Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (2): 234-241.DOI: 10.19852/j.cnki.jtcm.20210220.001
• Research Articles • Previous Articles Next Articles
Randomized controlled study of a diagnosis and treatment plan for moderate coronavirus disease 2019 that integrates Traditional Chinese and Western Medicine
XIA Wenguang1, ZHENG Chanjuan1, ZHANG Jixian2, HUANG Min1,3, LI Qinglin1, DUAN Can1, LI Zhengliang1, FAN Cunyu2, ZOU Yilong2, XU Bo2, YANG Fengwen4, LIU Qingquan5( )
)
- 1 Rehabilitation Department, Hubei Provincial Hospital of Integrated Traditional Chinese and Western Medicine, Wuhan 430000, China
2 Respiratory Department, Hubei Provincial Hospital of Integrated Traditional Chinese and Western Medicine, Wuhan 430000, China
3 Endocrinology Department, Hubei P rovincial Hospital of Integrated Traditional Chinese and Western Medicine, Wuhan 430000, China
4 Traditional Chinese Medicine Department, Tianjin University of Traditional Chinese Medicine, Tianjin 300000, China
5 Emergency Department, Beijing Hospital of Traditional Chinese Medicine Affiliated to Capital Medical University, Beijing 100000, China
-
Received:2021-01-06Accepted:2021-02-19Online:2022-04-01Published:2022-04-01 -
Contact:LIU Qingquan -
About author:LIU Qingquan, Emergency Department, Beijing Hospital of tra ditional Chinese Medicine, Beijing 100010, China. hxyykfzx@163.com, Telephone: +86-13910055687
-
Supported by:National Key R&D Program “Public Security Risk Prevention and Control and Emergency Technology and Equipment” Key Special Project(2020YFC0841600);Hubei Provincial Department of Science and Technology, 2020 Hubei Province New Pneumonia Emergency Science and Technology Project(2020FCA027);Hubei Provincial Health Commission, 2021-2022 TCM Scientific Research Project Key Project(ZY2021Z003);Hubei Provincial Health Commission Key Support Projects from 2021 to 2022(WJ2021Z009);Hubei Provincial Department of Science and Technology, 2020 Hubei Province Key R&D Program Project(2020BCB012)
Cite this article
XIA Wenguang, ZHENG Chanjuan, ZHANG Jixian, HUANG Min, LI Qinglin, DUAN Can, LI Zhengliang, FAN Cunyu, ZOU Yilong, XU Bo, YANG Fengwen, LIU Qingquan. Randomized controlled study of a diagnosis and treatment plan for moderate coronavirus disease 2019 that integrates Traditional Chinese and Western Medicine[J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 234-241.
share this article
| Characteristics | Total | Experimental group (n = 80) | Control group (n = 40) | P valuea | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Age (years, $\bar{x}± s$) | 55.5±13.1 | 55.1±13.7 | 56.3±11.9 | 0.649 | |||||||
| 18-40 | 20 (16.7) | 15 (18.8) | 5 (12.5) | 0.386 | |||||||
| 41-60 | 48 (40.0) | 31 (38.8) | 17 (42.5) | 0.693 | |||||||
| 61-80 | 52 (43.3) | 34 (42.5) | 18 (45.0) | 0.794 | |||||||
| Sex, n (%) | Female | 69 (57.5) | 45 (56.3) | 24 (60.0) | 0.695 | ||||||
| Male | 51 (42.5) | 35 (43.8) | 16 (40.0) | ||||||||
| Smokers (active/ex-) | 38 (31.7) | 26 (32.5) | 12 (30.0) | 0.781 | |||||||
| Alcohol drinkers | 34 (28.3) | 24 (30.0) | 10 (25.0) | 0.567 | |||||||
| Comorbidities [n (%)] | Pulmonary tuberculosis | 2 (1.7) | 1 (1.3) | 1 (2.5) | 1 | ||||||
| COPD | 2 (1.7) | 1 (1.3) | 1 (2.5) | 1 | |||||||
| Hypertension | 59 (49.2) | 40 (50.0) | 19 (47.5) | 0.796 | |||||||
| Diabetes | 28 (23.3) | 20 (25.0) | 8 (20.0) | 0.542 | |||||||
| CKD | 3 (2.5) | 2 (2.5) | 1 (2.5) | 1 | |||||||
| Chronic liver disease | 3 (2.5) | 2 (2.5) | 1 (2.5) | 1 | |||||||
| Stroke | 4 (3.3) | 3 (3.8) | 1 (2.5) | 1 | |||||||
| Time from illness onset to hospitalization (days, $\bar{x}± s$) Signs and symptoms [n (%)] | 8.6±3.8 | 8.4±4.0 | 9.0±3.2 | 0.394 | |||||||
| Fever | 105 (87.5) | 71 (88.8) | 34 (85.0) | 0.558 | |||||||
| Cough | 59 (49.2) | 37 (46.3) | 22 (55.0) | 0.366 | |||||||
| Fatigue | 16 (13.3) | 10 (12.5) | 6 (15.0) | 0.704 | |||||||
| Diarrhea | 8 (6.7) | 5 (6.3) | 3 (7.5) | 1 | |||||||
| Other | 32 (26.7) | 23 (28.7) | 9 (22.5) | 0.465 | |||||||
| TCM syndrome [n (%)] | 0.901 | ||||||||||
| Dampness and stagnation lung | 55 (45.8) | 37 (46.3) | 18 (45.0) | 0.892 | |||||||
| Dampness and heat-accumulation lung | 33 (27.5) | 21 (26.3) | 12 (30.0) | 0.665 | |||||||
| Cold dampness lung | 32 (26.7) | 22 (27.5) | 10 (25.0) | 0.770 | |||||||
| Temperature (℃, $\bar{x}± s$) | 38.1±0.7 | 38.1±0.7 | 38.2±0.7 | 0.380 | |||||||
| Heart rate (bpm, $\bar{x}± s$) | 81.2±12.3 | 80.8±12.6 | 82.1±11.8 | 0.570 | |||||||
| Respiratory rate (bpm, $\bar{x}± s$) | 20.5±1.7 | 20.4±1.8 | 20.6±1.7 | 0.604 | |||||||
| Systolic pressure (mm Hg, $\bar{x}± s$) | 122.7±15.2 | 122.8±15.2 | 122.5±15.4 | 0.923 | |||||||
| Finger blood oxygen saturation (%) | 96.5±2.8 | 96.3±2.6 | 97.0±3.2 | 0.232 | |||||||
Table 1 Baseline characteristics of the patients with moderate COVID-19
| Characteristics | Total | Experimental group (n = 80) | Control group (n = 40) | P valuea | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Age (years, $\bar{x}± s$) | 55.5±13.1 | 55.1±13.7 | 56.3±11.9 | 0.649 | |||||||
| 18-40 | 20 (16.7) | 15 (18.8) | 5 (12.5) | 0.386 | |||||||
| 41-60 | 48 (40.0) | 31 (38.8) | 17 (42.5) | 0.693 | |||||||
| 61-80 | 52 (43.3) | 34 (42.5) | 18 (45.0) | 0.794 | |||||||
| Sex, n (%) | Female | 69 (57.5) | 45 (56.3) | 24 (60.0) | 0.695 | ||||||
| Male | 51 (42.5) | 35 (43.8) | 16 (40.0) | ||||||||
| Smokers (active/ex-) | 38 (31.7) | 26 (32.5) | 12 (30.0) | 0.781 | |||||||
| Alcohol drinkers | 34 (28.3) | 24 (30.0) | 10 (25.0) | 0.567 | |||||||
| Comorbidities [n (%)] | Pulmonary tuberculosis | 2 (1.7) | 1 (1.3) | 1 (2.5) | 1 | ||||||
| COPD | 2 (1.7) | 1 (1.3) | 1 (2.5) | 1 | |||||||
| Hypertension | 59 (49.2) | 40 (50.0) | 19 (47.5) | 0.796 | |||||||
| Diabetes | 28 (23.3) | 20 (25.0) | 8 (20.0) | 0.542 | |||||||
| CKD | 3 (2.5) | 2 (2.5) | 1 (2.5) | 1 | |||||||
| Chronic liver disease | 3 (2.5) | 2 (2.5) | 1 (2.5) | 1 | |||||||
| Stroke | 4 (3.3) | 3 (3.8) | 1 (2.5) | 1 | |||||||
| Time from illness onset to hospitalization (days, $\bar{x}± s$) Signs and symptoms [n (%)] | 8.6±3.8 | 8.4±4.0 | 9.0±3.2 | 0.394 | |||||||
| Fever | 105 (87.5) | 71 (88.8) | 34 (85.0) | 0.558 | |||||||
| Cough | 59 (49.2) | 37 (46.3) | 22 (55.0) | 0.366 | |||||||
| Fatigue | 16 (13.3) | 10 (12.5) | 6 (15.0) | 0.704 | |||||||
| Diarrhea | 8 (6.7) | 5 (6.3) | 3 (7.5) | 1 | |||||||
| Other | 32 (26.7) | 23 (28.7) | 9 (22.5) | 0.465 | |||||||
| TCM syndrome [n (%)] | 0.901 | ||||||||||
| Dampness and stagnation lung | 55 (45.8) | 37 (46.3) | 18 (45.0) | 0.892 | |||||||
| Dampness and heat-accumulation lung | 33 (27.5) | 21 (26.3) | 12 (30.0) | 0.665 | |||||||
| Cold dampness lung | 32 (26.7) | 22 (27.5) | 10 (25.0) | 0.770 | |||||||
| Temperature (℃, $\bar{x}± s$) | 38.1±0.7 | 38.1±0.7 | 38.2±0.7 | 0.380 | |||||||
| Heart rate (bpm, $\bar{x}± s$) | 81.2±12.3 | 80.8±12.6 | 82.1±11.8 | 0.570 | |||||||
| Respiratory rate (bpm, $\bar{x}± s$) | 20.5±1.7 | 20.4±1.8 | 20.6±1.7 | 0.604 | |||||||
| Systolic pressure (mm Hg, $\bar{x}± s$) | 122.7±15.2 | 122.8±15.2 | 122.5±15.4 | 0.923 | |||||||
| Finger blood oxygen saturation (%) | 96.5±2.8 | 96.3±2.6 | 97.0±3.2 | 0.232 | |||||||
| Item | Normal range | Total (n = 108) | Experimental group (n = 73) | Control group (n = 35) | P value |
|---|---|---|---|---|---|
| WBC count (×109/L) | 4-10 | 6.00±2.59 | 6.17±2.80 | 5.68±2.11 | 0.290 |
| NEUT (%) | 50-70 | 62.98±11.62 | 62.40±11.31 | 64.15±12.30 | 0.439 |
| LYMPH (%) | 20-40 | 14.91±6.96 | 14.29±6.51 | 16.16±7.71 | 0.164 |
| SAA (mg/L) | < 10 | 151.01±109.22 | 150.98±105.51 | 151.07±117.70 | 0.997 |
| CRP (mg/L) | < 10 | 31.15±24.43 | 32.70±23.52 | 28.06±26.19 | 0.329 |
| ESR (mm/H) | < 20 | 30.55±17.98 | 29.92±19.56 | 31.82±14.43 | 0.549 |
| PCT (µg/L) | < 0.046 | 0.03±0.04 | 0.03±0.04 | 0.03±0.05 | 0.899 |
| CK-MB (U/L) | 0-24 | 25.83±9.94 | 25.23±7.17 | 27.05±13.98 | 0.441 |
| LDH (U/L) | 114-240 | 284.00±128.13 | 277.96±108.43 | 296.08±161.39 | 0.524 |
| ALT (U/L) | 7-40 | 45.37±35.71 | 45.90±36.08 | 44.30±35.39 | 0.818 |
| AST (U/L) | 13-35 | 42.07±22.94 | 41.03±20.66 | 44.18±27.11 | 0.481 |
| BUN (mmol/L) | 2.3-8.2 | 7.57±3.54 | 7.42±3.74 | 7.87±3.12 | 0.509 |
| D-dimer (mg/L) | < 0.5 | 2.50±6.97 | 2.52±7.22 | 2.46±6.54 | 0.960 |
Table 2 Comparison of laboratory findings on admission between the experimental and control groups of patients with moderate COVID-19
| Item | Normal range | Total (n = 108) | Experimental group (n = 73) | Control group (n = 35) | P value |
|---|---|---|---|---|---|
| WBC count (×109/L) | 4-10 | 6.00±2.59 | 6.17±2.80 | 5.68±2.11 | 0.290 |
| NEUT (%) | 50-70 | 62.98±11.62 | 62.40±11.31 | 64.15±12.30 | 0.439 |
| LYMPH (%) | 20-40 | 14.91±6.96 | 14.29±6.51 | 16.16±7.71 | 0.164 |
| SAA (mg/L) | < 10 | 151.01±109.22 | 150.98±105.51 | 151.07±117.70 | 0.997 |
| CRP (mg/L) | < 10 | 31.15±24.43 | 32.70±23.52 | 28.06±26.19 | 0.329 |
| ESR (mm/H) | < 20 | 30.55±17.98 | 29.92±19.56 | 31.82±14.43 | 0.549 |
| PCT (µg/L) | < 0.046 | 0.03±0.04 | 0.03±0.04 | 0.03±0.05 | 0.899 |
| CK-MB (U/L) | 0-24 | 25.83±9.94 | 25.23±7.17 | 27.05±13.98 | 0.441 |
| LDH (U/L) | 114-240 | 284.00±128.13 | 277.96±108.43 | 296.08±161.39 | 0.524 |
| ALT (U/L) | 7-40 | 45.37±35.71 | 45.90±36.08 | 44.30±35.39 | 0.818 |
| AST (U/L) | 13-35 | 42.07±22.94 | 41.03±20.66 | 44.18±27.11 | 0.481 |
| BUN (mmol/L) | 2.3-8.2 | 7.57±3.54 | 7.42±3.74 | 7.87±3.12 | 0.509 |
| D-dimer (mg/L) | < 0.5 | 2.50±6.97 | 2.52±7.22 | 2.46±6.54 | 0.960 |
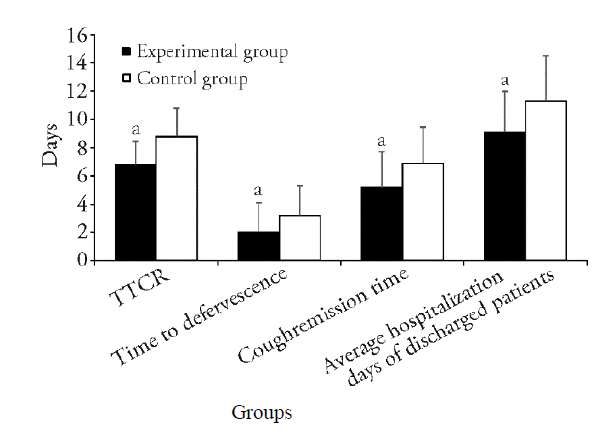
Figure 2 Comparison of efficacy endpoints for coronavirus disease 2019 patients who received combination treatment versus control treatment Experimental group: patients received Western Medicine treatment plus Traditional Chinese Medicine; control group: patients received only Western Medicine treatment. Significant differences compared with control group were designated as aP < 0.01.
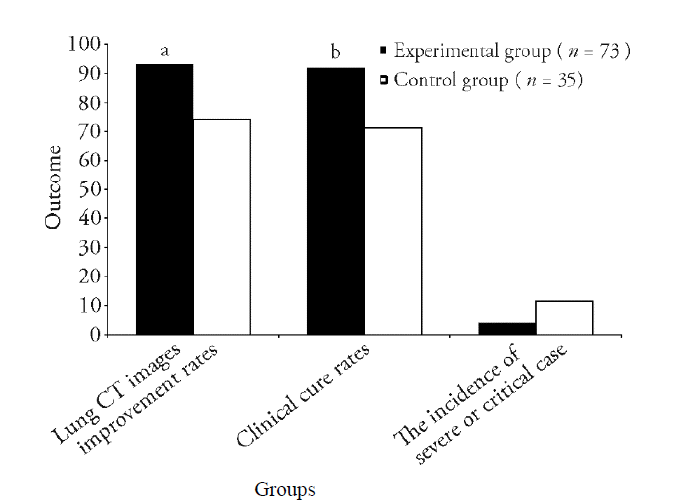
Figure 3 Comparison of efficacy endpoints for coronavirus disease 2019 patients who received combination treatment versus control treatment Experimental group: patients received Western Medicine treatment plus Traditional Chinese Medicine; control group: patients received only Western Medicine treatment. Significant differences compared with control group were designated as aP < 0.05 and bP < 0.01.

Figure 4 Hospital discharge rates in the experimental and control groups over 2 weeks following coronavirus disease 2019 diagnosis Experimental group: patients received Western Medicine treatment plus Traditional Chinese Medicine; control group: patients received only Western Medicine treatment. Significant differences compared with control group were designated as aP < 0.05.
| Group | n | Before treatment | After treatmenta | Z value | P value | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 0 | 1 | 2 | 3 | 4 | |||||
| Control | 35 | 2 | 4 | 13 | 16 | 0 | 7 | 10 | 10 | 6 | 2 | –3.810 | < 0.001 | |
| Experimental | 73 | 4 | 11 | 25 | 33 | 0 | 20 | 38 | 8 | 6 | 1 | –6.789 | < 0.001 | |
Table 3 Comparison of mMRC scale scores between the experimental and control groups of patients with moderate coronavirus disease 2019
| Group | n | Before treatment | After treatmenta | Z value | P value | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 0 | 1 | 2 | 3 | 4 | |||||
| Control | 35 | 2 | 4 | 13 | 16 | 0 | 7 | 10 | 10 | 6 | 2 | –3.810 | < 0.001 | |
| Experimental | 73 | 4 | 11 | 25 | 33 | 0 | 20 | 38 | 8 | 6 | 1 | –6.789 | < 0.001 | |
| Item | Experimental group (n = 73) | Control Group (n = 35) | P value |
|---|---|---|---|
| WBC count (×109/L) | 69 (94.5) | 26 (74.3) | 0.007 |
| NEUT (%) | 68 (93.2) | 31 (88.6) | 0.664 |
| LYMPH (%) | 67 (91.8) | 23 (65.7) | 0.001 |
| SAA (mg/L) | 64 (87.7) | 25 (71.4) | 0.038 |
| CRP (mg/L) | 66 (90.4) | 23 (65.7) | 0.002 |
| ESR (mm/H) | 70 (95.9) | 27 (77.1) | 0.007 |
| PCT (µg/L) | 69 (94.5) | 32 (91.4) | 0.847 |
| CK-MB (U/L) | 69 (94.5) | 28 (80.0) | 0.046 |
| LDH (U/L) | 70 (95.9) | 28 (80.0) | 0.021 |
| ALT (U/L) | 68 (93.2) | 26 (74.3) | 0.015 |
| AST (U/L) | 70 (95.9) | 28 (80.0) | 0.021 |
| BUN (mmol/L) | 67 (91.8) | 29 (82.9) | 0.292 |
| D-dimer (mg/L) | 70 (95.9) | 31 (88.6) | 0.304 |
Table 4 Comparison of the percentages of patients with normal values after treatment between the experimental and control groups of patients with moderate COVID-19 [n (%)]
| Item | Experimental group (n = 73) | Control Group (n = 35) | P value |
|---|---|---|---|
| WBC count (×109/L) | 69 (94.5) | 26 (74.3) | 0.007 |
| NEUT (%) | 68 (93.2) | 31 (88.6) | 0.664 |
| LYMPH (%) | 67 (91.8) | 23 (65.7) | 0.001 |
| SAA (mg/L) | 64 (87.7) | 25 (71.4) | 0.038 |
| CRP (mg/L) | 66 (90.4) | 23 (65.7) | 0.002 |
| ESR (mm/H) | 70 (95.9) | 27 (77.1) | 0.007 |
| PCT (µg/L) | 69 (94.5) | 32 (91.4) | 0.847 |
| CK-MB (U/L) | 69 (94.5) | 28 (80.0) | 0.046 |
| LDH (U/L) | 70 (95.9) | 28 (80.0) | 0.021 |
| ALT (U/L) | 68 (93.2) | 26 (74.3) | 0.015 |
| AST (U/L) | 70 (95.9) | 28 (80.0) | 0.021 |
| BUN (mmol/L) | 67 (91.8) | 29 (82.9) | 0.292 |
| D-dimer (mg/L) | 70 (95.9) | 31 (88.6) | 0.304 |
| 1. |
Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med 2020; 382: 1199-207.
DOI URL |
| 2. |
Phan LT, Nguyen TV, Luong QC, et al. Importation and human-to-human transmission of a novel coronavirus in Vietnam. N Engl J Med 2020; 382: 872-4.
DOI URL |
| 3. |
Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382: 1708-20.
DOI URL |
| 4. |
Alanagreh L, Alzoughool F, Atoum M. The human coronavirus disease COVID-19: its origin, characteristics, and insights into potential drugs and its mechanisms. Pathogens 2020; 9: 331.
DOI URL |
| 5. |
Lu H. Drug treatment options for the 2019-new coronavirus (2019-nCoV). Biosci Trends 2020, 14: 69-71.
DOI URL |
| 6. | National Health Commission of the People’s Republic of China. Diagnosis and treatment of New coronavirus pneumonia.(trial version 5). (in Chinese). Published February 4, 2020. Accessed February 20, 2020: screen 10-14. Available from URL: http://www.nhc.gov.cn/yzygj/s7653p/202002/3b09b894ac9b4204a79db5b8912d4440.shtml. |
| 7. | National Health Commission of the People's Republic of China. Diagnosis and treatment of new coronavirus pneumonia. (trial version 6). (in Chinese). Published February 18, 2020. Accessed February 20, 2020: screen 8-15. Available from URL: news.sciencenet.cn/htmlnews/2020/2/435927.shtm. |
| 8. |
Lau AC, So LK, Miu FP, et al. Outcome of coronavirus-associated severe acute respiratory syndrome using a standard treatment protocol. Respirology 2004; 9: 173-83.
DOI URL |
| 9. |
Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An Official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med 2019; 200: e45-67.
DOI URL |
| 10. |
Yang Y, Wang SC, Bai WJ, Li RL, Ai J. Evaluation by survival analysis on effect of Traditional Chinese Medicine in treating children with respiratory syncytial viral pneumonia of phlegm-heat blocking Fei syndrome. Chin J Integr Med 2009; 15: 95-100.
DOI PMID |
| 11. |
Stets R, Popescu M, Gonong JR, et al. Omadacycline for community-acquired bacterial pneumonia. N Engl J Med 2019; 380: 517-27.
DOI URL |
| 12. | Sandberg J, Johnson MJ, Currow DC, Ekström M. Validation of the dyspnea exertion scale of breathlessness in people with life-limiting illness. J Pain Symptom Manage 2018; 56: 430-5.e2. |
| 13. |
Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 2020; 323: 1061-9.
DOI URL |
| 14. |
Abo-Hagar HH, Abo-Elezz AAE, Mehrez M, et al. Diagnostic efficacy of serum amyloid a protein and soluble intercellular adhesion molecule 1 in pediatric ventilator-associated pneumonia. J Intensive Care Med 2019; 34: 503-10.
DOI PMID |
| 15. |
Wong JP, Viswanathan S, Wang M, Sun LQ, Clark GC, D'Elia RV. Current and future developments in the treatment of virus-induced hypercytokinemia. Future Med Chem 2017; 9: 169-78.
DOI URL |
| [1] | WANG Jiabao, ZHANG Lishuang, NIU Baihan, YU Yajun, YANG Fengwen, MIAO Lin, CHAI Lijuan, DING Xinya, SUN Yingjie, WANG Yujing, WANG Lin, ZHANG Han, WANG Yi, LI Lin. Efficacy and safety of Weichang’ an pill (胃肠安丸) combined with Western Medicine on gastrointestinal diseases: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1057-1067. |
| [2] | FAN Rong, HE Haoyu, TANG Tao, CUI Hanjin. Long-term effects of Qingfei Paidu decoction (清肺排毒汤) in patients with coronavirus disease 2019 acute pneumonia after treatment: a protocol for systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1068-1071. |
| [3] | SUN Wu, ZHAO Yuwei, LIAO Liang, ZHAO Zhonghui, CHEN Shiqi, YAN Xiaoling, WANG Xueyao, CHAO Guojun, ZHOU Jian. Effectiveness and safety of Xuebijing injection for patients with coronavirus disease 2019: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 631-639. |
| [4] | LI He, SU Wenquan, LI Shanshan, JI Hanrui, CHENG Jiangyan, CUI Fangyuan, TANG Lu, ZHOU Li, GAO Ying, DONG Xinglu. Supplementing Qi and activating blood circulation method to treat vertebrobasilar dolichoectasia with posterior circulatory watershed infarction: a case report of two patients [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 824-828. |
| [5] | ZHANG Yuehong, SHAO Xianzhi, ZHAO Qianlong, ZHAN Hualong, ZHANG Jianhua, DU Sisi, CHEN Jing, LIU Yingfang, ZHOU Haiwang, CHEN Xinsheng, HONG Ying, LIAN Fengmei, TONG Xiaolin, BA Yuanming. Effectiveness of Xiangsha Liujun pills (香砂六君丸) on decreased digestive function in convalescent patients of coronavirus disease 2019: a randomized, double blind, placebo controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 552-558. |
| [6] | HOU Chao, ZHANG Yusen, YANG Die, LI Yifei, ZHANG Xiaochun, LIU Yanqing. Effects of Traditional Chinese Medicine on the survival of patients with stage I gastric cancer and high-risk factors: a real-world retrospective study [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 568-573. |
| [7] | XU Guihua, CHEN Feifei, ZHANG Wei, WU Yingen, CHEN Xiaorong, SHI Kehua, WANG Zhenwei, SHI Miaoyan, ZHANG Xing, LU Yunfei, YUAN Weian, LYU Hua, CHEN Xuan. Effectiveness of Traditional Chinese Medicine on coronavirus disease 2019 in 92 patients: a retrospective study [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 582-587. |
| [8] | HU Xingyao, LIU Hongning, YAN Xiaojun, CHEN Zhong, FU Liu, LIU Ge, CHEN Xuan, SHANG Guangbin. Liver metabolomic characteristics in three different rat models of Yin deficiency based on ultra-performance liquid chromatography coupled to quadrupole time-of-flight mass spectrometry [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 274-285. |
| [9] | YANG Cunqing, LIAN Fengmei, YANG Guiping, HUANG Yufeng, ZHANG Shuangbin, WANG Jianghua, ZHOU Jing, GUO Dongqing, SHEN Chuanyun, YE Tiansong, FU Aojie, LI Xiaoli, CHEN Le, ZHANG Huifeng, TU Qiyin, WANG Ying, YANG Wenzhe, TONG Xiaolin, BA Yuanming. Effectiveness of Xiaoyao capsule (逍遥丸) on sleep disorders and mood disturbance in patients in recovery from coronavirus disease 2019: a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 343-351. |
| [10] | ZHANG Yuehong, DONG Dandan, YAN Youqin, ZHANG Hao, WANG Guangli, ZHOU Wei, LI Wei, QIU Li, LI Tingming, LIU Quan, XIA Ping, MAO Lina, YANG Danlin, YANG Lu, LIAN Fengmei, TONG Xiaolin, BA Yuanming. Effectiveness and safety of Jinshuibao capsules (金水宝胶囊) in treatment of residual cardiopulmonary symptoms in convalescent patients of coronavirus disease 2019: a pilot randomized, double-blind, placebo-controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 134-139. |
| [11] | AN Xuedong, MAO Lina, XIA Ping, SU Wen, WANG Beibei, KOU Leiya, ZHANG Zequan, QI Meng, HU Song, CHEN Jing, LI Xiujuan, LIU Jinwei, ZHOU Juan, QIAO Jie, LUO Dan, LUO Guangwei, YAN Youqin, YANG Guiping, DONG Dandan, ZHOU Wei, TAO Junxiu, JIN De, TONG Xiaolin, WEI Li. Effects of Shengmai Yin (生脉饮) on pulmonary and cardiac function in coronavirus disease 2019 convalescent patients with cardiopulmonary symptoms: a randomized, double blind, multicenter control trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 140-145. |
| [12] | LI Ximeng, KANG Yuan, LI Wenjing, LIU Zhuangzhuang, XU Zhenlu, ZHANG Xiaoyu, CAI Runlan, GAO Yuan, QI Yun. Comparing the effects of three decoctions for coronavirus disease 2019 on severe acute respiratory syndrome coronavirus 2-related toll-like receptors-mediated inflammations [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 51-59. |
| [13] | ZHANG Wen, CAO Min, SUN Ding, SHI Li, LU Wei, XU Xiangru, ZHOU Shuang, FANG Bangjiang. Tongue and pulse features of 668 asymptomatic patients infected with the severe acute respiratory syndrome coronavirus 2 omicron variant in Shanghai [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 1006-1011. |
| [14] | XU Qing, WANG Yang, LI Zhongyu, YAN Jiaxing, ZHAO Yingpan, WANG Ping, WEN Yandong. Therapeutic mechanisms of integrated traditional Chinese and conventional medicine underlying its treatment of precancerous lesions of gastric cancer [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 1023-1028. |
| [15] | ZHU Qingguang, ZHANG Shuaipan, LI Jingxian, SUN Wuquan, CHENG Wei, ZHAN Chao, CHENG Yanbin, FANG Lei, FANG Min. Effectiveness of Liu-zi-jue exercise on coronavirus disease 2019 in the patients: a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 997-10053. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||