Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (5): 732-740.DOI: 10.19852/j.cnki.jtcm.20220719.001
• Research Articles • Previous Articles Next Articles
Electroacupuncture at Tianshu (ST25) and Zusanli (ST36) alleviates stress-induced irritable bowel syndrome in mice by modulating gut microbiota and corticotropin-releasing factor
SUN Mengzhu1, ZHANG Yujie1, SONG Yafang1, GUO Jing1, ZHAO Tingting1, WANG Yuhang1, PEI Lixia2( ), SUN Jianhua2(
), SUN Jianhua2( )
)
- 1 Department of Acupuncture and Rehabilitation, the Affiliated Hospital of Nanjing University of Chinese Meidicne, Nanjing 210023, China
2 Department of Acupuncture and Rehabilitation, Jiangsu Province Hospital of Chinese Medicine, Nanjing 210029, China
-
Received:2021-07-06Accepted:2021-10-14Online:2022-10-15Published:2022-07-19 -
Contact:PEI Lixia,SUN Jianhua -
About author:Prof. PEI Lixia, Department of Acupuncture and Rehabilitation, Jiangsu Province Hospital of Chinese Medicine, Nanjing 210029, China. plx11801758@163.com Telephone: +86-13851402838; +86-13914722816; +86-18851182170
Prof. SUN Jianhua, Department of Acupuncture and Rehabilitation, Jiangsu Province Hospital of Chinese Medicine, Nanjing 210029, China. drjhsun@sina.com;
-
Supported by:Natural Science Foundation-funded Project: Mechanism of Electroacupuncture Regulating CRF-NLRP6 Inflammasome-Related Pathways in Intestinal Flora Immune Dialogue(81804193);Leading Talents of Jiangsu Provincial Administration of TCM-funded Project: Clinical Effect and Mechanism of Acupuncture and Moxibustion on Irritable Bowel Syndrome(SLJ0206);eak Talent of Jiangsu Province Hospital of Chinese Medicine-funded Project: Clinical Effect and Mechanism of Acupuncture and Moxibustion in the Treatment of Functional Gastrointestinal Diseases(y2018rc05);Research and Innovation Project for Postgraduates of Jiangsu Province-funded Project: Mechanism of Electroacupuncture Regulating Bile Acid Receptor to Alleviate Visceral Hypersensitivity in Irritable Bowel Syndrome(YCX20_1469)
Cite this article
SUN Mengzhu, ZHANG Yujie, SONG Yafang, GUO Jing, ZHAO Tingting, WANG Yuhang, PEI Lixia, SUN Jianhua. Electroacupuncture at Tianshu (ST25) and Zusanli (ST36) alleviates stress-induced irritable bowel syndrome in mice by modulating gut microbiota and corticotropin-releasing factor[J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 732-740.
share this article
| Groups | n | AWR scores | |||
|---|---|---|---|---|---|
| 15 mm Hg | 30 mm Hg | 45 mm Hg | 60 mm Hg | ||
| Normal | 10 | 0.57±0.39 | 1.40±0.44 | 2.03±0.46 | 3.00±0.57 |
| WAS | 10 | 1.30±0.33a | 2.27±0.49a | 2.80±0.59c | 3.70±0.43d |
| WAS+EA | 10 | 0.80±0.48b | 1.77±0.52b | 2.27±0.49b | 3.30±0.46 |
Table 1 AWR scores at different stimulation pressures in normal, WAS and WAS+EA groups ($\bar{x}$ ± s)
| Groups | n | AWR scores | |||
|---|---|---|---|---|---|
| 15 mm Hg | 30 mm Hg | 45 mm Hg | 60 mm Hg | ||
| Normal | 10 | 0.57±0.39 | 1.40±0.44 | 2.03±0.46 | 3.00±0.57 |
| WAS | 10 | 1.30±0.33a | 2.27±0.49a | 2.80±0.59c | 3.70±0.43d |
| WAS+EA | 10 | 0.80±0.48b | 1.77±0.52b | 2.27±0.49b | 3.30±0.46 |
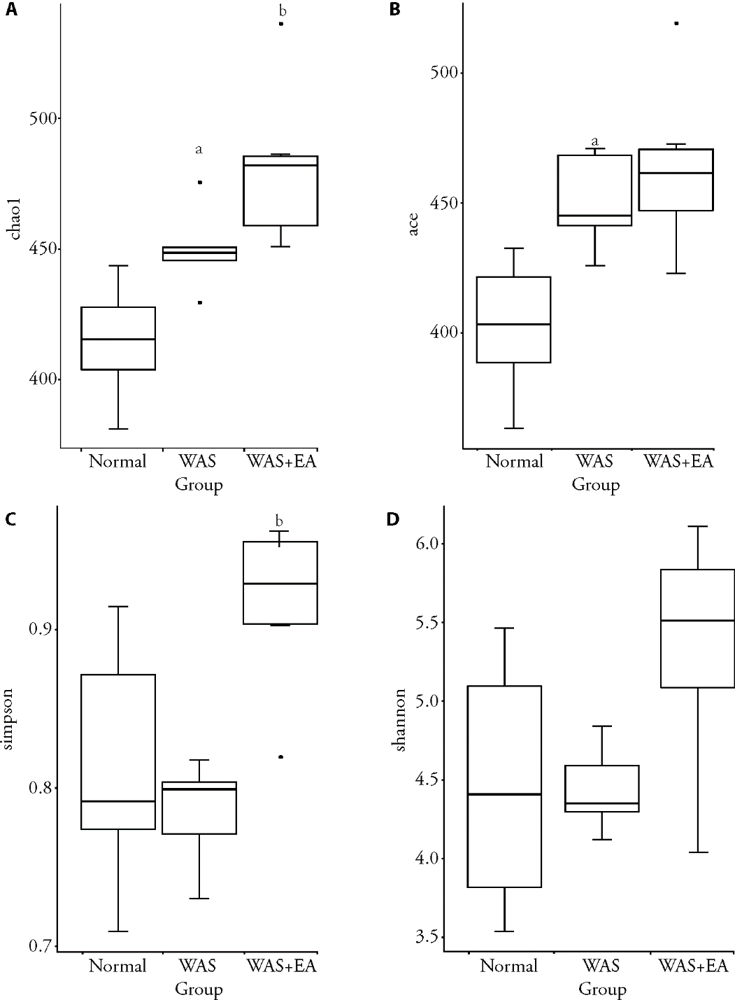
Figure 1 α-diversity of the gut microbiota A: Chao1 index in normal, WAS and WAS + EA groups; B: Ace index in normal, WAS and WAS + EA groups; C: Simpson index in normal, WAS and WAS + EA groups; D: Shannon index in normal, WAS and WAS + EA groups. Normal group: without treatment; WAS group: subjected to WAS for 1 h daily on 10 consecutive days; WAS + EA group: subjected to WAS for 1 h daily on 10 consecutive days and EA at bilateral Tianshu (ST25) and Zusanli (ST36) for 7 consecutive days. WAS: water avoidance stress; EA: electroacupuncture. aP < 0.01 versus normal group; bP < 0.01 versus WAS group.
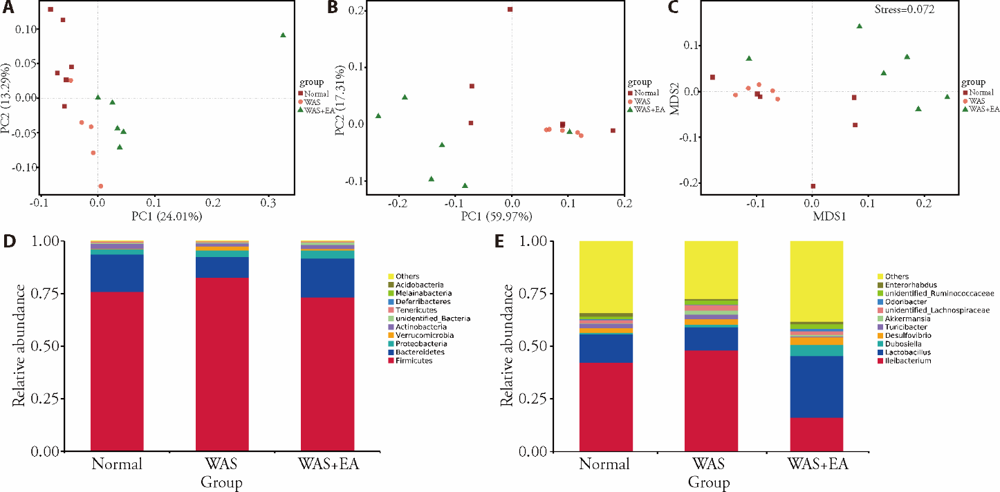
Figure 2 Structure of the gut microbiota A, B: PCoA plots of the unweighted and weighted UniFrac distances in normal, WAS and WAS + EA groups; C: NMDS ordination in normal, WAS and WAS + EA groups. Stress < 0.2 shows that NMDS can accurately reflect the difference between samples; D-E: the relative abundances of the microbial communities for each group at the phylum and genus levels. Normal group: without treatment; WAS group: subjected to WAS for 1 h daily on 10 consecutive days; WAS+EA group: subjected to WAS for 1 h daily on 10 consecutive days and EA at bilateral Tianshu (ST25) and Zusanli (ST36) for 7 consecutive days. WAS: water avoidance stress; EA: electroacupuncture; PCoA: principal co-ordinate analysis; NMDS: non-metric multidimensional scaling.
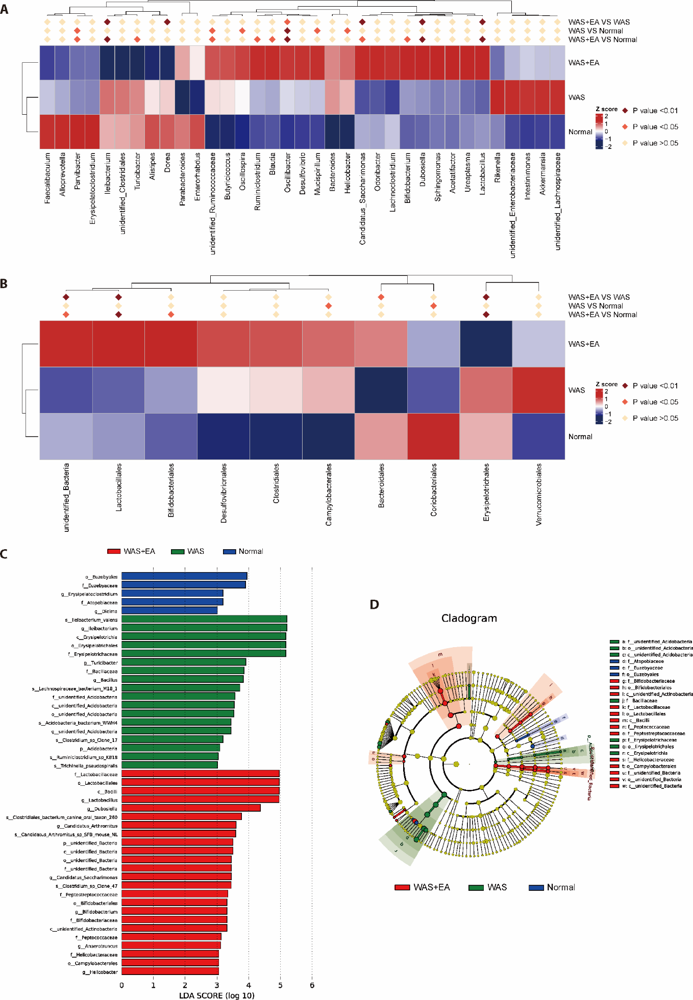
Figure 3 Composition of the gut microbiota A-B: MetaStat analysis at the genus and order level in normal, WAS and WAS+EA groups; C: histogram of LEfSe in normal, WAS and WAS+EA groups; D: cladogram of LEfSe in normal, WAS and WAS+EA groups. LDA score > 3.0. The red, green, or blue shading in the cladogram depicts bacterial taxa that were significantly higher in either the normal, WAS or WAS+EA groups, as indicated. p: phylum; c: class; o: order; f: family; g: genus; s: species. Normal group: without treatment; WAS group: subjected to WAS for 1 h daily on 10 consecutive days; WAS+EA group: subjected to WAS for 1 h daily on 10 consecutive days and EA at bilateral Tianshu (ST25) and Zusanli (ST36) for 7 consecutive days. WAS: water avoidance stress; EA: electroacupuncture; LDA: linear discriminant analysis.

Figure 4 Effect of EA on the expression of CRF in colon tissues (magnification, × 200) A: relative mRNA expression of CRF in normal, WAS and WAS + EA groups; B: relative fluorescence intensity of CRF in normal, WAS and WAS + EA groups; C1-C3: confocal images of CRF, DAPI and merge (CRF + DAPI) in normal group; C4-C6: confocal images of CRF, DAPI and merge (CRF + DAPI) in WAS group; C7-C9: confocal images of CRF, DAPI and merge (CRF + DAPI) in WAS + EA group. Micrographs represent labeling of CRF (green) in mice colon tissues. Nuclear staining was conducted with DAPI (blue). Data were mean ± standard deviation (n = 3/ group). Normal group: without treatment; WAS group: subjected to WAS for 1 h daily on 10 consecutive days; WAS + EA group: subjected to WAS for 1 h daily on 10 consecutive days and EA at bilateral Tianshu (ST25) and Zusanli (ST36) for 7 consecutive days. EA: electroacupuncture; WAS: water avoidance stress; CRF: corticotropin-releasing factor; DAPI: 4',6-diamidino-2-phenylindole. aP < 0.001, cP < 0.01 versus normal group; bP < 0.001, dP < 0.05 versus WAS group.
| 1. |
Sultan S, Malhotra A. Irritable bowel syndrome. Ann Intern Med 2017; 166: ITC81-96.
DOI URL |
| 2. |
Canavan C, West J, Card T. Review article: the economic impact of the irritable bowel syndrome. Aliment Pharmacol Ther 2014; 40: 1023-34.
DOI URL |
| 3. |
Koloski NA, Jones M, Talley NJ. Evidence that independent gut-to-brain and brain-to-gut pathways operate in the irritable bowel syndrome and functional dyspepsia: a 1-year population-based prospective study. Aliment Pharmacol Ther 2016; 44: 592-600.
DOI URL |
| 4. | Tache Y, Larauche M, Yuan PQ, Million M. Brain and gut CRF signaling: biological actions and role in the gastrointestinal tract. Curr Mol Pharmacol 2018; 11: 51-71. |
| 5. |
Gué M, Del Rio-Lacheze C, Eutamene H, Théodorou V, Fioramonti J, Buéno L. Stress-induced visceral hypersensitivity to rectal distension in rats: role of CRF and mast cells. Neurogastroenterol Motil 1997; 9: 271-9.
DOI URL |
| 6. |
Schwetz I, McRoberts JA, Coutinho SV, et al. Corticotropin-releasing factor receptor 1 mediates acute and delayed stress-induced visceral hyperalgesia in maternally separated Long-Evans rats. Am J Physiol Gastrointest Liver Physiol 2005; 289: G704-12.
DOI URL |
| 7. |
Lynch SV, Pedersen O. The human intestinal microbiome in health and disease. N Engl J Med 2016; 375: 2369-79.
DOI URL |
| 8. |
Maharshak N, Ringel Y, Katibian D, et al. Fecal and mucosa-associated intestinal microbiota in patients with diarrhea-predominant irritable bowel syndrome. Dig Dis Sci 2018; 63: 1890-99.
DOI URL |
| 9. |
Botschuijver S, Roeselers G, Levin E, et al. Intestinal fungal dysbiosis is associated with visceral hypersensitivity in patients with irritable bowel syndrome and rats. Gastroenterology 2017; 153: 1026-39.
DOI PMID |
| 10. |
Crouzet L, Gaultier E, Del'Homme C, et al. The hypersensitivity to colonic distension of IBS patients can be transferred to rats through their fecal microbiota. Neurogastroenterol Motil 2013; 25: e272-82.
DOI URL |
| 11. |
Preston K, Krumian R, Hattner J, de Montigny D, Stewart M, Gaddam S. Lactobacillus acidophilus CL1285, Lactobacillus casei LBC80R and Lactobacillus rhamnosus CLR2 improve quality-of-life and IBS symptoms: a double-blind, randomised, placebo-controlled study. Benef Microbes 2018; 9: 697-706.
DOI PMID |
| 12. |
Ait-Belgnaoui A, Payard I, Rolland C, et al. Bifidobacterium longum and Lactobacillus helveticus synergistically suppress stress-related visceral hypersensitivity through hypothalamic-pituitary-adrenal axis modulation. J Neurogastroenterol Motil 2018; 24: 138-46.
DOI URL |
| 13. | Burokas A, Moloney RD, Dinan TG, Cryan JF. Microbiota regulation of the Mammalian gut-brain axis. Adv Appl Microbiol 2015; 91:1-62. |
| 14. |
Moloney RD, Johnson AC, O'Mahony SM, Dinan TG, Greenwood-Van Meerveld B, Cryan JF. Stress and the microbiota-gut-brain axis in visceral pain: relevance to irritable bowel syndrome. CNS Neurosci Ther 2016; 22: 102-17.
DOI PMID |
| 15. | Chen Y, Zhao Y, Luo DN, Zheng H, Li Y, Zhou SY. Electroacupuncture regulates disorders of gut-brain interaction by decreasing corticotropin-releasing factor in a rat model of IBS. Gastroenterol Res Pract 2019; 2019: 1759842. |
| 16. |
Song YF, Pei LX, Chen L, et al. Electroacupuncture relieves irritable bowel syndrome by regulating IL-18 and gut microbial dysbiosis in a trinitrobenzene sulfonic acid-induced post-inflammatory animal model. Am J Chin Med 2020; 48: 77-90.
DOI URL |
| 17. | Pei LX, Zhang W, Song YF, et al. Electroacupuncture of “Tianshu” (ST25) suppresses visceral pain possibly by down-regulating mast cell activation, and tryptase and SP expression in rats with post-infectious irritable bowel syndrome. Zhen Ci Yan Jiu 2018; 43: 419-23. |
| 18. |
Fourie NH, Wang D, Abey SK, et al. Structural and functional alterations in the colonic microbiome of the rat in a model of stress induced irritable bowel syndrome. Gut Microbes 2017; 8: 33-45.
DOI URL |
| 19. |
Al-Chaer ED, Kawasaki M, Pasricha PJ. A new model of chronic visceral hypersensitivity in adult rats induced by colon irritation during postnatal development. Gastroenterology 2000; 119: 1276-85.
PMID |
| 20. | Song Y, Geng H, Chen L, et al. Electroacupuncture relieves irritable bowel syndrome by increasing expression of nucleotide-binding oligomerization domain protein-like receptor family pyrin domain containing 6 in water-avoidance stress mice. J Tradit Chin Med 2020; 40: 407-13. |
| 21. |
Han JS. Acupuncture: neuropeptide release produced by electrical stimulation of different frequencies. Trends Neurosci 2003; 26: 17-22.
DOI URL |
| 22. |
Backhed F, Fraser CM, Ringel Y, et al. Defining a healthy human gut microbiome: current concepts, future directions, and clinical applications. Cell Host Microbe 2012; 12: 611-22.
DOI URL |
| 23. |
Rajilić-Stojanović M, Jonkers DM, Salonen A, et al. Intestinal microbiota and diet in IBS: causes, consequences, or epiphenomena? Am J Gastroenterol 2015; 110: 278-87.
DOI PMID |
| 24. |
Jeffery IB, O'Toole PW, Öhman L, et al. An irritable bowel syndrome subtype defined by species-specific alterations in faecal microbiota. Gut 2012; 61: 997-1006.
DOI PMID |
| 25. |
Gagliardi A, Totino V, Cacciotti F, et al. Rebuilding the gut microbiota ecosystem. Int J Environ Res Public Health 2018; 15: 1679.
DOI URL |
| 26. |
Tap J, Derrien M, Törnblom H, et al. Identification of an intestinal microbiota signature associated with severity of irritable bowel syndrome. Gastroenterology 2017; 152: 111-23.
DOI URL |
| 27. |
Wang JM, Yang MX, Wu QF, et al. Improvement of intestinal flora: accompany with the antihypertensive effect of electroacupuncture on stage 1 hypertension. Chin Med 2021; 16: 7.
DOI URL |
| 28. |
Gorbach SL. Probiotics in the third millennium. Dig Liver Dis 2002; 34 Suppl 2: S2-7.
DOI URL |
| 29. |
Preston K, Krumian R, Hattner J, de Montigny D, Stewart M, Gaddam S. Lactobacillus acidophilus CL1285, Lactobacillus casei LBC80R and Lactobacillus rhamnosus CLR2 improve quality-of-life and IBS symptoms: a double-blind, randomised, placebo-controlled study. Benef Microbes 2018; 9: 697-706.
DOI PMID |
| 30. |
Zhang L, Chen X, Wang H, et al. "Adjusting internal organs and dredging channel" electroacupuncture ameliorates insulin resistance in type 2 diabetes mellitus by regulating the intestinal flora and inhibiting inflammation. Diabetes Metab Syndr Obes 2021; 14: 2595-607.
DOI URL |
| 31. | Hou TS, Han XX, Yang Y, et al. Effect of electroacupuncture intervention on enteric microecology in ulcerative colitis rats. Zhen Ci Yan Jiu 2014; 39: 27-34. |
| 32. | Gracie DJ, Hamlin PJ, Ford AC. The influence of the brain-gut axis in inflammatory bowel disease and possible implications for treatment. Lancet Gastroenterol Hepatol 2019; 4: 632-42. |
| [1] | QIN Xiaoyu, WANG Chunai, XUE Jianjun, ZHANG Jie, LU Xiaoting, DING Shengshuang, GE Long, WANG Minzhen. Efficacy of electroacupuncture on myocardial protection and postoperative rehabilitation in patients undergoing cardiac surgery with cardiopulmonary bypass: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 1-15. |
| [2] | SUN Qianhui, CHENG Kai, DAI Xingye, YANG Zhiwen, WU Xiaoling, XU Chang, QIU Xinghua, GAO Xiaofeng, LIU Daonan, YANG Qirui. Effect of electroacupuncture at Neiguan (PC6) at different time points on myocardial ischemia reperfusion arrhythmia in rats [J]. Journal of Traditional Chinese Medicine, 2024, 44(1): 113-121. |
| [3] | DU Zhongheng, CONG Wenjie, TANG Kejing, ZHENG Qiqi, SONG Zhiwei, CHEN Yong, YANG Su, ZHANG Chunwu, YE Tianshen. Electroacupuncture stimulating Zusanli (ST36), Sanyinjiao (SP6) in mice with collagen-induced arthritis leads to adenosine A2A receptor-mediated alteration of p38α mitogen-activated protein kinase signaling and inhibition of osteoclastogenesis [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1103-1109. |
| [4] | LI Chaoran, YANG Yan, FENG Chuwen, LI Heng, QU Yuanyuan, WANG Yulin, WANG Delong, WANG Qingyong, GUO Jing, SHI Tianyu, SUN Xiaowei, WANG Xue, HOU Yunlong, SUN Zhongren, YANG Tiansong. Integrated 'omics analysis for the gut microbiota response to moxibustion in a rat model of chronic fatigue syndrome [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1176-1189. |
| [5] | JIANG Jianzhen, ZHANG Xin, LUO Zhenguo, SU Chengguo, ZHOU Haiyan, JIANG Yuqing, XIAO Xianjun, CHEN Yunfei, ZHU Jun. Efficacy of electroacupuncture stimulating Zusanli (ST36) and Xuanzhong (GB39) on synovial angiogenesis in rats with adjuvant arthritis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 955-962. |
| [6] | LI Zhihao, HAN Wenjun, SONG Xiuling, LI Yan, CHEN Yuelai. Electroacupuncture stimulating Zhongji (CV3), Guanyuan (CV4), and bilateral Dahe (KI12) attenuates inflammation in rats with chronic nonbacterial prostatitis induced by estradiol through inhibiting toll-like receptor 4 pathway [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 963-972. |
| [7] | DING Luobin, WANG Huajun, LI Yao, LI Jia, LI Ling, GAO Yangping, GUAN Jian, GENG Weiqiang. Electroacupuncture stimulating Neixiyan (EX-LE5) and Dubi (ST35) alleviates osteoarthritis in rats induced by anterior cruciate ligament transaction via affecting DNA methylation regulated transcription of miR-146a and miR-140-5p [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 983-990. |
| [8] | ZHOU Jun, WANG Junhua, LI Xiaobing, WAN Chenyi, LI Fangjun, Lü Yanni, CHEN Hao, SUN Meiying. Efficacy of Heshouwu (Radix Polygoni Multiflori) on gut mircobiota in mice with autoimmune encephalomyelitis [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 676-685. |
| [9] | JIANG Yiqian, ZHOU Xibin, PU Wenyuan, ZHOU Chunxiang. Sanwu Baisan decoction (三物白散) inhibits colorectal cancer progression in mice by remodeling gut microbiota and tumorigenesis [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 466-473. |
| [10] | SUN Mengzhu, ZHANG Yujie, SONG Yafang, GUO Jing, WANG Yuhang, XIN Chen, GU Dongmei, SUN Jianhua, PEI Lixia. Electroacupuncture alleviates water avoidance stress-induced irritable bowel syndrome in mice by improving intestinal barrier functions and suppressing the expression of inflammatory cytokines [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 494-500. |
| [11] | YANG Yang, YUAN Haining, JIA Hongxiao, NING Yanzhe, WANG Di, ZHANG Lei, YAN Kaijuan, GUO Yumeng, WANG Fei, SUN Weishuang, CHEN Pei. Therapy of replenishing Yin and regulating Yang for manic episode in bipolar disorder: study protocol for a prospective, double-blind, randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 594-601. |
| [12] | HAN Rui, CHANG Junzhao, LIU Qianqian, LIU Haitao, LI Junwei. Efficacy of electroacupunture at Zusanli (ST36) on jumping-injured muscle based on transcriptome sequencing and genes analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 322-328. |
| [13] | Minh Duc Nguyen, Thanh Van Tran, Quoc Vinh Nguyen, Ninh Khac Nguyen, Son Truong Vu, Luu Trong Nguyen, Linh Vu Phuong Dang. Effectiveness on post-stroke hemiplegia in patients: electroacupuncture plus cycling vs electroacupuncture alone [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 352-358. |
| [14] | XU Chang, LI Na, WU Xiaoling, DAI Xingye, YANG Zhiwen, SUN Qianhui, SHI Tianyu, CHAI Yemao, PANG Dandan, CHENG Kai. Effect of electroacupuncture on inflammatory signal expression in local tissues of rats with chronic pelvic pain syndrome based on purinergic 2X7 receptor/NOD-like receptor pyrin domain-containing 3 signal pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 965-971. |
| [15] | HU Xijiao, CHENG Yinglong, KANG Huanan, LI Shuoxi, WANG Yawen, LIU Jinzhe, SUN Yiming, LIU Li. Electroacupuncture attenuates chronic salpingitis via transforming growth factor-β1/p38 mitogen-activated protein kinase signaling pathway [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 781-787. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

