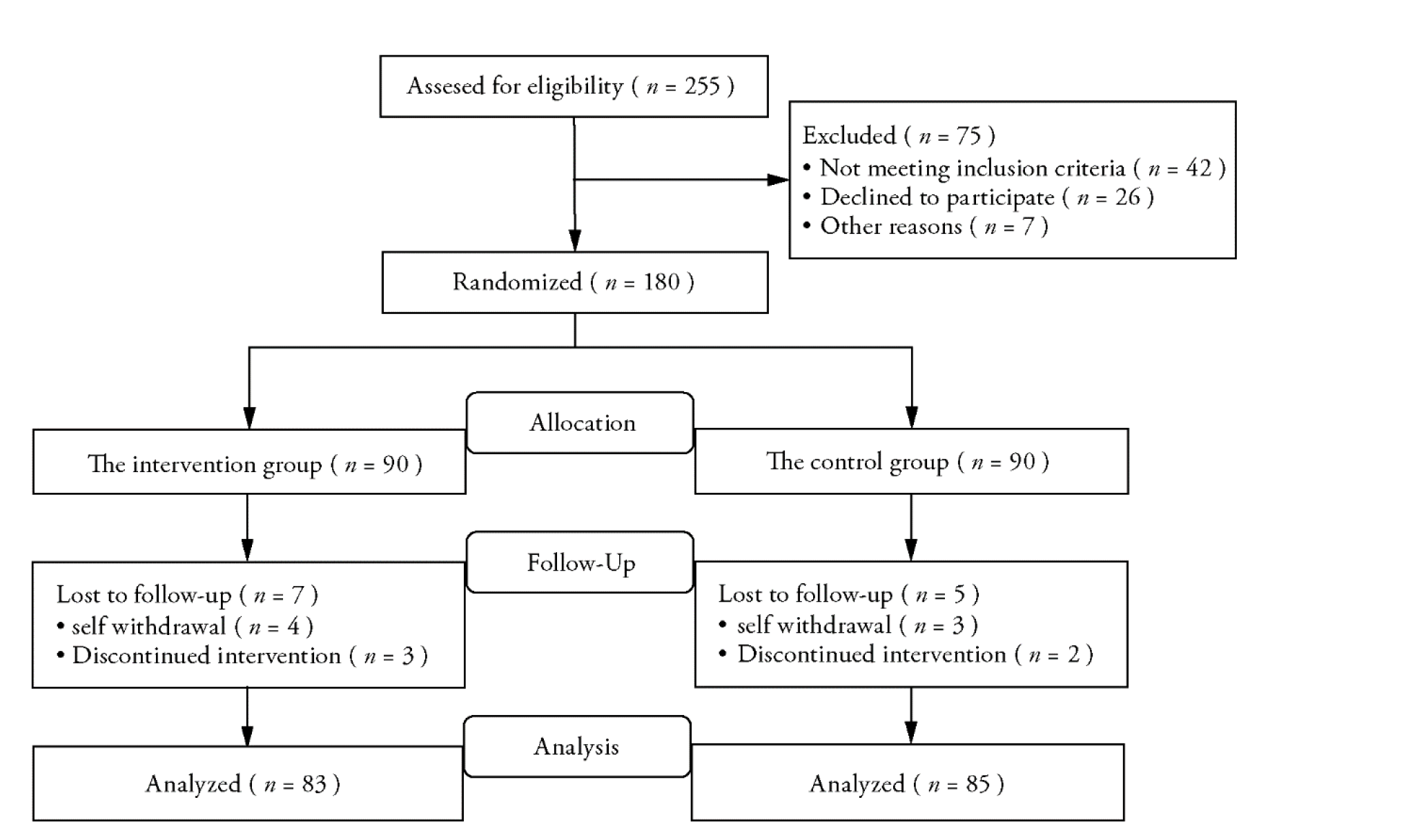
Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (5): 1010-1018.DOI: 10.19852/j.cnki.jtcm.20230713.002
Previous Articles Next Articles
Clinical efficacy of Buzhong Yiqi decoction (补中益气汤) in the treatment of hospital-acquired pneumonia with multi-drug resistant bacteria: a prospective, randomized, multicenter controlled trial
XU Xiangru1, ZHOU Yi2, CHEN Gang3, LEI Ming3, ZHANG Wen1, WU Xinxin1, PU Yuting1, CHEN Caiyu1, SUN Yuting1, ZHOU Shuang4( ), FANG Bangjiang1,5(
), FANG Bangjiang1,5( )
)
- 1 Department of Emergency, Longhua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai 200032, China
2 School of Basic Medical Sciences, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China
3 Department of Intensive Care Medicine, Seventh People’s Hospital of Shanghai University of Traditional Chinese Medicine, Shanghai 200137, China
4 Acupuncture and Massage College, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China
5 Institute of Critical Care, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China
-
Received:2022-12-12Accepted:2023-02-04Online:2023-10-15Published:2023-07-13 -
Contact:Prof. FANG Bangjiang, Department of Emergency, Longhua Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai 200032, China.fangbji@163.com ; ZHOU Shuang, Acupuncture and Massage College, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China.zhoushuang8008@163.com . Telephone: +86-21-64385700 -
Supported by:Basic Research Program of Shanghai Municipal Science and Technology Commission: A Multicenter, Randomized, Controlled Clinical Study on Fuzheng Quxie Jing Prescription "Buzhong Yiqi Decoction" in the Treatment of Hospital-acquired Pneumonia caused by Multi-drug Resistant Bacteria(18401971600);Three-year Action Plan for the Development of Traditional Chinese Medicine in Shanghai: National TCM Emergency Medical Rescue Base Construction(ZY(2021-2023)-0101-01);East China Area and Municipal TCM Specialty Disease Alliance Construction(ZY(2021-2023)-0302)
Cite this article
XU Xiangru, ZHOU Yi, CHEN Gang, LEI Ming, ZHANG Wen, WU Xinxin, PU Yuting, CHEN Caiyu, SUN Yuting, ZHOU Shuang, FANG Bangjiang. Clinical efficacy of Buzhong Yiqi decoction (补中益气汤) in the treatment of hospital-acquired pneumonia with multi-drug resistant bacteria: a prospective, randomized, multicenter controlled trial[J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1010-1018.
share this article
| Variable | Control group (n = 90) | Intervention group (n = 90) | P value | |
|---|---|---|---|---|
| Age [years, n (%)] | <65 | 12 (13.3) | 20 (22.2) | 0.827 |
| ≥65 | 78 (86.7) | 70 (77.8) | ||
| $\bar{x}±s$ | 76.5±10.2 | 75.9±12.3 | ||
| Sex [n (%)] | Female | 32 (35.6) | 41 (45.6) | 0.172 |
| Male | 58 (64.4) | 49 (54.4) | ||
| Mechanical ventilation [n (%)] | 32 (35.6) | 32 (35.6) | 0.999 | |
| Hospitalization duration [days, Median (IQR)] | 4.5 (2.0-7.0) | 5.0 (3.0-8.0) | 0.160 | |
| Vital signs ($\bar{x}±s$) | Temperature (℃) | 37.5±0.8 | 37.3±0.8 | 0.079 |
| Respiratory rate (bpm) | 21.5±12.9 | 19.1±3.3 | 0.048 | |
| Heart rate (bpm) | 89.4±17.1 | 90.5±14.2 | 0.959 | |
| Systolic blood pressure (mm Hg) | 132.4±20.9 | 135.7±20.3 | 0.319 | |
| Diastolic blood pressure (mm Hg) | 76.0±11.0 | 77.0±11.2 | 0.717 | |
| APACHE Ⅱ score | <15 [n (%)] | 9 (10.7) | 12 (14.0) | 0.279 |
| ≥15 [n (%)] | 75 (89.3) | 74 (86.0) | ||
| $\bar{x}±s$ | 19.1±4.6 | 20.5±6.1 | ||
| CPIS score | <6, n (%) | 56 (62.2) | 61 (67.8) | 0.197 |
| ≥6, n (%) | 34 (37.8) | 29 (32.2) | ||
| $\bar{x}±s$ | 5.5±1.3 | 5.3±1.6 | ||
| Baseline pathogen [n (%)] | Acinetobacter baumannii | 21 (23.3) | 31 (34.4) | 0.100 |
| Klebsiella pneumoniae | 31 (34.4) | 24 (26.7) | 0.257 | |
| Pseudomonas aeruginosa | 8 (8.9) | 9 (10.0) | 0.799 | |
| Staphylococcus aureus | 25 (27.8) | 15 (16.7) | 0.073 | |
| Escherichia coli | 0 | 7 (7.8) | 0.014a | |
| Others resistant bacteria [n (%)] | 5 (5.6) | 4 (4.4) | 0.999 | |
| Comorbidities [n (%)] | Cerebrovascular disease | 59 (65.6) | 51 (56.7) | 0.221 |
| Hypertension | 56 (62.2) | 57 (63.3) | 0.877 | |
| Coronary heart disease | 55 (61.1) | 48 (53.3) | 0.292 | |
| Diabetes mellitus | 34 (37.8) | 37 (41.1) | 0.647 | |
| Chronic kidney disease | 11 (12.2) | 12 (13.3) | 0.823 | |
| Chronic pulmonary disease | 6 (6.7) | 9 (10.0) | 0.418 | |
| Hypoproteinemia | 44 (48.9) | 48 (53.3) | 0.551 |
Table 1 Baseline demographic data and clinical characteristics
| Variable | Control group (n = 90) | Intervention group (n = 90) | P value | |
|---|---|---|---|---|
| Age [years, n (%)] | <65 | 12 (13.3) | 20 (22.2) | 0.827 |
| ≥65 | 78 (86.7) | 70 (77.8) | ||
| $\bar{x}±s$ | 76.5±10.2 | 75.9±12.3 | ||
| Sex [n (%)] | Female | 32 (35.6) | 41 (45.6) | 0.172 |
| Male | 58 (64.4) | 49 (54.4) | ||
| Mechanical ventilation [n (%)] | 32 (35.6) | 32 (35.6) | 0.999 | |
| Hospitalization duration [days, Median (IQR)] | 4.5 (2.0-7.0) | 5.0 (3.0-8.0) | 0.160 | |
| Vital signs ($\bar{x}±s$) | Temperature (℃) | 37.5±0.8 | 37.3±0.8 | 0.079 |
| Respiratory rate (bpm) | 21.5±12.9 | 19.1±3.3 | 0.048 | |
| Heart rate (bpm) | 89.4±17.1 | 90.5±14.2 | 0.959 | |
| Systolic blood pressure (mm Hg) | 132.4±20.9 | 135.7±20.3 | 0.319 | |
| Diastolic blood pressure (mm Hg) | 76.0±11.0 | 77.0±11.2 | 0.717 | |
| APACHE Ⅱ score | <15 [n (%)] | 9 (10.7) | 12 (14.0) | 0.279 |
| ≥15 [n (%)] | 75 (89.3) | 74 (86.0) | ||
| $\bar{x}±s$ | 19.1±4.6 | 20.5±6.1 | ||
| CPIS score | <6, n (%) | 56 (62.2) | 61 (67.8) | 0.197 |
| ≥6, n (%) | 34 (37.8) | 29 (32.2) | ||
| $\bar{x}±s$ | 5.5±1.3 | 5.3±1.6 | ||
| Baseline pathogen [n (%)] | Acinetobacter baumannii | 21 (23.3) | 31 (34.4) | 0.100 |
| Klebsiella pneumoniae | 31 (34.4) | 24 (26.7) | 0.257 | |
| Pseudomonas aeruginosa | 8 (8.9) | 9 (10.0) | 0.799 | |
| Staphylococcus aureus | 25 (27.8) | 15 (16.7) | 0.073 | |
| Escherichia coli | 0 | 7 (7.8) | 0.014a | |
| Others resistant bacteria [n (%)] | 5 (5.6) | 4 (4.4) | 0.999 | |
| Comorbidities [n (%)] | Cerebrovascular disease | 59 (65.6) | 51 (56.7) | 0.221 |
| Hypertension | 56 (62.2) | 57 (63.3) | 0.877 | |
| Coronary heart disease | 55 (61.1) | 48 (53.3) | 0.292 | |
| Diabetes mellitus | 34 (37.8) | 37 (41.1) | 0.647 | |
| Chronic kidney disease | 11 (12.2) | 12 (13.3) | 0.823 | |
| Chronic pulmonary disease | 6 (6.7) | 9 (10.0) | 0.418 | |
| Hypoproteinemia | 44 (48.9) | 48 (53.3) | 0.551 |
| Variable | Control group (n = 85) | Intervention group (n = 83) | P value |
|---|---|---|---|
| Primary outcome | |||
| Clinical success rate [n (%)] | 28 (32.9) | 40 (48.2) | 0.044a |
| Pathogen eradication rate [n (%)] | 33 (38.9) | 49 (59.0) | 0.009a |
| CPIS score ($\bar{x}±s$) | 9.6±2.5 | 8.9±1.7 | 0.003a |
| Secondary outcome | |||
| 28-day ACM [n (%)] | 18 (21.2) | 11 (13.3) | 0.174 |
| Ventilator weaning rate [n (%)] | 18 (56.3) | 22 (68.8) | 0.302 |
| Length of MV [days, median (IQR)] | 17.2±7.2 | 13.7±6.4 | 0.038a |
| Length of hospital stay [days, median (IQR)] | 24.6±8.6 | 23.1±8.3 | 0.271 |
| APACHE II score ($\bar{x}±s$) | 3.1±1.5 | 2.4±1.5 | 0.288 |
Table 2 Clinical outcome between the intervention and control groups on day 28
| Variable | Control group (n = 85) | Intervention group (n = 83) | P value |
|---|---|---|---|
| Primary outcome | |||
| Clinical success rate [n (%)] | 28 (32.9) | 40 (48.2) | 0.044a |
| Pathogen eradication rate [n (%)] | 33 (38.9) | 49 (59.0) | 0.009a |
| CPIS score ($\bar{x}±s$) | 9.6±2.5 | 8.9±1.7 | 0.003a |
| Secondary outcome | |||
| 28-day ACM [n (%)] | 18 (21.2) | 11 (13.3) | 0.174 |
| Ventilator weaning rate [n (%)] | 18 (56.3) | 22 (68.8) | 0.302 |
| Length of MV [days, median (IQR)] | 17.2±7.2 | 13.7±6.4 | 0.038a |
| Length of hospital stay [days, median (IQR)] | 24.6±8.6 | 23.1±8.3 | 0.271 |
| APACHE II score ($\bar{x}±s$) | 3.1±1.5 | 2.4±1.5 | 0.288 |
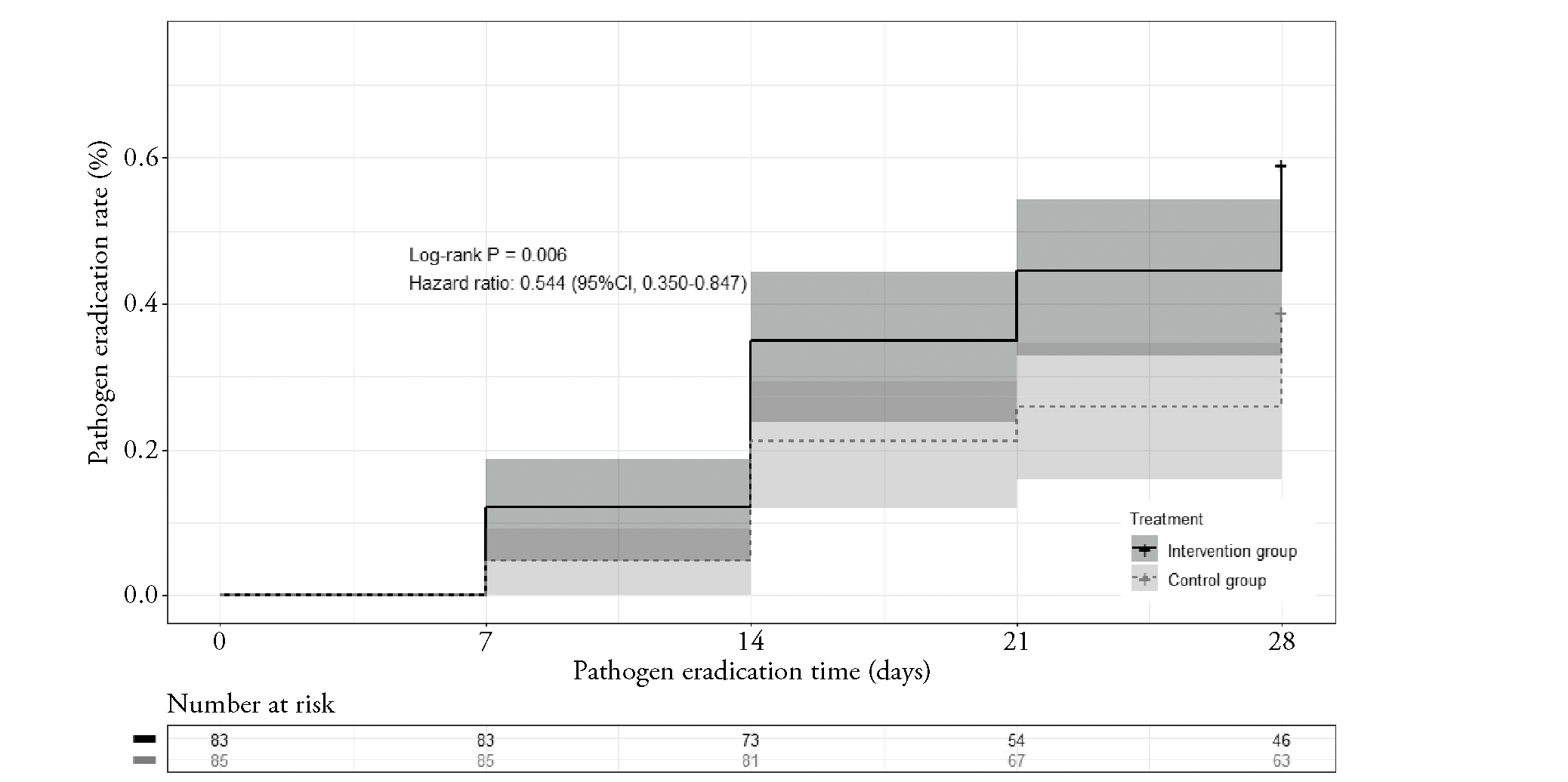
Figure 2 Kaplan-Meier curves of pathogen eradication rate after medication CI: confidence interval. Intervention group: conventional Western Medicine therapy and BZYQ (200 mL, oral or nasal feeding, once a day for 28 d); control group: conventional Western Medicine therapy. The group comparison was performed using the long-rank test. BZYQ: Buzhong Yiqi decoction.
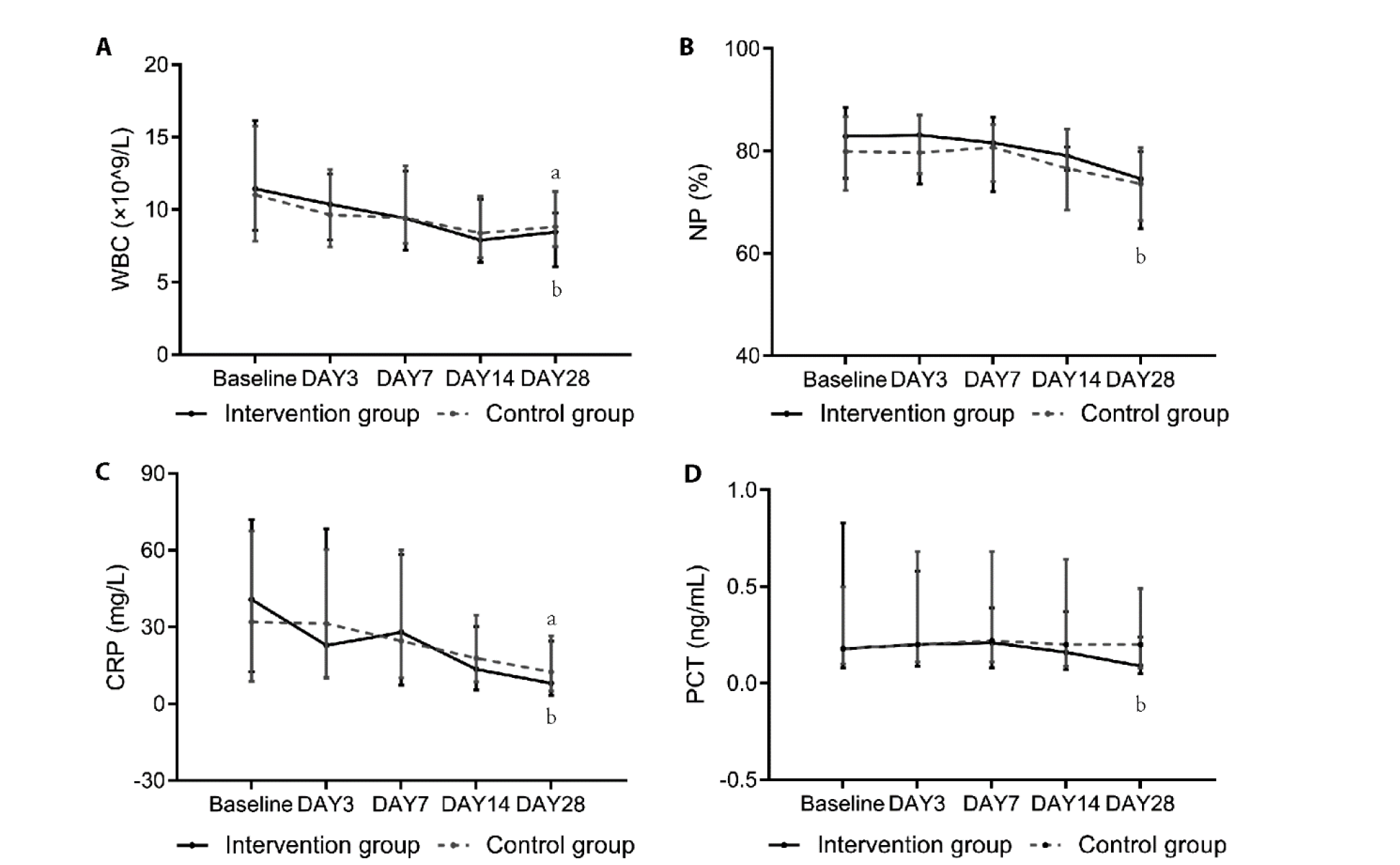
Figure 3 Dynamic changes of infection indicators [Medium (IQR)] IQR: interquartile range; WBC: white blood cell; NP: neutrophil percentage; CRP: C-reactive protein; PCT: procalcitonin; Intervention group: conventional Western Medicine therapy and BZYQ (200 mL, oral or nasal feeding, once a day for 28 d) (n = 83); control group: conventional Western Medicine therapy (n = 85). Difference between day 28 and baseline was tested by a linear mixed model. Comparison of baseline vs post-treatment, aP < 0.05 for the control group. bP < 0.05 for the intervention group.
| Any adverse events | Control group (n = 85) | Intervention group (n = 83) |
|---|---|---|
| Stomachache | 2 (2.4) | 0 |
| Diarrhea | 3 (3.5) | 0 |
| Leukopenia | 7 (8.2) | 5 (6.0) |
| Anemia | 10 (11.8) | 12 (14.5) |
| Thrombocytopenia | 6 (7.1) | 5 (6.0) |
| Hypoproteinemia | 18 (21.2) | 17 (20.5) |
| Increased alanine aminotransferase | 13 (15.3) | 13 (15.7) |
| Increased aspartate aminotransferase | 12 (14.1) | 14 (16.9) |
| Increased blood bilirubin | 4 (4.7) | 4 (4.8) |
| Increased creatinine | 4 (4.7) | 7 (8.4) |
Table 3 Summary of adverse events [n (%)]
| Any adverse events | Control group (n = 85) | Intervention group (n = 83) |
|---|---|---|
| Stomachache | 2 (2.4) | 0 |
| Diarrhea | 3 (3.5) | 0 |
| Leukopenia | 7 (8.2) | 5 (6.0) |
| Anemia | 10 (11.8) | 12 (14.5) |
| Thrombocytopenia | 6 (7.1) | 5 (6.0) |
| Hypoproteinemia | 18 (21.2) | 17 (20.5) |
| Increased alanine aminotransferase | 13 (15.3) | 13 (15.7) |
| Increased aspartate aminotransferase | 12 (14.1) | 14 (16.9) |
| Increased blood bilirubin | 4 (4.7) | 4 (4.8) |
| Increased creatinine | 4 (4.7) | 7 (8.4) |
| 1. |
Torres A, Niederman MS, Chastre J, et al. International ERS/ESICM/ESCMID/ALAT guidelines for the management of hospital-acquired pneumonia and ventilator-associated pneumonia: Guidelines for the management of hospital-acquired pneumonia (HAP)/ventilator-associated pneumonia (VAP) of the European Respiratory Society (ERS), European Society of Intensive Care Medicine (ESICM), European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and Asociación Latinoamericana del Tórax (ALAT). Eur Respir J 2017; 50: 1700582.
DOI URL |
| 2. |
Scott RD, Culler SD, Rask KJ. Understanding the economic impact of health care-associated infections: a cost perspective analysis. J Infus Nurs 2019; 42: 61-9.
DOI PMID |
| 3. |
Cilloniz C, Dominedo C, Torres A. An overview of guidelines for the management of hospital-acquired and ventilator-associated pneumonia caused by multidrug-resistant Gram-negative bacteria. Curr Opin Infect Dis 2019; 32: 656-62.
DOI PMID |
| 4. | Centers for Disease Control and Prevention. Antimicrobial Resistance. 2022-10-05, cited 2023-01-20. Available from URL: https://www.cdc.gov/drugresistance/about.html. |
| 5. | China Antimicrobial Resistence Surveillance System. National Bacterial Resistance Surveillance Report. 2020-11-19, cited 2022-05-14. Available from URL: http://www.carss.cn/Report/Details/770. |
| 6. |
Lakbar I, Medam S, Ronflé R, et al. Association between mortality and highly antimicrobial-resistant bacteria in intensive care unit-acquired pneumonia. Sci Rep 2021; 11: 16497.
DOI PMID |
| 7. |
Paramythiotou E, Routsi C. Association between infections caused by multidrug-resistant gram-negative bacteria and mortality in critically ill patients. World J Crit Care Med 2016; 5: 111-20.
DOI PMID |
| 8. | World Health Organization. Antibiotic resistance. 2020-07-31, cited 2022-05-14. 2022-05-14. Available from URL: https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance. |
| 9. |
Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis 2016; 63: e61-111.
DOI URL |
| 10. |
Martin-Loeches I, Torres A, Rinaudo M, et al. Resistance patterns and outcomes in intensive care unit (ICU)-acquired pneumonia. Validation of European Centre for Disease Prevention and Control (ECDC) and the Centers for Disease Control and Prevention (CDC) classification of multidrug resistant organisms. J Infect 2015; 70: 213-22.
DOI PMID |
| 11. | Yang WQ, Zhao ZX, Xu Y, Liu J, Xue JX, Guan SL. Advances in the use of constituents of Traditional Chinese Medicines to inhibit multidrug-resistant bacteria. Zhong Guo Bing Yuan Sheng Wu Xue Za Zhi 2021; 16: 734-7. |
| 12. |
Su T, Qiu Y, Hua XS, et al. Novel opportunity to reverse antibiotic resistance: to explore Traditional Chinese Medicine with potential activity against antibiotics-resistance bacteria. Front Microbiol 2020; 11: 610070.
DOI URL |
| 13. | Yin YY, Yue ZD, Ding Y, Yu SH. Study on the mechanism of Buzhong Yiqi decoction on the recovery period of COVID-19 based on network pharmacology. Shandong Zhong Yi Yao Da Xue Xue Bao 2021; 45: 685-94. |
| 14. | An HM. Efficiency of Traditional Chinese Medicine modified Buzhong Yiqi decoction on the prognosis of patients with ventilator-associated pneumonia. Zhong Xi Yi Jie He Xin Xue Guan Bing Dian Zi Za Zhi 2020; 8: 171. |
| 15. | Kou LJ, Liu QQ, Jiang QM, et al. Clinical study on the effect of modified Buzhong Yiqi decoction on the prognosis of patients with ventilator-associated pneumonia. Zhong Guo Zhong Yi Ji Zheng 2011; 20: 202+208. |
| 16. | Peng MH, Chen XB, Ruan XM, Zhang T. Curative effect of Buzhong Yiqi decoction in the treatment of multidrug-resistant pneumonia after stroke. Shi Yong Zhong Yi Yao Za Zhi 2019; 35: 1457-8. |
| 17. |
Kohno J, Kawamura T, Kikuchi A, Akaishi T, Takayama S, Ishii T. A Japanese traditional medicine Hochuekkito promotes negative conversion of vancomycin-resistant Enterococci. Sci Rep 2021; 11: 11300.
DOI PMID |
| 18. |
Deng D, Chen ZY, Jia LY, et al. Treatment of hospital-acquired pneumonia with multi-drug resistant organism by Buzhong Yiqi decoction based on Fuzheng Quxie classical prescription: study protocol for a randomized controlled trial. Trials 2019; 20: 817.
DOI PMID |
| 19. | Infections Group, Chinese Society of Respiratory, Chinese Medical Association. Guidelines for the diagnosis and treatment of hospital-acquired pneumonia and ventilator-associated pneumonia in Chinese adults (2018 edition). Zhong Hua Jie He He Hu Xi Za Zhi 2018; 41: 255-80. |
| 20. | Lyu HH. Clinical study on Buzhong Yiqi decoction combined with Western Medicine in treating stroke-associated pneumonia. Zhong Guo Zhong Yi Yao Xian Dai Yuan Cheng Jiao Yu 2018, 16: 115-7. |
| 21. |
Gulick AM. Nonribosomal peptide synthetase biosynthetic clusters of ESKAPE pathogens. Nat Prod Rep 2017; 34: 981-1009.
DOI PMID |
| 22. |
Yin Y, Zhao C, Li H, et al. Clinical and microbiological characteristics of adults with hospital-acquired pneumonia: a 10-year prospective observational study in China. Eur J Clin Microbiol Infect Dis 2021; 40: 683-90.
DOI |
| 23. |
Martin-Loeches I, Rodriguez AH, Torres A. New guidelines for hospital-acquired pneumonia/ventilator-associated pneumonia: USA vs Europe. Curr Opin Crit Care 2018; 24: 347-52.
DOI PMID |
| 24. | Chen G, Xu KL, Sun FY, Sun YX, Kong ZY, Fang BJ. Risk factors of multidrug-resistant bacteria in lower respiratory tract infections: a systematic review and Meta-analysis. Can J Infect Dis Med Microbiol 2020; 2020: 7268519. |
| 25. |
Torres A, Ewig S, Lode H, Carlet J. Defining, treating and preventing hospital acquired pneumonia: European perspective. Intensive Care Med 2009; 35: 9-29.
DOI PMID |
| 26. |
Wetzel C, Lonneman M, Wu C. Polypharmacological drug actions of recently FDA approved antibiotics. Eur J Med Chem 2021; 209: 112931.
DOI URL |
| 27. |
Brown DG, Wobst HJ. A decade of FDA-approved drugs (2010-2019): trends and future directions. J Med Chem 2021; 64: 2312-38.
DOI PMID |
| 28. |
Khaksarian M, Bahmani M, Taherikalani M, Ashrafi B, Rafieian-Kopaei M, Abbasi N. Biosynthesis of titanium dioxide nanoparticles using Hypericum perforatum and Origanum vulgare extracts and their main components, hypericin and carvacrol as promising antibacterial agents. J Tradit Chin Med 2022; 42: 167-75.
DOI PMID |
| 29. | Cai Y, Zhang Q, Fu Y, et al. Effectiveness of Chinese herbal medicine combined with antibiotics for extensively drug-resistant enterobacteria and nonfermentative bacteria infection: real-life experience in a retrospective cohort. Biomed Res Int 2017; 2017: 2897045. |
| 30. |
Yamada T, Wajima T, Nakaminami H, Kobayashi K, Ikoshi H, Noguchi N. The modified Gingyo-san, a Chinese herbal medicine, has direct antibacterial effects against respiratory pathogens. BMC Complement Altern Med 2016; 16: 463.
DOI URL |
| 31. | Wei Q, Bhasme P, Wang Z, et al. Chinese medicinal herb extract inhibits PQS-mediated quorum sensing system in Pseudomonas aeruginosa. BMC Complement Altern Med 2020; 248: 112272. |
| 32. |
Hou Y, Nie Y, Cheng B, et al. Qingfei Xiaoyan Wan, a Traditional Chinese Medicine formula, ameliorates Pseudomonas aeruginosa-induced acute lung inflammation by regulation of PI3K/AKT and Ras/MAPK pathways. Acta pharmaceutica Sinica B 2016; 6: 212-21.
DOI PMID |
| 33. |
Baillie JK, Digard P. Influenza--time to target the host? N Engl J Med 2013; 369: 191-3.
DOI URL |
| 34. | Takayama S, Kikuchi A, Makino T, Kainuma M, Namiki T, Ito T. Basic pharmacological mechanisms and clinical evidence of the efficacy of hochuekkito against infectious diseases and its potential for use against COVID-19. Trad Kampo Med 2021; 8: 3-21. |
| [1] | QIN Xiaoyu, WANG Chunai, XUE Jianjun, ZHANG Jie, LU Xiaoting, DING Shengshuang, GE Long, WANG Minzhen. Efficacy of electroacupuncture on myocardial protection and postoperative rehabilitation in patients undergoing cardiac surgery with cardiopulmonary bypass: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 1-15. |
| [2] | DAI Xiaoling, ZHANG Anming, LIN Hui, SHI Bei, REN Yi, WEN Hongzhu, FEI Xiaoyan, LIN Jiang. Qingchang suppositry (清肠栓) induced remission in patients with mild-to-moderate ulcerative proctitis: a multicenter, prospective, randomized, parallel-controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 156-162. |
| [3] | WANG Yichen, WU Shiyi, WANG Zhengyan, CHANG Wenling, XIE Zhihao, TANG Xing, ZHAO Songmei, ZHOU Jing, CHEN Zehong, WANG Chao, YANG Chunxia. Efficacy of Zhumian Tang formula granules (助眠汤配方颗粒) combined with eszopiclone for the treatment of poor sleep quality: a multi-center, randomized controlled, superiority trial [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 163-171. |
| [4] | YANG Yi, YE Huijun, ZHENG Huiling, JIN Lihua. Clinical observation on 90 cases of primary dysmenorrhea treated by buccal acupuncture therapy: a randomized controlled study [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 172-181. |
| [5] | DAI Zeqi, LIAO Xing, GUAN Yueyue, ZENG Zixiu, TANG Jun, HU Jing. Bloodletting puncture in the treatment of acute ischemic stroke: protocol for a mixed-method study of a multi-center randomized controlled trial and focus group [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1259-1267. |
| [6] | XU Yani, ZHANG Yutong, HE Weile, DAI Linglin, TANG Ding, WANG Jialing, ZHANG Xufen, CHEN Qin, CHEN Lifang, WANG Zhanglian, ZHAN Mingjie. Efficiency and safety of acupuncture for women with premature ovarian insufficiency: study protocol for a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1268-1274. |
| [7] | ZHAO Ming, LUO Yimiao, WANG Huichan, CAO Yu, MA Lina, PEI Hui, LI Hao. Guilingji capsule (龟龄集胶囊) for Alzheimer's disease: secondary analysis of a randomized non-inferiority controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1019-1025. |
| [8] | ZHANG Meizhen, HAO Xiaohui, TANG Yiting, CHEN Yupeng, HE Puyu, ZHAO Liming, PANG Bing, NI Qing. Efficacy and safety of Buyang Huanwu decoction (补阳还五汤) for diabetic peripheral neuropathy: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 841-850. |
| [9] | YANG Yuqing, CHEN Yuhuan, LI Chunxiao, LING Xiao, WANG Panpan, GUO Jing, ZHANG Yingying. Effectiveness and safety of Pingxiao capsule (平消胶囊) as adjuvant therapy in treatment of breast cancer: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 851-859. |
| [10] | LI Zhihao, HAN Wenjun, SONG Xiuling, LI Yan, CHEN Yuelai. Electroacupuncture stimulating Zhongji (CV3), Guanyuan (CV4), and bilateral Dahe (KI12) attenuates inflammation in rats with chronic nonbacterial prostatitis induced by estradiol through inhibiting toll-like receptor 4 pathway [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 963-972. |
| [11] | WANG Chao, WU Qiong, LI Ping, WANG Zhigang, LOU Xusheng, LI Yuanyuan, ZHANG Lin. Effect of Traditional Chinese Medicine combined with Western Medicine on blood lipid levels and inflammatory factors in patients with angina pectoris in coronary heart disease identified as intermingled phlegm and blood stasis syndrome: a network Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 640-649. |
| [12] | HAN Yunpeng, YU Wentao, ZHANG Ying, XU Huazhou, DENG Guoxing, FANG Chaoyi. Qinghua decoction (清化饮) improves chronic nonbacterial prostatitis possibly via regulating the chromogranin A/nerve growth factor/tyrosine kinase A signaling pathway mediated by inflammatory factors [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 695-703. |
| [13] | ZHANG Xinghe, LI Qifu, YI Rong, XING Chonghui, JIN Yuhao, MENG Jiangqiong, FENG Jialei, ZHAO Siwen, LIANG Fanrong, GUO Taipin. Effect of catgut embedding at acupoints versus non-acupoints in abdominal obesity: a randomized clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 780-786. |
| [14] | JIA Lihua, KUANG Haodan, XU Yuan. Efficacy of Buzhong Yiqi decoction (补中益气汤) on benign prostatic hyperplasia and its possible mechanism [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 533-541. |
| [15] | ZHANG Yuehong, DONG Dandan, YAN Youqin, ZHANG Hao, WANG Guangli, ZHOU Wei, LI Wei, QIU Li, LI Tingming, LIU Quan, XIA Ping, MAO Lina, YANG Danlin, YANG Lu, LIAN Fengmei, TONG Xiaolin, BA Yuanming. Effectiveness and safety of Jinshuibao capsules (金水宝胶囊) in treatment of residual cardiopulmonary symptoms in convalescent patients of coronavirus disease 2019: a pilot randomized, double-blind, placebo-controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 134-139. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||