Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (5): 944-954.DOI: 10.19852/j.cnki.jtcm.20230727.001
Previous Articles Next Articles
Electroacupuncture ameliorates cardiac dysfunction in myocardial ischemia model rats: a potential role of the hypothalamic-pituitary-adrenal axis
WANG Kun1, ZHOU Jie2, CUI Shuai3, WU Xin3, ZHU Guoqi1, WU Shengbing1( ), ZHOU Meiqi4(
), ZHOU Meiqi4( )
)
- 1 Key Laboratory of Xin’an Medicine, Ministry of Education, Anhui University of Chinese Medicine, Hefei 230038, China
2 Department of Acupuncture and Moxibustion, the Third Affiliated Hospital of Zhejiang Chinese Medical University, Hangzhou 310053, China
3 Colleges of Acupuncture and Massage, Anhui University of Chinese Medicine, Hefei 230038, China
4 Bozhou Institute of Chinese Medicine, Anhui Academy of Traditional Chinese Medicine, Bozhou 236800, China
-
Received:2022-06-26Accepted:2022-09-14Online:2023-10-15Published:2023-06-27 -
Contact:Prof. ZHOU Meiqi, Bozhou Institute of Chinese Medicine, Anhui Academy of Traditional Chinese Medicine,meiqizhou@163.com ; Prof. WU Shengbing, Key Laboratory of Xin’an Medicine, Ministry of Education, Anhui University of Chinese Medicine,wsb922@126.com . Telephone: +86-551-65169752 -
Supported by:Natural Science Foundation-funded Project: Exploring the Mechanism of Anti-Ischemic Effect of Acupuncture Based on the Regulation of Hippocampal-HPA Axis by Glutamatergic Neurons(82004462);Opening Project of Zhejiang Provincial Preponderant and Characteristic Subject of Key University (Chinese Traditional Medicine): Study of the Mechanism of Action of Acupuncture in the Treatment of Myocardial Ischemia in Rats Based on the Amygdala Glutamatergic System-Mediated Affective Changes(ZYXYB2019002);National Key Research and Development Program of China: Influence of Heart and Lung Meridians on Heart and Lung Function(SQ2018YFC170298);National Natural Science Foundation of Anhui: Mechanism of Action of Intestinal Flora Involved in the Intervention of Myocardial Ischemia by Acupuncture of the Heart Meridian(1908085MH289)
Cite this article
WANG Kun, ZHOU Jie, CUI Shuai, WU Xin, ZHU Guoqi, WU Shengbing, ZHOU Meiqi. Electroacupuncture ameliorates cardiac dysfunction in myocardial ischemia model rats: a potential role of the hypothalamic-pituitary-adrenal axis[J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 944-954.
share this article
| Group | n | Low shear rates (1/s)(/mPa.s) | Low shear rates (5/s)(/mPa.s) | Middle shear rates (30/s)(/mPa.s) | High shear rates (200/s)(/mPa.s) | Plasma viscosity (/mPa.s) | RBC aggregation index |
|---|---|---|---|---|---|---|---|
| Sham | 6 | 32.86±1.99 | 13.14±0.61 | 6.84±0.23 | 4.79±0.11 | 1.42±0.06 | 6.24±0.33 |
| Model | 6 | 47.41±2.83a | 17.86±1.01c | 8.95±0.50c | 6.03±0.35a | 1.64±0.02c | 7.44±0.15c |
| EA | 6 | 33.14±3.20b | 13.22±1.28d | 7.08±0.57d | 4.89±0.11d | 1.36±0.06b | 6.32±0.35d |
| Control | 6 | 44.41±1.98 | 17.88±0.78 | 9.13±0.28 | 6.32±0.14 | 1.55±0.04 | 7.56±0.05 |
Table 1 Comparison of hemorheology in each group
| Group | n | Low shear rates (1/s)(/mPa.s) | Low shear rates (5/s)(/mPa.s) | Middle shear rates (30/s)(/mPa.s) | High shear rates (200/s)(/mPa.s) | Plasma viscosity (/mPa.s) | RBC aggregation index |
|---|---|---|---|---|---|---|---|
| Sham | 6 | 32.86±1.99 | 13.14±0.61 | 6.84±0.23 | 4.79±0.11 | 1.42±0.06 | 6.24±0.33 |
| Model | 6 | 47.41±2.83a | 17.86±1.01c | 8.95±0.50c | 6.03±0.35a | 1.64±0.02c | 7.44±0.15c |
| EA | 6 | 33.14±3.20b | 13.22±1.28d | 7.08±0.57d | 4.89±0.11d | 1.36±0.06b | 6.32±0.35d |
| Control | 6 | 44.41±1.98 | 17.88±0.78 | 9.13±0.28 | 6.32±0.14 | 1.55±0.04 | 7.56±0.05 |

Figure 1 HR and ECG-ST levels in the study rats A-D: ECGs of each group rats. A: Sham group; B: Model group; C: EA group; D: Control group; E: HR in the Sham, Model, EA and Control groups; F: ECG-ST level in the Sham, Model, EA and Control groups. Sham: performed with sham surgery without ligation; Model: received surgery to ligate the LAD; EA: model rats with EA at Shenmen (HT7)-Tongli (HT5) segment; Control: model rats with EA at points lying 5 and 10 mm from the base of the tail. ECG: electrocardiogram; EA: electroacupunture; HR: heart rate; ST: ST-segment; LAD: left anterior descending artery. aP < 0.01, compared with the Sham group; bP < 0.01, compared with the Model group; Data are presented as mean ± standard error of mean (n = 6). Analysis of variance was performed to compare the differences among the groups.

Figure 2 Histopathological comparison of myocardial tissue in each group (HE staining method, × 200) A-D: representative images of HE staining of myocardial tissue to identify pathological damage. A: Sham group; B: Model group; C: EA group; D: Control group; E: GPBB levels in the Sham, Model, EA and Control groups; F: H-FABP levels in the Sham, Model, EA and Control groups. Sham: performed with sham surgery without ligation; Model: received surgery to ligate the LAD; EA: model rats with EA at Shenmen (HT7)-Tongli (HT5) segment; Control: model rats with EA at points lying 5 and 10 mm from the base of the tail. HE: hematoxylin-eosin; EA: electroacupunture; GPBB: recombinant human glycogen phosphorylase; H-FABP: heart-type fatty acid binding protein; LAD: left anterior descending artery. Data are presented as mean± standard error of mean (n = 6). Analysis of variance was performed to compare the differences among the groups. aP < 0.01, compared with the Sham group; bP < 0.05, compared with the Model group; cP < 0.01, compared with the Model group.
| Group | n | ACTH level (pg/mL) | CORT level (ng/mL) |
|---|---|---|---|
| Sham | 6 | 43.56± 1.39 | 10.01± 0.63 |
| Model | 6 | 71.99± 2.96a | 15.9± 0.80a |
| EA | 6 | 49.22± 2.38b | 10.7± 0.22b |
| Control | 6 | 67.73± 0.92 | 15.1± 0.31 |
Table 2 Levels of ACTH and CORT in the study rats
| Group | n | ACTH level (pg/mL) | CORT level (ng/mL) |
|---|---|---|---|
| Sham | 6 | 43.56± 1.39 | 10.01± 0.63 |
| Model | 6 | 71.99± 2.96a | 15.9± 0.80a |
| EA | 6 | 49.22± 2.38b | 10.7± 0.22b |
| Control | 6 | 67.73± 0.92 | 15.1± 0.31 |

Figure 3 Expression of Vim and GFAP in the hippocampus in the study rats (immunofluorescence staining, × 400) A: representative Western blots of Vim and GFAP; B: Vimentin/β-actin quantification; C: GFAP/β-actin quantification of the blots in the Sham, Model, EA and Control groups; D: representative of immunofluorescence images of GFAP in the hippocampus. D1, D2, D3: Sham group; D4, D5, D6: Model group; D7, D8, D9: EA group; D10, D11, D12: Control group; D1, D4, D7, D10: merged images of GFAP and DAPI; D2, D5, D8, D11: expressed GFAP in the hippocampus; D3, D6, D9, D12: expressed DAPI in the hippocampus. Sham: performed with sham surgery without ligation; Model: received surgery to ligate the LAD; EA: model rats with EA at Shenmen (HT7)-Tongli (HT5) segment; Control: model rats with EA at points lying 5 and 10 mm from the base of the tail. EA: electroacupunture; Vim: vimentin; GFAP: glial fibrillary acidic protein; DAPI: 4',6-diamidino-2-phenylindole; LAD: left anterior descending artery. Data are presented as mean ± standard error of mean (n = 3). Analysis of variance was performed to compare the differences among the groups. aP < 0.01, compared with the Sham group; bP < 0.01, compared with the Model group.
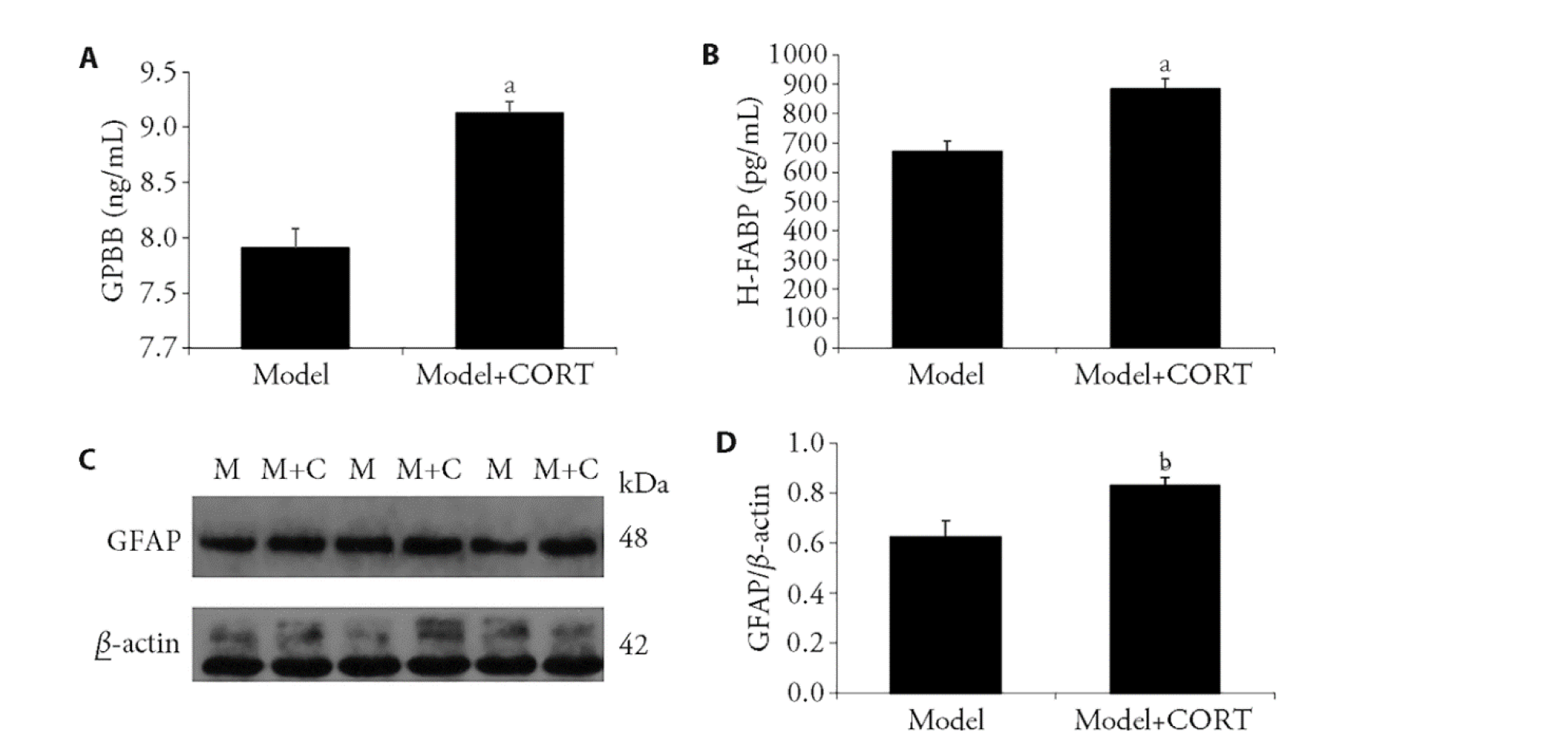
Figure 4 Intraperitoneal injection of CORT aggravated the expression of GPBB, H-FABP and GFAP A: the levels of GPBB in Model and Model + CORT rats (n = 6); B: the levels of H-FABP in Model and Model + CORT rats (n = 6); C: Western blotting of GFAP in the Model and Model+CORT groups showing expression of GFAP (n = 3); D: GFAP/β-actin quantification of the blots in the Model and Model + CORT groups (n = 3). Model: received surgery to ligate the LAD; Model + CORT: injected with CORT at a dose of 20 mg/kg in a saline vehicle. GPBB: recombinant human glycogen phosphorylase; H-FABP: heart-type fatty acid binding protein; CORT: corticosterone; GFAP: glial fibrillary acidic protein; LAD: left anterior descending artery. Data are presented as mean ± standard error of mean. Analysis of variance was performed to compare the differences among the groups. aP < 0.01, compared with the Model group; bP < 0.05, compared with the Model group.
| 1. |
DeFilippis AP, Chapman AR, Mills NL, et al. Assessment and treatment of patients with type 2 myocardial infarction and acute nonischemic myocardial injury. Circulation 2019; 140: 1661-78.
DOI PMID |
| 2. |
Silvis MJM, Demkes EJ, Fiolet ATL, et al. Immunomodulation of the NLRP3 inflammasome in atherosclerosis, coronary artery disease, and acute myocardial infarction. J Cardiovasc Transl Res 2021; 14: 23-34.
DOI |
| 3. |
Wang Y, Chuo WJ, Li C, et al. Energy metabolism disorder and myocardial injury in chronic myocardial ischemia with Qi deficiency and blood stasis syndrome based on 2-DE proteomics. Chin J Integr Med 2013; 19: 616-20.
DOI URL |
| 4. | Fu Y, Li J, Wu S, Wang H. Electroacupuncture pretreatment promotes angiogenesis via hypoxia-inducible factor 1α and vascular endothelial growth factor in a rat model of chronic myocardial ischemia. Zhen Ci Yan Jiu 2021; 39: 367-75. |
| 5. | Han Y, Chen S, Wang H, Peng XM. Electroacupuncture pretreatment regulates apoptosis of myocardial ischemia-reperfusion injury in rats through RhoA/p38MAPK pathway mediated by miR-133a-5p. Evid Based Complement Alternat Med 2021; 2021: 8827891. |
| 6. | Cui S, Xu J, Wang J, Wu SB, Zhou YP, Zhou MQ. Effect of electroacupuncture stimulation of heart meridian on autonomic nervous activities in acute myocardial ischemia rats. Zhen Ci Yan Jiu 2016; 41: 515-20. |
| 7. |
Meloux A, Rigal E, Rochette L, Cottin Y, Bejot Y, Vergely C. Ischemic stroke increases heart vulnerability to ischemia-reperfusion and alters myocardial cardioprotective pathways. Stroke 2018; 49: 2752-60.
DOI PMID |
| 8. |
Infanger DW, Cao X, Butler SD, et al. Silencing nox4 in the paraventricular nucleus improves myocardial infarction-induced cardiac dysfunction by attenuating sympathoexcitation and periinfarct apoptosis. Circ Res 2010; 106: 1763-74.
DOI PMID |
| 9. |
O'Leary OF, Ogbonnaya ES, Felice D, et al. The vagus nerve modulates BDNF expression and neurogenesis in the hippocampus. Eur Neuropsychopharmacol 2018; 28: 307-16.
DOI PMID |
| 10. | Bellavance MA, Rivest S. The HPA-Immune axis and the immunomodulatory actions of glucocorticoids in the brain. Front Immunol 2014; 5: 136. |
| 11. |
Lowry CA. Functional subsets of serotonergic neurones: implications for control of the hypothalamic-pituitary-adrenal axis. J Neuroendocrinol 2002; 14: 911-23.
PMID |
| 12. |
Battaglini D, Robba C, Lopes da Silva A, et al. Brain-heart interaction after acute ischemic stroke. Crit Care 2020; 24: 163.
DOI |
| 13. |
Sjörs Dahlman A, Jonsdottir IH, Hansson C. The hypothalamo-pituitary-adrenal axis and the autonomic nervous system in burnout. Handb Clin Neurol 2021; 182: 83-94.
DOI PMID |
| 14. |
Trifunovic S, Stevanovic I, Milosevic A, et al. The function of the hypothalamic-pituitary-adrenal axis during experimental autoimmune encephalomyelitis: involvement of oxidative stress mediators. Front Neurosci 2021; 15: 649485.
DOI URL |
| 15. |
Portaluppi F. The circadian organization of the cardiovascular system in health and disease. Indian J Exp Biol 2014; 52: 395-8.
PMID |
| 16. |
Bernardi J, Aromolaran KA, Aromolaran AS. Neurological disorders and risk of arrhythmia. Int J Mol Sci 2020; 22: 188.
DOI URL |
| 17. |
Tan X, Pan Y, Su W, et al. Acupuncture therapy for essential hypertension: a network Meta-analysis. Ann Transl Med 2019; 7: 266.
DOI PMID |
| 18. | Robinson N, Ye T, Ronan P, Garbelli PE, Smithard D. Acupuncture for stroke: perceptions and possibilities. Zhen Ci Yan Jiu 2020; 38: 158-68. |
| 19. |
Yang Q, Mao H, Chen X, et al.Neiguan (PC6)-based acupuncture pretreatment for myocardial ischemia reperfusion injury: a protocol for preclinical systematic review and Meta-analysis. Medicine (Baltimore) 2020; 99: e20792.
DOI URL |
| 20. |
Li H, Wu C, Yan C, et al. Cardioprotective effect of transcutaneous electrical acupuncture point stimulation on perioperative elderly patients with coronary heart disease: a prospective, randomized, controlled clinical trial. Clin Interv Aging 2019; 14: 1607-14.
DOI PMID |
| 21. | Huang H, Zhong Z, Chen J, et al. Effect of acupuncture at HT7 on heart rate variability: an exploratory study. Zhen Ci Yan Jiu 2015; 33: 30-5. |
| 22. |
Wang N, Lu SF, Chen H, et al. A protocol of histone modification-based mechanistic study of acupuncture in patients with stable angina pectoris. BMC Complement Altern Med 2015; 15: 139.
DOI URL |
| 23. |
Zhang HR, Tao JL, Bai H, et al. Changes in the serum metabolome of acute myocardial ischemia rat pretreatment with electroacupuncture. Am J Chin Med 2019; 47: 1025-41.
DOI URL |
| 24. |
Zhang T, Yang WX, Wang YL, et al. Electroacupuncture preconditioning attenuates acute myocardial ischemia injury through inhibiting NLRP 3 inflammasome activation in mice. Life Sci 2020; 248: 117451.
DOI URL |
| 25. |
Cui S, Wang K, Wu SB, et al. Electroacupuncture modulates the activity of the hippocampus-nucleus tractus solitarius-vagus nerve pathway to reduce myocardial ischemic injury. Neural Regen Res 2018; 13: 1609-18.
DOI PMID |
| 26. | Cui S, Zhou Y, Wu S, Cao J, Zhu G, Zhou M. Electroacupuncture improved the function of myocardial ischemia involved in the hippocampus-paraventricular nucleus-sympathetic nerve pathway. Evid Based Complement Alternat Med 2018; 2018: 2870676. |
| 27. | Yuan J, Wang JM, Cai Y, Fu SP, Zhu BM, Lu SF. Effects of electroacupuncture on sympathetic nerve-related substance in myocardial tissue in mice with myocardial ischemia. Zhong Guo Zhen Jiu 2019; 39: 501-6. |
| 28. | Wang K, Wu SB, Cui S, Xiang SY, Wu X, Zhou MQ. Effect of electroacupuncture on hippocampal IL-6, IL-1 β, TNF-α and norepinephrine levels in acute myocardial ischemia rats. Zhen Ci Yan Jiu 2018; 43: 365-9. |
| 29. | Wu S, Cao J, Zhang T, et al. Electroacupuncture ameliorates the coronary occlusion related tachycardia and hypotension in acute rat myocardial ischemia model: potential role of hippocampus. Evid Based Complement Alternat Med 2015; 2015: 925987. |
| 30. |
Wang XP, Wang PF, Bai JQ, et al. Investigating the effects and possible mechanisms of Danshen-Honghua herb pair on acute myocardial ischemia induced by isoproterenol in rats. Biomed Pharmacother 2019; 118: 109268.
DOI URL |
| 31. |
Popovic B, Agrinier N, Bouchahda N, et al. Coronary embolism among ST-Segment-elevation myocardial infarction patients: mechanisms and management. Circ Cardiovasc Interv 2018; 11: e005587.
DOI URL |
| 32. |
Baskurt OK, Meiselman HJ. Blood rheology and hemodynamics. Semin Thromb Hemost 2003; 29: 435-50.
DOI PMID |
| 33. |
Javadi E, Jamali S. Hemorheology: the critical role of flow type in blood viscosity measurements. Soft Matter 2021; 17: 8446-58.
DOI PMID |
| 34. |
Okahara S, Zu S, Takahashi S, Sueda T, Tsuji T. Blood viscosity monitoring during cardiopulmonary bypass based on pressure-flow characteristics of a Newtonian fluid. Annu Int Conf IEEE Eng Med Biol Soc 2016; 2016: 2331-4.
DOI PMID |
| 35. |
Kim Y, Kim H, Kim SY, et al. Automated heart-type fatty acid-binding protein assay for the early diagnosis of acute myocardial infarction. Am J Clin Pathol 2010; 134: 157-62.
DOI PMID |
| 36. |
Dobric M, Ostojic M, Giga V, et al. Glycogen phosphorylase BB in myocardial infarction. Clin Chim Acta 2015; 438: 107-11.
DOI PMID |
| 37. |
Zhong Y, Wang N. Erratum to "Traditional acupuncture and myocardial ischemia". Int J Cardiol 2017; 235: 208.
DOI |
| 38. | Zeng Q, He H, Wang XB, et al. Electroacupuncture preconditioning improves myocardial infarction injury via enhancing AMPK-dependent autophagy in rats. Biomed Res Int 2018; 2018: 1238175. |
| 39. |
Micale V, Drago F. Endocannabinoid system, stress and HPA axis. Eur J Pharmacol 2018; 834: 230-9.
DOI PMID |
| 40. |
Speer KE, Semple S, Naumovski N, D'Cunha NM, McKune AJ. HPA axis function and diurnal cortisol in post-traumatic stress disorder: a systematic review. Neurobiol Stress 2019; 11: 100180.
DOI URL |
| 41. | Zhang ZJ, Yao JP, Guo ZY, Li D. Advances in study of icarrin for anti-aging and anti-depression: a review. Zhong Guo Shi Yan Fang Ji Xue Za Zhi 2022; 28: 276-282. |
| 42. |
Headrick JP, Peart JN, Budiono BP, Shum DHK, Neumann DL, Stapelberg NJC. The heartbreak of depression: 'Psycho-cardiac' coupling in myocardial infarction. J Mol Cell Cardiol 2017; 106: 14-28.
DOI PMID |
| 43. |
Goico A, Chandrasekaran M, Herrera CJ. Novel developments in stress cardiomyopathy: from pathophysiology to prognosis. Int J Cardiol 2016; 223: 1053-8.
DOI PMID |
| 44. |
Smith GD, Ben-Shlomo Y, Beswick A, Yarnell J, Lightman S, Elwood P. Cortisol, testosterone, and coronary heart disease: prospective evidence from the Caerphilly study. Circulation 2005; 112: 332-40.
DOI PMID |
| 45. |
Zhu LJ, Liu MY, Li H, et al. The different roles of glucocorticoids in the hippocampus and hypothalamus in chronic stress-induced HPA axis hyperactivity. PLoS One 2014; 9: e97689.
DOI URL |
| 46. |
Azuma K, Zhou Q, Niwa M, Kubo KY. Association between mastication, the hippocampus, and the HPA Axis: a comprehensive review. Int J Mol Sci 2017; 18 : 1687.
DOI URL |
| 47. |
Snyder JS, Soumier A, Brewer M, Pickel J, Cameron HA. Adult hippocampal neurogenesis buffers stress responses and depressive behaviour. Nature 2011; 476: 458-61.
DOI |
| 48. |
Herman JP, McKlveen JM, Ghosal S, et al. Regulation of the hypothalamic-pituitary-adrenocortical stress response. Compr Physiol 2016; 6: 603-21.
DOI PMID |
| 49. |
Chen Z, Venkat P, Seyfried D, Chopp M, Yan T, Chen J. Brain-Heart interaction: cardiac complications after stroke. Circ Res 2017; 121: 451-68.
DOI PMID |
| 50. |
Iob E, Kirschbaum C, Steptoe A. Persistent depressive symptoms, HPA-axis hyperactivity, and inflammation: the role of cognitive-affective and somatic symptoms. Mol Psychiatry 2020; 25: 1130-40.
DOI |
| 51. |
Goshen I, Yirmiya R. Interleukin-1 (IL-1): a central regulator of stress responses. Front Neuroendocrinol 2009; 30: 30-45.
DOI URL |
| 52. |
Qiu J, Wang M, Zhang J, et al. The neuroprotection of sinomenine against ischemic stroke in mice by suppressing NLRP3 inflammasome via AMPK signaling. Int Immunopharmacol 2016; 40: 492-500.
DOI URL |
| 53. |
Silverman MN, Sternberg EM. Glucocorticoid regulation of inflammation and its functional correlates: from HPA axis to glucocorticoid receptor dysfunction. Ann NY Acad Sci 2012; 1261: 55-63.
DOI PMID |
| 54. |
Girotti M, Donegan JJ, Morilak DA. Influence of hypothalamic IL-6/gp130 receptor signaling on the HPA axis response to chronic stress. Psychoneuroendocrinology 2013; 38: 1158-69.
DOI PMID |
| 55. | Himmerich H, Binder EB, Künzel HE, et al. Successful antidepressant therapy restores the disturbed interplay between TNF-alpha system and HPA axis. Biol Psychiatry 2006; 60: 882-8. |
| 56. |
Gądek-Michalska A, Tadeusz J, Rachwalska P, Bugajski J. Cytokines, prostaglandins and nitric oxide in the regulation of stress-response systems. Pharmacol Rep 2013; 65: 1655-62.
DOI PMID |
| 57. |
Shi Y, Guo X, Zhang J, Zhou H, Sun B, Feng J. DNA binding protein HMGB1 secreted by activated microglia promotes the apoptosis of hippocampal neurons in diabetes complicated with OSA. Brain Behav Immun 2018; 73: 482-92.
DOI PMID |
| 58. |
Pekny M, Wilhelmsson U, Bogestal YR, Pekna M. The role of astrocytes and complement system in neural plasticity. Int Rev Neurobiol 2007; 82: 95-111.
PMID |
| 59. |
Iglesias J, Morales L, Barreto GE. Metabolic and inflammatory adaptation of reactive astrocytes: role of PPARs. Mol Neurobiol 2017; 54: 2518-38.
DOI PMID |
| 60. |
Liddelow SA, Guttenplan KA, Clarke LE, et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 2017; 541: 481-7.
DOI |
| 61. |
Norden DM, Trojanowski PJ, Villanueva E, Navarro E, Godbout JP. Sequential activation of microglia and astrocyte cytokine expression precedes increased Iba-1 or GFAP immunoreactivity following systemic immune challenge. Glia 2016; 64: 300-16.
DOI PMID |
| 62. |
Eng LF, Ghirnikar RS, Lee YL. Glial fibrillary acidic protein: GFAP-thirty-one years (1969-2000). Neurochem Res 2000; 25: 1439-51.
DOI PMID |
| 63. |
Wilhelmsson U, Pozo-Rodrigalvarez A, Kalm M, et al. The role of GFAP and vimentin in learning and memory. Biol Chem 2019; 400: 1147-56.
DOI PMID |
| 64. |
Kamphuis W, Kooijman L, Orre M, Stassen O, Pekny M, Hol EM. GFAP and vimentin deficiency alters gene expression in astrocytes and microglia in wild-type mice and changes the transcriptional response of reactive glia in mouse model for Alzheimer's disease. Glia 2015; 63: 1036-56.
DOI PMID |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

