Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (3): 484-493.DOI: 10.19852/j.cnki.jtcm.20230214.005
Previous Articles Next Articles
Protective effect of Tongdu Tiaoshen acupuncture combined with Xiaoxuming decoction (小续命汤) on dopaminergic neurons in Parkinson’s disease model
WU Haiyang1, WANG Ying1( ), HAN Wei2(
), HAN Wei2( ), LI Huihui3, JI Haisheng3, LIU Xiuxiu3
), LI Huihui3, JI Haisheng3, LIU Xiuxiu3
- 1 Department of encephalopathy, the Second Affiliated Hospital of Anhui University of Traditional Chinese Medicine, Hefei 230061, China
2 Deparment of acupuncture and Moxibustion, The Affiliated Brain Hospital of Guangzhou Medical University, Guangzhou 510370, China
3 Department of acupuncture and massage, Anhui University of Chinese Medicine, Hefei 230061, China
-
Received:2022-08-03Accepted:2022-11-23Online:2023-06-15Published:2023-02-14 -
Contact:HAN Wei, Deparment of acupuncture and Moxibustion, the Affiliated Brain Hospital of Guangzhou Medical University, Guangzhou 510370, China. 13956060099@163.com. Telephone: +86-13956060099
WANG Ying, Department of Encephalopathy, the Second Affiliated Hospital of Anhui University of Traditional Chinese Medicine, Hefei 230061, China. zhenjiu205@126.com -
Supported by:Study on the Mechanism of Regulating miR-124 by Tongdu Tiaoshen Acupuncture to Promote Neuroprotection in Cerebral Ischemia Reperfusion Injury(81973933);Study on the Regulatory Mechanism of Tongdu Tiaoshen Acupuncture on Dopaminergic Neurons in Parkinson's Disease Model Mice Based on Mitochondrial Autophagy(KJ2019A0475)
Cite this article
WU Haiyang, WANG Ying, HAN Wei, LI Huihui, JI Haisheng, LIU Xiuxiu. Protective effect of Tongdu Tiaoshen acupuncture combined with Xiaoxuming decoction (小续命汤) on dopaminergic neurons in Parkinson’s disease model[J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 484-493.
share this article

Figure 1 Representative images of substantia nigra tissues and tyrosine hydroxylase (TH) in substantia nigra A1-H1: representative images of substantia nigra tissues (hematoxylin-eosin staining, ×200). A1: blank group; B1: model group; C1: XXMD-H group; D1: XXMD-L group; E1: acupuncture group; F1: A + H group; G1: A + L group; H1: medication group. Red arrows indicate severely damaged dopamine neurons. A2-H2: representative histologic images of TH in substantia nigra (immumohistochemical staining, ×400). A2: blank group; B2: model group; C2: XXMD-H group; D2: XXMD-L group; E2: acupuncture group; F2: A + H group; G2: A+L group; H2: medication group. The brown or brownish yellow in the picture is positive staining. Blank group: without any treatment. Model group: only modeled without any treatment. XXMD-H and XXMD-L group: treated with Xiaoxunming decotion by intragastric administration (6 weeks, 60 and 15 g·kg-1·d-1, respectively). A + H and A + L group: treated with Xiaoxunming decotion by intragastric administration (6 weeks, 60 and 15 g·kg-1·d-1, respectively) and corresponding acupuncture. Medication group: treated with dopasrazide (0.5 mg/10 g, 3 times/d, 6 weeks) by gavage. XXMD-H: high-dose Xiaoxunming decotion; XXMD-L: low-dose Xiaoxunming decotion; A + H: acupuncture combined with high-dose Xiaoxunming decotion; A + L: acupuncture combined with low-dose Xiaoxunming decotion.
| Group | n | Locomotor counts | Pole test scores | |||||
|---|---|---|---|---|---|---|---|---|
| Pre-modeling | Post-modeling | Post-treatment | Pre-modeling | Post-modeling | Post-treatment | |||
| Blank | 12 | 148.143±17.332 | 135.600±15.231 | 143.773±14.121 | 0.073±0.0328 | 0.074±0.033 | 0.073±0.032 | |
| Model | 12 | 148.791±18.508 | 81.237±9.830a | 84.782±11.725a | 0.057±0.041 | 1.924±0.208a | 1.936±0.311a | |
| XXMD-H | 12 | 136.218±6.112 | 89.471±8.536b | 114.070±9.051bc | 0.072±0.032 | 1.751±0.192a | 0.592±0.109bc | |
| XXMD-L | 12 | 139.212±9.537 | 78.207±10.738a | 99.718±8.741a | 0.059±0.034 | 1.729±0.136a | 0.711±0.086bd | |
| Acupuncture | 12 | 132.031±10.229 | 91.288±10.126b | 118.187±10.513c | 0.064±0.043 | 1.691±0.110a | 0.721±0.127ad | |
| A+H | 12 | 140.677±11.141 | 80.132±8.224a | 139.033±15.862d | 0.069±0.031 | 1.743±0.082a | 0.514±0.122bd | |
| A+L | 12 | 135.571±14.329 | 92.379±8.010b | 120.881±15.722c | 0.058±0.027 | 1.677±0.142a | 0.583±0.120ad | |
| Medication | 12 | 132.031±13.717 | 97.521±7.626b | 136.338±17.206c | 0.062±0.032 | 1.713±0.130a | 0.389±0.133bd | |
Table 1 Locomotor counts and pole test scores of mice in each group
| Group | n | Locomotor counts | Pole test scores | |||||
|---|---|---|---|---|---|---|---|---|
| Pre-modeling | Post-modeling | Post-treatment | Pre-modeling | Post-modeling | Post-treatment | |||
| Blank | 12 | 148.143±17.332 | 135.600±15.231 | 143.773±14.121 | 0.073±0.0328 | 0.074±0.033 | 0.073±0.032 | |
| Model | 12 | 148.791±18.508 | 81.237±9.830a | 84.782±11.725a | 0.057±0.041 | 1.924±0.208a | 1.936±0.311a | |
| XXMD-H | 12 | 136.218±6.112 | 89.471±8.536b | 114.070±9.051bc | 0.072±0.032 | 1.751±0.192a | 0.592±0.109bc | |
| XXMD-L | 12 | 139.212±9.537 | 78.207±10.738a | 99.718±8.741a | 0.059±0.034 | 1.729±0.136a | 0.711±0.086bd | |
| Acupuncture | 12 | 132.031±10.229 | 91.288±10.126b | 118.187±10.513c | 0.064±0.043 | 1.691±0.110a | 0.721±0.127ad | |
| A+H | 12 | 140.677±11.141 | 80.132±8.224a | 139.033±15.862d | 0.069±0.031 | 1.743±0.082a | 0.514±0.122bd | |
| A+L | 12 | 135.571±14.329 | 92.379±8.010b | 120.881±15.722c | 0.058±0.027 | 1.677±0.142a | 0.583±0.120ad | |
| Medication | 12 | 132.031±13.717 | 97.521±7.626b | 136.338±17.206c | 0.062±0.032 | 1.713±0.130a | 0.389±0.133bd | |
| Group | n | IL-1β | IL-6 | IL-10 | TNF-α |
|---|---|---|---|---|---|
| Blank | 12 | 27±3 | 38±5 | 145±12 | 109±8 |
| Model | 12 | 106±12a | 126±11a | 40±5a | 339±26a |
| XXMD-H | 12 | 63±9bc | 73±7bc | 71±8bc | 243±29bc |
| XXMD-L | 12 | 73±8bc | 85±9bd | 64±9bc | 256±37b |
| Acupuncture | 12 | 67±8bc | 73±6bc | 67±5bc | 242±34bc |
| A+H | 12 | 41±7d | 54±5de | 104±12cd | 146±25d |
| A+L | 12 | 48±6de | 63±6de | 81±10bc | 212±20bd |
| medication | 12 | 39±5d | 52±7d | 107±8cd | 141±24d |
Table 2 Comparison of serum levels of IL-1β, IL-6, IL-10 and TNF-α in each group (pg/mL)
| Group | n | IL-1β | IL-6 | IL-10 | TNF-α |
|---|---|---|---|---|---|
| Blank | 12 | 27±3 | 38±5 | 145±12 | 109±8 |
| Model | 12 | 106±12a | 126±11a | 40±5a | 339±26a |
| XXMD-H | 12 | 63±9bc | 73±7bc | 71±8bc | 243±29bc |
| XXMD-L | 12 | 73±8bc | 85±9bd | 64±9bc | 256±37b |
| Acupuncture | 12 | 67±8bc | 73±6bc | 67±5bc | 242±34bc |
| A+H | 12 | 41±7d | 54±5de | 104±12cd | 146±25d |
| A+L | 12 | 48±6de | 63±6de | 81±10bc | 212±20bd |
| medication | 12 | 39±5d | 52±7d | 107±8cd | 141±24d |
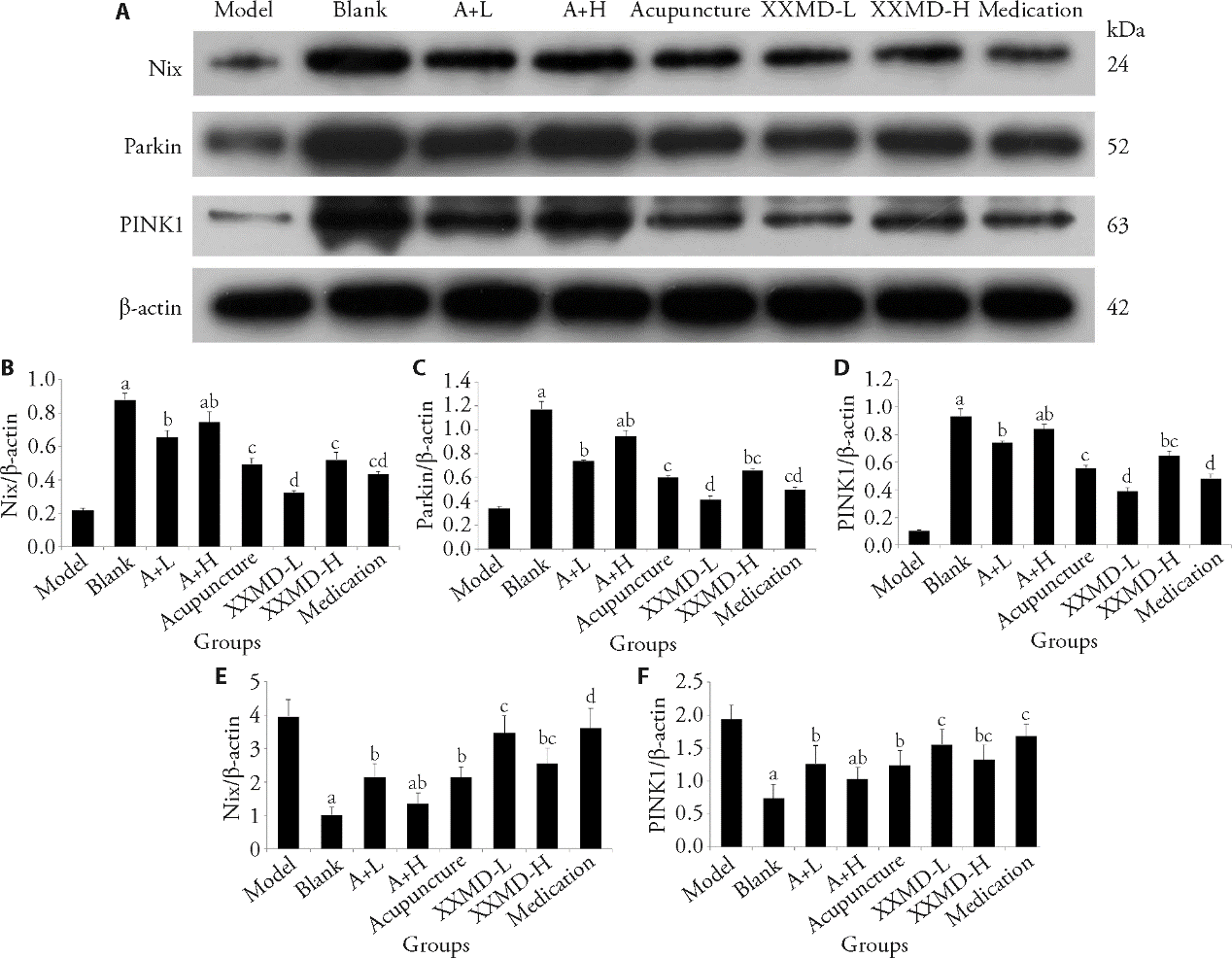
Figure 2 Expression of Nix, Parkin and PINK1 in the substantia nigra of mice after different treatment A: epresentative images of protein bands of Nix, Parkin and PINK1. B-D: results of quantitative analyses of Nix (B), Parkin (C) and PINK1 (D) in protein level. E, F: results of relative mRNA levels of PINK1 (E) and Parkin (F). Compared with model group, aP<0.0001, bP<0.001, cP<0.01, dP<0.05; compared with black group, aP<0.0001, bP<0.001, cP<0.01, dP<0.05. Blank group: without any treatment. Model group: only modeled without any treatment. XXMD-H and XXMD-L group: treated with Xiaoxunming decotion by intragastric administration (60 and 15 g·kg-1·d-1, respectively). A + H and A + L group: treated with Xiaoxunming decotion by intragastric administration (60 and 15 g·kg-1·d-1, respectively) and corresponding acupuncture. Medication group: treated with dopasrazide (0.5 mg/10 g) by gavage. XXMD-H: high-dose Xiaoxunming decotion; XXMD-L: low-dose Xiaoxunming decotion; A + H: acupuncture combined with high-dose Xiaoxunming decotion; A + L: acupuncture combined with low-dose Xiaoxunming decotion. 1: model; 2: blank 3: A + L; 4: A + H; 5: acupuncture; 6: XXMD-L; 7: XXMD-H; 8: medication. Statistical significance between the groups was determined by one-way analysis of variance. The sample size for each group was 12.
| [1] |
Le Bras A. Development of a new preclinical model of Parkinson’s disease. Lab Animal 2020; 49: 219.
DOI |
| [2] |
Liu L, Peritore C, Ginsberg J, Shih J, Arun S, Donmez G. Protective role of SIRT5 against motor deficit and dopaminergic degeneration in MPTP-induced mice model of Parkinson’s disease. Behav Brain Res 2015; 281: 215-21.
DOI URL |
| [3] |
Aarsland D, Batzu L, Halliday GM, et al. Parkinson disease-associated cognitive impairment. Nat Rev Dis Primers 2021; 7: 47.
DOI PMID |
| [4] | Hou X, Watzlawik JO, Fiesel FC, Springer W. Autophagy in Parkinson’s Disease. J Mol Biol 2020; 432: 2651-72. |
| [5] | Zhu H, Toan S, Mui D, Zhou H. Mitochondrial quality surveillance as a therapeutic target in myocardial infarction. Acta Physiol 2021; 231: e13590. |
| [6] |
Twig G, Hyde B, Shirihai OS. Mitochondrial fusion, fission and autophagy as a quality control axis: the bioenergetic view. BBA-Bioenergetics 2008; 1777: 1092-7.
DOI URL |
| [7] |
Chen H, Chan DC. Mitochondrial dynamics-fusion, fission, movement, and mitophagy-in neurodegenerative diseases. Hum Mol Genet 2009; 18: R169-76.
DOI URL |
| [8] |
Geisler S, Holmström KM, Skujat D, et al. PINK1/Parkin-mediated mitophagy is dependent on VDAC1 and p62/SQSTM1. Nat Cell Biol 2010; 12: 119-31.
DOI PMID |
| [9] |
Diwan A, Wansapura J, Syed FM, Matkovich SJ, Lorenz JN, Dorn GW. Nix-mediated apoptosis links myocardial fibrosis, cardiac remodeling, and hypertrophy decompensation. Circulation 2008; 117: 396-404.
DOI PMID |
| [10] | Kabat AM, Harrison OJ, Riffelmacher T, et al. The autophagy gene Atg16l 1 differentially regulates Treg and TH2 cells to control intestinal inflammation. eLife 2016; 5: e12444. |
| [11] |
Colín-Castelán D, Zaina S. Associations between atherosclerosis and neurological diseases, beyond ischemia-induced cerebral damage. Rev Endocr Metab Dis 2019; 20: 15-25.
DOI PMID |
| [12] |
Sharma S, Singh S, Sharma V, Singh VP, Deshmukh R. Neurobiology of l-DOPA induced dyskinesia and the novel therapeutic strategies. Biomed Pharmacother 2015; 70: 283-93.
DOI PMID |
| [13] | Xing BF, Zhou X, Deng XQ. Effect of “Tongdu Tiaoshen” needling combined with swallowing training on dysphagia, cerebral blood flow and serum BDNF and NGF levels in ischemic stroke patients. Zhen Ci Yan Jiu 2019; 44: 506-11. |
| [14] | Sun PY, Li PF, Wang T, et al. Effect of Tongdu Tiaoshen acupuncture on PI3K/Akt/mTOR signaling pathway and autophagy-related proteins of hippocampus in rats with post-stroke depression. Zhong Guo Zhen Jiu 2020; 40: 1205-10. |
| [15] |
Shen Y, Zhang B, Pang X, et al. Network pharmacology-based analysis of Xiao-Xu-Ming decoction on the treatment of Alzheimer’s disease. Front Pharmacol 2020; 11: 595254
DOI URL |
| [16] |
Wu ZQ, Qian SY, Zhao LC, et al. Metabolomics-based study of the potential interventional effects of Xiao-Xu-Ming decoction on cerebral ischemia/reperfusion rats. J Ethnopharmacol 2022; 295: 115379.
DOI URL |
| [17] |
Lu KT, Ko MC, Chen BY, et al. Neuroprotective effects of resveratrol on MPTP-induced neuron loss mediated by free radical scavenging. J Agr Food Chem 2008; 56: 6910-3.
DOI URL |
| [18] |
Paul SS, Canning CG, Sherrington C, Fung VSC. Reduced muscle strength is the major determinant of reduced leg muscle power in Parkinson’s disease. Parkinsonism Relat D 2012; 18: 974-7.
DOI PMID |
| [19] | Lan R, Xiang J, Wang GH, et al. Xiao-Xu-Ming decoction protects against blood-brain barrier disruption and neurological injury induced by cerebral ischemia and reperfusion in rats. Evid-Based Compl Alt 2013; 2013: 629782. |
| [20] |
Kim SN, Doo AR, Park JY, et al. Acupuncture enhances the synaptic dopamine availability to improve motor function in a mouse model of Parkinson’s disease. PLoS One 2011; 6: e27566.
DOI URL |
| [21] | He X, Yang S, Zhang R, et al. Smilagenin protects dopaminergic neurons in chronic MPTP/probenecid-lesioned Parkinson’s disease models. Front Cell Neurosci 2019; 13: 18. |
| [22] |
Kastner A, Hirsch EC, Agid Y, Javoy-Agid F. Tyrosine hydroxylase protein and messenger RNA in the dopaminergic nigral neurons of patients with Parkinson’s disease. Brain Res 1993; 606: 341-5.
PMID |
| [23] |
Klemann CJHM, Martens GJM, Poelmans G, Visser JE. Validity of the MPTP-treated mouse as a model for Parkinson’s disease. Mol Neurobiol 2015; 53: 1625-36.
DOI URL |
| [24] | Liu J, Gao J, Tu S, et al. Neuroprotective effects of Jitai tablet, a Traditional Chinese Medicine, on the MPTP-induced acute model of Parkinson’s disease: involvement of the dopamine system. Evid-Based Compl Alt 2014; 2014: 542383. |
| [25] |
Chen P, Zhang J, Wang C, et al. The pathogenesis and treatment mechanism of Parkinson’s disease from the perspective of traditional Chinese medicine. Phytomedicine 2022; 100: 154044.
DOI URL |
| [26] |
Ko JH, Lee H, Kim SN, Park HJ. Does acupuncture protect dopamine neurons in Parkinson’s disease rodent model? A systematic review and Meta-analysis. Front Aging Neurosci 2019; 11: 102.
DOI URL |
| [27] |
Rubinsztein David C, Mariño G, Kroemer G. Autophagy and aging. Cell 2011; 146: 682-95.
DOI PMID |
| [28] |
Schapira AHV, Olanow CW, Greenamyre JT, Bezard E. Slowing of neurodegeneration in Parkinson’s disease and Huntington's disease: future therapeutic perspectives. Lancet 2014; 384: 545-55.
DOI PMID |
| [29] | Reeve AK, Grady JP, Cosgrave EM, et al. Mitochondrial dysfunction within the synapses of substantia nigra neurons in Parkinson’s disease. NPJ Parkinson’s Dis 2018; 4: 1-9. |
| [30] |
Youle RJ, Narendra DP. Mechanisms of mitophagy. Nat Rev Mol Cell Bio 2011; 12: 9-14.
DOI PMID |
| [31] | Narendra DP, Jin SM, Tanaka A, et al. PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol 2010; 8: e1000298. |
| [32] |
Santos D, Cardoso SM. Mitochondrial dynamics and neuronal fate in Parkinson’s disease. Mitochondrion 2012; 12: 428-37.
DOI PMID |
| [33] |
Bingol B, Sheng M. Mechanisms of mitophagy: PINK1, Parkin, USP30 and beyond. Free Radical Bio Med 2016; 100: 210-22.
DOI PMID |
| [34] | Dorn GW. Mitochondrial pruning by Nix and BNip3: an essential function for cardiac-expressed death factors. J Cardiovasc Transl 2010; 3: 374-83. |
| [35] |
Sandoval H, Thiagarajan P, Dasgupta SK, et al. Essential role for Nix in autophagic maturation of erythroid cells. Nature 2008; 454: 232-5.
DOI |
| [36] |
Lan R, Zhang Y, Xiang J, et al. Xiao-Xu-Ming decoction preserves mitochondrial integrity and reduces apoptosis after focal cerebral ischemia and reperfusion via the mitochondrial p53 pathway. J Ethnopharmacol 2014; 151: 307-16.
DOI URL |
| [37] |
Tian T, Sun Y, Wu H, et al. Acupuncture promotes mTOR-independent autophagic clearance of aggregation-prone proteins in mouse brain. Sci Rep 2016; 6: 19714.
DOI PMID |
| [38] |
Fakhoury M. Microglia and astrocytes in Alzheimer’s disease: implications for therapy. curr neuropharmacol 2018; 16: 508-18.
DOI PMID |
| [39] |
Ip WKE, Hoshi N, Shouval DS, Snapper S, Medzhitov R. Anti-inflammatory effect of IL-10 mediated by metabolic reprogramming of macrophages. Science 2017; 356: 513-9.
DOI PMID |
| [40] | Fang EF, Hou Y, Palikaras K, et al. Mitophagy inhibits amyloid-β and tau pathology and reverses cognitive deficits in models of Alzheimer’s disease. Nat Neurosci 2019; 22: 401-12. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

