Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (1): 124-133.DOI: 10.19852/j.cnki.jtcm.2023.01.011
• Original articles • Previous Articles Next Articles
Efficacy of active ingredients in Qingdai (Indigo Naturalis) on ulcerative colitis: a network pharmacology-based evaluation
LI Yue1, WEN Shuting2, ZHAO Runyuan2, FAN Dongmei4, ZHAO Dike3, LIU Fengbin4, MI Hong( )
)
- 1 Department of orthopedics, the First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510405, China
2 The First School of Clinical Medicine, Guangzhou University of Chinese Medicine, Guangzhou 510405, China
3 Basic Medical College, Henan University of Chinese Medicine, Zhengzhou 450046, China
4 Department of Gastroenterology, the First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510405, China
-
Received:2021-11-29Accepted:2022-02-11Online:2023-02-15Published:2023-01-10 -
Contact:MI Hong -
About author:Dr. MI Hong, Department of Gastroenterology, the First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510405, China. mihong10000@163.com. Telephone:+86-15920583726
-
Supported by:Mechanism of Chang-An Decotion in Neuropeptide Spexin related GSK-3 β Regulating Intestinal Nerve Immune Network in Ulcerative Colitis(2018A030310614);Mchanism of Chang-An Decotion in Intestinal Mucosal Immunity of Ulcerative Colitis on Exocrine Mediated Rab27(81903963);Mchanism of Chang-An decotion of Ulcerative Colitis on Exocrine Mediated GSK-3 β Regulating Th17/Treg in Ulcerative Colitis(2017KQNCX045)
Cite this article
LI Yue, WEN Shuting, ZHAO Runyuan, FAN Dongmei, ZHAO Dike, LIU Fengbin, MI Hong. Efficacy of active ingredients in Qingdai (Indigo Naturalis) on ulcerative colitis: a network pharmacology-based evaluation[J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 124-133.
share this article
| MOL_ID | molecule_name | OB (%) | DL |
|---|---|---|---|
| MOL000358 | beta-sitosterol | 36.91 | 0.75 |
| MOL001781 | Indigo | 38.20 | 0.26 |
| MOL001810 | 6-(3-oxoindolin-2-ylidene)indolo[2,1-b]quinazolin-12-one | 45.28 | 0.89 |
| MOL002309 | indirubin | 48.59 | 0.26 |
| MOL002322 | isovitexin | 31.29 | 0.72 |
| MOL011100 | bisindigotin | 41.66 | 0.39 |
| MOL011105 | indican | 34.90 | 0.23 |
| MOL011332 | 10h-indolo,[3,2-b],quinoline | 54.57 | 0.22 |
| MOL011335 | Isoindigo | 94.30 | 0.26 |
Table 1 Active compounds identified in QD
| MOL_ID | molecule_name | OB (%) | DL |
|---|---|---|---|
| MOL000358 | beta-sitosterol | 36.91 | 0.75 |
| MOL001781 | Indigo | 38.20 | 0.26 |
| MOL001810 | 6-(3-oxoindolin-2-ylidene)indolo[2,1-b]quinazolin-12-one | 45.28 | 0.89 |
| MOL002309 | indirubin | 48.59 | 0.26 |
| MOL002322 | isovitexin | 31.29 | 0.72 |
| MOL011100 | bisindigotin | 41.66 | 0.39 |
| MOL011105 | indican | 34.90 | 0.23 |
| MOL011332 | 10h-indolo,[3,2-b],quinoline | 54.57 | 0.22 |
| MOL011335 | Isoindigo | 94.30 | 0.26 |
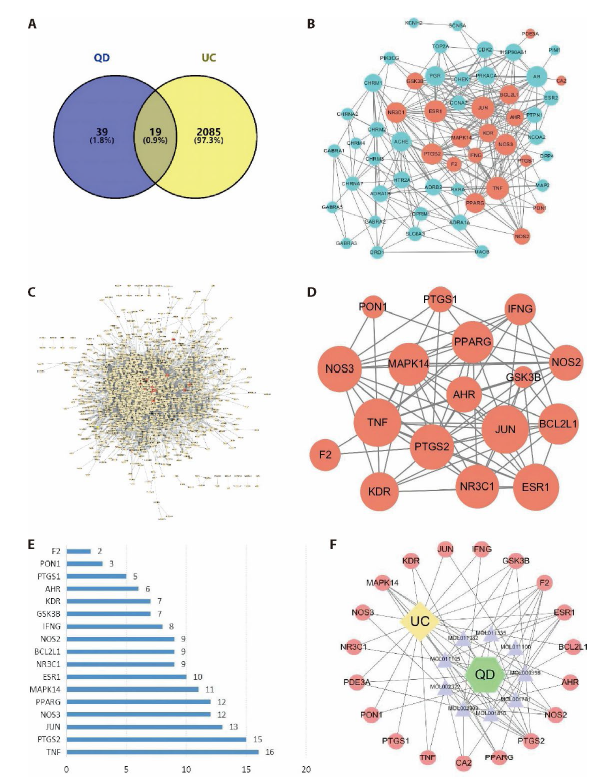
Figure 1 Results of network pharmacology-based analyses A: after searching from a variety of databases, the Venn diagram summarizes the targets in UC and QD; B: by using the Cytoscape 3.7.1 visualization software, the PPI network of QD was generated with 56 nodes and 272 edges; C: the PPI network of UC analysis was generated with 1423 nodes and 13335 edges; D: the network diagram of core targets of QD treatment against UC, which include 17 nodes and 77 edges; E: 17 targets identified according to the degrees of the nodes presented in the previous figure; F: the ultimate TCM-active ingredients-targets-disease network diagram was generated upon the above analyses. BCL2L1: apoptosis regulator Bcl-2; CA2: carbonic anhydrase II; ESR1: estrogen receptor; F2: thrombin; GSK3B: glycogen synthase kinase-3 beta; JUN: transcription factor AP-1; MAPK14: mitogen-activated protein kinase 14; NOS2: nitric oxide synthase, inducible; NR3C1: glucocorticoid receptor; PDE3A: CGMP-inhibited 3',5'-cyclic phosphodiesterase A; PON1: serum paraoxonase/arylesterase 1; PPARG: peroxisome proliferator activated receptor gamma; PTGS2: prostaglandin G/H synthase 2; ESR1: estrogen receptor; KDR: vascular endothelial growth factor receptor 2; MAPK14: mitogen-activated protein kinase 14; AHR: aryl hydro-carbon receptor; CA2: carbonic anhydrase II; IFNG: interferon gamma; NOS3: nitric-oxide synthase, endothelial; TNF: tumor necrosis factor.
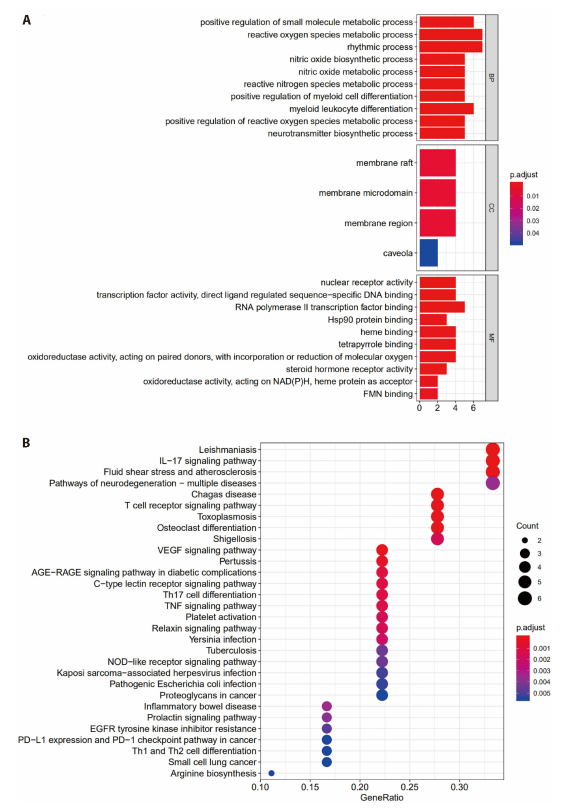
Figure 2 Results of enrichment analyses A: a total of 1028 GO entries were identified from 19 potential targets upon the GO enrichment analysis; FDR < 0.05; B: a total of 78 signal pathways were obtained upon the KEGG enrichment analysis, R language was used to plot the KEGG histogram. GO: gene ontology; KEGG: Kyoto Encyclopedia of Genes and Genomes; FDR: false discovery rate; VEGF: vascular endothlial growth factor; PD-L1: programmed cell death-Ligand 1; TNF: tumor necrosis factor; NAD (P) H: dehydrogenase, quinone 2; FMN: flavinmononucleotide.
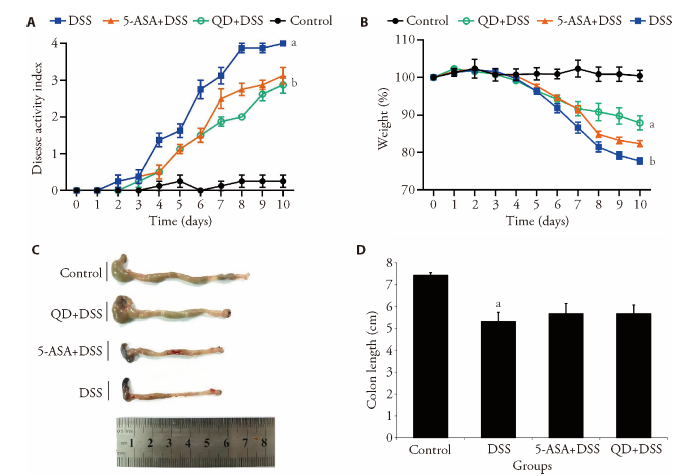
Figure 3 Ameliorative effect of QD on experimental colitis A: change of body weight of each group was calculated as the percent difference between the original body weight at day 0 and the weight on any particular day during the experimental period. Values represent the averages of 8 mice; B: DAI scores of different experimental groups of mice; C: lengths of colons were measured at the time of sacrifice. Control group treating saline treatment control for 10 d. DSS group consuming 2.5% DSS water for 7 d followed by an additional consumption period of normal drinking water for 3 d. (5-ASA+DSS) group fed with 2.5% DSS, and respectively given with 5-ASA (o.g.; 100 mg·kg-1·d-1) from day 0 till the end of experiment for a total of 10 d. (QD+DSS) group fed with 2.5% DSS, and respectively given with QD (o.g.; 1.3 g raw herbs·kg-1·d-1) from day 0 till the end of experiment for a total of 10 d. QD: Qingdai (Indigo Naturalis); DAI: disease activity index; DSS: dextran sodium sulfate; 5-ASA: 5-aminosalicylic acid. aP < 0.005, DSS group compared with Control groups; bP < 0.05, (5-ASA+DSS) group compared with DSS groups.
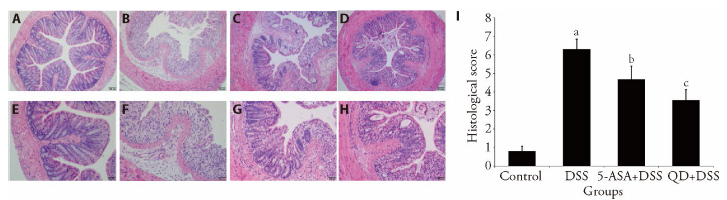
Figure 4 HE images revealing mucosal integrity in tissues in 10 d A, H: mucosal integrity in tissues (× 100 and × 200) of control group treating saline treatment control for 10 d. B, E: mucosal integrity in tissues (×100 and ×200) of DSS group consuming 2.5% DSS water for 7 d followed by an additional consumption period of normal drinking water for 3 d. C, G: mucosal integrity in tissues (× 100 and × 200) of (5-ASA+DSS) group fed with 2.5% DSS, and respectively given with 5-ASA o.g.; 100 mg·kg-1·d-1) from day 0 till the end of experiment for a total of 10 d. D, H: mucosal integrity in tissues (× 100 and × 200) of (QD + DSS) group fed with 2.5% DSS, and respectively given with QD (o.g.; 1.3 g raw herbs·kg-1·d-1) from day 0 till the end of experiment for a total of 10 d. I: Histological scores were given based on the severity of colonic damages, lymphocyte infiltration and crypt disruption (n = 8/group). HE: hematoxylin-eosin; QD: Qingdai (Indigo Naturalis); DSS: dextran sodium sulfate; 5-ASA: 5-aminosalicylic acid. aP < 0.005 DSS group compared with Control groups; bP < 0.05, (5-ASA + DSS) group compared with DSS groups; cP < 0.05, (QD + DSS) group compared with DSS groups.
| Group | n | FOXP3 | GSK-3β | p-GSK-3β | |
|---|---|---|---|---|---|
| Control | 8 | 0.10±0.04 | 0.05±0.14 | 0.10±0.04 | |
| DSS | 8 | 0.55±0.95a | 0.38±0.08 | 0.55±0.95a | |
| 5-ASA+DSSS | 8 | 1.46±0.18b | 0.15±0.61b | 0.27±0.08b | |
| QD+DSS | 8 | 1.37±0.18c | 0.13±0.50c | 0.31±0.08c | |
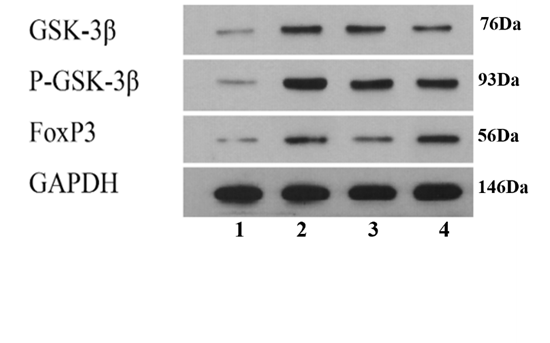
Table 2 Expression levels of FOXP3, GSK-3β and p-GSK-3β in colonic by means of Western blotting ($\bar{x}\pm s$)
| Group | n | FOXP3 | GSK-3β | p-GSK-3β | |
|---|---|---|---|---|---|
| Control | 8 | 0.10±0.04 | 0.05±0.14 | 0.10±0.04 | |
| DSS | 8 | 0.55±0.95a | 0.38±0.08 | 0.55±0.95a | |
| 5-ASA+DSSS | 8 | 1.46±0.18b | 0.15±0.61b | 0.27±0.08b | |
| QD+DSS | 8 | 1.37±0.18c | 0.13±0.50c | 0.31±0.08c | |
| Group | n | GSK-3β | TNF-α | FOXP3 | |
|---|---|---|---|---|---|
| Control | 8 | 1.00±0.07 | 1.03±0.13 | 1.04±0.13 | |
| DSS | 8 | 2.15±0.22a | 4.29±0.60a | 0.43±0.06a | |
| 5-ASA+DSS | 8 | 1.46±0.18b | 2.65±0.73b | 0.82±0.06b | |
| QD+DSS | 8 | 1.37±0.18c | 2.20±0.51c | 0.70±0.16c | |
Table 3 qRT-PCR results showed the regulation of mRNA levels of GSK-3β, TNF-α and FOXP3 in the colonic tissues of experimental mice (%,$\bar{x}\pm s$)
| Group | n | GSK-3β | TNF-α | FOXP3 | |
|---|---|---|---|---|---|
| Control | 8 | 1.00±0.07 | 1.03±0.13 | 1.04±0.13 | |
| DSS | 8 | 2.15±0.22a | 4.29±0.60a | 0.43±0.06a | |
| 5-ASA+DSS | 8 | 1.46±0.18b | 2.65±0.73b | 0.82±0.06b | |
| QD+DSS | 8 | 1.37±0.18c | 2.20±0.51c | 0.70±0.16c | |
| Group | n | IL-1β | TNF-α | IL-17A | |
|---|---|---|---|---|---|
| Control | 8 | 4.15±0.84 | 14.30±1.2 | 2.63±0.78 | |
| DSS | 8 | 19.11±1.75a | 97.25±9.68a | 8.59±2.41a | |
| 5-ASA+DSS | 8 | 10.93±0.99b | 37.83±8.41b | 3.83±0.98b | |
| QD+DSS | 8 | 11.20±0.55c | 47.24±6.05c | 3.24±1.32c | |
Table 4 Colonic levels of pro-inflammatory cytokines TNF-α, IL-1β and IL-17A were evaluated using ELISAs (pg/mL mg per tissue, $\bar{x}\pm s$)
| Group | n | IL-1β | TNF-α | IL-17A | |
|---|---|---|---|---|---|
| Control | 8 | 4.15±0.84 | 14.30±1.2 | 2.63±0.78 | |
| DSS | 8 | 19.11±1.75a | 97.25±9.68a | 8.59±2.41a | |
| 5-ASA+DSS | 8 | 10.93±0.99b | 37.83±8.41b | 3.83±0.98b | |
| QD+DSS | 8 | 11.20±0.55c | 47.24±6.05c | 3.24±1.32c | |
| Scoring | Change of body weight | Consistency of stool | Fecal occult blood |
|---|---|---|---|
| 0 | <1 % | Normal | Negative |
| 1 | 1-5% | ||
| 2 | 6-10% | Loose | Moderate |
| 3 | 11-15% | ||
| 4 | >15% | Intensively loose | Severe |
Table S1 Scoring of DAI of each group of animals
| Scoring | Change of body weight | Consistency of stool | Fecal occult blood |
|---|---|---|---|
| 0 | <1 % | Normal | Negative |
| 1 | 1-5% | ||
| 2 | 6-10% | Loose | Moderate |
| 3 | 11-15% | ||
| 4 | >15% | Intensively loose | Severe |
| Gene name | Primer | Sequence |
|---|---|---|
| β-actin | Forward | 5′-GGGAAATCGTGCGTGAC-3′ |
| Reverse | 5′-AGGCTGGAAAAGAGCCT-3′ | |
| GSK-3β | Forward | 5′-CCCACCATCACCATTAAGA-3′ |
| Reverse | 5′-AATCCACCTTGCTTTCCA-3′ | |
| Foxp3 | Forward | 5′-ACCATTGGTTTACTCGCATGT-3′ |
| Reverse | 5′-TCCACTCGCACAAAGCACTT-3′ | |
| TNF-α | Forward | 5′-CTGAACTTCGGGGTGATCGG-3′ |
| Reverse | 5′-GGCTTGTCACTCGAATTTTGAGA-3′ |
Table S2 The list of primer sequences used in the qRT-PCR experiment
| Gene name | Primer | Sequence |
|---|---|---|
| β-actin | Forward | 5′-GGGAAATCGTGCGTGAC-3′ |
| Reverse | 5′-AGGCTGGAAAAGAGCCT-3′ | |
| GSK-3β | Forward | 5′-CCCACCATCACCATTAAGA-3′ |
| Reverse | 5′-AATCCACCTTGCTTTCCA-3′ | |
| Foxp3 | Forward | 5′-ACCATTGGTTTACTCGCATGT-3′ |
| Reverse | 5′-TCCACTCGCACAAAGCACTT-3′ | |
| TNF-α | Forward | 5′-CTGAACTTCGGGGTGATCGG-3′ |
| Reverse | 5′-GGCTTGTCACTCGAATTTTGAGA-3′ |
| 1 |
Ungaro R, Mehandru S, Allen PB, Peyrin-Biroulet L, Colombel JF. Ulcerative colitis. Lancet 2017; 389: 1756-70.
DOI PMID |
| 2 |
Doherty G, Katsanos KH, Burisch J, et al. European crohn's and colitis organisation topical review on treatment withdrawal ['exit strategies'] in inflammatory bowel disease. J Crohns Colitis 2018; 12: 17-31.
DOI PMID |
| 3 | Zhao L, Zhang S, He P. Mechanistic understanding of herbal therapy in inflammatory bowel disease. Curr Pharm Des 2017; 23: 5173-79. |
| 4 |
Yang Y, Zhang Z, Li S, Ye X, Li X, He K. Synergy effects of herb extracts: pharmacokinetics and pharmacodynamic basis. Fitoterapia 2014; 92: 133-47.
DOI PMID |
| 5 | Shimada F, Yoshimatsu Y, Sujino T, Fukuda T, Naganuma M, Kanai T. Su473 natural history after the induction therapy in uc patients with indigo naturalis. Gastroenterology 2021; 160: S-708. |
| 6 | Antonio-Cisneros CM, Dávila-Jiménez MM, Elizalde-González MP, García-Díaz E. Tio2 immobilized on manihot carbon: Optimal preparation and evaluation of its activity in the decomposition of indigo carmine. Int J Mol Sci 2015; 16: 1590-612. |
| 7 | Li S. Framework and practice of network-based studies for Chinese herbal formula. Zhong Xi Yi Jie He Xue Bao 2007; 5: 489-93. |
| 8 | Ru J, Li P, Wang J, et al. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. J Cheminform 2014; 6: 13. |
| 9 |
Liu Z, Guo F, Wang Y, et al. Batman-TCM: a bioinformatics analysis tool for molecular mechanism of Traditional Chinese Medicine. Sci Rep 2016; 6: 21146.
DOI PMID |
| 10 | Xue R, Fang Z, Zhang M, Yi Z, Wen C, Shi T. TCMID: Traditional Chinese Medicine integrative database for herb molecular mechanism analysis. Nucleic Acids Res 2013; 41: D1089-95. |
| 11 | Daina A, Michielin O, Zoete V. Swisstargetprediction: updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Res 2019; 47: W357-64. |
| 12 | Liu X, Ouyang S, Yu B, et al. Pharmmapper server: a web server for potential drug target identification using pharmacophore mapping approach. Nucleic Acids Res 2010; 38: W609-14. |
| 13 |
Shannon P, Markiel A, Ozier O, et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 2003; 13: 2498-504.
DOI PMID |
| 14 |
Hamosh A, Scott AF, Amberger J, Bocchini C, Valle D, McKusick VA. Online mendelian inheritance in man (omim), a knowledgebase of human genes and genetic disorders. Nucleic Acids Res 2002; 30: 52-5.
DOI PMID |
| 15 | Yang H, Qin C, Li YH, et al. Therapeutic target database update 2016: enriched resource for bench to clinical drug target and targeted pathway information. Nucleic Acids Res 2016; 44: D1069-74. |
| 16 |
Thorn CF, Klein TE, Altman RB. Pharmgkb: the pharmacogenomics knowledge base. Methods Mol Biol 2013; 1015: 311-20.
DOI PMID |
| 17 | Kim J, So S, Lee HJ, Park JC, Kim JJ, Lee H. Digsee: disease gene search engine with evidence sentences (version cancer). Nucleic Acids Res 2013; 41: W510-7. |
| 18 |
Mandloi S, Chakrabarti S. Palm-ist: pathway assembly from literature mining--an information search tool. Sci Rep 2015; 5: 10021.
DOI PMID |
| 19 | Liu Y, Liang Y, Wishart D. Polysearch2: a significantly improved text-mining system for discovering associations between human diseases, genes, drugs, metabolites, toxins and more. Nucleic Acids Res 2015; 43: W535-42. |
| 20 |
de Leeuw N, Dijkhuizen T, Hehir-Kwa JY, et al. Diagnostic interpretation of array data using public databases and internet sources. Hum Mutat 2012; 33: 930-40.
DOI PMID |
| 21 | Szklarczyk D, Morris JH, Cook H, et al. The string database in 2017: quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res 2017; 45: D362-8. |
| 22 | Sanchez-Munoz F, Dominguez-Lopez A, Yamamoto-Furusho JK. Role of cytokines in inflammatory bowel disease. World J Gastroenterol 2008; 14: 4280-8. |
| 23 |
Schubert LA, Jeffery E, Zhang Y, Ramsdell F, Ziegler SF. Scurfin (foxp3) acts as a repressor of transcription and regulates t cell activation. J Biol Chem 2001; 276: 37672-9.
DOI PMID |
| 24 |
Fontenot JD, Gavin MA, Rudensky AY. Foxp3 programs the development and function of CD4+CD25+ regulatory t cells. Nat Immunol 2003; 4: 330-6.
DOI PMID |
| 25 |
Naganuma M. Treatment with indigo naturalis for inflammatory bowel disease and other immune diseases. Immunol Med 2019; 42: 16-21.
DOI PMID |
| 26 | Leclerc S, Garnier M, Hoessel R, et al. Indirubins inhibit glycogen synthase kinase-3 beta and cdk5/p25, two protein kinases involved in abnormal tau phosphorylation in alzheimer's disease. A property common to most cyclin-dependent kinase inhibitors? J Biol Chem 2001; 276: 251-60. |
| 27 |
Eisenbrand G, Hippe F, Jakobs S, Muehlbeyer S. Molecular mechanisms of indirubin and its derivatives: Novel anticancer molecules with their origin in traditional Chinese phytomedicine. J Cancer Res Clin Oncol 2004; 130: 627-35.
DOI PMID |
| 28 | Xiao HT, Peng J, Wen B, et al. Indigo naturalis suppresses colonic oxidative stress and th1/th 17 responses of dss-induced colitis in mice. Oxid Med Cell Longev 2019;2019: 9480945. |
| 29 | Hofmann C, Dunger N, Schölmerich J, Falk W, Obermeier F. Glycogen synthase kinase 3-β: a master regulator of toll-like receptor-mediated chronic intestinal inflammation. Inflamm Bowel Dis 2010; 16: 1850-8. |
| 30 | Whittle BJ, Varga C, Pósa A, Molnár A, Collin M, Thiemermann C. Reduction of experimental colitis in the rat by inhibitors of gly-cogen synthase kinase-3beta. Br J Pharmacol 2006; 147: 575-82. |
| 31 | Beurel E, Grieco SF, Jope RS. Glycogen synthase kinase-3 (gsk3): regulation, actions, and diseases. Pharmacol Ther 2015; 148: 114-31. |
| 32 |
Elson CO, Cong Y, Weaver CT, et al. Monoclonal anti-interleukin 23 reverses active colitis in a t cell-mediated model in mice. Gastroenterology 2007; 132: 2359-70.
DOI PMID |
| 33 |
Takahashi-Yanaga F. Activator or inhibitor? Gsk-3 as a new drug target. Biochem Pharmacol 2013; 86: 191-9.
DOI PMID |
| 34 | Cortés-Vieyra R, Bravo-Patiño A, Valdez-Alarcón JJ, Juárez MC, Finlay BB, Baizabal-Aguirre VM. Role of glycogen synthase kinase-3 beta in the inflammatory response caused by bacterial pathogens. J Inflamm (Lond) 2012; 9: 23. |
| 35 | Mi H, Liu FB, Li HW, Hou JT, Li PW. Anti-inflammatory effect of chang-an-shuan on tnbs-induced experimental colitis in rats. BMC Complement Altern Med 2017; 17: 315. |
| 36 |
Harada K, Shimoda S, Sato Y, Isse K, Ikeda H, Nakanuma Y. Periductal interleukin-17 production in association with biliary innate immunity contributes to the pathogenesis of cholangiopathy in primary biliary cirrhosis. Clin Exp Immunol 2009; 157: 261-70.
DOI PMID |
| 37 | Graham JA, Fray M, de Haseth S, et al. Suppressive regulatory t cell activity is potentiated by glycogen synthase kinase 3{beta} inhibition. J Biol Chem 2010;285: 32852-59. |
| 38 |
Cosmi L, Santarlasci V, Maggi L, Liotta F, Annunziato F. Th17 plasticity: pathophysiology and treatment of chronic inflammatory disorders. Curr Opin Pharmacol 2014; 17: 12-6.
DOI PMID |
| 39 |
Romagnani S, Maggi E, Liotta F, Cosmi L, Annunziato F. Properties and origin of human th17 cells. Mol Immunol 2009; 47: 3-7.
DOI PMID |
| 40 |
Annunziato F, Cosmi L, Liotta F, Maggi E, Romagnani S. The phenotype of human th17 cells and their precursors, the cytokines that mediate their differentiation and the role of th17 cells in inflammation. Int Immunol 2008; 20: 1361-8.
DOI PMID |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||