Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (3): 582-587.DOI: 10.19852/j.cnki.jtcm.20230208.001
Previous Articles Next Articles
Effectiveness of Traditional Chinese Medicine on coronavirus disease 2019 in 92 patients: a retrospective study
XU Guihua1, CHEN Feifei1, ZHANG Wei1( ), WU Yingen2, CHEN Xiaorong3, SHI Kehua4, WANG Zhenwei5, SHI Miaoyan1, ZHANG Xing1, LU Yunfei3, YUAN Weian1, LYU Hua1, CHEN Xuan1(
), WU Yingen2, CHEN Xiaorong3, SHI Kehua4, WANG Zhenwei5, SHI Miaoyan1, ZHANG Xing1, LU Yunfei3, YUAN Weian1, LYU Hua1, CHEN Xuan1( )
)
- 1 Shuguang Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China
2 Longhua Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai 200032, China
3 Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, China
4 Shanghai Hospital of Traditional Chinese Medicine, Shanghai 200071, China
5 Yueyang Hospital of Integrated Traditional Chinese and Western Medicine, Shanghai 200437, China
-
Received:2022-01-21Accepted:2022-04-10Online:2023-06-15Published:2023-02-08 -
Contact:CHEN Xuan, Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China. chen77xuan@aliyun.com. Telephone: +86-21-20256375
ZHANG Wei, Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China. zhangw1190@sina.com -
Supported by:Shanghai Science and Technology Committee, Clinical Study on the Treatment of COVID-19 with Traditional Chinese Medicine(2041195020002);Shanghai Infectious Disease Prevention and Treatment Ability Cultivation Project of Traditional Chinese Medicine(ZYYB-NLPY-09);Shanghai Infectious Disease Prevention and Treatment Ability Cultivation Project of Traditional Chinese Medicine(ZYYB-NLPY-15);Shanghai Key Clinical Specialty(shslczdzk05101);Shanghai Key Clinical Laboratory of Traditional Chinese Medicine(20dz2272200);Shanghai Health Commission, Shanghai Chinese Medicine Administration Emergency Scientific Research Project, Clinical Study on Prevention and Treatment of New Coronavirus Pneumonia with Integrated Chinese And Western Medicine(2020YJ01)
Cite this article
XU Guihua, CHEN Feifei, ZHANG Wei, WU Yingen, CHEN Xiaorong, SHI Kehua, WANG Zhenwei, SHI Miaoyan, ZHANG Xing, LU Yunfei, YUAN Weian, LYU Hua, CHEN Xuan. Effectiveness of Traditional Chinese Medicine on coronavirus disease 2019 in 92 patients: a retrospective study[J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 582-587.
share this article
| Baseline characteristic | Treatment group I (n = 45) | Treatment group Ⅱ (n = 47) | P value |
|---|---|---|---|
| Male [n (%)] | 27 (60.00) | 21 (44.68) | 0.141 |
| Age (years, mean ± SD) | 48±16 | 54±15 | 0.618 |
| Time interval between onset to admission [days, M (Q1, Q3)] | 5.00 (3.00, 6.00) | 4.00 (3.00, 6.00) | 0.355 |
| Armpit temperature [℃, M (Q1, Q3)] | 38.00 (37.60, 38.45) | 38.20 (37.80, 38.70) | 0.190 |
| Use of antibiotics [n (%)] | 20 (44.44) | 30 (63.83) | 0.062 |
| Use of hormones [n (%)] | 12 (26.67) | 21 (44.68) | 0.072 |
| Underlying diseases [n (%)] | 12 (26.67) | 17 (36.17) | 0.327 |
| Severity of the disease | |||
| Mild [n (%)] | 2 (4.44) | 0 (0.00) | 0.699 |
| Moderate [n (%)] | 41 (91.11) | 42 (89.36) | |
| Severe [n (%)] | 2 (4.44) | 3 (6.38) | |
| Critical [n (%)] | 0 (0.00) | 2 (4.26) |
Table 1 Clinical characteristics of 92 COVID-19 patients
| Baseline characteristic | Treatment group I (n = 45) | Treatment group Ⅱ (n = 47) | P value |
|---|---|---|---|
| Male [n (%)] | 27 (60.00) | 21 (44.68) | 0.141 |
| Age (years, mean ± SD) | 48±16 | 54±15 | 0.618 |
| Time interval between onset to admission [days, M (Q1, Q3)] | 5.00 (3.00, 6.00) | 4.00 (3.00, 6.00) | 0.355 |
| Armpit temperature [℃, M (Q1, Q3)] | 38.00 (37.60, 38.45) | 38.20 (37.80, 38.70) | 0.190 |
| Use of antibiotics [n (%)] | 20 (44.44) | 30 (63.83) | 0.062 |
| Use of hormones [n (%)] | 12 (26.67) | 21 (44.68) | 0.072 |
| Underlying diseases [n (%)] | 12 (26.67) | 17 (36.17) | 0.327 |
| Severity of the disease | |||
| Mild [n (%)] | 2 (4.44) | 0 (0.00) | 0.699 |
| Moderate [n (%)] | 41 (91.11) | 42 (89.36) | |
| Severe [n (%)] | 2 (4.44) | 3 (6.38) | |
| Critical [n (%)] | 0 (0.00) | 2 (4.26) |
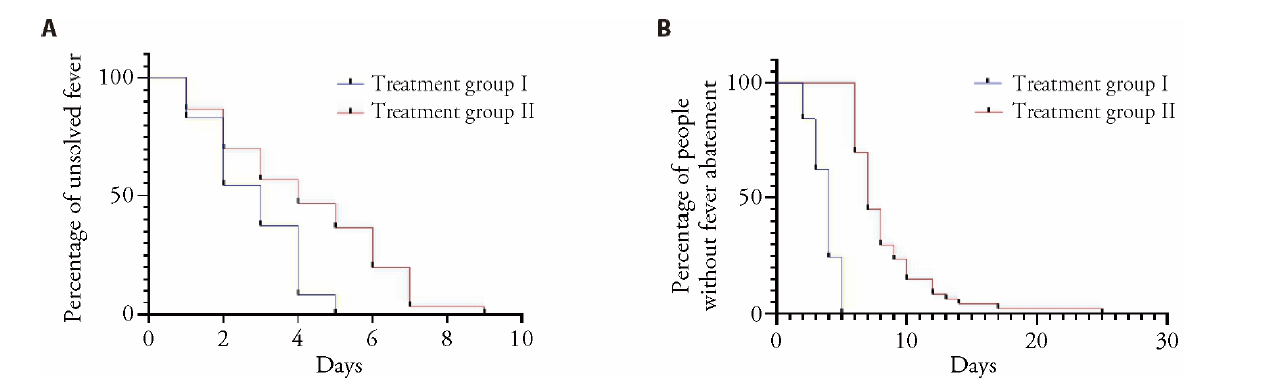
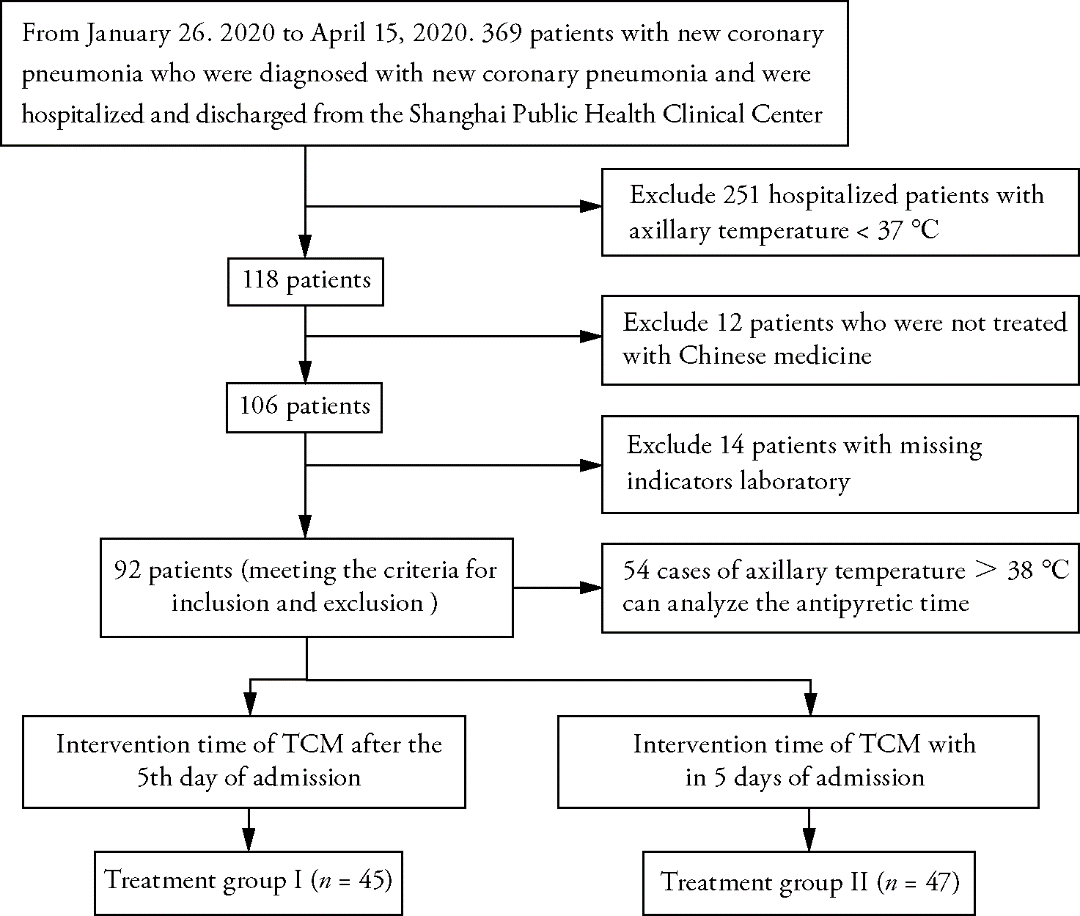
Figure 2 Comparison of body temperature between two groups A: comparison of antipyretic time between the two groups (Kaplan Meier survival curve); B: comparison of fever duration between the two groups (Kaplan Meier survival curve).
| Item | Time | Treatment group I (n = 45) | Treatment group Ⅱ (n = 47) | P value |
|---|---|---|---|---|
| WBC (×109/L) | Admission day | 4.32 (3.74, 5.12) | 4.28 (3.33, 5.66) | 0.542 |
| 3rd day | 4.63 (4.01, 5.88) | 4.01 (3.07, 6.03) | 0.052 | |
| 6th day | 4.87 (4.18, 6.17) | 4.86 (3.72, 7.16) | 0.873 | |
| Discharged day | 5.60 (4.70, 6.40)a | 5.58 (4.05, 6.62)a | 0.755 | |
| LYMPH (×109/L) | Admission day | 0.96 (0.78, 1.40) | 0.85 (0.67, 1.26) | 0.108 |
| 3rd day | 1.28 (0.88, 1.58) | 1.03 (0.76, 1.33)b | 0.022 | |
| 6th day | 1.55 (1.15, 1.96) | 1.06 (0.78, 1.39)b | <0.001 | |
| Discharged day | 1.74 (1.49, 2.18)a | 1.52 (1.21, 1.97)ab | 0.036 | |
| EOS (×109/L) | Admission day | 0.00 (0.00, 0.01) | 0.00 (0.00, 0.01) | 0.483 |
| 3rd day | 0.01 (0.00, 0.03)b | 0.00 (0.00, 0.01)b | 0.004 | |
| 6th day | 0.04 (0.01, 0.08)b | 0.02 (0.00, 0.06)b | 0.016 | |
| Discharged day | 0.08 (0.06, 0.13)a | 0.09 (0.06, 0.14)a | 0.591 | |
| NLR | Admission day | 2.89 (1.73, 3.80) | 3.11 (2.20, 3.91) | 0.341 |
| 3rd day | 2.53 (1.75, 3.52) | 2.71 (1.82, 4.14) | 0.426 | |
| 6th day | 1.77 (1.28, 2.41) b | 2.98 (1.99, 5.89)b | <0.001 | |
| Discharged day | 1.84 (1.49, 2.33)a | 2.04 (1.53, 2.82)a | 0.085 |
Table 2 Comparison of inflammatory indexes between the two groups
| Item | Time | Treatment group I (n = 45) | Treatment group Ⅱ (n = 47) | P value |
|---|---|---|---|---|
| WBC (×109/L) | Admission day | 4.32 (3.74, 5.12) | 4.28 (3.33, 5.66) | 0.542 |
| 3rd day | 4.63 (4.01, 5.88) | 4.01 (3.07, 6.03) | 0.052 | |
| 6th day | 4.87 (4.18, 6.17) | 4.86 (3.72, 7.16) | 0.873 | |
| Discharged day | 5.60 (4.70, 6.40)a | 5.58 (4.05, 6.62)a | 0.755 | |
| LYMPH (×109/L) | Admission day | 0.96 (0.78, 1.40) | 0.85 (0.67, 1.26) | 0.108 |
| 3rd day | 1.28 (0.88, 1.58) | 1.03 (0.76, 1.33)b | 0.022 | |
| 6th day | 1.55 (1.15, 1.96) | 1.06 (0.78, 1.39)b | <0.001 | |
| Discharged day | 1.74 (1.49, 2.18)a | 1.52 (1.21, 1.97)ab | 0.036 | |
| EOS (×109/L) | Admission day | 0.00 (0.00, 0.01) | 0.00 (0.00, 0.01) | 0.483 |
| 3rd day | 0.01 (0.00, 0.03)b | 0.00 (0.00, 0.01)b | 0.004 | |
| 6th day | 0.04 (0.01, 0.08)b | 0.02 (0.00, 0.06)b | 0.016 | |
| Discharged day | 0.08 (0.06, 0.13)a | 0.09 (0.06, 0.14)a | 0.591 | |
| NLR | Admission day | 2.89 (1.73, 3.80) | 3.11 (2.20, 3.91) | 0.341 |
| 3rd day | 2.53 (1.75, 3.52) | 2.71 (1.82, 4.14) | 0.426 | |
| 6th day | 1.77 (1.28, 2.41) b | 2.98 (1.99, 5.89)b | <0.001 | |
| Discharged day | 1.84 (1.49, 2.33)a | 2.04 (1.53, 2.82)a | 0.085 |
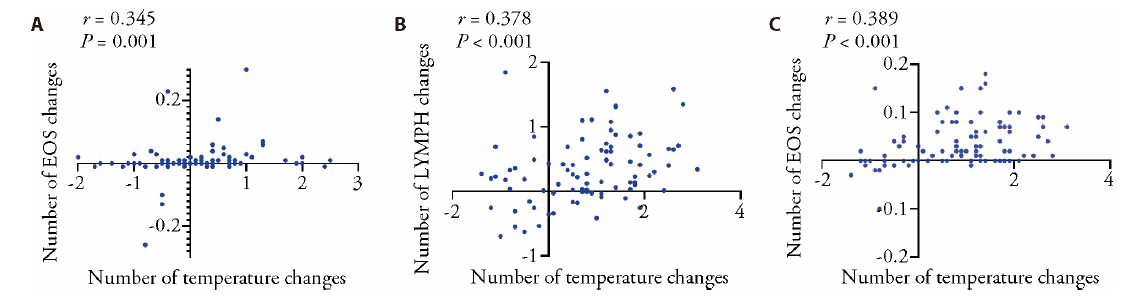
Figure 3 Comparison of laboratory indicators A: correlation between temperature difference and eosinophil value on the third day after admission; B: correlation between temperature difference and lymphocyte changes on the sixth day after admission; C: correlation between temperature difference and eosinophil changes on the sixth day after admission.
| [1] | Health emergency office of National Health Commission. Update on New Coronavirus pneumonia at 24 January 29th, 2022-1-30, cited 2022-03-10. Available from URL: http://www.nhc.gov.cn/xcs/yqtb/202201/a19ffdd25c32479893822addee618669.shtml. |
| [2] | National Health Commission of the People’s Republic of China. Protocol for Diagnosis and Treatment of 2019 Novel Coronavirus Pneumonia (trial 8th edition), 2020-08-18, cited 2022-03-10. Available from URL: http://www.nhc.gov.cn/yzygj/s7653p/202008/0a7bdf12bd4b46e5bd28ca7f9a7f5e5a/files/a449a3e2e2c94d9a856d5faea2ff0f94.pdf. |
| [3] |
Wang ZW, Chen XR, Lu YF, Chen FF, Zhang W. Clinical characteristics and therapeutic procedure for four cases with 2019 novel coronavirus pneumonia receiving combined Chinese and Western medicine treatment. Biosci Trends 2020; 14: 64-8.
DOI PMID |
| [4] | Zhang W, Wu YG, Zhang HY, Chen W, Shi KH, Wang ZW. Interpretation of TCM diagnosis and treatment plan for pneumonia infected by New Coronavirus in Shanghai city (trial edition). Shanghai Zhong Yi Yao Za Zhi 2020; 54: 1-4. |
| [5] | Pan F. China's deep involvement novel coronavirus pneumonia diagnosis and treatment process: interview with Professor Zhang Boli, member of the Chinese Academy of engineering and the central steering group expert group. Zhong Guo Yi Yao Dao Bao 2020; 17: 1-3. |
| [6] | Protocol for Diagnosis and Treatment of 2019 Novel Coronavirus Pneumonia (trial 4th edition), 2020-01-27, cited 2022-03-10, Available from URL: http://www.nhc.gov.cn/yzygj/s7653p/202001/4294563ed35b43209b31739bd0785e67.shtml. |
| [7] |
Chen Z, Nakamura T. Statistical evidence for the usefulness of Chinese medicine in the treatment of SARS. Phytother Res 2004; 18: 592-94.
PMID |
| [8] | Huang Di Nei Jing Su Wen. Beijing: People's Health Publishing House, 1963: 183-98. |
| [9] | Chen G, Yang HJ, Zhang Y, Dong S, Zhang BR, Zhai SQ. TCM innovation summarized from development of TCM fever theory. Shi Jie Zhong Yi Yao 2015; 10: 1250-3. |
| [10] | Liu N. Professor Liu Jingyuan’s experience in exogenous fever based on pattern differentiation. Xian Dai Zhong Yi Lin Chuang 2018; 25: 31-5. |
| [11] | Wang YJ, Wang L, Guo TT, Sun LP. Professor Wang Lie's academic thought on treating children's exogenous fever from "heat toxin". Zhong Guo She Qu Yi Shi 2021; 37: 66-8. |
| [12] | Han QR, Zhang YK, Liu MY. Research progress on antipyretic mechanism of Traditional Chinese Medicine. Zhong Yi Xue Bao 2013; 28: 1018-20. |
| [13] | Ma LL, Liu HM, Luo CH, et al. Fever and antipyretic supported by Traditional Chinese Medicine: a multi-pathway regulation. Front Pharmacol 2021; 12: 583279. |
| [14] |
Zhang W, Du RH, Li B, et al. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg Microbes Infect 2020; 9: 386-9.
DOI URL |
| [15] |
Xu X, Yu MQ, Shen Q, et al. Analysis of inflammatory parameters and disease severity for 88 hospitalized COVID-19 patients in Wuhan China. Int J Med Sci 2020; 17: 2052-62.
DOI URL |
| [16] |
Xie GG, Ding FM, Han L, Yin DN, Lu HZ, Zhang L. The role of peripheral blood eosinophil counts in COVID-19 patients. Allergy 2020; 76: 1-12.
DOI URL |
| [17] | Yang AP, Liu J, Tao W, Tao WQ, Li HM. The diagnostic and predictive role of NLR, d-NLR and PLR in COVID-19 patients. Int Immunopharmacol 2020; 84: 106504. |
| [18] | Nalbant A, Kaya T, Varim C, Selçuk Y, Tamer A, Cinemre H. Can the neutrophil/lymphocyte ratio (NLR) have a role in the diagnosis of coronavirus 2019 disease (COVID-19)? Revista da Associao Médica Brasileira 2020; 66: 746-51. |
| [19] |
Liu Y, Du X, Chen J, et al. Neutrophil-to-lymphocyte ratio as an independent risk factor for mortality in hospitalized patients with COVID-19. J Infect 2020; 81: e6-12.
DOI PMID |
| [1] | FAN Rong, HE Haoyu, TANG Tao, CUI Hanjin. Long-term effects of Qingfei Paidu decoction (清肺排毒汤) in patients with coronavirus disease 2019 acute pneumonia after treatment: a protocol for systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(6): 1068-1071. |
| [2] | SUN Wu, ZHAO Yuwei, LIAO Liang, ZHAO Zhonghui, CHEN Shiqi, YAN Xiaoling, WANG Xueyao, CHAO Guojun, ZHOU Jian. Effectiveness and safety of Xuebijing injection for patients with coronavirus disease 2019: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 631-639. |
| [3] | ZHANG Yuehong, SHAO Xianzhi, ZHAO Qianlong, ZHAN Hualong, ZHANG Jianhua, DU Sisi, CHEN Jing, LIU Yingfang, ZHOU Haiwang, CHEN Xinsheng, HONG Ying, LIAN Fengmei, TONG Xiaolin, BA Yuanming. Effectiveness of Xiangsha Liujun pills (香砂六君丸) on decreased digestive function in convalescent patients of coronavirus disease 2019: a randomized, double blind, placebo controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 552-558. |
| [4] | YANG Cunqing, LIAN Fengmei, YANG Guiping, HUANG Yufeng, ZHANG Shuangbin, WANG Jianghua, ZHOU Jing, GUO Dongqing, SHEN Chuanyun, YE Tiansong, FU Aojie, LI Xiaoli, CHEN Le, ZHANG Huifeng, TU Qiyin, WANG Ying, YANG Wenzhe, TONG Xiaolin, BA Yuanming. Effectiveness of Xiaoyao capsule (逍遥丸) on sleep disorders and mood disturbance in patients in recovery from coronavirus disease 2019: a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(2): 343-351. |
| [5] | ZHANG Yuehong, DONG Dandan, YAN Youqin, ZHANG Hao, WANG Guangli, ZHOU Wei, LI Wei, QIU Li, LI Tingming, LIU Quan, XIA Ping, MAO Lina, YANG Danlin, YANG Lu, LIAN Fengmei, TONG Xiaolin, BA Yuanming. Effectiveness and safety of Jinshuibao capsules (金水宝胶囊) in treatment of residual cardiopulmonary symptoms in convalescent patients of coronavirus disease 2019: a pilot randomized, double-blind, placebo-controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 134-139. |
| [6] | AN Xuedong, MAO Lina, XIA Ping, SU Wen, WANG Beibei, KOU Leiya, ZHANG Zequan, QI Meng, HU Song, CHEN Jing, LI Xiujuan, LIU Jinwei, ZHOU Juan, QIAO Jie, LUO Dan, LUO Guangwei, YAN Youqin, YANG Guiping, DONG Dandan, ZHOU Wei, TAO Junxiu, JIN De, TONG Xiaolin, WEI Li. Effects of Shengmai Yin (生脉饮) on pulmonary and cardiac function in coronavirus disease 2019 convalescent patients with cardiopulmonary symptoms: a randomized, double blind, multicenter control trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 140-145. |
| [7] | LI Ximeng, KANG Yuan, LI Wenjing, LIU Zhuangzhuang, XU Zhenlu, ZHANG Xiaoyu, CAI Runlan, GAO Yuan, QI Yun. Comparing the effects of three decoctions for coronavirus disease 2019 on severe acute respiratory syndrome coronavirus 2-related toll-like receptors-mediated inflammations [J]. Journal of Traditional Chinese Medicine, 2023, 43(1): 51-59. |
| [8] | ZHU Qingguang, ZHANG Shuaipan, LI Jingxian, SUN Wuquan, CHENG Wei, ZHAN Chao, CHENG Yanbin, FANG Lei, FANG Min. Effectiveness of Liu-zi-jue exercise on coronavirus disease 2019 in the patients: a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 997-10053. |
| [9] | AN Xuedong, ZHANG Qing, TAO Junxiu, LI Li, CHEN Yun, LI Kejian, HE Jing, LIU Ru, GUO Juan, ZHANG Jia, ZHU Hui, LIAN Fengmei, LI Xiaodong. Shugan Jieyu capsule (舒肝解郁胶囊) improve sleep and emotional disorder in coronavirus disease 2019 convalescence patients: a randomized, double-blind, placebo-controlled trial [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 803-809. |
| [10] | CHEN Huang, SHI Lushaobo, SHI Zengping, XIA Yi, WANG Dong. Factors influencing physician's behavioral intention to use Traditional Chinese Medicine to treat coronavirus disease 2019 based on the theory of planned behavior [J]. Journal of Traditional Chinese Medicine, 2022, 42(4): 633-6400. |
| [11] | ZHAO Yufeng, PANG Huaxin, Lü Lanting, ZHOU Pei, WANG Kaining, CAI Shengxing, ZHANG Huifeng, LI Kun. Risk assessment and analysis of Traditional Chinese Medicine intervention in coronavirus disease [J]. Journal of Traditional Chinese Medicine, 2022, 42(3): 472-478. |
| [12] | XIA Wenguang, ZHENG Chanjuan, ZHANG Jixian, HUANG Min, LI Qinglin, DUAN Can, LI Zhengliang, FAN Cunyu, ZOU Yilong, XU Bo, YANG Fengwen, LIU Qingquan. Randomized controlled study of a diagnosis and treatment plan for moderate coronavirus disease 2019 that integrates Traditional Chinese and Western Medicine [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 234-241. |
| [13] | YAN Shixing, Lü Yi, LIU Ziqing, REN Meng, HE Haiyang, XIAO Li, GUO Feng, PENG Miao, LI Xiaoxia, WANG Yong, XU Xi, YANG Tao, SHAO Zuoyu, HUANG Jingjing, XIAO Mingzhong. Mining intrinsic information of convalescent patients after suffering coronavirus disease 2019 in Wuhan [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 279-288. |
| [14] | Kamal Dawood, Roohullah, Rabbi Fazle, Naz Attiqa, Bilal Muhammad. In-vitro and in-vivo pharmacological screening of Iris albicans [J]. Journal of Traditional Chinese Medicine, 2022, 42(1): 9-16. |
| [15] | LI Li, AN Xuedong, ZHANG Qing, TAO Junxiu, HE Jing, CHEN Yun, LI Kejian, LIU Ru, GUO Juan, ZHANG Hao, TONG Xiaolin, BA Yuanming. Shumian capsule(舒眠胶囊) improves symptoms of sleep mood disorder in convalescent patients of Corona Virus Disease 2019 [J]. Journal of Traditional Chinese Medicine, 2021, 41(6): 974-981. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||