Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (4): 762-769.DOI: 10.19852/j.cnki.jtcm.20240515.002
• Original articles • Previous Articles Next Articles
Tuina alleviates neuropathic pain through regulate the activation of microglia and the secretion of inflammatory cytokine in spinal cord
WU Zhiwei1,2, ZHU Qingguang1,2, KONG Lingjun3, SONG Pengfei3, ZHOU Xin1,2, GUO Guangxin4, ZHANG Shuaipan3, HE Tianxiang3, CHENG Yanbin1,2, FANG Min1,2,3( )
)
- 1 Yueyang Hospital of Integrated Traditional Chinese and Western Medicine, Shanghai University of Traditional Chinese Medicine, Shanghai 200437, China
2 Institute of Tuina, Shanghai Institute of Traditional Chinese Medicine, Shanghai 200437, China
3 Shuguang Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai 200021, China
4 Department of Acupuncture and Tuina, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China
-
Received:2023-02-23Accepted:2023-07-14Online:2024-08-15Published:2024-05-15 -
Contact:FANG Min, Yueyang Hospital of Integrated Traditional Chinese and Western Medicine, Shanghai University of Traditional Chinese Medicine, Shanghai 200437, China; Institute of Tuina, Shanghai Institute of Traditional Chinese Medicine, Shanghai 200437, China; Shuguang Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China.fm-tn0510@shutcm.edu.cn Telephone: +86-21-65161782 -
Supported by:National Natural Science Foundation of China: Study on the Mechanism of Tuina Inhibiting the Spinal Cord Central Sensitization of Neuropathic Pain through Regulates Interleukin-10/β-Endorphin Pathway in Spinal Microglia(82205302);Study on the Mechanism of Peripheral and Central Pain in Lumbar disc herniation with Nerve Injury and the Analgesia Mechanism of Tuina(82030121);Shanghai Sailing Program: Study on the Analgesic Mechanism of Tuina Inhibiting Spinal Cord Central Sensitization based on Microglia and Related Pro-inflammatory Factors(20YF1450900);Science Foundation of Yueyang Hospital of Integrated Traditional Chinese and Western Medicine: Study on the Mechanism of Tuina Analgesia based on Spinal Cord Microglia and Inflammatory Factors(2021yygq03)
Cite this article
WU Zhiwei, ZHU Qingguang, KONG Lingjun, SONG Pengfei, ZHOU Xin, GUO Guangxin, ZHANG Shuaipan, HE Tianxiang, CHENG Yanbin, FANG Min. Tuina alleviates neuropathic pain through regulate the activation of microglia and the secretion of inflammatory cytokine in spinal cord[J]. Journal of Traditional Chinese Medicine, 2024, 44(4): 762-769.
share this article
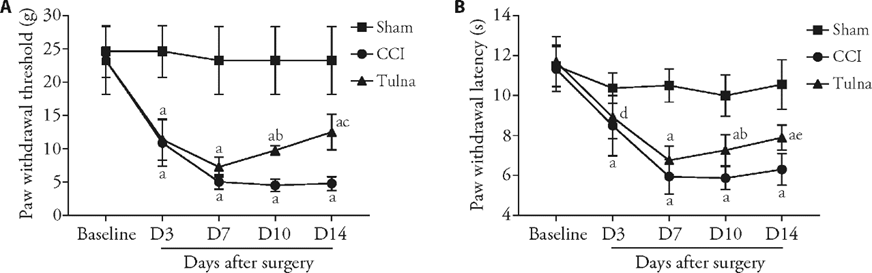
Figure 1 Analgesic effects of tuina on PWT and PWL in SD rats with CCI A: the PWT in response to von Frey filaments; B: the PWL to a noxious thermal beam. Tuina treatment was performed each day from days 4 to 13 (total of 10 times). Both PWT and PWL were tested and evaluated among sham group, CCI-induced NPP model group (CCI group), and CCI-induced NPP model with tuina treatment group (Tuina group) at days -2, -1, 3, 7, 10 and 14. PWT: paw withdrawal threshold; PWL: paw withdrawal latency; SD: Sprague-Dawley; CCI: chronic constrictive injury; NPP: neuropathic pain. Two-way repeated measures analysis of variance followed by Bonferroni’s multiple comparison tests was used for PWT and PWL test. Data are expressed as the mean ± standard error of mean (n = 8 per group). aP < 0.001, dP < 0.05 vs the sham group; bP < 0.05, eP < 0.01, cP < 0.001 vs the CCI group.
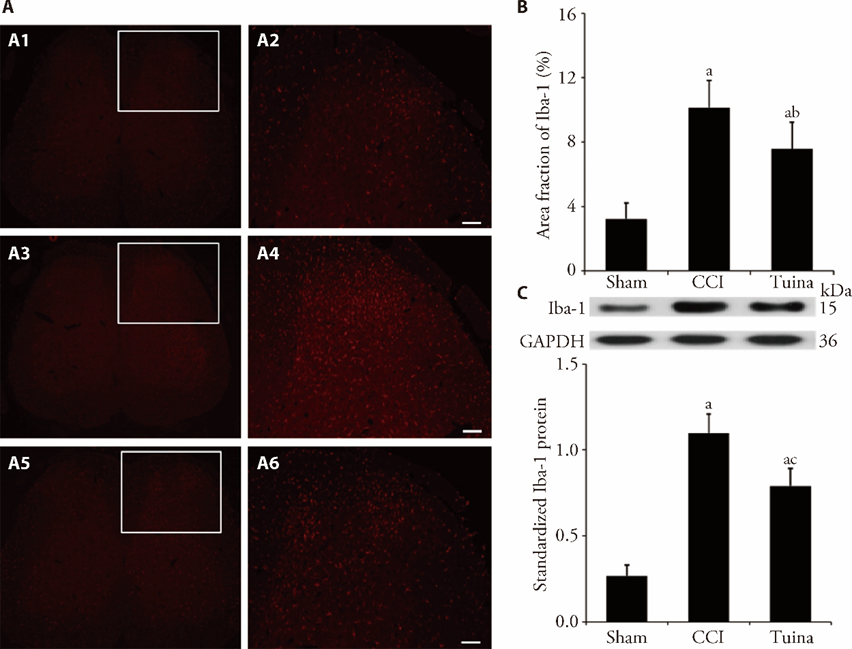
Figure 2 Effect of Tuina on the activation of microglia in the ipsilateral SDH of CCI rats A: representative Immunofluorescence staining images of Iba1 in the ipsilateral SDH, Scale bar, 100 μm; A1: Iba1 expression of Sham in low magnification mirror; A2: Iba1 expression of Sham in high magnification mirror; A3: Iba1 expression of CCI in low magnification mirror; A4: Iba1 expression of CCI in high magnification mirror; A5: Iba1 expression of Tuina in low magnification mirror; A6: Iba1 expression of Tuina in high magnification mirror; B: area fraction of Iba1 in the ipsilateral SDH; C: protein levels of Iba-1 standardized by GAPDH. Sham group: CCI except for the ligation of the sciatic nerve; CCI group: CCI-induced NPP model group; Tuina group: CCI-induced NPP model with Tuina treatment group. Iba-1: Ionised calcium binding adaptor molecule 1; SDH: spinal dorsal horn; CCI: chronic constrictive injury; GAPDH: glyceraldehyde-3?phosphate dehydrogenase. One-way analysis of variance was utilized for area fraction and protein levels of Iba1. Data are expressed as the mean ± standard error of mean (n = 8 per group). aP < 0.001 vs the sham group; bP < 0.01, cP < 0.001 vs the CCI group.
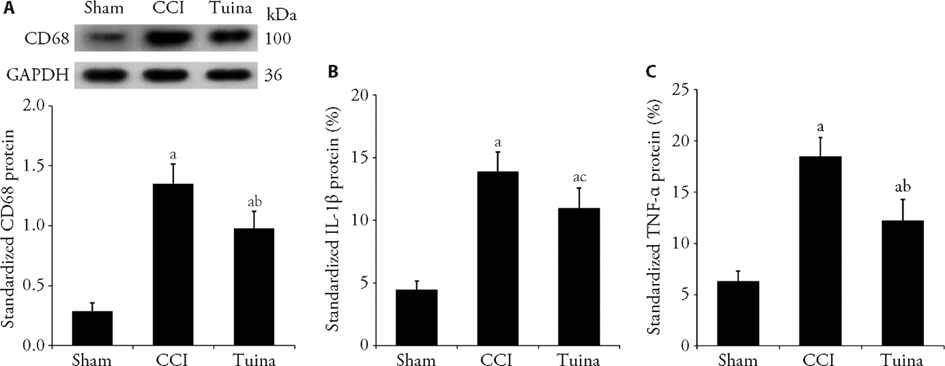
Figure 3 Effect of Tuina on the polarization of activated microglial A: protein levels of CD68 standardized by GAPDH; B: concentration of IL-1β standardized by total protein (IL-1β / total protein × 100%); C: concentration of TNF-α standardized by total protein (TNF-α / total protein × 100%). Sham group: CCI except for the ligation of the sciatic nerve; CCI group: CCI-induced NPP model group; Tuina group: CCI-induced NPP model with Tuina treatment group. GAPDH: glyceraldehyde-3?phosphate dehydrogenase; IL-1β: interleukin-1β; TNF-α: tumor necrosis factor-α; CCI: chronic constrictive injury. One-way analysis of variance was utilized for protein levels of CD68, concentration of IL-1β and TNF-α. Data are expressed as the mean ± standard error of mean (n = 8 per group). aP < 0.001 vs the sham group; bP < 0.001, cP < 0.01 vs the CCI group.
| 1. |
Jensen TS, Baron R, Haanpää M, et al. A new definition of neuropathic pain. Pain 2011; 152: 2204-5.
DOI PMID |
| 2. | Liu ZY, Song ZW, Guo SW, et al. CXCL12/CXCR4 signaling contributes to neuropathic pain via central sensitization mechanisms in a rat spinal nerve ligation model. CNS Neurosci Ther 2019; 25: 922-36. |
| 3. |
Haanpää M, Attal N, Backonja M, et al. NeuPSIG guidelines on neuropathic pain assessment. Pain 2011; 152: 14-27.
DOI PMID |
| 4. | Cvijanovic M, Simic S, Banic HS, et al. Contemporary treatment neuropathic pain. Med Pregl 2011; 64: 443-7. |
| 5. |
van Hecke O, Austin SK, Khan RA, et al. Neuropathic pain in the general population: a systematic review of epidemiological studies. Pain 2014; 155: 654-62.
DOI PMID |
| 6. | Alles SRA, Smith PA. Etiology and pharmacology of neuropathic pain. Pharmacol Rev 2018;70: 315-47. |
| 7. |
Khan WU, Michelini G, Battaglia M. Twin studies of the covariation of pain with depression and anxiety: a systematic review and re-evaluation of critical needs. Neurosci Biobehav Rev 2020; 111: 135-48.
DOI PMID |
| 8. |
Kirtley OJ, Rodham K, Crane C. Understanding suicidal ideation and behaviour in individuals with chronic pain: a review of the role of novel transdiagnostic psychological factors. Lancet Psychiatry 2020; 7: 282-90.
DOI PMID |
| 9. |
Toth C, Lander J, Wiebe S. The prevalence and impact of chronic pain with neuropathic pain symptoms in the general population. Pain Med 2009; 10: 918-29.
DOI PMID |
| 10. |
Yekkirala AS, Roberson DP, Bean BP, et al. Breaking barriers to novel analgesic drug development. Nat Rev Drug Discov 2017; 16: 545-64.
DOI PMID |
| 11. |
Mitsi V, Terzi D, Purushothaman I, et al. RGS9-2--controlled adaptations in the striatum determine the onset of action and efficacy of antidepressants in neuropathic pain states. Proc Natl Acad Sci USA 2015; 112: 5088-97.
DOI PMID |
| 12. |
Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and Meta-analysis. Lancet Neurol 2015; 14: 162-73.
DOI PMID |
| 13. | Yao CJ, Cheng YB, Zhu QG, et al. Clinical evidence for the effects of manual therapy on cancer pain: a systematic review and meta-analysis. Evid Based Complement Alternat Med 2021; 2021: 6678184. |
| 14. | Li T, Li X, Huang F, et al. Clinical efficacy and safety of acupressure on low back pain: a systematic review and meta-analysis. Evid Based Complement Alternat Med 2021; 2021: 8862399. |
| 15. | Wang MY, Tsai PS, Lee PH, et al. Systematic review and Meta-analysis of the efficacy of Tuina for cervical spondylosis. J Clin Nurs 2008; 17: 2531-8. |
| 16. | Mo Z, Li D, Zhang R, et al. Comparisons of the effectiveness and safety of Tuina, acupuncture, traction, and Chinese herbs for lumbar disc herniation: a systematic review and network Meta-analysis. Evid Based Complement Alternat Med 2019; 2019: 6821310. |
| 17. |
Niu K, Li Y, Guan H, et al. Optimization of traditional Chinese medicine rolling manipulation and pressure attenuation. Am J Transl Res 2021; 13: 7654-66.
PMID |
| 18. |
Jiang SC, Zhang H, Fang M, et al. Analgesic effects of Chinese Tuina massage in a rat model of pain. Exp Ther Med 2016; 11: 1367-74.
PMID |
| 19. |
Song P, Sun W, Zhang H, et al. Possible mechanism underlying analgesic effect of Tuina in rats may involve piezo mechanosensitive channels within dorsal root ganglia axon. J Tradit Chin Med 2018; 38: 834-41.
PMID |
| 20. | Lin R, Zhou Y, Yan T, et al. Directed evolution of adeno-associated virus for efficient gene delivery to microglia. Nat Methods 2022; 19: 976-85. |
| 21. |
Mika J, Zychowska M, Popiolek-Barczyk K, et al. Importance of glial activation in neuropathic pain. Eur J Pharmacol 2013; 716: 106-19.
DOI PMID |
| 22. |
Flor H. Psychological pain interventions and neurophysiology: implications for a mechanism-based approach. Am Psychol 2014; 69: 188-96.
DOI PMID |
| 23. | Ma JY, Zhao ZQ. The involvement of glia in long-term plasticity in the spinal dorsal horn of the rat. Neuroreport 2002; 13: 1781-4. |
| 24. | Cheng CF, Cheng JK, Chen CY, et al. Mirror-image pain is mediated by nerve growth factor produced from tumor necrosis factor alpha-activated satellite glia after peripheral nerve injury. Pain 2014; 155: 906-20. |
| 25. |
Liu T, Gao YJ, Ji RR. Emerging role of toll-like receptors in the control of pain and itch. Neurosci Bull 2012; 28: 131-44.
DOI PMID |
| 26. |
Zhang H, Yoon SY, Zhang H, et al. Evidence that spinal astrocytes but not microglia contribute to the pathogenesis of Paclitaxel-induced painful neuropathy. J Pain 2012; 13: 293-303.
DOI PMID |
| 27. |
Raghavendra V, Tanga F, DeLeo JA. Inhibition of microglial activation attenuates the development but not existing hypersensitivity in a rat model of neuropathy. J Pharmacol Exp Ther 2003; 306: 624-30.
DOI PMID |
| 28. |
Ledeboer A, Sloane EM, Milligan ED, et al. Minocycline attenuates mechanical allodynia and proinflammatory cytokine expression in rat models of pain facilitation. Pain 2005; 115: 71-83.
DOI PMID |
| 29. | Chu YX, Zhang Y, Zhang YQ, et al. Involvement of microglial P2X7 receptors and downstream signaling pathways in long-term potentiation of spinal nociceptive responses. Brain Behav Immun 2010; 24: 1176-89. |
| 30. | Liu Z, Wang H, Yu T, et al. A review on the mechanism of Tuina promoting the recovery of peripheral nerve injury. Evid Based Complement Alternat Med 2021; 2021: 6652099. |
| 31. | Wu JJ, Lu YC, Hua XY, et al. Cortical remodeling after electroacupuncture therapy in peripheral nerve repairing model. Brain Res 2018; 1690: 61-73. |
| 32. | Xing XX, Zheng MX, Hua XY, et al. Brain plasticity after peripheral nerve injury treatment with massage therapy based on resting-state functional magnetic resonance imaging. Neural Regen Res 2021; 16: 388-93. |
| 33. |
Aboodarda SJ, Greene RM, Philpott DT, et al. The effect of rolling massage on the excitability of the corticospinal pathway. Appl Physiol Nutr Metab 2018; 43: 317-23.
DOI PMID |
| 34. | Lv TT, Mo YJ, Yu TY, et al. Using RNA-Seq to explore the repair mechanism of the three methods and three-acupoint technique on DRGs in sciatic nerve injured rats. Pain Res Manag 2020; 2020: 7531409. |
| 35. |
Guo X, Yu TY, Steven W, et al. "Three Methods and Three Points" regulates p38 mitogen-activated protein kinase in the dorsal horn of the spinal cord in a rat model of sciatic nerve injury. Neural Regen Res 2016; 11: 2018-24.
DOI PMID |
| 36. | Li YZ, Miao RP, Yu TY, et al. Mild mechanic stimulate on acupoints regulation of CGRP-positive cells and microglia morphology in spinal cord of sciatic nerve injured rats. Front Integr Neurosci 2019; 13: 58. |
| 37. |
Yamato K, Kataoka T, Nishiyama Y, et al. Preventive and curative effects of radon inhalation on chronic constriction injury-induced neuropathic pain in mice. Eur J Pain 2013; 17: 480-92.
DOI PMID |
| 38. |
Bennett GJ, Xie YK. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 1988; 33: 87-107.
DOI PMID |
| 39. |
Roeska K, Doods H, Arndt K, et al. Anxiety-like behaviour in rats with mononeuropathy is reduced by the analgesic drugs morphine and gabapentin. Pain 2008; 139: 349-57.
DOI PMID |
| 40. | Levin S, Pearsall G, Ruderman RJ. Von Frey's method of measuring pressure sensibility in the hand: an engineering analysis of the Weinstein-Semmes pressure aesthesiometer. J Hand Surg Am 1978; 3: 211-6. |
| 41. | Austin PJ, Wu A, Moalem-Taylor G. Chronic constriction of the sciatic nerve and pain hypersensitivity testing in rats. J Vis Exp 2012; 61: 3393. |
| 42. | Chaplan SR, Bach FW, Pogrel JW, et al. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods 1994; 53: 55-63. |
| 43. |
Hargreaves K, Dubner R, Brown F, et al. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 1988; 32: 77-88.
DOI PMID |
| 44. | Yao CJ, Ren J, Huang RX, et al. Transcriptome profiling of microRNAs reveals potential mechanisms of manual therapy alleviating neuropathic pain through microRNA-547-3p-mediated Map4k4/NF-κb signaling pathway. J Neuroinflammation 2022; 19: 211. |
| 45. | Spångeus A, Willerton C, Enthoven P, et al. Patient education improves pain and health-related quality of life in patients with established spinal osteoporosis in primary care-a pilot study of short- and long-term effects. Int J Environ Res Public Health 2023; 20: 4933. |
| 46. | McGinnis A, Ji RR. The similar and distinct roles of satellite glial cells and spinal astrocytes in neuropathic pain. Cells 2023; 12: 965. |
| 47. | Yousefpour N, Locke S, Deamond H, et al. Time-dependent and selective microglia-mediated removal of spinal synapses in neuropathic pain. Cell Rep 2023; 42: 112010. |
| 48. | Guan X, Cao F, Ren L, et al. STAT1 as a downstream mediator of ERK signaling contributes to bone cancer pain by regulating MHC Ⅱ expression in spinal microglia. Brain Behav Immun 2017; 60: 161-73. |
| 49. | Andreeva D, Murashova L, Burzak N, et al. Satellite glial cells: morphology, functional heterogeneity, and role in pain. Front Cell Neurosci 2022; 16: 1019449. |
| 50. | Jeffrey-Gauthier R, Bouyer J, Piché M, et al. Locomotor deficits induced by lumbar muscle inflammation involve spinal microglia and are independent of KCC 2 expression in a mouse model of complete spinal transection. Exp Neurol 2021: 113592. |
| 51. | Vega-Avelaira D, Ballesteros JJ, López-García JA. Inflammation-induced hyperalgesia and spinal microglia reactivity in neonatal rats. Eur J Pain 2013; 17: 1180-8. |
| 52. | Calvello R, Porro C, Lofrumento DD, et al. Decoy receptors regulation by resveratrol in lipopolysaccharide-activated microglia. Cells 2023; 12: 681. |
| 53. | Vergne-Salle P, Bertin P. Chronic pain and neuroinflammation. Joint Bone Spine 2021; 88: 105222. |
| 54. |
Iqubal A, Ahmed M, Iqubal MK, et al. Polyphenols as potential therapeutics for pain and inflammation in spinal cord injury. Curr Mol Pharmacol 2021; 14: 714-30.
DOI PMID |
| 55. | Liu Z, Chen S, Qiu C, et al. Fractalkine/CX3CR1 contributes to endometriosis- induced neuropathic pain and mechanical hypersensitivity in rats. Front Cell Neurosci 2018; 12: 495. |
| 56. |
Tan Y, Wang Z, Liu T, et al. RNA interference-mediated silencing of DNA methyltransferase 1 attenuates neuropathic pain by accelerating microglia M2 polarization. BMC Neurol 2022; 22: 376.
DOI PMID |
| 57. |
Yang X, Chen S, Shao Z, et al. Apolipoprotein E deficiency exacerbates spinal cord injury in mice: inflammatory response and oxidative stress mediated by NF-κB signaling pathway. Front Cell Neurosci 2018; 12: 142.
DOI PMID |
| 58. |
Xu J, E X, Liu H, et al. Tumor necrosis factor-alpha is a potential diagnostic biomarker for chronic neuropathic pain after spinal cord injury. Neurosci Lett 2015; 595: 30-4.
DOI PMID |
| 59. |
Kiguchi N, Maeda T, Kobayashi Y, et al. Macrophage inflammatory protein-1alpha mediates the development of neuropathic pain following peripheral nerve injury through interleukin-1beta up-regulation. Pain 2010; 149: 305-15.
DOI PMID |
| 60. |
Choi BM, Lee SH, An SM, et al. The time-course and RNA interference of TNF-α, IL-6, and IL-1β expression on neuropathic pain induced by L5 spinal nerve transection in rats. Korean J Anesthesiol 2015; 68: 159-69.
DOI PMID |
| 61. |
Karam MC, Merckbawi R, Salman S, et al. Atenolol reduces leishmania major-induced hyperalgesia and TNF-α without affecting IL-1β or keratinocyte derived chemokines (KC). Front Pharmacol 2016; 7: 22.
DOI PMID |
| 62. |
Lee J, Lee G, Ko G, et al. Nerve injury-induced gut dysbiosis contributes to spinal cord TNF-α expression and nociceptive sensitization. Brain Behav Immun 2023; 110: 155-61.
DOI PMID |
| 63. | Rivaz M, Rahpeima M, Khademian Z, et al. The effects of aromatherapy massage with lavender essential oil on neuropathic pain and quality of life in diabetic patients: a randomized clinical trial. Complement Ther Clin Pract 2021; 44: 101430. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

