Journal of Traditional Chinese Medicine ›› 2022, Vol. 42 ›› Issue (5): 687-692.DOI: 10.19852/j.cnki.jtcm.20220426.001
• Research Articles • Previous Articles Next Articles
Sanguinarine suppresses cell proliferation, migration and invasion in nasopharyngeal carcinoma via inhibiting mTOR signaling
YANG Mengzhe1, ZHANG Beibei2, LIANG Zhenqiang3, CHENG Nannan1, Lü Anqiao1, YANG Jianyu1, GUO Xingzhe1, BAI Xianyu1, HUANG Yuanjiao4, JIAO Aijun5( ), XU Ning6(
), XU Ning6( )
)
- 1 School of Basic Medical Sciences, Guangxi Medical University, Nanning 530021, China
2 Institute of Biomedical Research, Yunnan University, Kunming 650500, China
3 Department of Otolaryngology-Head and Neck Surgery, the Affiliated Tumor Hospital of Guangxi Medical University, Nanning 530021, China
4 Life Science Institute, Guangxi Medical University, Nanning 530021, China
5 Pharmaceutical College, Guangxi Medical University, Nanning 530021, China
6 Department of Neurology, the Fifth Affiliated Hospital of Guangxi Medical University, Nanning 530021, China
-
Received:2021-06-27Accepted:2021-09-22Online:2022-10-15Published:2022-04-26 -
Contact:JIAO Aijun,XU Ning -
About author:XU Ning, Department of Neurology, The Fifth Affiliated Hospital of Guangxi Medical University, Nanning 530021, China, m189770838191@163.com; Telephone: +86-771-5358128
JIAO Aijun, Pharmaceutical College, Guangxi Medical University, Nanning 530021, China, 807249683@qq.com;
-
Supported by:Department of Science and Technology of Guangxi Zhuang Autonomous Region, China, Screening, Product Development and Mechanism Research of Anti-Nasopharyngeal Cancer Chinese Medicine Monomers(AB19110052);Natural Science Foundation of Guangxi, China, Epstein-Barr Virus Infection and Up-Regulation Mechanism of Invasion and Migration-Related Proteins S100A8 and S100A9 in Nasopharyngeal Carcinoma Cells(2015GXNSFAA139215);National Natural Science Foundation of China, Quantification, Validation, Functional Analysis and Construction of Regulatory Pathway of iTRAQ Serum Protein Molecular Markers in Nasopharyngeal Carcinoma(81260405)
Cite this article
YANG Mengzhe, ZHANG Beibei, LIANG Zhenqiang, CHENG Nannan, Lü Anqiao, YANG Jianyu, GUO Xingzhe, BAI Xianyu, HUANG Yuanjiao, JIAO Aijun, XU Ning. Sanguinarine suppresses cell proliferation, migration and invasion in nasopharyngeal carcinoma via inhibiting mTOR signaling[J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 687-692.
share this article
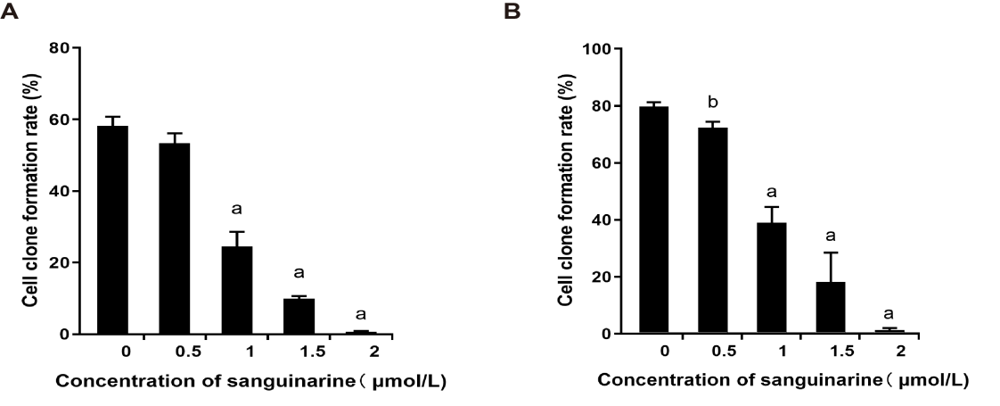
Figure 1 Effects of sanguinarine (SA) treatment on cloning ability of nasopharyngeal carcinoma (NPC) cells A: quantifications of cell cloning ability in CNE2 NPC cells after different doses of sanguinarine (SA) treatment. n = 3, aP < 0.001, which indicated the comparison with Control (0 μmol/L); B: quantifications of cell cloning ability in 5-8F NPC cells after different doses of sanguinarine (SA) treatment. n = 3, aP < 0.001, bP < 0.05, which indicated the comparison with Control (0 μmol/L). The results showed that as the concentration increased, the cloning rate of CNE2 and 5-8F both decreased significantly. At a concentration of 2 μmol/L, only a few macroscopic cell clones have formed.
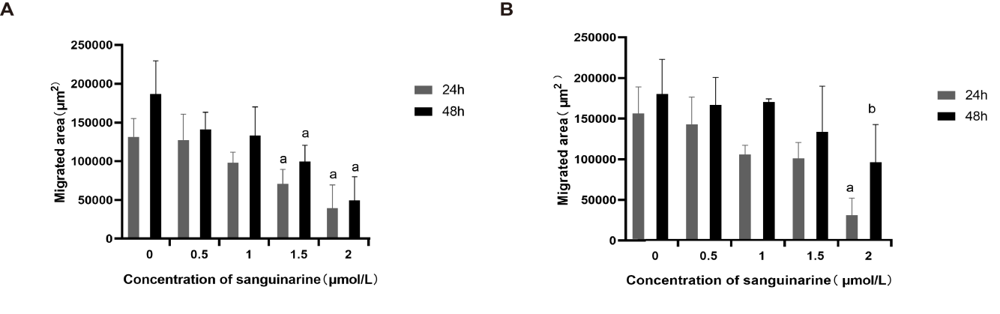
Figure 2 Effects of sanguinarine (SA) treatment on metastasis ability of nasopharyngeal carcinoma (NPC) cells A: quantifications of cell migrated area in CNE2 NPC cells after different doses of sanguinarine (SA) treatment for 24 h and 48 h. n = 3, aP < 0.05, which indicated the comparison with Control (0 μmol/L); B: quantifications of cell migrated area in 5-8F NPC cells after different doses of sanguinarine (SA) treatment for 24 h and 48 h. n = 3, aP < 0.05, bP < 0.01, which indicated the comparison with Control (0 μmol/L). Collectively, the results showed that compared with the control group, as the concentration increased, the migration area of CNE2 and 5-8F cells both showed a significant concentration-dependent decrease.
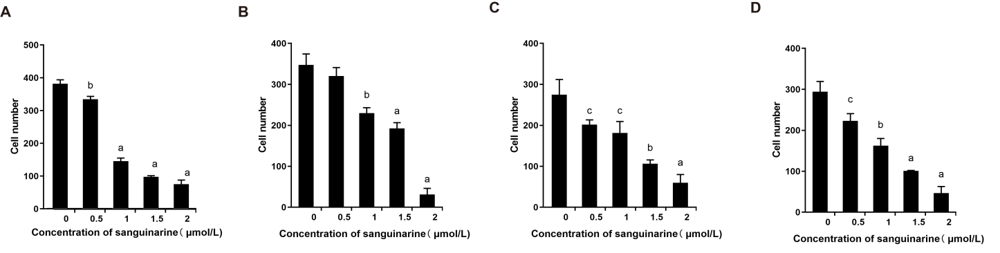
Figure 3 Effects of sanguinarine (SA) treatment on migration and invasion ability of nasopharyngeal carcinoma (NPC) cells A: quantifications of migration ability in NPC cells including CNE2 cells and 5-8F cells after different doses of sanguinarine (SA) treatment. n = 3, aP < 0.001, bP < 0.01, which indicated the comparison with Control (0 μmol/L); B: Quantifications of invasion ability in NPC cells including CNE-2 cells and 5-8F cells after different doses of sanguinarine (SA) treatment. n = 3, aP < 0.001, bP < 0.01, cP < 0.05, which indicated the comparison with Control (0 μmol/L); C: quantifications of invasion ability in NPC cells including CNE-2 cells after different doses of sanguinarine (SA) treatment. n = 3, aP < 0.001, bP < 0.01, cP < 0.05, which indicated the comparison with Control (0 μmol/L). D: quantifications of invasion ability in NPC cells including 5-8F cells after different doses of sanguinarine (SA) treatment. n = 3, aP < 0.001, bP < 0.01, cP < 0.05, which indicated the comparison with Control (0 μmol/L). Clearly, SA treatment on migration and invasion ability of NPC cells.
| CNE2 | 5-8F | ||||
|---|---|---|---|---|---|
| mTOR | p-mTOR | mTOR | p-mTOR | ||
| 0 μmol/L | 0.589±0.032 | 0.445±0.060 | 0.726±0.043 | 0.786±0.050 | |
| 1 μmol/L | 0.639±0.030 | 0.499±0.015 | 0.623±0.041 | 0.575±0.040 | |
| 2 μmol/L | 0.589±0.032 | 0.445±0.060 | 0.510±0.006a | 0.444±0.011a | |
| 3 μmol/L | 0.493±0.013a | 0.372±0.044a | 0.490±0.023a | 0.374±0.068a | |
| 4 μmol/L | 0.466±0.016b | 0.372±0.030a | 0.426±0.013a | 0.321±0.026b | |
| 5 μmol/L | 0.362±0.012b | 0.287±0.048a | 0.277±0.029b | 0.230±0.023b | |
Table 1 Relative protein levels of mTOR and p-mTOR with sanguinarine (SA) treatment
| CNE2 | 5-8F | ||||
|---|---|---|---|---|---|
| mTOR | p-mTOR | mTOR | p-mTOR | ||
| 0 μmol/L | 0.589±0.032 | 0.445±0.060 | 0.726±0.043 | 0.786±0.050 | |
| 1 μmol/L | 0.639±0.030 | 0.499±0.015 | 0.623±0.041 | 0.575±0.040 | |
| 2 μmol/L | 0.589±0.032 | 0.445±0.060 | 0.510±0.006a | 0.444±0.011a | |
| 3 μmol/L | 0.493±0.013a | 0.372±0.044a | 0.490±0.023a | 0.374±0.068a | |
| 4 μmol/L | 0.466±0.016b | 0.372±0.030a | 0.426±0.013a | 0.321±0.026b | |
| 5 μmol/L | 0.362±0.012b | 0.287±0.048a | 0.277±0.029b | 0.230±0.023b | |
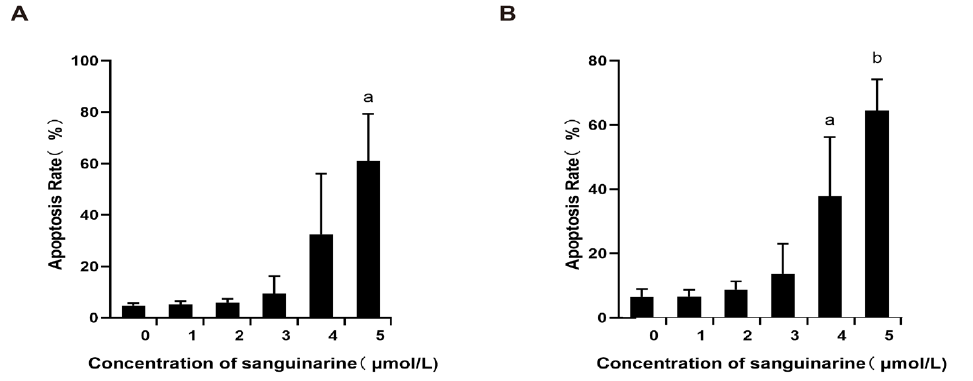
Figure 4 Effects of sanguinarine (SA) treatment on the apoptosis of nasopharyngeal carcinoma (NPC) cells A: Quantifications of cell apoptosis in CNE2 cells with different dose of sanguinarine (SA) treatment; B: Quantifications of cell apoptosis in 5-8F cells with different dose of sanguinarine (SA) treatment. n = 3, aP < 0.01, bP < 0.001, which indicated the comparison with Control (0 μmol/L), respectively. The treatment of SA obviously increased the cell apoptosis in both NPC cells.
| 1. |
Brennan B. Nasopharyngeal carcinoma. Orphanet J Rare Dis 2006; 1: 23.
DOI URL |
| 2. | Cao SM, Simons MJ, Qian CN. The prevalence and prevention of nasopharyngeal carcinoma in China. Chin J Cancer 2011; 30: 114-9. |
| 3. | Lee HM, Okuda KS, González FE, et al. Current perspectives on nasopharyngeal carcinoma. Adv Exp Med Biol 2019; 1164: 11-34. |
| 4. |
Peng L, Liu JQ; Chen YP, et al. The next decade of clinical trials in locoregionally advanced nasopharyngeal carcinoma. Br J Radiol 2019; 92: 20181031.
DOI URL |
| 5. |
Saini VK, Sewal RK, Ahmad Y, et al. Prospective observational study of adverse drug reactions of anticancer drugs used in cancer treatment in a Tertiary Care Hospital. Indian J Pharm Sci 2015; 77: 687-93.
PMID |
| 6. |
Orlikova B, Legrand N, Panning J, et al. Anti-inflammatory and anticancer drugs from nature. Cancer Treat Res 2014; 159: 123-43.
DOI PMID |
| 7. |
Mackraj I, Govender T, Gathiram P. Sanguinarine. Cardiovasc Ther 2008; 26: 75-83.
DOI PMID |
| 8. |
Zhang R, Wang G, Zhang PF, et al. Sanguinarine inhibits growth and invasion of gastric cancer cells via regulation of the DUSP4/ERK pathway. J Cell Mol Med 2017; 21: 1117-27.
DOI PMID |
| 9. |
Singh CK; Kaur S; George J, et al. Molecular signatures of sang-uinarine in human pancreatic cancer cells: a large scale label-free comparative proteomics approach. Oncotarget 2015; 6: 10335-48.
DOI URL |
| 10. | Kalogris C, Garulli C, Pietrella L, et al. Sanguinarine suppresses basal-like breast cancer growth through dihydrofolate reductase inhibition. Biochem Pharmacol 2014; 90: 226-34. |
| 11. |
Mao YP, Tang LL, Chen L, et al. Prognostic factors and failure patterns in non-metastatic nasopharyngeal carcinoma after intensity-modulated radiotherapy. Chin J Cancer 2016; 35: 103.
DOI URL |
| 12. |
Tian YM, Huang WZ, Yuan X, et al. The challenge in treating locally recurrent T3-4 nasopharyngeal carcinoma: the survival benefit and severe late toxicities of re-irradiation with intensity-modulated radiotherapy. Oncotarget 2017; 8: 43450-7.
DOI URL |
| 13. |
Gangemi R, Paleari L, Orengo AM, et al. Cancer stem cells: a new paradigm for understanding tumor growth and progression and drug resistance. Curr Med Chem 2009; 16: 1688-703.
DOI URL |
| 14. |
Sinnung S, Janvilisri T, Kiatwuthinon P. Reversal of cisplatin sensitization and abrogation of cisplatin-enriched cancer stem cells in 5-8F nasopharyngeal carcinoma cell line through a suppression of Wnt/beta-catenin-signaling pathway. Mol Cell Biochem 2021; 476: 1663-72.
DOI URL |
| 15. |
Lee AW, Ma BB, Ng WT, et al. Management of nasopharyngeal carcinoma: current practice and future perspective. J Clin Oncol 2015; 33: 3356-64.
DOI URL |
| 16. |
Lee TK, Park C, Jeong SJ, et al. Sanguinarine induces apoptosis of human oral squamous cell carcinoma KB cells via inactivation of the PI3K/Akt signaling pathway. Drug Dev Res 2016; 77: 227-40.
DOI URL |
| 17. |
Galadari S, Rahman A, Pallichankandy S, et al. Molecular targets and anticancer potential of sanguinarine-a benzophenanthridine alkaloid. Phytomedicine 2017; 34: 143-53.
DOI URL |
| 18. |
Achkar IW, Mraiche F, Mohammad RM, et al. Anticancer potential of sanguinarine for various human malignancies. Future Med Chem 2017; 9: 933-50.
DOI PMID |
| 19. |
Wu GS. Role of mitogen-activated protein kinase phosphatases (MKPs) in cancer. Cancer Metastasis Rev 2007; 26: 579-85.
DOI URL |
| 20. |
Vogt A, Tamewitz A, Skoko J, et al. The benzo[c]phenanthridine alkaloid, sanguinarine, is a selective, cell-active inhibitor of mitogen-activated protein kinase phosphatase-1. J Biol Chem 2005; 280: 19078-86.
DOI URL |
| 21. |
Burotto M, Chiou VL, Lee JM, et al. The MAPK pathway across different malignancies: a new perspective. Cancer 2014; 120: 3446-56.
DOI URL |
| 22. |
Gaziano R, Moroni G, Buè C, et al. Antitumor effects of the benzophenanthridine alkaloid sanguinarine: Evidence and perspectives. World J Gastrointest Oncol 2016; 8: 30-9.
DOI URL |
| 23. |
Ahmad N, Gupta S, Husain MM, et al. Differential antiproliferative and apoptotic response of sanguinarine for cancer cells versus normal cells. Clin Cancer Res 2000; 6: 1524-8.
PMID |
| 24. |
Sarbassov DD, Guertin DA, Ali SM, et al. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 2005; 307: 1098-101.
DOI PMID |
| 25. |
Saxton RA, Sabatini DM. mTOR Signaling in Growth, Metabolism, and Disease. Cell, 2017; 168: 960-76.
DOI URL |
| 26. |
Heitman J, Movva NR, Hall MN. Targets for cell cycle arrest by the immunosuppressant rapamycin in yeast. Science 1991; 253: 905-9.
PMID |
| 27. |
Jiang BH, Liu LZ. Role of mTOR in anticancer drug resistance: perspectives for improved drug treatment. Drug Resist Updat 2008; 11: 63-76.
DOI URL |
| 28. | Robb VA, Karbowniczek M, Klein-Szanto AJ, et al. Activation of the mTOR signaling pathway in renal clear cell carcinoma. J Urol 2007; 177: 346-52. |
| 29. |
Siu MK, Tsai YC, Chang YS, et al. Transforming growth factor-β promotes prostate bone metastasis through induction of microRNA-96 and activation of the mTOR pathway. Oncogene 2015; 34: 4767-76.
DOI PMID |
| 30. | Ghayad SE, Bieche I, Vendrell JA, et al. mTOR inhibition reverses acquired endocrine therapy resistance of breast cancer cells at the cell proliferation and gene-expression levels. Cancer Sci 2008; 99: 1992-2003. |
| 31. |
Ponziani F, Ojetti V, Tortora A, et al. The metabolic and toxico-logical considerations for mTOR inhibitors in the treatment of hepatocarcinoma. Expert Opin Drug Metab Toxicol 2011; 7: 1535-46.
DOI URL |
| 32. |
Vivanco I; Sawyers CL. The phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nature rev Cancer 2002; 2: 489-501.
DOI URL |
| 33. | Zhong JT, Yu J, Wang HJ, et al. Effects of endoplasmic reticulum stress on the autophagy, apoptosis, and chemotherapy resistance of human breast cancer cells by regulating the PI3K/ AKT/mTOR signaling pathway. Tumour Biol 2017; 39: 1010428317697562. |
| 34. |
Guertin DA, Sabatini DM. Defining the role of mTOR in cancer. Cancer cell 2007; 12: 9-22.
DOI PMID |
| 35. |
Hara K, Yonezawa K, Weng QP, et al. Amino acid sufficiency and mTOR regulate p70 S6 kinase and eIF-4E BP1 through a common effector mechanism. J Biol Chem 1998; 273: 14484-94.
DOI PMID |
| 36. |
Pópulo H, Lopes JM, Soares P. The mTOR signalling pathway in human cancer. Int J Mol Sci 2012; 13: 1886-1918.
DOI URL |
| 37. |
Eid SY, El-Readi MZ, Wink M. Synergism of three-drug combinations of sanguinarine and other plant secondary metabolites with digitonin and doxorubicin in multi-drug resistant cancer cells. Phytomedicine 2012; 19: 1288-97.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

