Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (4): 770-783.DOI: 10.19852/j.cnki.jtcm.20240610.002
• Original articles • Previous Articles Next Articles
Uncovering the action mechanism of Shenqi Tiaoshen formula (参芪调肾方) in the treatment of chronic obstructive pulmonary disease through network pharmacology, molecular docking, and experimental verification
YANG Qinjun1,2, YIN Dandan3, WANG Hui1, GAO Yating2,4,5, WANG Xinheng1,2, WU Di1,2, TONG Jiabing2,4,5, WANG Chuanbo6( ), LI Zegeng2,4,5(
), LI Zegeng2,4,5( )
)
- 1 School of Chinese Medicine, Anhui University of Chinese Medicine, Hefei 230038, China
2 Anhui Province Key Laboratory of the Application and Transformation of Traditional Chinese Medicine in the Prevention and Treatment of Major Pulmonary Diseases, Hefei 230031, China
3 Department of Pharmacy, the First Affiliated Hospital of Anhui Medical University, Hefei 230022, China
4 Institute of Traditional Chinese Medicine Respiratory Disease Prevention and Control, Anhui Academy of Traditional Chinese Medicine, Hefei 230031, China
5 Department of Respiratory, the First Affiliated Hospital of Anhui University of Chinese Medicine, Hefei 230031, China
6 Department of Chinese Medicine, the Second Affiliated Hospital of Anhui Medical University, Hefei 230601, China
-
Received:2023-02-22Accepted:2023-06-15Online:2024-08-15Published:2024-06-10 -
Contact:Prof. LI Zegeng, Department of Respiratory, the First Affiliated Hospital of Anhui University of Chinese Medicine, Hefei 230031, China.ahzyfb@sina.com ; Chief physician WANG Chuanbo, Department of Chinese Medicine, the Second Affiliated Hospital of Anhui Medical University, Hefei 230601, China.wangcb120@163.com Telephone: +86-13805516609 -
Supported by:Joint Key Project of the National Natural Science Foundation of China: the Molecular Mechanism Research on Xin'an Medical's Syndrome Differentiation of "Guben Peiyuan" in Improving Self-Perception of Chronic Obstructive Pulmonary Disease (COPD) Patients by the Regulation of Inflammation-Immunity Network(U20A20398);Major Science and Technology Project of Anhui Province: Development of a Comprehensive Management Platform for COPD based on Cloud Data and Research on the Promotion of Traditional Chinese Medicine Techniques(201903a0702015);Collaborative Innovation Project of Public Health in Provincial Medical Colleges: Research on the Intervention Mechanisms and Product Development of Xinan Medical's “Guben Peiyuan” Method and “Yangyin Tongluo” Method for Major Chronic Diseases based on Evidence-based Medicine Evaluation(GXXT-2020-025)
Cite this article
YANG Qinjun, YIN Dandan, WANG Hui, GAO Yating, WANG Xinheng, WU Di, TONG Jiabing, WANG Chuanbo, LI Zegeng. Uncovering the action mechanism of Shenqi Tiaoshen formula (参芪调肾方) in the treatment of chronic obstructive pulmonary disease through network pharmacology, molecular docking, and experimental verification[J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 770-783.
share this article
| MOL Id | Name | Degree | BC | CC | TC | Herbs |
|---|---|---|---|---|---|---|
| MOL000098 | Quercetin | 72 | 0.04720241 | 0.45654835 | 0.12767094 | Renshen (Radix Ginseng), Huangqi (Radix Astragali Mongolici), Shanzhuyu (Fructus Macrocarpiii) |
| MOL000006 | Luteolin | 42 | 0.0199764 | 0.41306755 | 0.15736827 | Semen Persicae |
| MOL005828 | Nobiletin | 38 | 0.00902303 | 0.401507 | 0.16578947 | Chenpi (Pericarpium Citri Reticulataei) |
| MOL000422 | Kaempferol | 30 | 0.01607944 | 0.41398446 | 0.18351648 | Renshen (Radix Ginseng), Huangqi (Radix Astragali Mongolici),Taoren (Semen Persicae) |
| MOL005320 | Arachidonic Acid | 29 | 0.02892894 | 0.41398446 | 0.163746 | Renshen (Radix Ginseng), Baijiezi (Semen Sinapis) |
| MOL000546 | Diosgenin | 24 | 0.00554195 | 0.39554613 | 0.18587824 | Renshen (Radix Ginseng) |
| MOL000358 | Sitosterol | 21 | 0.01046093 | 0.38414006 | 0.16056166 | Chenpi (Pericarpium Citri Reticulataei), Yizhiren (Fructus Alpiniae Oxyphyllae) |
| MOL001924 | Paeoniflorin | 21 | 0.00274493 | 0.3569378 | 0.22208884 | Huangqi (Radix Astragali Mongolici) |
| MOL008583 | Beta-Sitosterol | 20 | 0.01385171 | 0.40411701 | 0.19143646 | Renshen (Radix Ginseng), Huangqi (Radix Astragali Mongolici), Chenpi (Pericarpium Citri Reticulataei), Taoren ( Semen Persicae) |
| MOL007088 | Cryptotanshinone | 19 | 0.00420619 | 0.39139559 | 0.20705957 | Renshen (Radix Ginseng) |
| MOL001638 | Phorbol | 19 | 0.00257052 | 0.35899904 | 0.21307301 | Renshen (Radix Ginseng) |
| MOL005344 | Ginsenoside Rh2 | 18 | 0.00369042 | 0.38813736 | 0.22120596 | Renshen (Radix Ginseng) |
| MOL000392 | Formononetin | 18 | 0.00318226 | 0.38100102 | 0.22580645 | Huangqi (Radix Astragali Mongolici) |
| MOL007424 | Artemisinin | 17 | 0.00337229 | 0.37412237 | 0.19847939 | Renshen (Radix Ginseng), Huangqi (Radix Astragali Mongolici) |
| MOL005320 | Arachidonate | 17 | 0.00283547 | 0.38493292 | 0.21191085 | Renshen (Radix Ginseng) |
| MOL000492 | (+)-Catechin | 16 | 0.00888835 | 0.39139559 | 0.22904412 | Taoren (Semen Persicae), Rougui (Cortex Cinnamomi Cassia) |
| MOL002341 | Hesperetin | 15 | 0.0014985 | 0.37714863 | 0.27407407 | Chenpi (Pericarpium Citri Reticulataei) |
| MOL000354 | Isorhamnetin | 12 | 0.00370155 | 0.3786802 | 0.26265823 | Huangqi (Radix Astragali Mongolici), Shanzhuyu (Shanzhuyu (Fructus Macrocarpiii) |
| MOL000737 | Morin | 11 | 0.0019528 | 0.3786802 | 0.30074328 | Renshen (Radix Ginseng) |
| MOL004328 | naringenin | 10 | 9.95E-04 | 0.36532811 | 0.25774648 | Chenpi (Pericarpium Citri Reticulataei) |
Table 1 Key active ingredients and their topological parameters of SQTS in the treatment of COPD
| MOL Id | Name | Degree | BC | CC | TC | Herbs |
|---|---|---|---|---|---|---|
| MOL000098 | Quercetin | 72 | 0.04720241 | 0.45654835 | 0.12767094 | Renshen (Radix Ginseng), Huangqi (Radix Astragali Mongolici), Shanzhuyu (Fructus Macrocarpiii) |
| MOL000006 | Luteolin | 42 | 0.0199764 | 0.41306755 | 0.15736827 | Semen Persicae |
| MOL005828 | Nobiletin | 38 | 0.00902303 | 0.401507 | 0.16578947 | Chenpi (Pericarpium Citri Reticulataei) |
| MOL000422 | Kaempferol | 30 | 0.01607944 | 0.41398446 | 0.18351648 | Renshen (Radix Ginseng), Huangqi (Radix Astragali Mongolici),Taoren (Semen Persicae) |
| MOL005320 | Arachidonic Acid | 29 | 0.02892894 | 0.41398446 | 0.163746 | Renshen (Radix Ginseng), Baijiezi (Semen Sinapis) |
| MOL000546 | Diosgenin | 24 | 0.00554195 | 0.39554613 | 0.18587824 | Renshen (Radix Ginseng) |
| MOL000358 | Sitosterol | 21 | 0.01046093 | 0.38414006 | 0.16056166 | Chenpi (Pericarpium Citri Reticulataei), Yizhiren (Fructus Alpiniae Oxyphyllae) |
| MOL001924 | Paeoniflorin | 21 | 0.00274493 | 0.3569378 | 0.22208884 | Huangqi (Radix Astragali Mongolici) |
| MOL008583 | Beta-Sitosterol | 20 | 0.01385171 | 0.40411701 | 0.19143646 | Renshen (Radix Ginseng), Huangqi (Radix Astragali Mongolici), Chenpi (Pericarpium Citri Reticulataei), Taoren ( Semen Persicae) |
| MOL007088 | Cryptotanshinone | 19 | 0.00420619 | 0.39139559 | 0.20705957 | Renshen (Radix Ginseng) |
| MOL001638 | Phorbol | 19 | 0.00257052 | 0.35899904 | 0.21307301 | Renshen (Radix Ginseng) |
| MOL005344 | Ginsenoside Rh2 | 18 | 0.00369042 | 0.38813736 | 0.22120596 | Renshen (Radix Ginseng) |
| MOL000392 | Formononetin | 18 | 0.00318226 | 0.38100102 | 0.22580645 | Huangqi (Radix Astragali Mongolici) |
| MOL007424 | Artemisinin | 17 | 0.00337229 | 0.37412237 | 0.19847939 | Renshen (Radix Ginseng), Huangqi (Radix Astragali Mongolici) |
| MOL005320 | Arachidonate | 17 | 0.00283547 | 0.38493292 | 0.21191085 | Renshen (Radix Ginseng) |
| MOL000492 | (+)-Catechin | 16 | 0.00888835 | 0.39139559 | 0.22904412 | Taoren (Semen Persicae), Rougui (Cortex Cinnamomi Cassia) |
| MOL002341 | Hesperetin | 15 | 0.0014985 | 0.37714863 | 0.27407407 | Chenpi (Pericarpium Citri Reticulataei) |
| MOL000354 | Isorhamnetin | 12 | 0.00370155 | 0.3786802 | 0.26265823 | Huangqi (Radix Astragali Mongolici), Shanzhuyu (Shanzhuyu (Fructus Macrocarpiii) |
| MOL000737 | Morin | 11 | 0.0019528 | 0.3786802 | 0.30074328 | Renshen (Radix Ginseng) |
| MOL004328 | naringenin | 10 | 9.95E-04 | 0.36532811 | 0.25774648 | Chenpi (Pericarpium Citri Reticulataei) |
| No. | Genes | MCC Score | Degree | BC | CC | No. | Genes | MCC Score | Degree | BC | CC |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | IL6 | 35984 | 37 | 1372.918 | 0.088484 | 16 | MMP3 | 15176 | 18 | 630.5442 | 0.086957 |
| 2 | IL-10 | 35462 | 35 | 1809.186 | 0.08755 | 17 | SRC | 14002 | 57 | 5644.229 | 0.090909 |
| 3 | STAT3 | 31522 | 61 | 5542.987 | 0.090909 | 18 | PTPN11 | 12264 | 47 | 3391.096 | 0.08941 |
| 4 | FGF2 | 30368 | 23 | 479.668 | 0.087632 | 19 | HGF | 12036 | 26 | 536.7057 | 0.088015 |
| 5 | MMP9 | 30354 | 20 | 776.8301 | 0.087388 | 20 | EGFR | 11516 | 34 | 1353.179 | 0.089241 |
| 6 | MMP1 | 30270 | 19 | 1158.218 | 0.086689 | 21 | TGFB1 | 10140 | 17 | 667.4372 | 0.088042 |
| 7 | VEGFA | 21138 | 33 | 2017.926 | 0.089184 | 22 | HRAS | 8178 | 37 | 1310.815 | 0.088847 |
| 8 | CREBBP | 20170 | 40 | 6223.5 | 0.089952 | 23 | CTNNB1 | 7684 | 40 | 2212.381 | 0.089552 |
| 9 | EP300 | 19440 | 46 | 3923.157 | 0.089952 | 24 | TNF | 7342 | 38 | 2301.604 | 0.089156 |
| 10 | ESR1 | 18860 | 27 | 592.0106 | 0.088959 | 25 | MAPK1 | 7254 | 50 | 4258.772 | 0.090269 |
| 11 | AKT1 | 18046 | 51 | 5545.408 | 0.090211 | 26 | TP53 | 6782 | 48 | 4923.421 | 0.089638 |
| 12 | MYC | 16968 | 21 | 437.7729 | 0.088291 | 27 | IL1B | 6708 | 26 | 1063.439 | 0.087878 |
| 13 | HIF1A | 15504 | 23 | 755.6755 | 0.088374 | 28 | CXCL8 | 6672 | 17 | 281.1361 | 0.087225 |
| 14 | FOXO3 | 15434 | 24 | 1057.897 | 0.088624 | 29 | ERBB2 | 5858 | 20 | 298.4991 | 0.087687 |
| 15 | MMP2 | 15200 | 17 | 326.8929226 | 0.086769231 | 30 | JAK21 | 5259 | 20 | 298.4991 | 0.087687 |
Table 2 Hub genes and topological parameters of SQTS in the treatment of COPD
| No. | Genes | MCC Score | Degree | BC | CC | No. | Genes | MCC Score | Degree | BC | CC |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | IL6 | 35984 | 37 | 1372.918 | 0.088484 | 16 | MMP3 | 15176 | 18 | 630.5442 | 0.086957 |
| 2 | IL-10 | 35462 | 35 | 1809.186 | 0.08755 | 17 | SRC | 14002 | 57 | 5644.229 | 0.090909 |
| 3 | STAT3 | 31522 | 61 | 5542.987 | 0.090909 | 18 | PTPN11 | 12264 | 47 | 3391.096 | 0.08941 |
| 4 | FGF2 | 30368 | 23 | 479.668 | 0.087632 | 19 | HGF | 12036 | 26 | 536.7057 | 0.088015 |
| 5 | MMP9 | 30354 | 20 | 776.8301 | 0.087388 | 20 | EGFR | 11516 | 34 | 1353.179 | 0.089241 |
| 6 | MMP1 | 30270 | 19 | 1158.218 | 0.086689 | 21 | TGFB1 | 10140 | 17 | 667.4372 | 0.088042 |
| 7 | VEGFA | 21138 | 33 | 2017.926 | 0.089184 | 22 | HRAS | 8178 | 37 | 1310.815 | 0.088847 |
| 8 | CREBBP | 20170 | 40 | 6223.5 | 0.089952 | 23 | CTNNB1 | 7684 | 40 | 2212.381 | 0.089552 |
| 9 | EP300 | 19440 | 46 | 3923.157 | 0.089952 | 24 | TNF | 7342 | 38 | 2301.604 | 0.089156 |
| 10 | ESR1 | 18860 | 27 | 592.0106 | 0.088959 | 25 | MAPK1 | 7254 | 50 | 4258.772 | 0.090269 |
| 11 | AKT1 | 18046 | 51 | 5545.408 | 0.090211 | 26 | TP53 | 6782 | 48 | 4923.421 | 0.089638 |
| 12 | MYC | 16968 | 21 | 437.7729 | 0.088291 | 27 | IL1B | 6708 | 26 | 1063.439 | 0.087878 |
| 13 | HIF1A | 15504 | 23 | 755.6755 | 0.088374 | 28 | CXCL8 | 6672 | 17 | 281.1361 | 0.087225 |
| 14 | FOXO3 | 15434 | 24 | 1057.897 | 0.088624 | 29 | ERBB2 | 5858 | 20 | 298.4991 | 0.087687 |
| 15 | MMP2 | 15200 | 17 | 326.8929226 | 0.086769231 | 30 | JAK21 | 5259 | 20 | 298.4991 | 0.087687 |
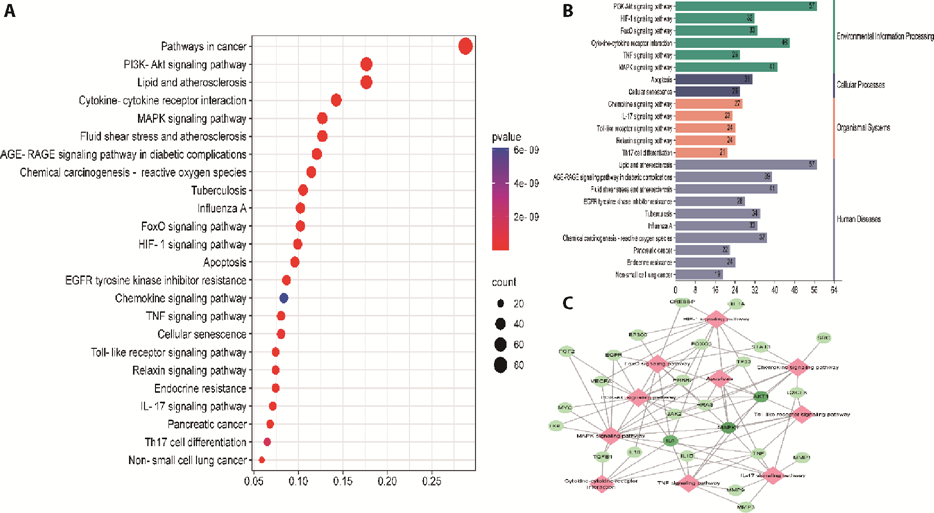
Figure 1 KEGG enrichment analysis, pathway classification summary, and the key pathway core genes A: KEGG enrichment analysis. B: classification summary of the KEGG-enrichment pathway. C: core genes of the key enrichment pathways. KEGG: Kyoto Encyclopedia of Genes and Genomes.
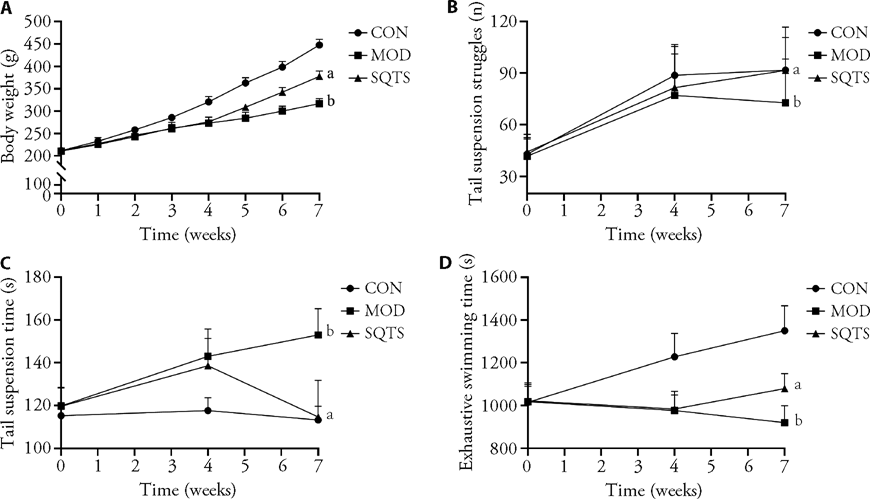
Figure 2 Effects of body weight, exhaustive swimming test and tail suspension test of rats in each group A: body weight; B: tail suspension struggle times; C: tail suspension immobility time. D: Exhaustive swimming time. CON group: normal saline 1 mL/kg, 21 d; MOD group: normal saline 1 mL/kg, 21 d; SQTS group: SQTS 14.94 g/kg, 21 d. CON: Control group; MOD: model group; SQTS: Shenqi Tiaoshen formula. The data represent the mean ± standard deviation using one-way analysis of variance (n = 6). aP < 0.05 vs model group; bP < 0.05 vs control group.

Figure 3 Changes of lung tissue pathology, lung functions and plasma inflammatory factors of the rats in each group A: HE staining (×200, Scale bar = 100 μm). A1: CON group; A2: MOD group; A3: SQTS group. B: the airway inflammation score; C: the MAN score; D: the MLI score; E: the FVC change; F: the FEV0.1/FVC% change; G: the Cdyn change; H: the RE change; I: plasma levels of IL-13; J: plasma levels of IFN-γ; K: plasma levels of TNF-α; L: plasma levels of interleukin-1β; M: plasma levels of IL-10. CON group: normal saline 1 mL/kg, 21 d; MOD group: normal saline 1 mL/kg, 21 d; SQTS group: SQTS 14.94 g/kg, 21 d. HE: hematoxylin-eosin; CON: control; MOD: model; SQTS: Shenqi Tiaoshen Formula; MAN: mean alveolar numbers score; MLI: mean linear interval; FVC: forced expiratory vital capacity; FEV0.1/FVC%: 0.1 s forced expiratory volume/forced expiratory vital capacity; Cdyn: pulmonary dynamic compliance; RE: resistance of expiration; IL-13: interleukin-13; IFN-γ: interferon-γ; TNF-α: tumor necrosis factor-α; IL-1β: interleukin-1β; IL-10: interleukin-10. The Data represent the mean ± standard deviation using one-way analysis of variance (n = 6). aP < 0.05 vs control group; bP < 0.05 vs model group.

Figure 4 Core proteins of the KEGG enrichment pathway were detected by Western blotting A: Western blotting was performed to analyze the expression of p-PI3K, p-AKT, p-mTOR, HIF-1α, FoXO3α, TLR4, VEGFA, caspase-3 and MMP9 in the lung tissues. B-J: image J software was used to evaluate the protein expression quantitatively. B: relative expression of p-PI3K; C: relative expression of p-AKT; D: relative expression of p-mTOR; E: relative expression of HIF-1α; F: relative expression of FoxO3α; G: relative expression of TLR4; H: relative expression of VEGFA; I: relative expression of caspase-3; J: relative expression of MMP9. CON group: normal saline 1mL/kg, 21 d; MOD group: normal saline 1 mL/kg, 21 d; SQTS group: SQTS 14.94 g/kg, 21 d. CON: control; MOD: model; SQTS: Shenqi Tiaoshen Formula; p-PI3K: phospho-phosphatidylinositol 3 kinase; p-AKT: phospho-protein kinase B; p-mTOR: phospho-mammalian target of rapamycin; HIF-1α: hypoxia-inducible factor-1α; FoXO3α: forkhead box O3α; TLR4: toll-like receptor 4; VEGFA: vascular endothelial growth factor A; MMP9: matrix metalloproteinase-9. The Data represent the mean ± standard deviation using one-way analysis of variance (n = 6). aP < 0.05 vs control group; bP < 0.05 vs model group.
| 1. | Rassam P, Pazzianotto-Forti EM, Matsumura U, et al. Impact of cognitive capacity on physical performance in chronic obstructive pulmonary disease patients: a scoping review. Chron Respir Dis 2023; 20: 14799731231163874. |
| 2. |
Christenson SA, Smith BM, Bafadhel M, Putcha N. Chronic obstructive pulmonary disease. Lancet 2022; 399: 2227-42.
DOI PMID |
| 3. | Singh D, Agusti A, Anzueto A, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease:the gold science committee report 2019. Eur Respir J 2019; 53: 1900164. |
| 4. | Nikolaou V, Massaro S, Fakhimi M, Stergioulas L, Price D. COPD phenotypes and machine learning cluster analysis: a systematic review and future research agenda. Respir Med 2020; 171: 106093. |
| 5. |
Wang C, Xu J, Yang L, et al. Prevalence and risk factors of chronic obstructive pulmonary disease in China (the China Pulmonary Health [CPH] study): a national cross-sectional study. Lancet 2018; 391: 1706-17.
DOI PMID |
| 6. | Fens T, Zhou G, Postma MJ, van Puijenbroek EP, van Boven JFM. Economic evaluations of chronic obstructive pulmonary disease pharmacotherapy: how well are the real-world issues of medication adherence, comorbidities and adverse drug-reactions addressed? Expert Opin Pharmacother 2021; 22: 923-35. |
| 7. | Barnes PJ. Oxidative stress in chronic obstructive pulmonary disease. Antioxidants (Basel) 2022; 11: 5. |
| 8. | Chang X, Zhang T, Meng Q, et al. Quercetin improves cardiomyocyte vulnerability to hypoxia by regulating SIRT1/TMBIM6-related mitophagy and endoplasmic reticulum stress. Oxid Med Cell Longev 2021;2021:5529913. |
| 9. | Chang X, Zhao Z, Zhang W, et al. Natural antioxidants improve the vulnerability of cardiomyocytes and vascular endothelial cells under stress conditions: a focus on mitochondrial quality control. Oxid Med Cell Longev 2021; 2021: 6620677. |
| 10. | Ma J, Zheng J, Zhong N, et al. Effects of Yupingfeng granules on acute exacerbations of COPD: a randomized, placebo-controlled study. Int J Chron Obstruct Pulmon Dis 2018; 13: 3107-14. |
| 11. | Han MK, Barreto TA, Martinez FJ, Comstock AT, Sajjan US. Randomised clinical trial to determine the safety of quercetin supplementation in patients with chronic obstructive pulmonary disease. BMJ Open Respiratory Research 2020;7:e000392. |
| 12. | Wang CY, Ding HZ, Tang X, Li ZG. Effect of Liuweibuqi capsules on CD4(+)CD25(+)Foxp3(+) regulatory T cells, helper T cells and lung function in patients with stable chronic obstructive pulmonary disease complicated with lung Qi deficiency. J Thorac Dis 2018; 10: 2700-11. |
| 13. | Wang CB, Wang JQ, Li ZG, et al. Thinking and method of diagnosis and treatment of chronic obstructive pulmonary disease based on classification of Lung Qi deficiency syndrome. Zhong Yi Za Zhi 2013; 54: 1550-52. |
| 14. | Zeng SJ, Wang DY, Zhang ZQ, Hu J, Li ZG. Clinical study of Liuwei Buqi granules combined with seretide in the treatment of patients with copd at stable stage. Nanjing Zhong Yi Yao Da Xue Xue Bao 2019; 35: 16-22. |
| 15. | Wang C, Ding H, Xiao T, Li Z, Gan L. Effect of Liuweibuqi capsules on the balance between MMP-9 and TIMP1 and viability of alveolar macrophages in COPD. Biosci Rep 2017; |
| 16. |
Wang C, Ding H, Tang X, Li Z, Gan L. Effect of liuweibuqi capsules in pulmonary alveolar epithelial cells and copd through JAK/STAT pathway. Cell Physiol Biochem 2017; 43: 743-56.
DOI PMID |
| 17. |
Wu Q, Chen Y, Gu Y, et al. Systems pharmacology-based approach to investigate the mechanisms of Danggui-Shaoyao-san prescription for treatment of Alzheimer's disease. BMC Complement Med Ther 2020; 20: 282.
DOI PMID |
| 18. |
Zhang Y, Li X, Guo C, Dong J, Liao L. Mechanisms of Spica Prunellae against thyroid-associated Ophthalmopathy based on network pharmacology and molecular docking. BMC Complement Med Ther 2020; 20: 229.
DOI PMID |
| 19. | Li X, Tang H, Tang Q, Chen W. Decoding the mechanism of Huanglian Jiedu decoction in treating pneumonia based on network pharmacology and molecular docking. Front Cell Dev Biol 2021;9:638366. |
| 20. | Chen PY, Yu JW, Lu FL, Lin MC, Cheng HF. Differentiating parts of Cinnamomum cassia using LC-qTOF-MS in conjunction with principal component analysis. Biomed Chromatogr 2016; 30: 1449-57. |
| 21. | Missiuro PV, Liu K, Zou L, et al. Information flow analysis of interactome networks. PLoS Comput Biol 2009; 5: e1000350. |
| 22. | Wu F, Li Z, Dong C, et al. Qibai Pingfei capsule alleviates inflammation and oxidative stress in a chronic obstructive pulmonary disease rat model with syndromes of Qi deficiency and phlegm and blood stasis by regulating the SIRT1/FoxO3a pathway. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 2019; 35: 115-20. |
| 23. |
Kirkham PA, Barnes PJ. Oxidative stress in copd. Chest 2013; 144: 266-73.
DOI PMID |
| 24. | Barnes PJ. Oxidative stress-based therapeutics in COPD. Redox Biology 2020; 33: 101544. |
| 25. | Yao CW, Piao MJ, Kim KC, et al. Cytoprotective effects of 6′-O-galloylpaeoniflorin against ultraviolet B radiation-induced cell damage in human keratinocytes. In Vitro Cell Dev Biol Anim 2014; 50: 664-74. |
| 26. |
Wain LV, Shrine N, Artigas MS, et al. Genome-wide association analyses for lung function and chronic obstructive pulmonary disease identify new loci and potential druggable targets. Nat Genet 2017; 49: 416-25.
DOI PMID |
| 27. |
Morris GM, Huey R, Lindstrom W, et al. AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. J Comput Chem 2009; 30: 2785-91.
DOI PMID |
| 28. | Han L, Yan Z, Liu C. Effects of Anshen Dingzhi prescription on tryptophan metabolism pathway and exercise ability in rats with chronic fatigue syndrome. Zhong Yi Za Zhi 2021; 43: 3493-7. |
| 29. | Zhang XF, Xiang SY, Geng WY, et al. Electro-acupuncture regulates the cholinergic anti-inflammatory pathway in a rat model of chronic obstructive pulmonary disease. J Integr Med 2018; 16: 418-26. |
| 30. | Wang Z, Li R, Rui Z. Extracellular matrix promotes proliferation, migration and adhesion of airway smooth muscle cells in a rat model of chronic obstructive pulmonary disease via upregulation of the PI3K/AKT signaling pathway. Mol Med Rep 2018; 18: 3143-52. |
| 31. | Liu L, Qin Y, Cai Z, et al. Effective-components combination improves airway remodeling in COPD rats by suppressing M2 macrophage polarization via the inhibition of mTORC2 activity. Phytomedicine 2021 ;92: 153759. |
| 32. |
Barnes PJ. New anti-inflammatory targets for chronic obstructive pulmonary disease. Nat Rev Drug Discov 2013; 12: 543-59.
DOI PMID |
| 33. |
Nadel JA. Mucous hypersecretion and relationship to cough. Pulm Pharmacol Ther 2013; 26: 510-3.
DOI PMID |
| 34. | Wang W, Wu HB, Wang Q, Zhang YS, Han FX, Li JT. Clinical efficacy of TCM formula of replenishing Qi and resolving phlegm and dispelling stasis on patients with stable chronic obstructive pulmonary disease. Beijing Zhong Yi Yao Da Xue Xue Bao 2015; 38: 777-80. |
| 35. | Ru J, Li P, Wang J, et al. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. J Cheminform 2014; 6: 13. |
| 36. | Wu Y, Zhang F, Yang K, et al. SymMap: an integrative database of traditional Chinese medicine enhanced by symptom mapping. Nucleic Acids Res 2019; 47: D1110-7. |
| 37. |
Barnes PJ. Inflammatory mechanisms in patients with chronic obstructive pulmonary disease. J Allergy Clin Immunol 2016; 138: 16-27.
DOI PMID |
| 38. |
Nambiar S, How SB, Gummer J, Trengove R, Moodley Y. Metabolomics in chronic lung diseases. Respirology 2020; 25: 139-48.
DOI PMID |
| 39. | Ganesan S, Faris AN, Comstock AT, Chattoraj SS, Sajjan US. Quercetin prevents progression of disease in elastase/LPS-exposed mice by negatively regulating MMP expression. Respir Res 2010; 11: 131. |
| 40. | Tripathi A, Kumar B, Sagi SSK. Hypoxia-mediated alterations in pulmonary surfactant protein expressions: beneficial effects of quercetin prophylaxis. Respir Physiol Neurobiol 2021; 291: 103695. |
| 41. |
Liu B, Huang J, Zhang B. Nobiletin protects against murine l-arginine-induced acute pancreatitis in association with downregulating p38MAPK and AKT. Biomed Pharmacother 2016; 81: 104-10.
DOI PMID |
| 42. |
Chen C, Ono M, Takeshima M, Nakano S. Antiproliferative and apoptosis-inducing activity of nobiletin against three subtypes of human breast cancer cell lines. Anticancer Res 2014; 34: 1785-92.
PMID |
| 43. |
Rong X, Xu J, Jiang Y, et al. Citrus peel flavonoid nobiletin alleviates lipopolysaccharide-induced inflammation by activating IL-6/STAT3/FOXO3a-mediated autophagy. Food Funct 2021; 12: 1305-17.
DOI PMID |
| 44. |
Zhang R, Ai X, Duan Y, et al. Kaempferol ameliorates H9N2 swine influenza virus-induced acute lung injury by inactivation of TLR4/MyD88-mediated NF-κB and MAPK signaling pathways. Biomed Pharmacother 2017; 89: 660-72.
DOI PMID |
| 45. | Nam SY, Jeong HJ, Kim HM. Kaempferol impedes IL-32-induced monocyte-macrophage differentiation. Chem Biol Interact 2017; 274: 107-15. |
| 46. | Tang XL, Liu JX, Dong W, Li P, Ren JG. Protective effect of kaempferol on LPS plus ATP-induced inflammatory response in cardiac fibroblasts. Inflammation 2015; 38: 94-101. |
| 47. |
Zdrojewicz Z, Pachura E, Pachura P. The thymus: a forgotten, but very important organ. Adv Clin Exp Med 2016; 25: 369-75.
DOI PMID |
| 48. | Shen ML, Wang CH, Lin CH, Zhou N, Kao ST, Wu DC. Luteolin attenuates airway mucus overproduction via inhibition of the GABAergic system. Sci Rep 2016; 6: 32756. |
| 49. | Mathiyalagan R, Wang C, Kim YJ, et al. Preparation of polyethylene glycol-ginsenoside Rh1 and Rh2 conjugates and their efficacy against lung cancer and inflammation. Molecules 2019; 24; 23. |
| 50. | Yi L, Cui J, Wang W, et al. Formononetin attenuates airway inflammation and oxidative stress in murine allergic asthma. Front Pharmacol 2020; 11: 533841 |
| 51. | Huang J, Chen X, Xie A. Formononetin ameliorates IL‑13‑induced inflammation and mucus formation in human nasal epithelial cells by activating the SIRT1/Nrf2 signaling pathway. Mol Med Rep 2021; 24; 6. |
| 52. | Chukowry PS, Spittle DA, Turner AM. Small airways disease, biomarkers and COPD: where are we? Int J Chron Obstruct Pulmon Dis 2021; 16: 351-65. |
| 53. |
Liu CW, Lee TL, Chen YC, et al. PM(2.5)-induced oxidative stress increases intercellular adhesion molecule-1 expression in lung epithelial cells through the IL-6/AKT/STAT3/NF-κB-dependent pathway. Part Fibre Toxicol 2018; 15: 4.
DOI PMID |
| 54. | Trojani A, Di Camillo B, Bossi LE, et al. Identification of a candidate gene set signature for the risk of progression in IgM MGUS to smoldering/symptomatic waldenström macroglobulinemia (WM) by a comparative transcriptome analysis of B cells and plasma cells. Cancers (Basel) 2021; 13: 8. |
| 55. |
Sun X, Chen L, He Z. PI3K/Akt-Nrf2 and anti-inflammation effect of macrolides in chronic obstructive pulmonary disease. Current Drug Metabolism 2019; 20: 301-4.
DOI PMID |
| 56. | Tsai MJ, Chang WA, Jian SF, Chang KF, Kuo PL. Possible mechanisms mediating apoptosis of bronchial epithelial cells in chronic obstructive pulmonary disease - a next-generation sequencing approach. Pathol Res Pract 2018; 214: 1489-96. |
| 57. | Zhang HX, Yang JJ, Zhang SA, et al. HIF-1α promotes inflammatory response of chronic obstructive pulmonary disease by activating EGFR/PI3K/AKT pathway. Eur Rev Med Pharmacol Sci 2018;22; 18: 6077-84. |
| 58. | Gui DM, Li YF, Chen XL, Gao DW, Yang Y, Li X. HIF1 signaling pathway involving iNOS, COX2 and caspase9 mediates the neuroprotection provided by erythropoietin in the retina of chronic ocular hypertension rats. Mol Med Rep 2015; 11: 1490-6. |
| 59. |
Lu J, Xie L, Liu C, Zhang Q, Sun S. PTEN/PI3k/AKT regulates macrophage polarization in emphysematous mice. Scand J Immunol 2017; 85: 395-405.
DOI PMID |
| 60. | Ersahin T, Tuncbag N, Cetin-Atalay R. The PI3K/AKT/mTOR interactive pathway. Mol Biosyst 2015;11:1946-54. |
| 61. |
Zhang Z, Yao L, Yang J, Wang Z, Du G. PI3K/Akt and HIF‑1 signaling pathway in hypoxia‑ischemia (review). Mol Med Rep 2018; 18: 3547-54.
DOI PMID |
| 62. |
Płóciennikowska A, Hromada-Judycka A, Borzęcka K, Kwiatkowska K. Co-operation of TLR4 and raft proteins in LPS-induced pro-inflammatory signaling. Cell Mol Life Sci 2015; 72: 557-81.
DOI PMID |
| 63. | Chen LJ, Xu W, Li YP, et al. Lycium barbarum polysaccharide inhibited hypoxia-inducible factor 1 in COPD patients. Int J Chron Obstruct Pulmon Dis 2020; 15: 1997-04. |
| 64. |
Tran H, Brunet A, Grenier JM, et al. DNA repair pathway stimulated by the forkhead transcription factor FOXO3a through the Gadd45 protein. Science 2002; 296: 530-4.
DOI PMID |
| 65. |
Lee SH, Sang HL, Kim CH, Yang KS, Sang YL. Increased expression of vascular endothelial growth factor and hypoxia inducible factor-1α in lung tissue of patients with chronic bronchitis. Clin Biochem 2014; 47: 552-9.
DOI PMID |
| 66. |
Shimazu T, Hirschey MD, Newman J, et al. Suppression of oxidative stress by β-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science 2013; 339: 211-4.
DOI PMID |
| 67. | Song Q, Chen P, Liu XM. The role of cigarette smoke-induced pulmonary vascular endothelial cell apoptosis in COPD. Respir Res 2021; 22: 39. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

