| Study | 70 | 10 (2, 54) | 6.5 (1, 49) | 0.58 (0.07, 35) | 0.5 (0.5, 4.8) |
| Control | 70 | 7.5 (1, 65) | 7 (2, 35) | 0.5 (0.5, 18) | 0.5 (0.5, 7.48) |
| Z value | -1.929 | -0.198 | -1.296 | -0.644 | |
| P value | 0.054 | 0.843 | 0.195 | 0.520 | |
Table 4 Comparison of ESR and CRP levels between two groups before and after treatment
| Group | n | ESR | CRP | ||
|---|---|---|---|---|---|
| 0 week | 12 weeks | 0 week | 12 weeks | ||
| Study | 70 | 10 (2, 54) | 6.5 (1, 49) | 0.58 (0.07, 35) | 0.5 (0.5, 4.8) |
| Control | 70 | 7.5 (1, 65) | 7 (2, 35) | 0.5 (0.5, 18) | 0.5 (0.5, 7.48) |
| Z value | -1.929 | -0.198 | -1.296 | -0.644 | |
| P value | 0.054 | 0.843 | 0.195 | 0.520 | |
| Group | n | 0 week | 12 weeks | Z value | P value |
|---|---|---|---|---|---|
| Study | 70 | 5 (2, 9) | 2 (0, 7) | -0.759 | 0.448 |
| Control | 70 | 5 (2, 8) | 2 (0, 7) |
Table 5 Comparison of Geboes scores between two groups after treatment
| Group | n | 0 week | 12 weeks | Z value | P value |
|---|---|---|---|---|---|
| Study | 70 | 5 (2, 9) | 2 (0, 7) | -0.759 | 0.448 |
| Control | 70 | 5 (2, 8) | 2 (0, 7) |
| Group | n | 4 weeks | 8 weeks | 12 weeks |
|---|---|---|---|---|
| Study | 70 | 50 (71.42) | 55 (78.57) | 59 (84.29) |
| Control | 70 | 37 (52.85) | 46 (65.71) | 52 (74.29)a |
| P value | 0.023 | 0.090 | 0.144 |
Table 6 Comparison of mucosa healing rates at weeks 4,8,12 between two groups [n (%)]
| Group | n | 4 weeks | 8 weeks | 12 weeks |
|---|---|---|---|---|
| Study | 70 | 50 (71.42) | 55 (78.57) | 59 (84.29) |
| Control | 70 | 37 (52.85) | 46 (65.71) | 52 (74.29)a |
| P value | 0.023 | 0.090 | 0.144 |
| 1. |
Pineton dCG, Tassy B, Kollen L, et al. The treatment of refractory ulcerative colitis. Best Pract Res Clin Gastroenterol 2018; 32-33: 49-57.
DOI PMID |
| 2. | Chambrun GP, Danese S, Peyrin-Biroulet L. Time to include patients with ulcerative proctitis in clinical trials. Lancet Gastroenterol Hepatol 2019; 4: 900-2. |
| 3. |
Henriksen M, Jahnsen J, Lygren I, et al. Ulcerative colitis and clinical course: results of a 5-year population-based follow-up study (the IBSEN study). Inflamm Bowel Dis 2006; 12: 543-50.
DOI PMID |
| 4. |
Meucci G, Vecchi M, Astegiano M, et al. The natural history of ulcerative proctitis: a multicenter, retrospective study. Gruppo di Studio per le Malattie Infiammatorie Intestinali (GSMII). Am J Gastroenterol 2000; 95: 469-73.
DOI PMID |
| 5. | Gong YP, Li CN. Clinical and experimental research progress of Qingchang suppository in the treatment of ulcerative colitis. Lin Chuang Yi Yao Wen Xian Dian Zi Za Zhi 2018; 5: 181-3,5. |
| 6. |
Watanabe M, Nishino H, Sameshima Y, et al. Randomised clinical trial: evaluation of the efficacy of mesalazine (mesalamine) suppositories in patients with ulcerative colitis and active rectal inflammation-a placebo-controlled study. Aliment Pharmacol Ther 2013; 38: 264-73.
DOI URL |
| 7. |
Kato S, Ishibashi A, Kani K, Yakabi K. Optimized management of ulcerative proctitis: when and how to use mesalazine suppository. Digestion 2018; 97: 59-63.
DOI PMID |
| 8. | Gong YP, Ma GT, LIU W, et al. Qingchang suppository in the treatment of ulcerative colitis: a randomized controlled study. Shanghai Zhong Yi Yao Da Xue Xue Bao 2007; 21: 33-6. |
| 9. | Hao WW, Ma GT, Zhang XF, et al. Effects of Qingchangshuan on IL-4 and IL-10 mRNA expression in rat colon tissue of ulcerative colitis induced by TNBS. Chin J Integr Trad West Med Dig 2007; 15: 177-80. |
| 10. | Wang ZN, Tang ZP, Ma GT, et al. Effect of Qingchang suppository on cell proliferation of colonic mucous in trinitrobenzene sulfonic acid-induced colitis in rats. Zhong Guo Zhong Xi Yi Jie He Za Zhi 2006; 14: 383-6. |
| 11. |
Sun BY, Yuan JY, Wang SY, et al. Qingchang suppository ameliorates colonic vascular permeability in dextran-sulfate-sodium-induced colitis. Front Pharmacol 2018; 9: 1235.
DOI PMID |
| 12. |
D’Haens G, Sandborn WJ, Feagan BG, et al. A review of activity indices and efficacy end points for clinical trials of medical therapy in adults with ulcerative coliti. Gastroenterology 2007; 132: 763-86.
DOI URL |
| 13. | Zhang S, Shen H, Zheng K, et al. Expert consensus opinion of TCM diagnosis and treatment of ulcerative colitis (2017). Zhong Hua Zhong Yi Yao Za Zhi 2017; 32: 3585-9. |
| 14. |
Geboes K, Riddell R, Ost A, et al. A reproducible grading scale for histological assessment of inflammation in ulcerative colitis. Gut 2000; 47: 404-9.
DOI PMID |
| 15. |
Safroneeva E, Vavricka S, Fournier N, et al. Systematic analysis of factors associated with progression and regression of ulcerative colitis in 918 patients. Aliment Pharmacol Ther 2015; 42: 540-8.
DOI URL |
| 16. |
Iacucci M, Ghosh S. Mucosal healing-how deep is enough? Dig Dis 2016; 34: 160-4.
DOI URL |
| 17. |
Dal BA, Roda G, Argollo M, et al. Histological healing: should it be considered as a new outcome for ulcerative colitis? Expert Opin Biol Ther 2020; 20: 407-12.
DOI PMID |
| 18. |
Sands BE, Peyrin-Biroulet L, Loftus EV Jr, et al. Vedolizumab versus adalimumab for moderate-to-severe ulcerative colitis. N Engl J Med 2019; 381: 1215-26.
DOI URL |
| 19. |
Yoon H, Jangi S, Dulai PS, et al. Incremental benefit of achieving endoscopic and histologic remission in patients with ulcerative colitis: a systematic review and Meta-analysis. Gastroenterology 2020; 159: 1262-75. e7.
DOI PMID |
| 20. |
Magro F, Doherty G, Peyrin-Biroulet L, et al. ECCO position paper: harmonization of the approach to ulcerative colitis histopathology. J Crohns Colitis 2020; 14: 1503-11.
DOI PMID |
| 21. |
Marchal BA, Riddell RH, Boulagnon-Rombi C, et al. Review article: the histological assessment of disease activity in ulcerative colitis. Aliment Pharmacol Ther 2015; 42: 957-67.
DOI URL |
| 22. |
Yoon JY, Park SJ, Hong SP, et al. Correlations of C-reactive protein levels and erythrocyte sedimentation rates with endoscopic activity indices in patients with ulcerative colitis. Dig Dis Sci 2014; 59: 829-37.
DOI URL |
| 23. |
Vermeire S, Van Assche G, Rutgeerts P. Laboratory markers in IBD: useful, magic, or unnecessary toys? Gut 2006; 55: 426-31.
DOI PMID |
| [1] | QIN Xiaoyu, WANG Chunai, XUE Jianjun, ZHANG Jie, LU Xiaoting, DING Shengshuang, GE Long, WANG Minzhen. Efficacy of electroacupuncture on myocardial protection and postoperative rehabilitation in patients undergoing cardiac surgery with cardiopulmonary bypass: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 1-15. |
| [2] | WANG Yichen, WU Shiyi, WANG Zhengyan, CHANG Wenling, XIE Zhihao, TANG Xing, ZHAO Songmei, ZHOU Jing, CHEN Zehong, WANG Chao, YANG Chunxia. Efficacy of Zhumian Tang formula granules (助眠汤配方颗粒) combined with eszopiclone for the treatment of poor sleep quality: a multi-center, randomized controlled, superiority trial [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 163-171. |
| [3] | YANG Yi, YE Huijun, ZHENG Huiling, JIN Lihua. Clinical observation on 90 cases of primary dysmenorrhea treated by buccal acupuncture therapy: a randomized controlled study [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 172-181. |
| [4] | DAI Zeqi, LIAO Xing, GUAN Yueyue, ZENG Zixiu, TANG Jun, HU Jing. Bloodletting puncture in the treatment of acute ischemic stroke: protocol for a mixed-method study of a multi-center randomized controlled trial and focus group [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1259-1267. |
| [5] | XU Yani, ZHANG Yutong, HE Weile, DAI Linglin, TANG Ding, WANG Jialing, ZHANG Xufen, CHEN Qin, CHEN Lifang, WANG Zhanglian, ZHAN Mingjie. Efficiency and safety of acupuncture for women with premature ovarian insufficiency: study protocol for a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1268-1274. |
| [6] | XU Xiangru, ZHOU Yi, CHEN Gang, LEI Ming, ZHANG Wen, WU Xinxin, PU Yuting, CHEN Caiyu, SUN Yuting, ZHOU Shuang, FANG Bangjiang. Clinical efficacy of Buzhong Yiqi decoction (补中益气汤) in the treatment of hospital-acquired pneumonia with multi-drug resistant bacteria: a prospective, randomized, multicenter controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1010-1018. |
| [7] | ZHAO Ming, LUO Yimiao, WANG Huichan, CAO Yu, MA Lina, PEI Hui, LI Hao. Guilingji capsule (龟龄集胶囊) for Alzheimer's disease: secondary analysis of a randomized non-inferiority controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1019-1025. |
| [8] | ZHANG Meizhen, HAO Xiaohui, TANG Yiting, CHEN Yupeng, HE Puyu, ZHAO Liming, PANG Bing, NI Qing. Efficacy and safety of Buyang Huanwu decoction (补阳还五汤) for diabetic peripheral neuropathy: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 841-850. |
| [9] | YANG Yuqing, CHEN Yuhuan, LI Chunxiao, LING Xiao, WANG Panpan, GUO Jing, ZHANG Yingying. Effectiveness and safety of Pingxiao capsule (平消胶囊) as adjuvant therapy in treatment of breast cancer: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 851-859. |
| [10] | WANG Chao, WU Qiong, LI Ping, WANG Zhigang, LOU Xusheng, LI Yuanyuan, ZHANG Lin. Effect of Traditional Chinese Medicine combined with Western Medicine on blood lipid levels and inflammatory factors in patients with angina pectoris in coronary heart disease identified as intermingled phlegm and blood stasis syndrome: a network Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 640-649. |
| [11] | ZHANG Xinghe, LI Qifu, YI Rong, XING Chonghui, JIN Yuhao, MENG Jiangqiong, FENG Jialei, ZHAO Siwen, LIANG Fanrong, GUO Taipin. Effect of catgut embedding at acupoints versus non-acupoints in abdominal obesity: a randomized clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 780-786. |
| [12] | ZHANG Yuehong, DONG Dandan, YAN Youqin, ZHANG Hao, WANG Guangli, ZHOU Wei, LI Wei, QIU Li, LI Tingming, LIU Quan, XIA Ping, MAO Lina, YANG Danlin, YANG Lu, LIAN Fengmei, TONG Xiaolin, BA Yuanming. Effectiveness and safety of Jinshuibao capsules (金水宝胶囊) in treatment of residual cardiopulmonary symptoms in convalescent patients of coronavirus disease 2019: a pilot randomized, double-blind, placebo-controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 134-139. |
| [13] | YAO Yao, ZHAO Zhenni, CHEN Fengqin, LENG Yufei, PANG Xiangtian, XU Xiao, SUN Zhiling. Effectiveness of moxibustion alone on lumbar disc herniation: a Meta-analysis of randomized controlled trials [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 14-26. |
| [14] | JIN Yutong, WU Lingtao, GUO Yanglu, XIA Chong, YU Binyan, XUAN Lihua. Long-term efficacy of point application therapy on different acupoints and durations in the treatment of asthma: a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 146-153. |
| [15] | SHEN Mengxia, SHANG Wenfang, WU Jiangxia, YU Zelin, XUAN Lihua. Efficacy and safety of acupoint application for allergic rhinitis: a systematic review and Meta-analysis of randomized controlled trials [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 858-868. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||