Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (1): 9-16.DOI: 10.19852/j.cnki.jtcm.2022.01.001
• Research Articles • Previous Articles Next Articles
In-vitro and in-vivo pharmacological screening of Iris albicans
Kamal Dawood, Roohullah( ), Rabbi Fazle, Naz Attiqa, Bilal Muhammad
), Rabbi Fazle, Naz Attiqa, Bilal Muhammad
- Department of Pharmacy, Abasyn University Peshawar, Peshawar, 25000, Khyber Pakhtunkhwa, Pakistan
-
Received:2021-03-05Accepted:2021-08-20Online:2022-02-15Published:2022-01-25 -
Contact:Roohullah -
About author:Prof. Dr. Roohullah, Department of Pharmacy, Abasyn University Peshawar, Peshawar, 25000, Khyber Pakhtunkhwa, Pakistan. dr.roohullah2012@gmail.com
Cite this article
Kamal Dawood, Roohullah, Rabbi Fazle, Naz Attiqa, Bilal Muhammad. In-vitro and in-vivo pharmacological screening of Iris albicans[J]. Journal of Traditional Chinese Medicine, 2022, 42(1): 9-16.
share this article
| Sample | Conc. (µgm/ mL) | DPPH scavenging assay | ABTS Scavenging assay | Lipoxygenase | Cyclo-oxygenase-2 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| % inhibition (mean± SEM) | IC50 (µg/mL) | % Inhibition (mean±SEM) | IC50 (µg/ mL) | % Inhibition (mean±SEM) | IC50 (µg/mL) | % Inhibition (mean±SEM) | IC50 (µg/mL) | ||||||||||||||
Meth-anolic | 1000 500 250 125 | 54.43±0.61a 47.03±0.38a 41.33±0.33a 39.07±1.02a | 750 | 55.63±0.48a 51.66±0.88a 46.00±0.57a 41.96±0.12a | 525 | 51.43±0.64 43.46±0.73 41.56±0.64 39.40±1.21 | 830 | 31.60±0.73 26.47±0.59 21.73±1.50 19.43±1.50 | 2292 | ||||||||||||
n-Hexane | 1000 500 250 125 | 43.02±1.11a 36.13±0.88a 31.46±0.54a 26.53±0.61a | 1370 | 48.03±0.38a 44.46±0.54a 38.56±0.40a 32.20±0.91a | 1060 | 21.90±0.78 17.97±1.33 09.60±2.03 | 2100 | 21.90±0.78 17.97±1.33 09.60±2.03 | 2100 | ||||||||||||
Chlor-oform | 1000 500 250 125 | 69.96±0.21b 63.53±1.09a 57.00±0.57a 49.06±1.04a | 139 | 65.26±0.32a 53.33±0.33a 49.43±0.61a 45.07±1.02a | 330 | 49.40±0.65 42.37±0.72 36.87±1.04 31.80±0.55 | 960 | 75.52 ± 3.28 55.59 ± 3.28 50.83 ± 1.21 45.87 ± 0.85 | 240 | ||||||||||||
Ethyl acetate | 1000 500 250 125 | 71.56±1.24b 64.46±0.50a 56.60±0.41a 48.43±0.81a | 150 | 63.13±0.88a 54.00±0.57a 47.02±1.11a 42.66±0.58a | 410 | 41.97±1.25 33.53±0.58 27.37±0.67 21.77±1.54 | 1280 | 42.77±0.82 38.87±0.70 32.73±0.95 28.60±1.19 | 1423 | ||||||||||||
| Aqueous | 1000 500 250 125 | 47.86±0.85a 41.76±0.78a 33.16±0.86a 28.60±0.41a | 1050 | 45.16±0.85a 36.26±0.32a 31.20±0.91a 28.63±0.52a | 1240 | 28.03±2.08 22.63±0.75 13.53±1.87 | 2176 | 34.83±0.33 31.60±0.88 22.67±1.29 13.73±0.84 | 1610 | ||||||||||||
| Positive control | 1000 500 250 125 | 83.33±0.88 74.66±0.49 71.50±0.60 68.00±0.57 | 13 | 87.83±0.29 86.66±0.66 81.00±1.06 79.03±0.87 | < 0.1 | 87.62±1.42 84.79±1.88 80.79±1.08 75.12±0.54 | 02 | 87.49 ± 0.60 76.28 ± 1.94 70.08 ± 1.04 65.37 ± 0.56 | 32 | ||||||||||||
Table 1 Antioxidant and enzyme inhibition assay of various fractions of Iris albicans
| Sample | Conc. (µgm/ mL) | DPPH scavenging assay | ABTS Scavenging assay | Lipoxygenase | Cyclo-oxygenase-2 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| % inhibition (mean± SEM) | IC50 (µg/mL) | % Inhibition (mean±SEM) | IC50 (µg/ mL) | % Inhibition (mean±SEM) | IC50 (µg/mL) | % Inhibition (mean±SEM) | IC50 (µg/mL) | ||||||||||||||
Meth-anolic | 1000 500 250 125 | 54.43±0.61a 47.03±0.38a 41.33±0.33a 39.07±1.02a | 750 | 55.63±0.48a 51.66±0.88a 46.00±0.57a 41.96±0.12a | 525 | 51.43±0.64 43.46±0.73 41.56±0.64 39.40±1.21 | 830 | 31.60±0.73 26.47±0.59 21.73±1.50 19.43±1.50 | 2292 | ||||||||||||
n-Hexane | 1000 500 250 125 | 43.02±1.11a 36.13±0.88a 31.46±0.54a 26.53±0.61a | 1370 | 48.03±0.38a 44.46±0.54a 38.56±0.40a 32.20±0.91a | 1060 | 21.90±0.78 17.97±1.33 09.60±2.03 | 2100 | 21.90±0.78 17.97±1.33 09.60±2.03 | 2100 | ||||||||||||
Chlor-oform | 1000 500 250 125 | 69.96±0.21b 63.53±1.09a 57.00±0.57a 49.06±1.04a | 139 | 65.26±0.32a 53.33±0.33a 49.43±0.61a 45.07±1.02a | 330 | 49.40±0.65 42.37±0.72 36.87±1.04 31.80±0.55 | 960 | 75.52 ± 3.28 55.59 ± 3.28 50.83 ± 1.21 45.87 ± 0.85 | 240 | ||||||||||||
Ethyl acetate | 1000 500 250 125 | 71.56±1.24b 64.46±0.50a 56.60±0.41a 48.43±0.81a | 150 | 63.13±0.88a 54.00±0.57a 47.02±1.11a 42.66±0.58a | 410 | 41.97±1.25 33.53±0.58 27.37±0.67 21.77±1.54 | 1280 | 42.77±0.82 38.87±0.70 32.73±0.95 28.60±1.19 | 1423 | ||||||||||||
| Aqueous | 1000 500 250 125 | 47.86±0.85a 41.76±0.78a 33.16±0.86a 28.60±0.41a | 1050 | 45.16±0.85a 36.26±0.32a 31.20±0.91a 28.63±0.52a | 1240 | 28.03±2.08 22.63±0.75 13.53±1.87 | 2176 | 34.83±0.33 31.60±0.88 22.67±1.29 13.73±0.84 | 1610 | ||||||||||||
| Positive control | 1000 500 250 125 | 83.33±0.88 74.66±0.49 71.50±0.60 68.00±0.57 | 13 | 87.83±0.29 86.66±0.66 81.00±1.06 79.03±0.87 | < 0.1 | 87.62±1.42 84.79±1.88 80.79±1.08 75.12±0.54 | 02 | 87.49 ± 0.60 76.28 ± 1.94 70.08 ± 1.04 65.37 ± 0.56 | 32 | ||||||||||||
| Dosage | Paw edema induction by carrageenan | ||||
|---|---|---|---|---|---|
| 0 h | 1 h | 3 h | 5 h | ||
| Normal Saline | 0.094±0.002 | 0.190±0.006 | 0.194±0.006 | 0.178±0.005 | |
| Aspirin 150 mg/kg | 0.096±0.004 | 0.162±0.007a | 0.140±0.005c | 0.122±0.003c | |
Crude Extract | 100 mg/kg | 0.096±0.002 | 0.194±0.007 | 0.175±0.007 | 0.154±0.008 |
| 200 mg/kg | 0.096±0.004 | 0.168±0.009b | 0.154±0.008c | 0.126±0.006c | |
| 300 mg/kg | 0.094±0.002 | 0.155±0.005c | 0.132±0.002c | 0.110±0.003c | |
Table 2 Effect of crude extracts of Iris albicans administered intra peritoneal at 100, 200 and 300 mg/kg in paw edema induced by Carrageenan
| Dosage | Paw edema induction by carrageenan | ||||
|---|---|---|---|---|---|
| 0 h | 1 h | 3 h | 5 h | ||
| Normal Saline | 0.094±0.002 | 0.190±0.006 | 0.194±0.006 | 0.178±0.005 | |
| Aspirin 150 mg/kg | 0.096±0.004 | 0.162±0.007a | 0.140±0.005c | 0.122±0.003c | |
Crude Extract | 100 mg/kg | 0.096±0.002 | 0.194±0.007 | 0.175±0.007 | 0.154±0.008 |
| 200 mg/kg | 0.096±0.004 | 0.168±0.009b | 0.154±0.008c | 0.126±0.006c | |
| 300 mg/kg | 0.094±0.002 | 0.155±0.005c | 0.132±0.002c | 0.110±0.003c | |
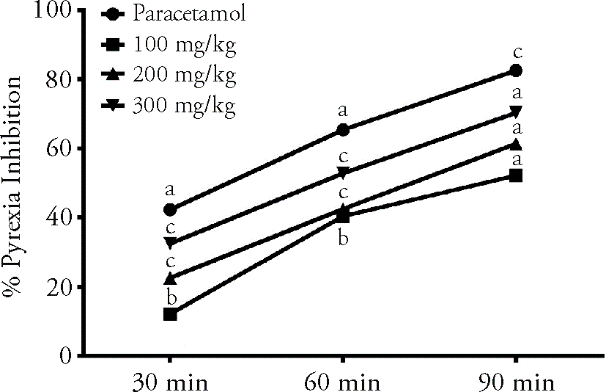
Figure 1 Anti-pyretic activity of Iris albicans crude extract using brewer’s yeast induced pyrexia model Positive control group received paracetamol (150 mg/kg), while sample groups received crude extract (100, 200 and 300 mg/kg) respectively. Each bar shows percent pyrexia inhibition (rectal) compared to vehicle control and aP ˂ 0.0001,bP ˂ 0.05 , cP ˂ 0.01 (one way analysis of variance followed by Dunnett’s post hoc analysis).

Figure 2 Percent inhibition potential of edema induced by Carrageenan Positive control group received aspirin (150 mg/kg, i.p.), while sample groups received crude extract (100, 200 and 300 mg/kg, i.p.) respectively. i.p: intraperitoneal injection. Represents the the mean ± standard error of mean (SEM) of n = 5 and values significant from control at aP ˂ 0.01, bP ˂ 0.05 and cP ˂ 0.0001 using one way analysis of variance followed by Dunnett’s post hoc analysis.
| Group | After 16 h of fasting | After 72 h of STZ | At 4th day | At 5th day | At 6th day | At 7th day | At 8th day | At 9th Day | At 10th Day | |
|---|---|---|---|---|---|---|---|---|---|---|
| Control | 68.5±5.0 | 80.2±5.0 | 74.8 ± 4.7 | 80.7±5.3 | 73.1±4.6 | 76.5 ± 5.8 | 76.3±4.6 | 78.2±4.2 | 71.7±4.8 | |
| STZ | 72.0±2.1 | 341.5±18.3 | 334.5±12.1 | 361.5±22.9 | 365.3±17.4 | 405.0±13.7 | 351.7±17.4 | 386.7±20.1 | 363.7±19.7 | |
| STZ +Metformin (25 mg/kg) | 66.7±2.9 | 330.7±10.6 | 295.5±20.3 | 295.2±8.5a | 272.0±5.7c | 241.3±9.0c | 223.2±13.5c | 184.2±9.7c | 148.2±10.5c | |
| STZ+Extract (100 mg/kg) | 60.0±1.7 | 354.7±25.0 | 333.3±19.3 | 309.8±22.7b | 291.2±22.9c | 285.3±15.1c | 239.5±18.2c | 232.8±16.1c | 211.5±17.2c | |
| STZ+Extract (200 mg/kg) | 63.5±5.0 | 369.8±32.0 | 339.8±11.2 | 260.7±13.9c | 254.8±9.6c | 243.7±5.6c | 205.0±5.2c | 169.3±4.4c | 144.5±7.3c | |
| STZ+Extract (300 mg/kg) | 58.8±3.0 | 385.6±18.1 | 323.2±12.6 | 245.0±13.3c | 243.5±2.3c | 212.0±4.7c | 183.8±12.9c | 154.2±3.4c | 127.3±5.7c | |
Table 3 Potential Effect of Iris albicans100 mg, 200 mg and 300 mg/kg intra peritoneal in streptozocin induced diabetes
| Group | After 16 h of fasting | After 72 h of STZ | At 4th day | At 5th day | At 6th day | At 7th day | At 8th day | At 9th Day | At 10th Day | |
|---|---|---|---|---|---|---|---|---|---|---|
| Control | 68.5±5.0 | 80.2±5.0 | 74.8 ± 4.7 | 80.7±5.3 | 73.1±4.6 | 76.5 ± 5.8 | 76.3±4.6 | 78.2±4.2 | 71.7±4.8 | |
| STZ | 72.0±2.1 | 341.5±18.3 | 334.5±12.1 | 361.5±22.9 | 365.3±17.4 | 405.0±13.7 | 351.7±17.4 | 386.7±20.1 | 363.7±19.7 | |
| STZ +Metformin (25 mg/kg) | 66.7±2.9 | 330.7±10.6 | 295.5±20.3 | 295.2±8.5a | 272.0±5.7c | 241.3±9.0c | 223.2±13.5c | 184.2±9.7c | 148.2±10.5c | |
| STZ+Extract (100 mg/kg) | 60.0±1.7 | 354.7±25.0 | 333.3±19.3 | 309.8±22.7b | 291.2±22.9c | 285.3±15.1c | 239.5±18.2c | 232.8±16.1c | 211.5±17.2c | |
| STZ+Extract (200 mg/kg) | 63.5±5.0 | 369.8±32.0 | 339.8±11.2 | 260.7±13.9c | 254.8±9.6c | 243.7±5.6c | 205.0±5.2c | 169.3±4.4c | 144.5±7.3c | |
| STZ+Extract (300 mg/kg) | 58.8±3.0 | 385.6±18.1 | 323.2±12.6 | 245.0±13.3c | 243.5±2.3c | 212.0±4.7c | 183.8±12.9c | 154.2±3.4c | 127.3±5.7c | |

Figure 3 Effect of crude extract of Iris albicans in streptozocin induced diabetes Positive control group received metformin (25 mg/kg, i.p.), while sample groups received extract (100, 200 and 300 mg/kg, i.p.) respectively. i.p: intraperitoneal injection; STZ: streptozotocin. d: the highest level of glucose in the blood. Values are expressed as mean blood glucose in mg/dL ± standard error of mean. P < 0.001 compared to that of control ( group), aP < 0.05, bP < 0.01, cP < 0.001 in comparison to the streptozotocin (STZ) only administered diabetic control group (two-way analysis of variance, followed by post hoc Bonferroni's multiple comparison test), n = 6 mice per group.
| Biochemical parameter | Control (Saline) | Positive control (Metformin) | Crude Extracts |
|---|---|---|---|
| Glycosylated hemoglobin (%) | 7.80±0.12 | 4.28±0.16 | 5.04±0.32 |
| Total proteins (mg/dL) | 4.46±0.54 | 6.85±0.36 | 4.80±0.48 |
| Total cholesterol (mg/dL) | 184.16±0.96 | 126.24±1.30 | 132.30±0.46 |
| Triglycerides (mg/dL) | 198.38±1.52 | 128.36±0.86 | 138.68±1.26 |
| Serum urea (mg/dL) | 25.65±0.96 | 10.30±0.41 | 12.37±0.42 |
| Creatinine (mg/dL) | 1.40±0.22 | 0.84±0.42 | 0.94±0.35 |
| HDL (mg/dL) | 38.30±0.45 | 48.43±0.96 | 46.23±0.32 |
| LDL (mg/dL) | 98.40±0.78 | 50.21±0.70 | 75.92±0.46 |
Table 4 Effect of treatments on biochemical parameters of diabetes in STZ-induced mice
| Biochemical parameter | Control (Saline) | Positive control (Metformin) | Crude Extracts |
|---|---|---|---|
| Glycosylated hemoglobin (%) | 7.80±0.12 | 4.28±0.16 | 5.04±0.32 |
| Total proteins (mg/dL) | 4.46±0.54 | 6.85±0.36 | 4.80±0.48 |
| Total cholesterol (mg/dL) | 184.16±0.96 | 126.24±1.30 | 132.30±0.46 |
| Triglycerides (mg/dL) | 198.38±1.52 | 128.36±0.86 | 138.68±1.26 |
| Serum urea (mg/dL) | 25.65±0.96 | 10.30±0.41 | 12.37±0.42 |
| Creatinine (mg/dL) | 1.40±0.22 | 0.84±0.42 | 0.94±0.35 |
| HDL (mg/dL) | 38.30±0.45 | 48.43±0.96 | 46.23±0.32 |
| LDL (mg/dL) | 98.40±0.78 | 50.21±0.70 | 75.92±0.46 |
| [1] | Ur Rahman S, Ismail M, Khurram M, Ullah I, Rabbi F, Iriti M. Bioactive steroids and saponins of the genus Trillium. Molecules 2017;22:2156-71. |
| [2] | Sofowora A, Ogunbodede E, Onayade A. The role and place of medicinal plants in the strategies for disease prevention. Afr J Tradit Complement Altern Med 2013;10:210-29. |
| [3] | Rabbi F, Zada A, Adhikari A, Nisar A, Khalil SK. Chemical constituent with cytotoxicity from Sterculia diversifolia. J Asian Nat Prod Res 2020;23:899-905. |
| [4] | Rabbi F, Zada A, Nisar A, Sohail M, Khalil SK, Shah WA. Bioassay-guided isolation, identification of compounds from sterculia diversifolia and investigation of their anti-glycation and antioxidant activities. Pharm Chem J 2020;53:1137-44. |
| [5] | Rabbi F, Zada A, Adhikari A, Jabeen A, Nisar A, Ullah I. Sterculia diversifolia bears anti-cancer and immunomodulatory activities. Bangladesh J Pharmacol 2017;12:51-5. |
| [6] | Basgedik B, Ugur A, Sarac N. Antimicrobial, antioxidant and antimutagenic properties of Iris albicans. Ind Crops Prod 2015;69:480-84. |
| [7] | Mothana RA, Abdo SA, Hasson S, Althawab F, Alaghbari SA, Lindequist U. Antimicrobial, antioxidant and cytotoxic activities and phytochemical screening of some yemeni medicinal plants. Evid Based Complement Atern Med 2010;7:323-30. |
| [8] | Hacıbekiroğlu I, Kolak U. Screening antioxidant and anticholinesterase potential of Iris albicans extracts. Arab J Chem 2015;8:264-8. |
| [9] | Abdel-Mageed WM, Al-Wahaibi LH, Al-Saleem MS, Gouda YG, Abdel-Kader MS, Ibraheim ZZ. Phytochemical and chemotaxonomic study on Iris albicans Lange leaves. Biochem Syst Ecol 2018;76:32-4. |
| [10] | Rabbi F, Zada A, Nisar A. Larvicidal, leishmanicidal, insecticidal and anthelmintic effects of Sterculia diversifolia stem bark and leaf. Bangladesh J Pharmacol 2020;15:32-8. |
| [11] | Rabbi F, Zada A, Adhikari A, et al. GC-MS analysis, metal analysis and antimicrobial investigation of sterculia diversifolia. Pharm Chem J 2020;54:943-53. |
| [12] | Rabbi F, Zada A, Nisar A, Sohail M, Khalil SK, Ahmad AA. In vivo laxative, anti-diarrheal, hepatoprotective and diuretic inves-tigations of Sterculia diversifolia and its isolated compounds. J Tradit Chin Med 2021;41:717-24. |
| [13] | Ahmad S, Ullah F, Sadiq A, et al. Chemical composition, antioxidant and anticholinesterase potentials of essential oil of Rumex hastatus D. Don collected from the North West of Pakistan. BMC Complement Altern Med 2016;16:29. |
| [14] | Ulusu NN, Ercil D, Sakar MK, Tezcan EF. Abietic acid inhibits lipoxygenase activity. Phytother Res 2002;16:88-90. |
| [15] | Nisar M, Kaleem WA, Khan I, et al. Molecular simulations probing Kushecarpin A as a new lipoxygenase inhibitor. Fitoterapia 2011;82:1008-11. |
| [16] | Ahmad S, Ullah F, Ayaz M, Sadiq A, Imran M. Antioxidant and anticholinesterase investigations of Rumex hastatus D. Don: potential effectiveness in oxidative stress and neurological disorders. Biol Res 2015;48:20. |
| [17] | Rabbi F, Zada A, Nisar A, et al. Evaluation of in-vivo biological activities of Sterculia diversifolia (G.Don) in relevance to the isolated secondary metabolites. J Anim Plant Sci 2021;31:296-306. |
| [18] | Khan I, Nisar M, Zarrelli A, et al. Molecular insights to explore abietane diterpenes as new LOX inhibitors. Med Chem Res 2013;22:5809-13. |
| [19] | Ghoul JE, Boughattas NA, Ben-Attia M. Antihyperglycemic and antihyperlipidemic activities of ethanolic extract of Zygophyllum album in streptozotocin-induced diabetic mice. Toxicol Indus Health 2013;29:43-51. |
| [20] | Alam F, us Saqib QN, Ashraf M. Zanthoxylum armatum DC extracts from fruit, bark and leaf induce hypolipidemic and hypoglycemic effects in mice-in vivo and in vitro study. BMC Complement Altern Med 2018;18:1-9. |
| [21] | Becerra-Verdín EM, Morales Ávila ÚM, García-Galindo HS, Montalvo-González R, Castañeda-Martínez A, Montalvo-González E. Evaluation of biochemical markers in diabetic rats fed diets supplemented with fruit purees. CyTA J Food 2019;17:307-15. |
| [22] | Jayaprakasha G, Girennavar B, Patil BS. Radical scavenging activities of Rio Red grapefruits and Sour orange fruit extracts in different in vitro model systems. Bioresour Technol 2008;99:4484-94. |
| [23] | Guo S, Song Z, Ma R, Yang Y, Yu M. Genome-wide identification and expression analysis of the lipoxygenase gene family during peach fruit ripening under different postharvest treatments. Acta Physiol Plant 2017;39:111. |
| [24] | Hadi F, Rahim A, Rabbi F. In vivo investigations of analgesic, antipyretic and anthelmintic activities of various extracts of fernandoa adenophylla. Pharm Chem J 2021;51:929-33. |
| [25] | Intahphuak S, Khonsung P, Panthong A. Anti-inflammatory, analgesic, and antipyretic activities of virgin coconut oil. Pharm Biol 2010;48:151-57. |
| [26] | PA VD. Antipyretic activity of ethanol and aqueous extract of root of Asparagus racemosus in yeast induced pyrexia. Asian J Pharm Clin Res 2013;6:190-93. |
| [27] | Nasir M, Tafess K, Abate D. Antimicrobial potential of the Ethiopian Thymus schimperi essential oil in comparison with others against certain fungal and bacterial species. BMC Complement Altern Med 2015;15:260. |
| [28] | Hussain Z, Khan JA, Arshad A, Asif P, Rashid H, Arshad MI. Protective effects of Cinnamomum zeylanicum L. (Darchini) in acetaminophen-induced oxidative stress, hepatotoxicity and nephrotoxicity in mouse model. Biomed Pharmacother 2019;109:2285-92. |
| [29] | Jarald E, Joshi S, Jain D, Edwin S. Biochemical evaluation of the hypoglycemic effects of extract and fraction of Cassia fistula Linn. in alloxan-induced diabetic rats. Ind J Pharm Sci 2013;75:427. |
| [30] | Ramadan KS, Alshamrani SA. Effects of Salvadora persica extract on the hematological and biochemical alterations against immobilization-induced rats. Scientifica 2015;2015:253195. |
| [1] | CHENG Huanbo, HU Hui, SUN Daihua, WANG Guangzhong. Anti-inflammatory, anti-tussive effects and toxicity evaluation of Qingfei Dayuan granules (清肺达原颗粒) [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1110-1117. |
| [2] | LIU Yue, ZHANG Fan, HAN Xiaomeng, XU Ningyang, ZHAO Yu, WANG Qige, WANG Jianan, LU Bingjiu, Zhang Yan. Jianpi Qutan Fang (健脾祛痰方) induces anti-atherosclerosis and ameliorates endothelial cell injury in high-fat diet rats via an anti-inflammatory and inhibiting Janus kinase/signal transducer and activator of transcription signaling pathway [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1168-1175. |
| [3] | LI Zhihao, HAN Wenjun, SONG Xiuling, LI Yan, CHEN Yuelai. Electroacupuncture stimulating Zhongji (CV3), Guanyuan (CV4), and bilateral Dahe (KI12) attenuates inflammation in rats with chronic nonbacterial prostatitis induced by estradiol through inhibiting toll-like receptor 4 pathway [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 963-972. |
| [4] | XU Guihua, CHEN Feifei, ZHANG Wei, WU Yingen, CHEN Xiaorong, SHI Kehua, WANG Zhenwei, SHI Miaoyan, ZHANG Xing, LU Yunfei, YUAN Weian, LYU Hua, CHEN Xuan. Effectiveness of Traditional Chinese Medicine on coronavirus disease 2019 in 92 patients: a retrospective study [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 582-587. |
| [5] | Esma Anissa Trad Khodja, Abd El Hamid Khabtane, Rabah Arhab, Djamila Benouchenne, Mohamed Sabri Bensaad, Chawki Bensouici, Ramazan Erenler. In vitro assessment of antioxidant, neuroprotective, anti-urease and anti-tyrosinase capacities of Tamarix africana leaves extracts [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 252-264. |
| [6] | Naser Mirazi, Sheida Hesami, Alireza Nourian, Abdolkarim Hosseini. Protective efficacy of dark chocolate in letrozole-induced ovary toxicity model rats: hormonal, biochemical, and histopathological investigation [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 741-748. |
| [7] | FENG Junfang, CHEN Ou, WANG Yibiao. Anti-inflammatory mechanism of rhein in treating asthma based on network pharmacology [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 296-303. |
| [8] | Rabbi Fazle, Zada Amir, Nisar Amna, Adhikari Achyut, Ullah Irfan, Ur Rahman Shafiq. Detailed approach toward the anti-hyperglycemic potential of Sterculia diversifolia G. Don against alloxan-induced in vivo hyperglycemia model [J]. Journal of Traditional Chinese Medicine, 2022, 42(1): 17-22. |
| [9] | Elham A.Abd-Allah, Nouf S.Al-Abbas, Mona M.Atia, Fawzia Alzahrani, El-Mokhtar M.Ahmed, Soad S.Ali, Soad K.Al Jaouni. Can Fig and Olive Ameliorate the toxicity Induced by 2-nitropropane in some organs of mice? role of inflammatory versus anti-inflammatory genes [J]. Journal of Traditional Chinese Medicine, 2021, 41(6): 891-899. |
| [10] | XIA Xichao, LI Bin, QIU Ju, TIAN Gang, CHEN Changdong, LA Ming, ZHANG Ke, QI Jinxu, LI Yanyan, GAO Huashan, SHAO Xiangyang, SU Congying, WANG Mengqi, OUYANG Jingfeng. Antioxidative and immunological effects of Cyclocarya paliurus polysaccharides on the spleen injury of diabetic rats [J]. Journal of Traditional Chinese Medicine, 2021, 41(5): 739-746. |
| [11] | TANG Chengfang, GAO Yang, Gulibairemu Yusuyin, MAO Yan, LI Yujun, WANG Yandong, GU Zhengyi. Anti-cataract effects of Dajizhi(Euphorbium) eye drops on selenite-induced cataracts in rats [J]. Journal of Traditional Chinese Medicine, 2021, 41(5): 747-752. |
| [12] | ZHANG Qiongzhi, FU Tingting, DAI Jianing, ZHOU Zhinan, SHEN Cuizhen. Sodium Danshensu promotes the healing of stage 2 pressure injury wounds in ischemia/reperfusion injury rat models: possible regulation of apoptosis and inflammatory response [J]. Journal of Traditional Chinese Medicine, 2021, 41(4): 571-580. |
| [13] | Mohsen Akbaribazm, Fatemeh Khazaei, Leila Naseri, Mona Pazhouhi, Mohammad Zamanian, Mozafar Khazaei. Pharmacological and therapeutic properties of the Red Clover(Trifolium pratense L.): an overview of the new findings [J]. Journal of Traditional Chinese Medicine, 2021, 41(4): 642-649. |
| [14] | Chien Yi Koay;Anna Pick Kiong Ling;Ying Pei Wong;Rhun Yian Koh;Sobri Hussein;. Anti-neuroinflammatory responses in lipopolysaccharide-stimulated BV2 microglial cells upon treatment with methanol extract of Panax ginseng root [J]. Journal of Traditional Chinese Medicine, 2021, 41(2): 185-193. |
| [15] | ?nder Aybast?er;. Efficacy of methanol-water extract of Inula helenium root against oxidative DNA damage [J]. Journal of Traditional Chinese Medicine, 2021, 41(2): 293-300. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

