Journal of Traditional Chinese Medicine ›› 2024, Vol. 44 ›› Issue (2): 260-267.DOI: 10.19852/j.cnki.jtcm.20240203.003
Previous Articles Next Articles
Anti-inflammatory mechanism of the non-volatile ingredients originated from Guanghuoxiang (Pogostemonis Herba) based on high performance liquid chromatography-heated electron spray ionization-high resolution mass spectroscope and cell metabolomics
JING Wenguang1, LIN Xiaoyu2, LI Chu2, ZHAO Xiaoliang3, CHENG Xianlong1, WANG Penglong2, WEI Feng1( ), MA Shuangcheng1(
), MA Shuangcheng1( )
)
- 1 Institute of Control of Chinese Traditional Medicine and Ethnic Medicine, National Institutes for Food and Drug Control, Beijing 102629, China
2 School of Traditional Chinese Medicine, Beijing University of Chinese Medicine, Beijing 102488, China
3 Beijing Key Laboratory of Traditional Chinese Medicine Basic Research on Prevention and Treatment of Major Diseases, Experimental Research Center, China Academy of Chinese Medical Sciences, Beijing 100700, China
-
Received:2023-04-22Accepted:2023-09-05Online:2024-04-15Published:2024-02-03 -
Contact:MA Shuangcheng, Institute of Control of Chinese Traditional Medicine and Ethnic Medicine, National Institutes for Food and Drug Control, Beijing 102629, China.masc@nifdc.org.cn ; WEI Feng, Institute of Control of Chinese Traditional Medicine and Ethnic Medicine, National Institutes for Food and Drug Control, Beijing 102629, China.weifeng@nifdc.org.cn Telephone: +86-10-53852020 -
Supported by:Institute of Chinese Medicine Discipline Construction Project of National Institutes for Food and Drug Control: Disciplinary Construction Program of Chinese Medicine Institute of NIFDC(1020050090116);Training Fund for Academic Leaders of NIFDC(2023X10);Program of State Drug Administraion-Key Laboratory of Quality Control of Chinese Medicinal Materials and Decoction Pieces(2022GSMPA-KL02)
Cite this article
JING Wenguang, LIN Xiaoyu, LI Chu, ZHAO Xiaoliang, CHENG Xianlong, WANG Penglong, WEI Feng, MA Shuangcheng. Anti-inflammatory mechanism of the non-volatile ingredients originated from Guanghuoxiang (Pogostemonis Herba) based on high performance liquid chromatography-heated electron spray ionization-high resolution mass spectroscope and cell metabolomics[J]. Journal of Traditional Chinese Medicine, 2024, 44(2): 260-267.
share this article
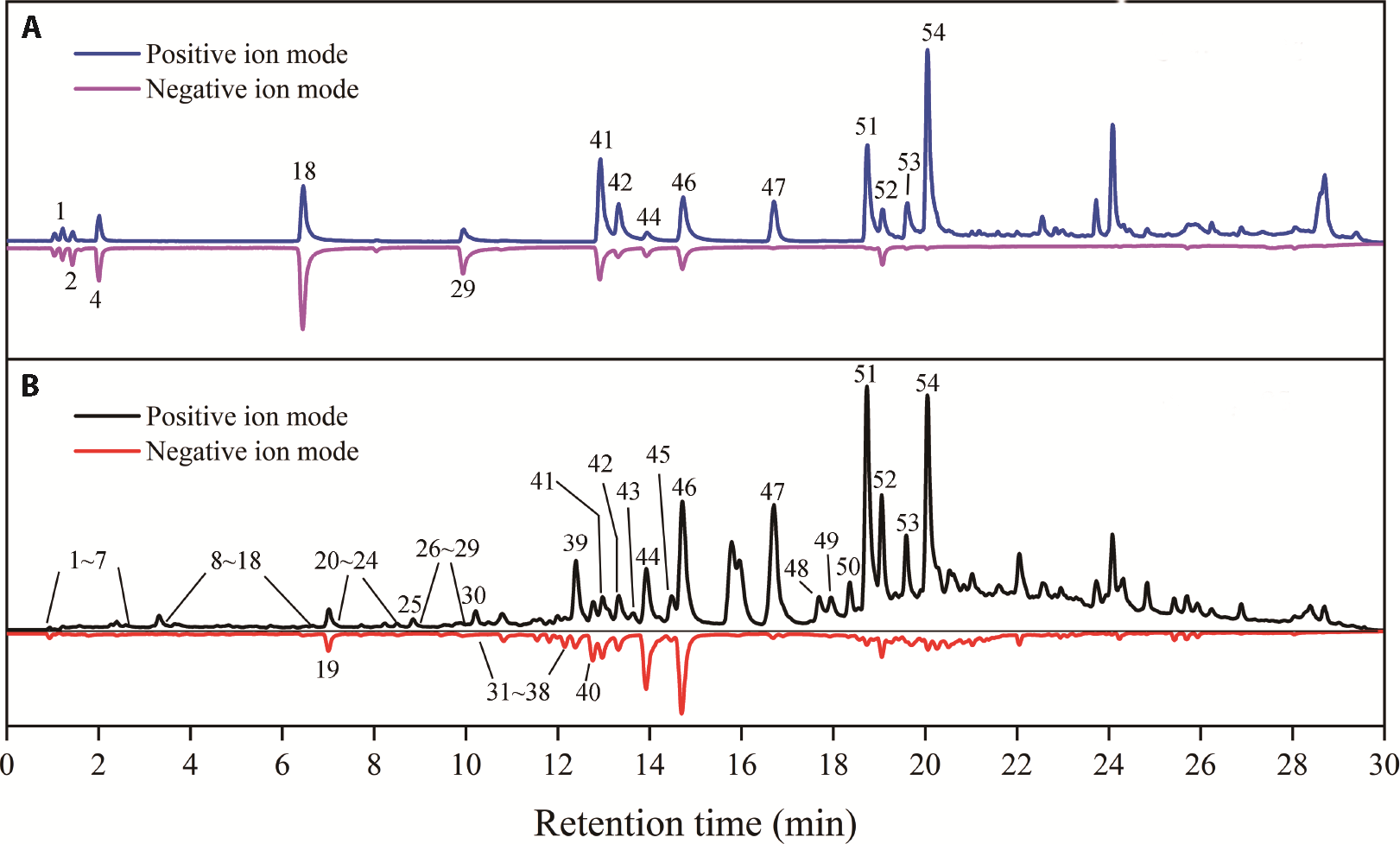
Figure 1 Total ion flow diagram of standard substances and the non-volatile ingredients of patchouli A: total ion flow diagram of the standard; B: total ion flow diagram of the sample.
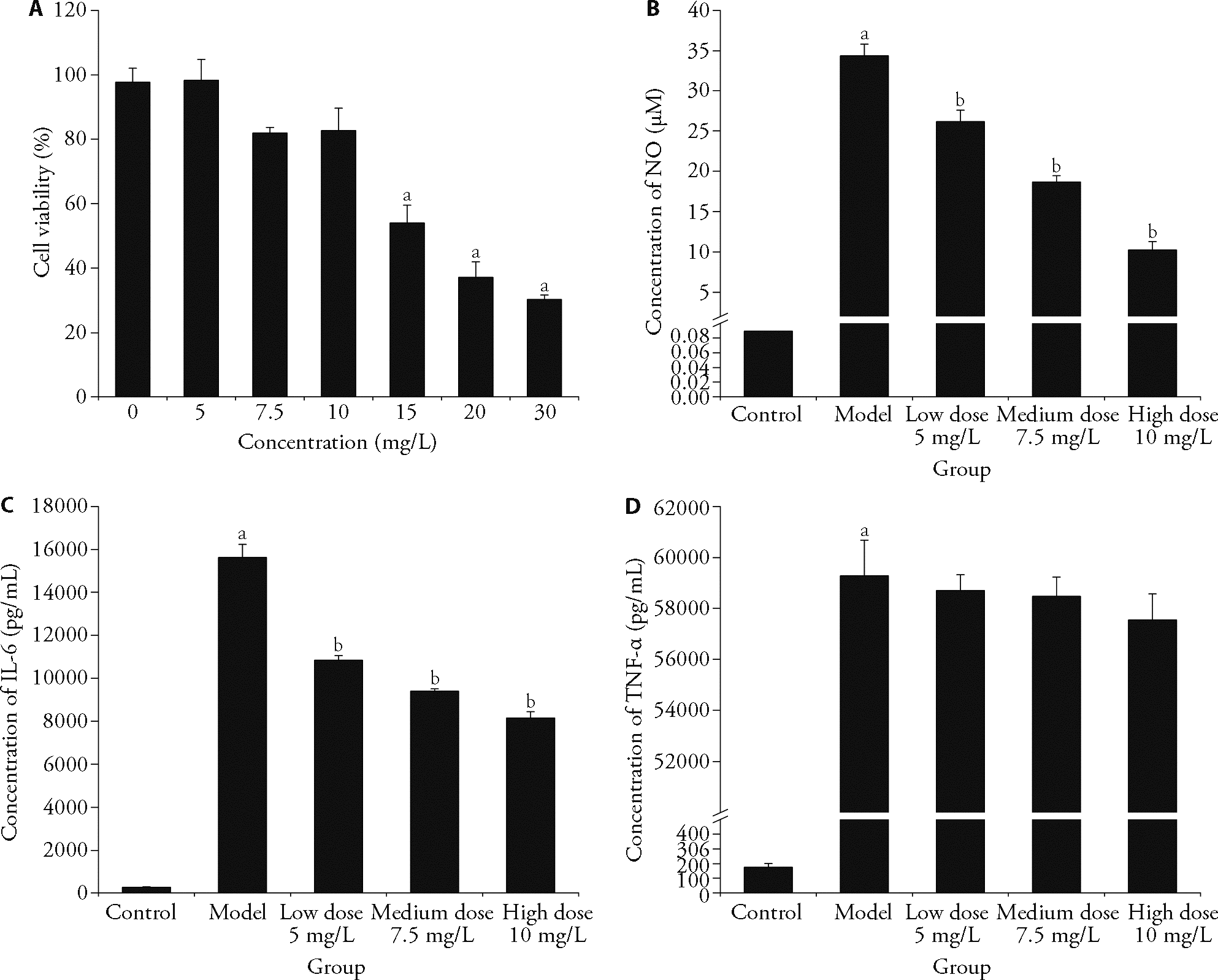
Figure 2 LPS induced cell inflammatory model A: toxicity of the samples to RAW264.7; B: NO content of each sample; C: IL-6 content of each sample; D: TNF-α content of each sample. LPS: lipid polysaccharide. Control group: without treatment; Model group: treated with lipopolysaccharide; Sample group: treated with lipopolysaccharide and non-volatile ingredients of patchouli of 5, 7.5, 10 mg/L. IL-6: interleukin-6; TNF-α: tumor necrosis factor-α. Student’s-test was used for statistical significance (P < 0.05); data are presented as mean ± standard deviation (n = 3). Significant differences compared with control group were designated as aP < 0.001 and with model group as bP < 0.001.
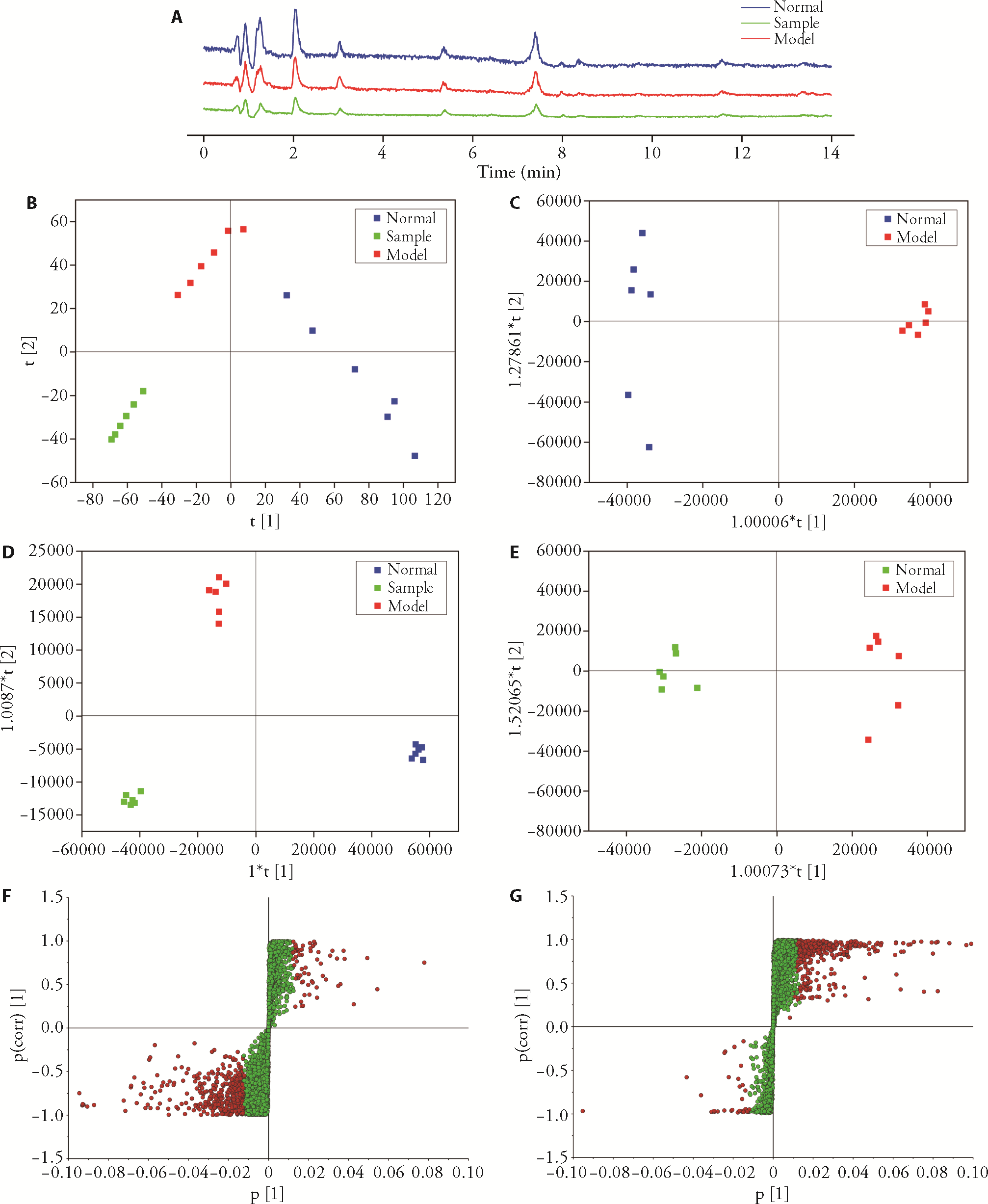
Figure 3 Metabolomic difference analysis A: total ion flow diagram of each group; B: PCA scores; C: OPLS-DA scores of normal group, model group and treated group; D: OPLS-DA scores of normal group and model group; E: OPLS-DA scores of the treated group and model group; F: S-plot of normal group and model group; G: S-plot of the treated group and model group; Control group: without treatment; Model group: treated with lipopolysaccharide; Sample group: treated with lipopolysaccharide and non-volatile ingredients of patchouli of 10 mg/L. PCA: principal component analysis; OPLS-DA: orthogonal partial least squares discriminant analysis. Student’s t-test was used for statistical significance (P < 0.05); Data are presented as mean ± standard deviation (n = 3).

Figure 4 Metabolomics pathway analysis A: clustering heat map of differential metabolites in the normal group and the model group; B: clustering heat map of differential metabolites in the treated group and the model group; C: arginine levels in cell samples; D: sorbitol levels in cell samples; E: KEGG enrichment pathway map of differential metabolic pathway between the normal group and the model group; F: KEGG enrichment pathway map of differential metabolic pathway between the treated group and the model group. Control group: without treatment; Model group: treated with lipopolysaccharide; Sample group: treated with lipopolysaccharide and non-volatile ingredients of patchouli of 10 mg/L. Student’s t-test was used for statistical significance (P < 0.05); Data are presented as mean ± standard deviation (n = 6). Significant differences compared with control group were designated as aP < 0.001 and with model group as bP < 0.001.
| 1. | Chinese Pharmacopoeia Commission. Pharmacopoeia of the People's Republic of China. Beijing: China Medical Science and Technology Press, 2020: 46. |
| 2. |
Yarovaya O, Salakhutdinov N. Mono- and sesquiterpenes as a starting platform for the development of antiviral drugs. Russ Chem Rev 2021; 90: 488-510.
DOI URL |
| 3. |
Da S, Figueried P, Byler G, et al. Essential oils as antiviral agents, potential of essential oils to treat SARS-CoV-2 infection: an in-silico investigation. Int J Mol Sci 2020; 21: 1-35.
DOI URL |
| 4. |
Zrieq R, Ahmad I, Snoussi M, et al. Tomatidine and patchouli alcohol as inhibitors of SARS-CoV-2 enzymes (3CLpro, PLpro and NSP15) by molecular docking and molecular dynamics simulations. Int J Mol Sci 2021; 22: 10693.
DOI URL |
| 5. |
Chen JR, Xie XF, Li MT, et al. Pharmacological activities and mechanisms of action of Pogostemon cablin Benth: a review. Chin Med 2021; 16: 1-20.
DOI |
| 6. |
Swamy MK, Sinniah UR. A Comprehensive review on the phytochemical constituents and pharmacological activities of Pogostemon cablin Benth: an aromatic medicinal plant of industrial importance. Molecules 2015; 20: 8521-47.
DOI PMID |
| 7. |
Verma R, Padalia R, Chauhan A, et al. Chemical composition of leaves, inflorescence, whole aerial-parts and root essential oils of patchouli {Pogostemon cablin (Blanco) Benth.}. J Essent Oil Res 2019; 31: 319-25.
DOI URL |
| 8. | Srivastava S, Lal R, Singh V, et al. Chemical investigation and biological activities of Patchouli [Pogostemon cablin (Blanco) Benth] essential oil. Ind Crop Prod 2022; 26: 328-37. |
| 9. |
Pandey S, Bhandari S, Sarma N, et al. Essential oil compositions, pharmacological importance and agro technological practices of Patchouli (Pogostemon cablin Benth.): a review. J Essent Oil Bear Pl 2021; 24: 1212-26.
DOI URL |
| 10. |
Jain P, Patel S, Desai M. Patchouli oil: an overview on extraction method, composition and biological activities. J Essent Oil Res 2022; 34: 1-11.
DOI URL |
| 11. |
Lee H, Lee J, Smolensky D, et al. Potential benefits of patchouli alcohol in prevention of human diseases: a mechanistic review. Int Immunopharmacol 2020; 89: 107056.
DOI URL |
| 12. |
Xu LQ, Huang QH, Tan XC, et al. Patchouli alcohol ameliorates acute liver injury via inhibiting oxidative stress and gut-origin LPS leakage in rats. Int Immunopharmacol 2021; 98: 107897.
DOI URL |
| 13. |
Ye QY, Ling QH, Shen J, et al. Protective effect of pogostone on murine norovirus infected-RAW264.7 macrophages through inhibition of NF-kappa B/NLRP3-dependent pyroptosis. J Ethnopharmacol 2021; 278: 114250.
DOI URL |
| 14. |
Peng F, Wan F, Xiong L, et al. In vitro and in vivo antibacterial activity of Pogostone. Chin Med J 2014, 127: 4001-5.
DOI URL |
| 15. |
Chen GR, Xie XF, Peng F, et al. Protective effect of the combination of essential oil from patchouli and tangerine peel against gastric ulcer in rats. J Ethnopharmacol 2022; 282: 114645.
DOI URL |
| 16. |
Xie L, Guo YL, Chen Y R, et al. A potential drug combination of omeprazole and patchouli alcohol significantly normalizes oxidative stress and inflammatory responses against gastric ulcer in ethanol-induced rat model. Int Immunopharmacol 2020; 85: 106660.
DOI URL |
| 17. |
Wu ZN, Zeng HR, Zhang LL, et al. Patchouli alcohol: a natural sesquiterpene against both inflammation and intestinal barrier damage of ulcerative colitis. Inflammation 2020; 43: 1423-35.
DOI PMID |
| 18. |
Lian DW, Xu YF, Ren WK, et al. Unraveling the novel protective effect of patchouli alcohol against helicobacter pylori-induced gastritis: insights into the molecular mechanism in vitro and in vivo. Front Pharmacol 2018; 9: 1347.
DOI URL |
| 19. | Galovicova L, Bortova P, Valkova V, et al. Biological activity of pogostemon cablin essential oil and its potential use for food preservation. Agronomy-Basel 2022; 12: 387. |
| 20. |
Zhong YZ, Tang LY, Deng QH, et al. Unraveling the novel effect of patchouli alcohol against the antibiotic resistance of helicobacter pylori. Front Microbiol 2021; 12: 674560.
DOI URL |
| 21. |
Xu YF, Deng QH, Zhong YZ, et al. Clinical strains of helicobacter pylori with strong cell invasiveness and the protective effect of patchouli alcohol by improving miR-30b/C nediated xenophagy. Front Pharmacol 2021; 12: 666903.
DOI URL |
| 22. |
Li D, Xing ZW, Yu TT, et al. Pogostone attenuates adipose tissue inflammation by regulating the adipocyte-macrophage crosstalk via activating SIRT1. Food Funct 2022; 13: 11853-64.
DOI URL |
| 23. |
Zhao YG, Yang YT, Zhang JX, et al. Lactoferrin-mediated macrophage targeting delivery and patchouli alcohol-based therapeutic strategy for inflammatory bowel diseases. Acta Pharmacol Sin B 2020; 10: 1966-76.
DOI URL |
| 24. |
Leong W, Huang GX, Liao WL, et al. Traditional Patchouli essential oil modulates the host's immune responses and gut microbiota and exhibits potent anti-cancer effects in Apc (Min /+) mice. Pharmacol Res 2022; 176: 106082.
DOI URL |
| 25. |
Song YR, Chang L, Wang XY, et al. Regulatory mechanism and experimental verification of patchouli alcohol on gastric cancer cell based on network pharmacology. Front Oncol 2021; 11: 711984.
DOI URL |
| 26. | Santos L, Brandao L, Martins R L, et al. Evaluation of the larvicidal potential of the essential oil pogostemon cablin (Blanco) Benth in the control of aedes aegypti. Pharmaceuticals-Base 2019; 12: 53. |
| 27. |
Xu FF, Cai WN, Ma T, et al. Traditional uses, phytochemistry, pharmacology, quality control, industrial application, pharmacokinetics and network pharmacology of pogostemon cablin: a comprehensive review. Am J Chinese Med 2022; 50: 691-721.
DOI URL |
| 28. |
Kim E, Kim J, Jeong S, et al. Pachypodol, a methoxyflavonoid isolated from pogostemon cablin bentham exerts antioxidant and cytoprotective effects in HepG2 cells: possible role of ERK-dependent Nrf2 activation. Int J Mol Sci 2019; 20: 4082.
DOI URL |
| 29. |
Chen M, Wang PL, Li T, et al. Comprehensive analysis of Huanglian Jiedu decoction: revealing the presence of a self-assembled phytochemical complex in its naturally-occurring precipitate. J Pharmaceut Biomed 2021; 195: 113820.
DOI URL |
| 30. | Hu Q, Chen M, Yan MM, et al. Comprehensive analysis of Sini decoction and investigation of acid-base self-assembled complexes using cold spray ionization mass spectrometry. Microchem J 2022; 173: 117008. |
| 31. |
Yang WH, Liu YH, Liang JL, et al. Beta-patchoulene, isolated from patchouli oil, suppresses inflammatory mediators in LPS-stimulated RAW264.7 macrophages. Eur J Inflamm 2017; 15: 136-41.
DOI URL |
| 32. |
Jeong J, Shin Y, Lee SH. Anti-inflammatory activity of patchouli alcohol in RAW264.7 and HT-29 cells. Food Chem Toxicol 2013; 55: 229-33.
DOI PMID |
| 33. |
Xian YF, Li YC, Ip SP, et al. Anti-inflammatory effect of patchouli alcohol isolated from Pogostemonis herba in LPS-stimulated RAW264.7 macrophages. Exp Ther Med 2011; 2: 545-50.
DOI URL |
| 34. |
Lindez A, Reith W. Arginine-dependent immune responses. Cell Mol Life Sci 2021; 78: 5303-24.
DOI |
| 35. |
Ulrich E, Donkt W. Cameo appearances of aminoacyl-tRNA in natural product biosynthesis. Curr Opin Chem Biol 2016; 35: 29-36.
DOI PMID |
| 36. |
Ramana K, Srivastava S. Aldose reductase: a novel therapeutic target for inflammatory pathologies. Int J Biochem Cell B 2010; 42: 17-20.
DOI URL |
| 37. |
Lanaspa M, Ishimoto T, Cicerchi C, et al. Endogenous fructose production and fructokinase activation mediate renal injury in diabetic nephropathy. J Am Soc Nephrol 2014; 25: 2526-38.
DOI PMID |
| 38. |
Curley S, Gall J, Byrne R, et al. Metabolic inflammation in obesity-at the crossroads between fatty acid and cholesterol metabolism. Mol Nutr Food Res 2021; 65: 1900482.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

