Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (5): 795-802.DOI: 10.19852/j.cnki.jtcm.20220519.005
• Research Articles • Previous Articles Next Articles
Wenshen Jianpi recipe (温肾健脾方) induced immune reconstruction and redistribution of natural killer cell subsets in immunological non-responders of human immunodeficiency virus/acquired immune deficiency syndrome: a randomized controlled trial
TAO Zhuang1, WANG Jian2, CHEN Xin3, LI Yonghong4, YAN Yuguang4, ZHANG Ao5, ZOU Wen2( ), LIU Ying2(
), LIU Ying2( )
)
- 1 Research Center of AIDS Treatment with Traditional Chinese Medicine, China Academy of Chinese Medical Sciences, Beijing 100700, China; Institute of Basic Theory for Chinese Medicine, China Academy of Chinese Medical Sciences, Beijing 100700, China
2 Research Center of AIDS Treatment with Traditional Chinese Medicine, China Academy of Chinese Medical Sciences, Beijing 100700, China
3 Department of Integrated Traditional Chinese and Wetern Medicine, Kunming Hospital of Traditional Chinese Medicine, Kunming 650000, China
4 Department of Infection, No.6 People's Hospital of Shenyang City, Shenyang 110006, China
5 University of Chinese Academy of SoCIal SCIences, Beijing, 100102, China
-
Received:2021-05-11Accepted:2021-08-20Online:2022-10-15Published:2022-05-19 -
Contact:ZOU Wen,LIU Ying -
About author:LIU Ying, Research Center of AIDS Treatment with Traditional Chinese Medicine, China Academy of Chinese Medical Sciences, Beijing 100700, China. liuying0516@163.com Telephone: +86-13552252730; +86-13021985109
ZOU Wen, Research Center of AIDS Treatment with Traditional Chinese Medicine, China Academy of Chinese Medical Sciences, Beijing 100700, China. veven@yeah.net;
-
Supported by:National Science and Technology Major Project of China During the 13th Five-year Plan Period: New Adjuvant Strategies of Traditional Chinese Medicine on Immune Reconstruction for Long-term ART-treated People Living with HIV(2017ZX10205501);Effects of Traditional Chinese Medicine on Intestinal Bacterial Community Structure of AIDS Patients(2017ZX10205501-001-010);National Natural Science Foundation of China: Immune Mechanism of Immunological Non-responders of HIV/AIDS Treated with Traditional Chinese Medicine from Immune Activation and Toll like Receptor Signal Pathway(81503571)
Cite this article
TAO Zhuang, WANG Jian, CHEN Xin, LI Yonghong, YAN Yuguang, ZHANG Ao, ZOU Wen, LIU Ying. Wenshen Jianpi recipe (温肾健脾方) induced immune reconstruction and redistribution of natural killer cell subsets in immunological non-responders of human immunodeficiency virus/acquired immune deficiency syndrome: a randomized controlled trial[J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 795-802.
share this article
Figure 1 Participants flowchart KHTCM: Kunming Hospital of Traditional Chinese Medicine; PHS: No. 6 People's Hospital of Shenyang City; INRs: immunological non-responders; ART: antiretroviral therapy; HIV: human immunodeficiency virus.
| Baseline information | Treatment group (n = 28) | Control group (n = 28) | Test | Statistic value | P value |
|---|---|---|---|---|---|
| Age | t | 1.416 | 0.163 | ||
| N (missing) | 28 (0) | 27 (1) | |||
| Mean ± SD | 47.0±10.8 | 42.8±11.2 | |||
| Sex | χ2 | 0.125 | 0.724 | ||
| N (missing) | 28 (0) | 27 (1) | |||
| Male [n (%)] | 24 (85.70 ) | 24 (88.90) | |||
| Female [n (%)] | 4 (14.30 ) | 3 (11.11 ) | |||
| Nationality | Fisher | - | 0.236 | ||
| N (missing) | 28 (0) | 27 (1) | |||
| Han [n (%)] | 28 (100.00) | 25 (92.59) | |||
| Minority [n (%)] | 0 (0.00 ) | 2 (7.41 ) | |||
| Allergy history | χ2 | <0.001 | 1.000 | ||
| N (missing) | 28 (0) | 28 (0) | |||
| Never [n (%)] | 27 (96.43 ) | 27 (96.43) | |||
| Before [n (%)] | 1 ( 3.57 ) | 1 (3.57 ) | |||
| Marriage | Fisher | - | 0.139 | ||
| N (missing) | 28 (0) | 27 (1) | |||
| Never married [n (%)] | 6 (21.43) | 12 (42.90) | |||
| Married/cohabitation [n (%)] | 21 (75.00) | 14 (50.00) | |||
| Divorced/separated [n (%)] | 1 (3.57 ) | 1 (3.57) | |||
| Smoking | Fisher | - | 0.420 | ||
| N (missing) | 28 (0) | 23 (5) | |||
| Always [n (%)] | 5 (17.86) | 1 (4.35) | |||
| Often [n (%)] | 1 (3.57) | 1 (4.35) | |||
| Seldom [n (%)] | 22 (78.57) | 21 (91.30) | |||
| Drinking | Fisher | - | 1.000 | ||
| N (missing) | 28 (0) | 22 (6) | |||
| Always [n (%)] | 0 (0.00) | 0 (0.00) | |||
| Often [n (%)] | 2 (7.14) | 1 (4.55) | |||
| Seldom [n (%)] | 26 (92.86 ) | 21(95.45) | |||
| BMI | t | 0.126 | 0.900 | ||
| N (missing) | 28 (0) | 28 (0) | |||
| Mean ± SD | 21.8 ± 2.3 | 21.7 ± 3.0 | |||
| Transmission route | Fisher | - | 0.470 | ||
| N (missing) | 28 (0) | 27 (1) | |||
| Drug injection [n (%)] | 3 (10.71 ) | 0 (0.00) | |||
| Homosexual sexual tranmission [n (%)] | 21 (75.00 ) | 22(78.57) | |||
| Heterosexual sexual transmission [n (%)] | 2 (7.14) | 2 (7.14) | |||
| Other [n (%)] | 2 (7.14) | 3 (10.71) | |||
| Duration of ART | t | 1.574 | 0.121 | ||
| N (missing) | 28 (0) | 28 (0) | |||
| Mean ± SD | 7.2±1.9 | 8.2±2.8 | |||
| Baseline CD4+T cell counts | t | 0.648 | 0.600 | ||
| N (missing) | 28 (0) | 28 (0) | |||
| Mean ± SD | 213.6 ± 67.8 | 224.5 ± 57.7 |
Table 1 Participants' characteristics
| Baseline information | Treatment group (n = 28) | Control group (n = 28) | Test | Statistic value | P value |
|---|---|---|---|---|---|
| Age | t | 1.416 | 0.163 | ||
| N (missing) | 28 (0) | 27 (1) | |||
| Mean ± SD | 47.0±10.8 | 42.8±11.2 | |||
| Sex | χ2 | 0.125 | 0.724 | ||
| N (missing) | 28 (0) | 27 (1) | |||
| Male [n (%)] | 24 (85.70 ) | 24 (88.90) | |||
| Female [n (%)] | 4 (14.30 ) | 3 (11.11 ) | |||
| Nationality | Fisher | - | 0.236 | ||
| N (missing) | 28 (0) | 27 (1) | |||
| Han [n (%)] | 28 (100.00) | 25 (92.59) | |||
| Minority [n (%)] | 0 (0.00 ) | 2 (7.41 ) | |||
| Allergy history | χ2 | <0.001 | 1.000 | ||
| N (missing) | 28 (0) | 28 (0) | |||
| Never [n (%)] | 27 (96.43 ) | 27 (96.43) | |||
| Before [n (%)] | 1 ( 3.57 ) | 1 (3.57 ) | |||
| Marriage | Fisher | - | 0.139 | ||
| N (missing) | 28 (0) | 27 (1) | |||
| Never married [n (%)] | 6 (21.43) | 12 (42.90) | |||
| Married/cohabitation [n (%)] | 21 (75.00) | 14 (50.00) | |||
| Divorced/separated [n (%)] | 1 (3.57 ) | 1 (3.57) | |||
| Smoking | Fisher | - | 0.420 | ||
| N (missing) | 28 (0) | 23 (5) | |||
| Always [n (%)] | 5 (17.86) | 1 (4.35) | |||
| Often [n (%)] | 1 (3.57) | 1 (4.35) | |||
| Seldom [n (%)] | 22 (78.57) | 21 (91.30) | |||
| Drinking | Fisher | - | 1.000 | ||
| N (missing) | 28 (0) | 22 (6) | |||
| Always [n (%)] | 0 (0.00) | 0 (0.00) | |||
| Often [n (%)] | 2 (7.14) | 1 (4.55) | |||
| Seldom [n (%)] | 26 (92.86 ) | 21(95.45) | |||
| BMI | t | 0.126 | 0.900 | ||
| N (missing) | 28 (0) | 28 (0) | |||
| Mean ± SD | 21.8 ± 2.3 | 21.7 ± 3.0 | |||
| Transmission route | Fisher | - | 0.470 | ||
| N (missing) | 28 (0) | 27 (1) | |||
| Drug injection [n (%)] | 3 (10.71 ) | 0 (0.00) | |||
| Homosexual sexual tranmission [n (%)] | 21 (75.00 ) | 22(78.57) | |||
| Heterosexual sexual transmission [n (%)] | 2 (7.14) | 2 (7.14) | |||
| Other [n (%)] | 2 (7.14) | 3 (10.71) | |||
| Duration of ART | t | 1.574 | 0.121 | ||
| N (missing) | 28 (0) | 28 (0) | |||
| Mean ± SD | 7.2±1.9 | 8.2±2.8 | |||
| Baseline CD4+T cell counts | t | 0.648 | 0.600 | ||
| N (missing) | 28 (0) | 28 (0) | |||
| Mean ± SD | 213.6 ± 67.8 | 224.5 ± 57.7 |
| Index | Treatment group (n = 28) | Control group (n = 28) | t value | P value |
|---|---|---|---|---|
| Baseline | -1.13 | 0.26 | ||
| N (missing) | 28 (0) | 28 (0) | ||
| Mean ± SD | 207±66 | 228±60 | ||
| 95% CI | 177.97, 236.85 | 203.49, 253.06 | ||
| After 12 weeks treatment | 0.51 | 0.61 | ||
| N (missing) | 22 (6) | 27 (1) | ||
| Mean ± SD | 238±77 | 228±68 | ||
| 95% CI | 205.96, 270.87 | 201.32, 254.91 | ||
| After 24 weeks treatment | 2.65 | 0.01b | ||
| N (missing) | 24 (4) | 23 (5) | ||
| Mean ± SD | 275±79a | 215±72 | ||
| 95% CI | 240.76, 309.23 | 182.82, 247.08 |
Table 2 CD4+T cell counts recovery before and after WJR treatment (cells/μL)
| Index | Treatment group (n = 28) | Control group (n = 28) | t value | P value |
|---|---|---|---|---|
| Baseline | -1.13 | 0.26 | ||
| N (missing) | 28 (0) | 28 (0) | ||
| Mean ± SD | 207±66 | 228±60 | ||
| 95% CI | 177.97, 236.85 | 203.49, 253.06 | ||
| After 12 weeks treatment | 0.51 | 0.61 | ||
| N (missing) | 22 (6) | 27 (1) | ||
| Mean ± SD | 238±77 | 228±68 | ||
| 95% CI | 205.96, 270.87 | 201.32, 254.91 | ||
| After 24 weeks treatment | 2.65 | 0.01b | ||
| N (missing) | 24 (4) | 23 (5) | ||
| Mean ± SD | 275±79a | 215±72 | ||
| 95% CI | 240.76, 309.23 | 182.82, 247.08 |
| Index | Treatment group (n = 28) | Control group (n = 28) | Z value | P value |
|---|---|---|---|---|
| CD56briCD16- | ||||
| Baseline | -1.225 | 0.221 | ||
| N (missing) | 28 (0) | 28 (0) | ||
| IQR | 3.50 (2.00, 6.00) | 4.00 (2.25, 8.00) | ||
| 95% CI | 2.51, 7.49 | 4.11, 7.31 | ||
| After 12 weeks treatment | -1.514 | 0.130 | ||
| N (missing) | 26 (2) | 27 (1) | ||
| IQR | 6.50 (2.00, 11.50) | 5.00 (2.00, 6.00) | ||
| 95% CI | 5.34, 10.81 | 3.44, 7.19 | ||
| After 24 weeks treatment | -0.567 | 0.571 | ||
| N (missing) | 24 (4) | 23 (5) | ||
| IQR | 6.78 (3.25, 8.50)* | 5.50 (4.00, 8.00) | ||
| 95% CI | 5.21, 8.35 | 4.97, 8.19 | ||
| CD56dimCD16+ | ||||
| Baseline | -0.387 | 0.699 | ||
| N (missing) | 28 (0) | 27 (1) | ||
| IQR | 197.00 (117.00, 359.00) | 176.00 (119.50, 271.50) | ||
| 95% CI | 177.24, 309.13 | 160.48, 298.59 | ||
| After 12 weeks treatment | -1.090 | 0.276 | ||
| N (missing) | 23 (5) | 27 (1) | ||
| IQR | 271.00 (151.25, 457.50) | 247.00 (147.75, 275.50) | ||
| 95% CI | 227.88, 404.91 | 181.83, 311.64 | ||
| After 24 weeks treatment | -2.246 | 0.025b | ||
| N (missing) | 24 (4) | 23 (5) | ||
| IQR | 342.00 (157.75, 412.75) | 193.50 (135.75, 267.75) | ||
| 95% CI | 252.31, 432.47 | 164.47, 272.39 | ||
| CD56negCD16+ | ||||
| Baseline | ||||
| N (missing) | 28 (0) | 28 (0) | -1.787 | 0.074 |
| IQR | 250.00 (212.00, 363.75) | 234.00 (186.00, 286.25) | ||
| 95% CI | 253.27, 369.66 | 203.77, 277.09 | ||
| After 12 weeks treatment | ||||
| N (missing) | 26 (2) | 27 (1) | -1.475 | 0.140 |
| IQR | 226.50 (180.25, 337.25) | 203.50 (148.25,265.00) | ||
| 95% CI | 211.81, 287.84 | 179.32, 252.03 | ||
| After 24 weeks treatment | ||||
| N (missing) | 24 (4) | 23 (5) | -2.278 | 0.023b |
| IQR | 172.50 (133.00, 265.00)a | 295.50 (205.50, 334.75) | ||
| 95% CI | 174.11, 245.39 | 233.91, 323.23 |
Table 3 Counts of NK cell subsets before and after WJR treatment (cells/mm3)
| Index | Treatment group (n = 28) | Control group (n = 28) | Z value | P value |
|---|---|---|---|---|
| CD56briCD16- | ||||
| Baseline | -1.225 | 0.221 | ||
| N (missing) | 28 (0) | 28 (0) | ||
| IQR | 3.50 (2.00, 6.00) | 4.00 (2.25, 8.00) | ||
| 95% CI | 2.51, 7.49 | 4.11, 7.31 | ||
| After 12 weeks treatment | -1.514 | 0.130 | ||
| N (missing) | 26 (2) | 27 (1) | ||
| IQR | 6.50 (2.00, 11.50) | 5.00 (2.00, 6.00) | ||
| 95% CI | 5.34, 10.81 | 3.44, 7.19 | ||
| After 24 weeks treatment | -0.567 | 0.571 | ||
| N (missing) | 24 (4) | 23 (5) | ||
| IQR | 6.78 (3.25, 8.50)* | 5.50 (4.00, 8.00) | ||
| 95% CI | 5.21, 8.35 | 4.97, 8.19 | ||
| CD56dimCD16+ | ||||
| Baseline | -0.387 | 0.699 | ||
| N (missing) | 28 (0) | 27 (1) | ||
| IQR | 197.00 (117.00, 359.00) | 176.00 (119.50, 271.50) | ||
| 95% CI | 177.24, 309.13 | 160.48, 298.59 | ||
| After 12 weeks treatment | -1.090 | 0.276 | ||
| N (missing) | 23 (5) | 27 (1) | ||
| IQR | 271.00 (151.25, 457.50) | 247.00 (147.75, 275.50) | ||
| 95% CI | 227.88, 404.91 | 181.83, 311.64 | ||
| After 24 weeks treatment | -2.246 | 0.025b | ||
| N (missing) | 24 (4) | 23 (5) | ||
| IQR | 342.00 (157.75, 412.75) | 193.50 (135.75, 267.75) | ||
| 95% CI | 252.31, 432.47 | 164.47, 272.39 | ||
| CD56negCD16+ | ||||
| Baseline | ||||
| N (missing) | 28 (0) | 28 (0) | -1.787 | 0.074 |
| IQR | 250.00 (212.00, 363.75) | 234.00 (186.00, 286.25) | ||
| 95% CI | 253.27, 369.66 | 203.77, 277.09 | ||
| After 12 weeks treatment | ||||
| N (missing) | 26 (2) | 27 (1) | -1.475 | 0.140 |
| IQR | 226.50 (180.25, 337.25) | 203.50 (148.25,265.00) | ||
| 95% CI | 211.81, 287.84 | 179.32, 252.03 | ||
| After 24 weeks treatment | ||||
| N (missing) | 24 (4) | 23 (5) | -2.278 | 0.023b |
| IQR | 172.50 (133.00, 265.00)a | 295.50 (205.50, 334.75) | ||
| 95% CI | 174.11, 245.39 | 233.91, 323.23 |
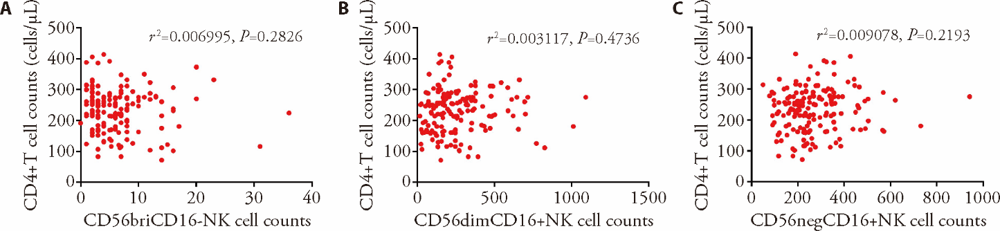
Figure 2 Correlation between the levels of NK cell subsets and CD4+T cell counts using Pearson correlation analysis A: correlation between CD56briCD16- NK cell counts and CD4+T cell counts (r2 = 0.006995, P = 0.2826); B: correlation between CD56dimCD16+ NK cell counts and CD4+T cell counts (r2 = 0.003117, P = 0.4736); C: correlation between CD56negCD16+ NK cell counts and CD4+T cell counts (r2 = 0.009078, P = 0.2193). Treatment group (n = 28): WJR (5.5 g/d, po, bid) + ART, control group (n = 28): simulant WJR (5.5 g/d, po, bid) + ART, the total treatment duration was 24 weeks for the two groups. NK: natural killer; WJR: Wenshen Jianpi recipe; ART: antiretroviral therapy.
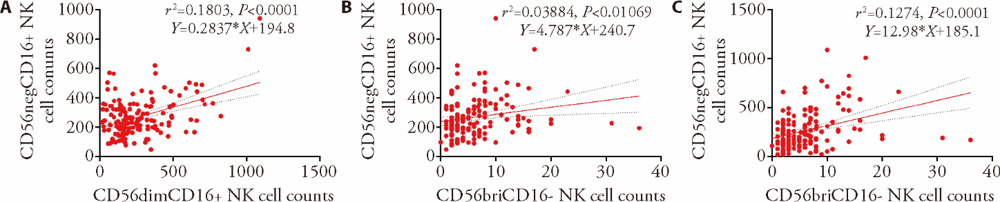
Figure 3 Correlation between the levels of different NK cell subsets using Pearson correlation analysis A: correlation between CD56dimCD16+ NK cell counts and CD56negCD16+ NK cell counts (r2 = 0.18, P < 0.001); B: correlation between CD56briCD16- NK cell counts and CD56negCD16+ NK cell counts (r2 = 0.04, P = 0.01); C: correlation between CD56briCD16- NK cell counts and CD56dimCD16+ NK cell counts (r2 = 0.13, P < 0.0001). Treatment group (n = 28): WJR (5.5 g/d, po, bid) + ART, control group (n = 28): simulant WJR (5.5 g/d, po, bid) + ART, the total treatment duration was 24 weeks for the two groups. NK: natural killer; WJR: Wenshen Jianpi recipe; ART: antiretroviral therapy.
| 1. |
Lizdany F-Á, Juan CH, Wildeman Z. NK cells in HIV-1 infection: from basic science to vaccine strategies. Front Immunol 2018; 9: 1-10.
DOI URL |
| 2. |
Anthony SF, Clifford LH. Four decades of HIV/AIDS - much accomplished, much to do. New Engl J Med 2020; 383: 1-4.
DOI URL |
| 3. |
Quillay H, Costa HE, Duriez M, et al. NK cells control HIV-1 infection of macrophages through soluble factors and cellular contacts in the human decidua. Retrovirology 2016; 13: 1-11.
DOI URL |
| 4. |
Marcus A, Lena F, Davor F, Nina B. DCs and NK cells: critical effectors in the immune response to HIV-1. Nat reviews Immunol 2011; 11: 176.
DOI URL |
| 5. |
Jiang YJ, Yang M, Sun XJ, et al. IL-10+ NK and TGF-β+NK cells play negative regulatory roles in HIV infection. BMC Infect Dis 2018; 18: 80.
DOI URL |
| 6. |
Wang YT, Lawrence L, Kyle G, et al. HIV-1-induced cytokines deplete homeostatic innate lymphoid cells and expand TCF7-dependent memory NK cells. Nat Immuno 2020; 21: 1-13.
DOI URL |
| 7. |
Liu ZB, Yang JP, Xu LR. Effectiveness and safety of Traditional Chinese Medicine in treating acquired immune deficiency syndrome: 2004-2014. Infect Dis Poverty 2015; 4: 59.
DOI URL |
| 8. | Tao Z, Huang XJ, Liu Y, et al. Efficacy of integrated Traditional Chinese Medicine and anti-retroviral therapy on immunological nonresponse in patients with human immunodeficiency virus/acquired immunodeficiency syndrome: a Meta-analysis of randomized controlled trials. J Tradit Chin Med 2021; 41: 669-676. |
| 9. |
Esposito F, Carli I, Vecchio CD, et al. Sennoside A, derived from the Traditional Chinese Medicine plant Rheum L., is a new dual HIV-1 inhibitor effective on HIV-1 replication. PhytoMedicine 2016; 23: 1383-91.
DOI URL |
| 10. | Xu LJ, Nicole G, Claudia DV, et al. From the Traditional Chinese Medicine plant Schisandra chinensis new scaffolds effective on HIV-1 reverse transcriptase resistant to non-nucleoside inhibitors. J microbio (Seoul, Korea) 2015; 53: 288-93. |
| 11. |
Li TS, Xie J, Li YJ, et al. Tripterygium wilfordii Hook F extract in cART-treated HIV patients with poor immune response: a pilot study to assess its immunomodulatory effects and safety. HIV Clin Trials 2015; 16: 49-56.
DOI URL |
| 12. | Sun WX, Liu Y, Zhou W, Li HW, Yang J, Chen ZB. Shikonin inhibits TNF-α production through suppressing PKC-NF-κB-dependent decrease of IL-10 in rheumatoid arthritis-like cell model. J Nat Med-Tokyo 2017; 71: 349-56. |
| 13. | Liu Y, Wang J, Zou W, et al. Effect of immune No. 2 granule on CD4+ T counts of AIDS patients with immune reconstitution deficiency after HAART. Zhong Yi Za Zhi 2017; 58: 34-7. |
| 14. |
Schulz KF, Altman DG, Moher D. CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. Int J Surg 2011; 9: 672-7.
DOI URL |
| 15. |
Giovanna QM, Alfonso N, Massimo S, LuCIana G, Benedetta M, Marina V. HIV-1 Nef impairs the dynamic of DC/NK crosstalk: different outcome of CD56 (dim) and CD56 (bright) NK cell subsets. FASEB J 2007; 21: 2323.
PMID |
| 16. | Carolyn R, A DK, P KE, et al. Generation of cellular immune memory and B-cell immunity is impaired by natural killer cells. Nat Commun 2015; 6: 1-14. |
| 17. | Wang J, Liu Y, He LY, et al. Distribution and development rules of Chinese Medicine syndromes in 2237 cases of HIV/AIDS. Zhong Yi Za Zhi 2012; 53: 948-51. |
| 18. | Wang W. Syndromes of the patients with HIV/AIDS before and after ART treatment and incomplete immune reconstruction. Beijing: China Academy of Medical SCIences, 2015: 67-74. |
| 19. | Li HJ, Guo HJ, Liu Y, et al. Thermal state data and clinical significance of cold or heat syndromes in HIV/AIDS patients. Beijing Zhong Yi Yao Da Xue Xue Bao (Zhong Yi Lin Chuang Ban) 2011; 18: 43-5. |
| 20. | Hou L, Li HJ, Wang J. Initial exploration of infrared imaging characteristics in HIV-infected patients. Shi Jie Zhong Xi Yi Jie He Za Zhi 2011; 6: 487-90. |
| 21. | Zou W, Wang J, Gao GJ, et al. Effects of TLR4 and TLR9 in immunological unresponsiveness AIDS patients treated with Yiqi Jianpi Granules combined with HAART. Zhong Hua Zhong Yi Yao Za Zhi 2019; 34: 4426-28. |
| 22. | Zou W, Wang J, Liu Y, Yang QL, Gao GJ, Zhang FC. Effects of Traditional Chinese Medicine combined with HAART therapy on the level of toll like receptors m RNA in AIDS patients with incomplete immune reconstitution. Zhong Hua Zhong Yi Yao Za Zhi 2017; 32: 5672-74. |
| 23. |
Milush JM, López-Vergès S, York VA, Deeks SG, Nixon DF. CD56negCD16+ NK cells are activated mature NK cells with impaired effector function during HIV-1 infection. Retrovirology 2013; 10: 158.
DOI URL |
| 24. |
Gregor FL, Cheng WJ, Yagmur F, et al. Virologically suppressed HIV patients show activation of NK cells and persistent innate immune activation. J Immuno 2012; 189: 1491-99.
DOI URL |
| 25. |
LetiCIa, Kuri-Cervantes, Salgado-Montes G, et al. Activation of NK cells is associated with HIV-1 disease progression. J Leukocyte Biol 2014; 96: 7-16.
DOI PMID |
| 26. |
Luo ZW, Li Z, Lisa M, et al. Increased natural killer cell activation in HIV-infected immunologic non-responders correlates with CD4+ T cell recovery after antiretroviral therapy and viral suppression. PLoS One 2017; 12: e0167640.
DOI URL |
| 27. |
Ilan A, Mahlab GK, Daniel E, et al. Characteristics and outcome of patients diagnosed With HIV at older age. Medicine 2016; 95: e2327.
DOI URL |
| 28. |
Mitchai M, Leeratanapetch N, Lulitanon DV, et al. Phenotypic characteristics and function of NK cell subsets in cART-treated HIV-1-infected individuals. World J AIDS 2014; 4: 293-300.
DOI URL |
| 29. |
Raquel T, Javier GC, Olga D, et al. Selective depletion of CD56(dim) NK cell subsets and maintenance of CD56 (bright) NK cells in treatment-naive HIV-1-seropositive individuals. J Clin Immun 2002; 22: 176-83.
DOI URL |
| 30. |
Xu J, Zhang J, Wang J. The application of Traditional Chinese Medicine against the tumor immune escape. J Transl Intern Med 2020; 8: 203-4.
DOI URL |
| 31. | Lan TJ, Luo XF, Mo MY, et al. Hydrolyzed seawater pearl tablet modulates the immunity via attenuating Th1/Th2 imbalance in an immunosuppressed mouse model. J Tradit Chin Med 2021; 41: 397-405. |
| 32. | Zhou ZJ, Liu XH, Wu TT, et al. Herbal formula of Bushen Jianpi (补肾健脾方) combined with sorafenib inhibits hepatocellular carCInoma growth by promoting cell apoptosis and blocking the cell cycle. J Tradit Chin Med 2021; 41: 194-202. |
| [1] | QIN Xiaoyu, WANG Chunai, XUE Jianjun, ZHANG Jie, LU Xiaoting, DING Shengshuang, GE Long, WANG Minzhen. Efficacy of electroacupuncture on myocardial protection and postoperative rehabilitation in patients undergoing cardiac surgery with cardiopulmonary bypass: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 1-15. |
| [2] | DAI Xiaoling, ZHANG Anming, LIN Hui, SHI Bei, REN Yi, WEN Hongzhu, FEI Xiaoyan, LIN Jiang. Qingchang suppositry (清肠栓) induced remission in patients with mild-to-moderate ulcerative proctitis: a multicenter, prospective, randomized, parallel-controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 156-162. |
| [3] | WANG Yichen, WU Shiyi, WANG Zhengyan, CHANG Wenling, XIE Zhihao, TANG Xing, ZHAO Songmei, ZHOU Jing, CHEN Zehong, WANG Chao, YANG Chunxia. Efficacy of Zhumian Tang formula granules (助眠汤配方颗粒) combined with eszopiclone for the treatment of poor sleep quality: a multi-center, randomized controlled, superiority trial [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 163-171. |
| [4] | YANG Yi, YE Huijun, ZHENG Huiling, JIN Lihua. Clinical observation on 90 cases of primary dysmenorrhea treated by buccal acupuncture therapy: a randomized controlled study [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 172-181. |
| [5] | DAI Zeqi, LIAO Xing, GUAN Yueyue, ZENG Zixiu, TANG Jun, HU Jing. Bloodletting puncture in the treatment of acute ischemic stroke: protocol for a mixed-method study of a multi-center randomized controlled trial and focus group [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1259-1267. |
| [6] | XU Yani, ZHANG Yutong, HE Weile, DAI Linglin, TANG Ding, WANG Jialing, ZHANG Xufen, CHEN Qin, CHEN Lifang, WANG Zhanglian, ZHAN Mingjie. Efficiency and safety of acupuncture for women with premature ovarian insufficiency: study protocol for a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1268-1274. |
| [7] | XU Xiangru, ZHOU Yi, CHEN Gang, LEI Ming, ZHANG Wen, WU Xinxin, PU Yuting, CHEN Caiyu, SUN Yuting, ZHOU Shuang, FANG Bangjiang. Clinical efficacy of Buzhong Yiqi decoction (补中益气汤) in the treatment of hospital-acquired pneumonia with multi-drug resistant bacteria: a prospective, randomized, multicenter controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1010-1018. |
| [8] | ZHAO Ming, LUO Yimiao, WANG Huichan, CAO Yu, MA Lina, PEI Hui, LI Hao. Guilingji capsule (龟龄集胶囊) for Alzheimer's disease: secondary analysis of a randomized non-inferiority controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 1019-1025. |
| [9] | ZHANG Meizhen, HAO Xiaohui, TANG Yiting, CHEN Yupeng, HE Puyu, ZHAO Liming, PANG Bing, NI Qing. Efficacy and safety of Buyang Huanwu decoction (补阳还五汤) for diabetic peripheral neuropathy: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 841-850. |
| [10] | YANG Yuqing, CHEN Yuhuan, LI Chunxiao, LING Xiao, WANG Panpan, GUO Jing, ZHANG Yingying. Effectiveness and safety of Pingxiao capsule (平消胶囊) as adjuvant therapy in treatment of breast cancer: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 851-859. |
| [11] | ZHOU Hua, LI Hui, WANG Haihua. Potential protective effects of the water-soluble Chinese propolis on experimental ulcerative colitis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 925-933. |
| [12] | WANG Chao, WU Qiong, LI Ping, WANG Zhigang, LOU Xusheng, LI Yuanyuan, ZHANG Lin. Effect of Traditional Chinese Medicine combined with Western Medicine on blood lipid levels and inflammatory factors in patients with angina pectoris in coronary heart disease identified as intermingled phlegm and blood stasis syndrome: a network Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 640-649. |
| [13] | ZHANG Xinghe, LI Qifu, YI Rong, XING Chonghui, JIN Yuhao, MENG Jiangqiong, FENG Jialei, ZHAO Siwen, LIANG Fanrong, GUO Taipin. Effect of catgut embedding at acupoints versus non-acupoints in abdominal obesity: a randomized clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 780-786. |
| [14] | LI Yue, WEN Shuting, ZHAO Runyuan, FAN Dongmei, ZHAO Dike, LIU Fengbin, MI Hong. Efficacy of active ingredients in Qingdai (Indigo Naturalis) on ulcerative colitis: a network pharmacology-based evaluation [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 124-133. |
| [15] | ZHANG Yuehong, DONG Dandan, YAN Youqin, ZHANG Hao, WANG Guangli, ZHOU Wei, LI Wei, QIU Li, LI Tingming, LIU Quan, XIA Ping, MAO Lina, YANG Danlin, YANG Lu, LIAN Fengmei, TONG Xiaolin, BA Yuanming. Effectiveness and safety of Jinshuibao capsules (金水宝胶囊) in treatment of residual cardiopulmonary symptoms in convalescent patients of coronavirus disease 2019: a pilot randomized, double-blind, placebo-controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 134-139. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

