Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (1): 39-48.DOI: 10.19852/j.cnki.jtcm.2022.01.004
• Research Articles • Previous Articles Next Articles
Shenweifang-containing serum inhibits transforming growth factor-β1-induced myofibroblast differentiation in normal rat kidney interstitial fibroblast cells
Jiaru LIN1,2,3, Li WANG4, Bo CHEN4, Santao OU1, Jianhua QIN1, Junming FAN1,3,4,5,6( )
)
- 1 Department of Nephrology, the Affiliated Hospital of Southwest Medical University, Luzhou 646000, China
2 Nephropathy Clinical Medical Research Center of Sichuan Province, Luzhou 646000, China
3 Chengdu University of Traditional Chinese Medicine, Chengdu 610075, China
4 Central Laboratory, Affiliated Chinese Medicine Hospital of Southwest Medical University, Luzhou, Sichuan 646000, China
5 Southwest Medical University, Luzhou 646000, China
6 Chengdu Medical College, Chengdu, Sichuan 610500, China
-
Received:2020-09-21Accepted:2021-02-16Online:2022-02-15Published:2022-01-25 -
Contact:Junming FAN -
About author:FAN Junming, Southwest Medical University, Luzhou 646000, China; Department of Nephrology, The Affiliated Hospital of Southwest Medical University, Luzhou 646000, China; Chengdu University of Traditional Chinese Medicine, Chengdu 610075, China; Central Laboratory, Affiliated Chinese Medicine Hospital of Southwest Medical University, Luzhou, Sichuan 646000, China; Chengdu Medical College, Chengdu, Sichuan 610500, China. junmingfan@163.com; mumuxiaoru@126.com
-
Supported by:Department of Science and Technology of Southwest Medical University(2017-ZRQN-072);Department of Science and Technology of Affiliated Hospital of Southwest Medical University(16231)
Cite this article
Jiaru LIN, Li WANG, Bo CHEN, Santao OU, Jianhua QIN, Junming FAN. Shenweifang-containing serum inhibits transforming growth factor-β1-induced myofibroblast differentiation in normal rat kidney interstitial fibroblast cells[J]. Journal of Traditional Chinese Medicine, 2022, 42(1): 39-48.
share this article
| Gene | Forward primer/reverse primer |
|---|---|
| α-SMA | F: 5'-CAGGGAGTGATGGTTGGAAT-3' |
| R: 5'-GGTGATGATGCCGTGTTCTA-3' | |
| Collagen Ⅰ | F: 5'TCCGACCTCTCTCCTCTGAA3' |
| R: 5'TGCTTTGTGCTTTGGGAAGT3' | |
| Fibronectin | F: 5'AGGAGAACCAGGAGAGCACA3' |
| R: 5'TCGGTCACTTCCACAAACTG3' | |
| PCNA | F: 5'ATCCTGAAGAAGGTGCTGGA3' |
| R: 5'GCTGCACTAAGGAGACGTGA3' | |
| Samd3 | F: 5' CTATGATGTCAAGTGCGTGCTG3' |
| R: 5'CGTTGTACTGCTGGGTGGTG3' | |
| MAPK10 | F: 5'TGCCAAGAGGGCTTACCG3' |
| R: 5'GCGTCCATCAGTTCCATCAC3' | |
| GAPDH | F: 5'ATCCCATCACCATCTTCCAG3' |
| R: 5'CCATCACGCCACAGTTTCC3' |
Table 1 Primers used in real-time polymerase chain reaction
| Gene | Forward primer/reverse primer |
|---|---|
| α-SMA | F: 5'-CAGGGAGTGATGGTTGGAAT-3' |
| R: 5'-GGTGATGATGCCGTGTTCTA-3' | |
| Collagen Ⅰ | F: 5'TCCGACCTCTCTCCTCTGAA3' |
| R: 5'TGCTTTGTGCTTTGGGAAGT3' | |
| Fibronectin | F: 5'AGGAGAACCAGGAGAGCACA3' |
| R: 5'TCGGTCACTTCCACAAACTG3' | |
| PCNA | F: 5'ATCCTGAAGAAGGTGCTGGA3' |
| R: 5'GCTGCACTAAGGAGACGTGA3' | |
| Samd3 | F: 5' CTATGATGTCAAGTGCGTGCTG3' |
| R: 5'CGTTGTACTGCTGGGTGGTG3' | |
| MAPK10 | F: 5'TGCCAAGAGGGCTTACCG3' |
| R: 5'GCGTCCATCAGTTCCATCAC3' | |
| GAPDH | F: 5'ATCCCATCACCATCTTCCAG3' |
| R: 5'CCATCACGCCACAGTTTCC3' |
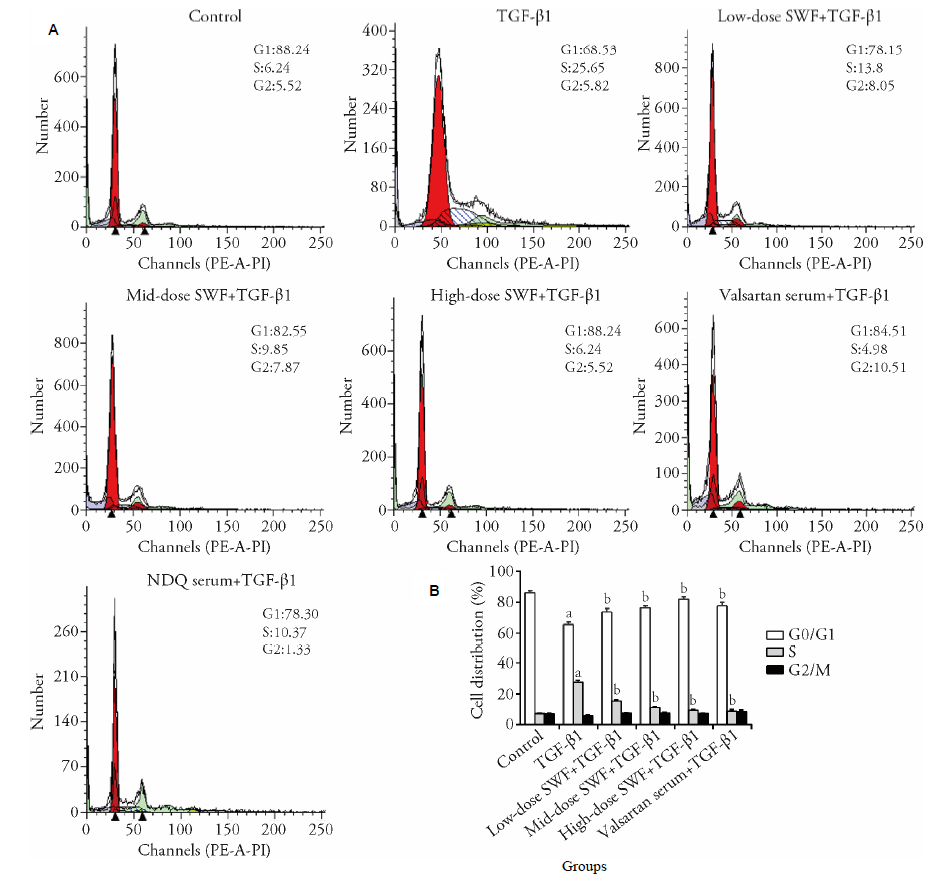
Figure 1 Effect of Shenweifang (SWF)-containing serum on the cell cycle distribution of transforming growth factor (TGF)-β1-treated normal rat kidney interstitial fibroblast cells (NRK-49F) As A and B shows control group, untreated cells (cultured in 10% fetal bovine serum). TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% normal rat serum (rats were gavaged saline). Low-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% low-dose SWF rat serum (rats were gavaged low-dose SWF). Medium-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% medium-dose SWF rat serum (rats were gavaged medium-dose SWF). High-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% high-dose SWF rat serum (rats were gavaged high-dose SWF). Valsartan serum+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% valsartan-containing rat serum (rats were gavaged valsartan). n = 3. aP ˂ 0.01 compared with control cells; bP < 0.01 compared with TGF-β1-treated cells.

Figure 2 Effect of Shenweifang (SWF)-containing serum on the protein expression of α-smooth muscle actin (α-SMA), fibronectin, and proliferating cell nuclear antigen (PCNA) in transforming growth factor (TGF)-β1-treated normal rat kidney interstitial fibroblast cells (NRK-49F) Control group, untreated cells (cultured in 10% fetal bovine serum). TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% normal rat serum (rats were gavaged saline). Low-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% low-dose-SWF rat serum (rats were gavaged low-dose SWF). Medium-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% medium-dose-SWF rat serum (rats were gavaged medium-dose SWF). High-dose SWF + TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% high-dose-SWF rat serum (rats were gavaged high-dose SWF). Valsartan serum + TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% valsartan-containing rat serum (rats were gavaged valsartan).
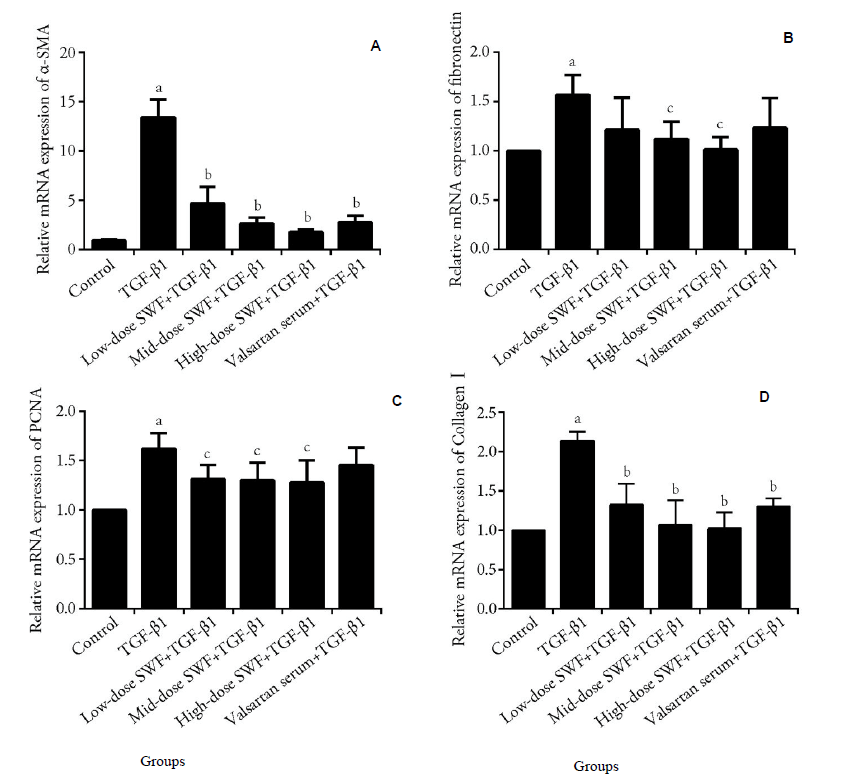
Figure 3 Effect of Shenweifang (SWF)-containing serum on the mRNA expression of α-smooth muscle actin (α-SMA), fibronectin, proliferating cell nuclear antigen (PCNA), and collagen Ⅰ in transforming growth factor (TGF)-β1-treated NRK-49F cells A: α-SMA; B: fibronectin; C: PCNA; D: collagen Ⅰ. Control group, untreated cells (cultured in 10% fetal bovine serum). TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% normal rat serum (rats were gavaged saline). Low-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% low-dose SWF rat serum (rats were gavaged low-dose SWF). Medium-dose SWF + TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% medium-dose SWF rat serum (rats were gavaged medium-dose SWF). High-dose SWF + TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% high-dose SWF rat serum (rats were gavaged high-dose SWF). Valsartan serum + TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% valsartan-containing rat serum (rats were gavaged valsartan). n = 3. aP ˂ 0.01, compared with control cells; bP < 0.01, compared with TGF-β1-treated cells. SWF: Shenweifang; α-SMA: α-smooth muscle actin; PCNA: proliferating cell nuclear antigen; TGF-β1: transforming growth factor-β1.
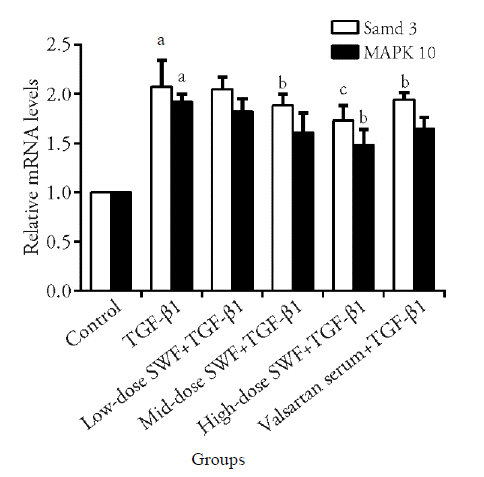
Figure 4 Effect of Shenweifang (SWF)-containing serum on the mRNA expression of Smad3 and MAPK-10 in transforming growth factor (TGF)-β1-treated NRK-49F cells Control group, untreated cells (cultured in 10% fetal bovine serum). TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% normal rat serum (rats were gavaged saline). Low-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% low-dose SWF rat serum (rats were gavaged low-dose SWF). Medium-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% medium-dose SWF rat serum (rats were gavaged medium-dose SWF). High-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% high-dose-SWF rat serum (rats were gavaged high-dose SWF). Valsartan serum+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% valsartan-containing rat serum (rats were gavaged valsartan). n = 3. aP ˂ 0.01, compared with control cells; bP < 0.01, cP < 0.05, compared with TGF-β1-treated cells.
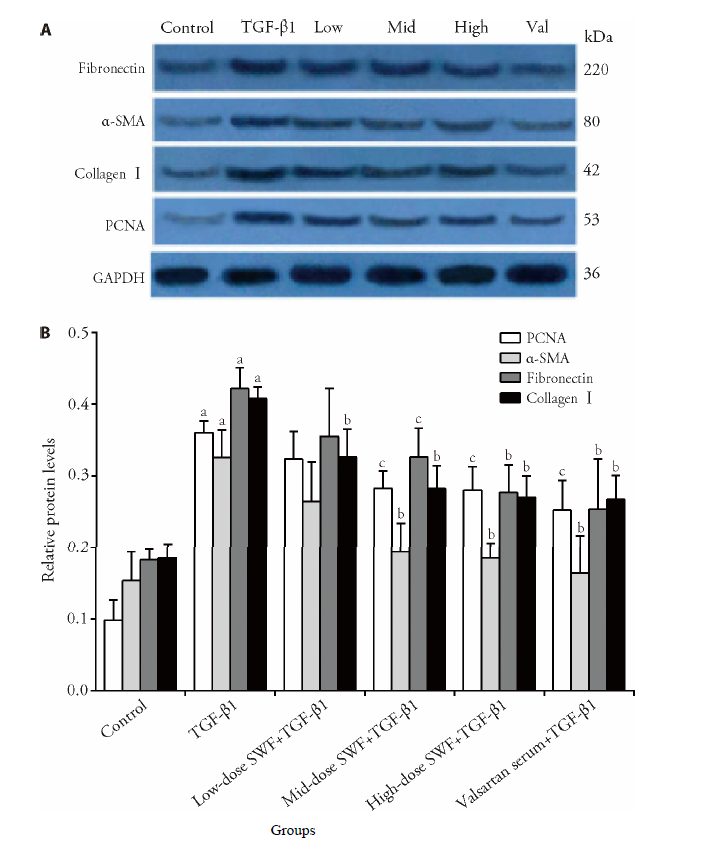
Figure 5 Effect of Shenweifang (SWF)-containing serum on the protein expression of α-smooth muscle actin (α-SMA), fibronectin, proliferating cell nuclear antigen (PCNA), and collagen Ⅰ in transforming growth factor (TGF)-β1-treated NRK-49F cells Control group, untreated cells (cultured in 10% fetal bovine serum). TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% normal rat serum (rats were gavaged saline). Low-dose SWF+TGF-β1 group (low-dose group), cells treated with TGF-β1 (10 ng/mL) and 10% low-dose SWF rat serum (rats were gavaged low-dose SWF). Medium-dose SWF+TGF-β1 group (medium-dose group), cells treated with TGF-β1 (10 ng/mL) and 10% medium-dose SWF rat serum (rats were gavaged medium-dose SWF). High-dose SWF+TGF-β1 group (high-dose group), cells treated with TGF-β1 (10 ng/mL) and 10% high-dose SWF rat serum (rats were gavaged high-dose SWF). Valsartan serum+TGF-β1 group (Val group), cells treated with TGF-β1 (10 ng/mL) and 10% valsartan-containing rat serum (rats were gavaged valsartan). n = 3. aP ˂ 0.01, compared with control cells; bP < 0.01, cP < 0.05, compared with TGF-β1-treated cells.
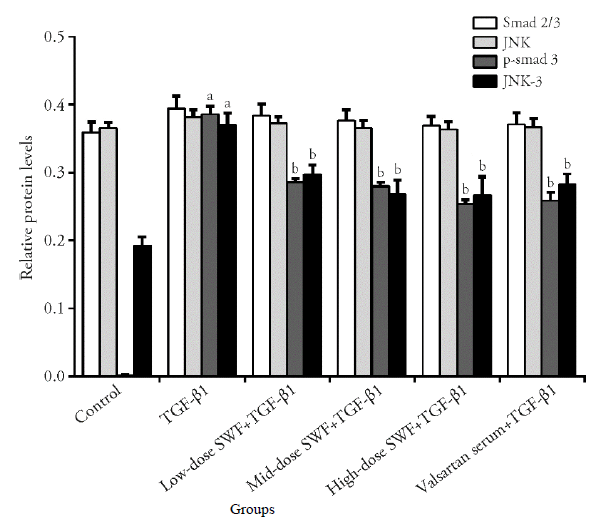
Figure 6 Effect of Shenweifang (SWF)-containing serum on the protein expression of Smad2/3, phosphorylated Smad3 (p-Smad3), c-Jun N-terminal kinase (JNK), and JNK-3 in transforming growth factor (TGF)-β1-treated NRK-49F cells Control group, untreated cells (cultured in 10% fetal bovine serum). TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% normal rat serum (rats were gavaged saline). Low-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% low-dose SWF rat serum (rats were gavaged low-dose SWF). Medium-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% medium-dose SWF rat serum (rats were gavaged medium-dose SWF). High-dose SWF+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% high-dose SWF rat serum (rats were gavaged high-dose SWF). Valsartan serum+TGF-β1 group, cells treated with TGF-β1 (10 ng/mL) and 10% valsartan-containing rat serum (rats were gavaged valsartan). n = 3. aP ˂ 0.01, compared with control cells; bP < 0.05, cP < 0.01, compared with TGF-β1-treated cells.
| [1] | Webster AC, Nagler EV, Morton RL, Masson P. Chronic kidney disease. Lancet 2017;389:1238-52. |
| [2] | Lane BR, Demirjian S, Derweesh IH, Riedinger CB, Fergany AF, Campbell SC. Is all chronic kidney disease created equal? Curr Opin Urol 2014;24:127-34. |
| [3] | Abubakar I, Tillmann T, Banerjee A. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015;385:117-71. |
| [4] | Levey A, Atkins R, Coresh J, et al. Chronic kidney disease as a global public health problem: approaches and initiatives-a position statement from Kidney Disease Improving Global Outcomes. Kidney Int 2007;72:247-59. |
| [5] | Foundation NK. KDOQI clinical practice guideline for diabetes and CKD: 2012 update. Am J Kidney Dis 2012;60:850-86. |
| [6] | Campanholle G, Ligresti G, Gharib SA, Duffield JS. Cellular mechanisms of tissue fibrosis. 3. Novel mechanisms of kidney fibrosis. Am J Physiol Cell Physiol 2013;304:C591-C603. |
| [7] | Fan J-M, Ng Y-Y, Hill PA, Nikolic-Paterson DJ, Mu W, Atkins RC, et al. Transforming growth factor-β regulates tubular epithelial-myofibroblast transdifferentiation in vitro. Kidney Int 1999;56:1455-67. |
| [8] | Lan HY. Diverse roles of TGF-β/Smads in renal fibrosis and inflammation. Int J Biol Sci 2011;7:1056. |
| [9] | Qin W, Chung AC, Huang XR, et al. TGF-β/Smad3 signaling promotes renal fibrosis by inhibiting miR-29. J Am Soc Nephrol 2011;22:1462-74. |
| [10] | Meng X-M, Chung AC, Lan HY. Role of the TGF-β/BMP-7/Smad pathways in renal diseases. Clin Sci 2013;124:243-54. |
| [11] | Meng XM, Huang XR, Chung AC, et al. Smad2 protects against TGF-β/Smad3-mediated renal fibrosis. J Am Soc Nephrol 2010;21:1477-87. |
| [12] | Ihn H. Pathogenesis of fibrosis: role of TGF-β and CTGF. Curr Opin Rheumatol 2002;14:681-5. |
| [13] | Leask A, Abraham DJ. TGF-β signaling and the fibrotic response. FASEB J 2004;18:816-27. |
| [14] | Biernacka A, Dobaczewski M, Frangogiannis NG. TGF-β signaling in fibrosis. Growth Factors 2011;29:196-202. |
| [15] | Nagai A, Shibamoto Y, Ogawa K. Therapeutic effects of saireito (Chai-Ling-Tang), a traditional Japanese herbal medicine, on lymphedema caused by radiotherapy: a case series study. Evid Based Complement Alternat Med 2013; 2013: 241629:1-6. |
| [16] | Chen Y, Deng Y, Ni Z, et al. Efficacy and safety of traditional chinese medicine (Shenqi particle) for patients with idiopathic membranous nephropathy: a multicenter randomized controlled clinical trial. Am J Kidney Dis 2013;62:1068-76. |
| [17] | Zhang J, Xie X, Li C, Fu P. Systematic review of the renal protective effect of Astragalus membranaceus (root) on diabetic nephropathy in animal models. J Ethnopharmacol 2009;126:189-96. |
| [18] | Wei Y, Fan J, Pan L. Effect of Panax notoginseng saponins on human kidney fibroblast. Zhong Guo Zhong Xi Yi Jie He Za Zhi 2002;22:47-9. |
| [19] | Xie X, Ping F, Liu X, Fan J, He C. Chinese medicine preventes the progression of chronic renal failure: clinical and molecular biology characteristics. Zhong Guo Lin Chuang Kang Fu Zazhi 2006;10:136-8. |
| [20] | Wang JL, Chen CW, Tsai MR, et al. Antifibrotic role of PGC-1α-siRNA against TGF-β1-induced renal interstitial fibrosis. Exp Cell Res 2018;370:160-7. |
| [21] | Suzuki H, Kanno Y, Sugahara S, Okada H, Nakamoto H. Effects of an angiotensin Ⅱ receptor blocker, valsartan, on residual renal function in patients on CAPD. Am J Kidney Dis 2004;43:1056-64. |
| [22] | Wu WP, Chang CH, Chiu YT, et al. A reduction of unilateral ureteral obstruction-induced renal fibrosis by a therapy combining valsartan with aliskiren. Am J Physiol-Renal 2010;299:F929-41. |
| [23] | Tang L, Yi R, Yang B, Li H, Chen H, Liu Z. Valsartan inhibited HIF-1α pathway and attenuated renal interstitial fibrosis in streptozotocin-diabetic rats. Diabetes Res Clin Pract 2012;97:125-31. |
| [24] | Lü LL, Liu BC. Role of non-classical renin-angiotensin system axis in renal fibrosis. Front Physiol 2015;6. |
| [25] | Liu X, Wang J, Lu Q. Inhibitory mechanism of Ginkgo giloba extract, captopril, and valsartan on kidney fibrosis of diabetic nephropathy rats and their comparison. Zhong Cao Yao 2008;39:560. |
| [26] | Liu Y. Cellular and molecular mechanisms of renal fibrosis. Nat Rev Nephrol 2011;7:684-96. |
| [27] | Verrecchia F, Chu M-L, Mauviel A. Identification of novel TGF-β/Smad gene targets in dermal fibroblasts using a combined cDNA microarray/promoter transactivation approach. J Biol Chem 2001;276:17058-62. |
| [28] | Sato M, Muragaki Y, Saika S, Roberts AB, Ooshima A. Targeted disruption of TGF-β1/Smad3 signaling protects against renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction. J Clin Invest 2003;112:1486. |
| [29] | Flanders KC, Sullivan CD, Fujii M, et al. Mice lacking Smad3 are protected against cutaneous injury induced by ionizing radiation. Am J Pathol 2002;160:1057-68. |
| [30] | Zhou L, Fu P, Huang XR, Liu F, et al. Mechanism of chronic aristolochic acid nephropathy: role of Smad3. Am J Physiol-Renal 2010;298:F1006-17. |
| [31] | Bonniaud P, Kolb M, Galt T, et al. Smad3 null mice develop airspace enlargement and are resistant to TGF-β-mediated pulmonary fibrosis. J Immunol 2004;173:2099-108. |
| [32] | Fujimoto M, Maezawa Y, Yokote K, et al. Mice lacking Smad3 are protected against streptozotocin-induced diabetic glome-rulopathy. Biochem Biophys Res Commun 2003;305:1002-7. |
| [33] | Isono M, Chen S, Hong SW, Iglesias-de la Cruz MC, Ziyadeh FN. Smad pathway is activated in the diabetic mouse kidney and Smad3 mediates TGF-β-induced fibronectin in mesangial cells. Biochem Biophys Res Commun 2002;296:1356-65. |
| [34] | Liu Z, Huang XR, Lan HY. Smad3 mediates ANG Ⅱ-induced hypertensive kidney disease in mice. Am J Physiol-Renal 2012;302:F986-97. |
| [35] | Yang F, Chung AC, Huang XR, Lan HY. Angiotensin Ⅱ induces connective tissue growth factor and collagen Ⅰ expression via transforming growth factor-β-dependent and-independent smad pathways. Hypertension 2009;54:877-84. |
| [36] | Gao P, Wei Y, Zhang Z, et al. Synergistic effects of c-Jun and SP1 in the promotion of TGFβ1-mediated diabetic nephropathy progression. Exp Mol Pathol 2016;100:441-50. |
| [37] | Macia MS, Halbritter J, Delous M, et al. Mutations in MAPKBP1 cause juvenile or late-onset cilia-independent nephronophthisis. Am J Hum Genet 2017;100:323-33. |
| [38] | Hung TW, Tsai JP, Lin SH, Lee CH, Hsieh YH, Chang HR. Pentraxin 3 activates JNK signaling and regulates the Epithelial-To-Mesenchymal transition in renal fibrosis. Cell Physiol Biochem 2016;40:1029-38. |
| [39] | Ren M, Zhang J, Wang B, et al. Qindan-capsule inhibits proliferation of adventitial fibroblasts and collagen synthesis. J Ethnopharmacol 2010;129:53-8. |
| [40] | Li JX, Wang ZB, Zhu LQ, Niu FL, Cui W. Effects of Radix notoginseng extracts drug-containing serum on expressions of Bcl-2, Bax and p21WAF1 proteins in MNNG transformed GES-1 cells. Zhong Xi Yi Jie He Xue Bao 2008;6:817-20. |
| [41] | Wang QL, Yuan JL, Tao YY, Zhang Y, Liu P, Liu CH. Fuzheng Huayu recipe and vitamin E reverse renal interstitial fibrosis through counteracting TGF-β1-induced epithelial-to-mesen-chymal transition. J Ethnopharmacol 2010;127:631-40. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

