Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (2): 418-426.DOI: 10.19852/j.cnki.jtcm.2026.02.014
• Original Articles • Previous Articles Next Articles
Efficacy and safety of Jiawei Simiaoyongan granules (加味四妙勇安颗粒) in the treatment of non-infectious anterior uveitis: a randomized open-label trial
SUN Wu1, CHU Liqun1, ZHOU Wanyu1( ), SHI Hang1, CHEN Shuiling1, LIANG Yu1, WU Jianguo1, WEI Dong1, LIU Yushen2, ZHANG Qi1
), SHI Hang1, CHEN Shuiling1, LIANG Yu1, WU Jianguo1, WEI Dong1, LIU Yushen2, ZHANG Qi1
- 1
Department of Ophthalmology ,Xiyuan Hospital of China Academy of Chinese Medical Sciences Beijing 100091, China
2Department of Encephalopathy ,Dongzhimen Hospital, Beijing University of Chinese Medicine Beijing 100007, China
-
Received:2025-02-07Accepted:2025-07-14Online:2026-04-15Published:2026-04-04 -
Contact:ZHOU Wanyu, Department of Ophthalmology, Xiyuan Hospital of China Academy of Chinese Medical Sciences, Beijing 100091, China. zhouwylx@sina.com; Telephone: +86-10-62835138 -
Supported by:Capital Health Development Research Project: Research on the Treatment of Non-infectious Uveitis Based on Disease-Syndrome Combination”(2022-2-4174);Beijing Natural Science Foundation(7244501);Study on the Mechanism of Liangxue Sanjie Decoction in Treating Subretinal Fibrosis Secondary to Neovascular Age-related Macular Degeneration Based on miR155-mediated Autophagy”(82405489);Hospital Capability Enhancement Project of Xiyuan Hospital, China Academy of Chinese Medical Sciences: a Randomized Controlled Trial of Acupuncture with Huoxue Tongluo Therapy for Neovascular Age-related Macular Degeneration(XYZX0202-10)
Cite this article
SUN Wu, CHU Liqun, ZHOU Wanyu, SHI Hang, CHEN Shuiling, LIANG Yu, WU Jianguo, WEI Dong, LIU Yushen, ZHANG Qi. Efficacy and safety of Jiawei Simiaoyongan granules (加味四妙勇安颗粒) in the treatment of non-infectious anterior uveitis: a randomized open-label trial[J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 418-426.
share this article
| Characteristic | TCM group (n = 49) | Control group (n = 49) | P value |
|---|---|---|---|
| Female [n (%)] | 23 (46.9) | 24 (49.0) | >0.999 |
| Age (years) | 45.51±9.73 | 41.92±10.62 | 0.084 |
| Disease course (days) | 2022±2540 | 1846±2557 | 0.733 |
| BCVA (LogMAR) | 0.38±0.41 | 0.33±0.29 | 0.481 |
| IOP (mm Hg) | 13.49±3.56 | 14.24±4.34 | 0.350 |
| Scoring of conjunctival hyperemia (scores) | 1.53±0.74 | 1.41±0.70 | 0.403 |
| The number of patients with kp (+) | 48 | 48 | >0.999 |
| Scoring of anterior chamber cells (scores) | 1.53±0.65 | 1.63±0.73 | 0.465 |
| Scoring of eye pain (scores) | 2.49±1.94 | 1.20±1.66 | 0.149 |
| Scoring of photophobia (scores) | 4.41±3.67 | 4.45±3.04 | 0.952 |
| Scoring of lacrimation (scores) | 2.90±3.44 | 1.80±2.59 | 0.076 |
| Scoring of bitter taste (scores) | 1.78±1.65 | 1.35±1.38 | 0.1669 |
| Insomnia scoring (scores) | 2.00±1.58 | 1.31±1.50 | 0.028 |
| Headache scoring (scores) | 1.27±1.40 | 1.06±1.36 | 0.147 |
| Upset scoring (scores) | 1.47±1.40 | 1.51±1.71 | 0.439 |
Table 1 Baseline characteristics of participants
| Characteristic | TCM group (n = 49) | Control group (n = 49) | P value |
|---|---|---|---|
| Female [n (%)] | 23 (46.9) | 24 (49.0) | >0.999 |
| Age (years) | 45.51±9.73 | 41.92±10.62 | 0.084 |
| Disease course (days) | 2022±2540 | 1846±2557 | 0.733 |
| BCVA (LogMAR) | 0.38±0.41 | 0.33±0.29 | 0.481 |
| IOP (mm Hg) | 13.49±3.56 | 14.24±4.34 | 0.350 |
| Scoring of conjunctival hyperemia (scores) | 1.53±0.74 | 1.41±0.70 | 0.403 |
| The number of patients with kp (+) | 48 | 48 | >0.999 |
| Scoring of anterior chamber cells (scores) | 1.53±0.65 | 1.63±0.73 | 0.465 |
| Scoring of eye pain (scores) | 2.49±1.94 | 1.20±1.66 | 0.149 |
| Scoring of photophobia (scores) | 4.41±3.67 | 4.45±3.04 | 0.952 |
| Scoring of lacrimation (scores) | 2.90±3.44 | 1.80±2.59 | 0.076 |
| Scoring of bitter taste (scores) | 1.78±1.65 | 1.35±1.38 | 0.1669 |
| Insomnia scoring (scores) | 2.00±1.58 | 1.31±1.50 | 0.028 |
| Headache scoring (scores) | 1.27±1.40 | 1.06±1.36 | 0.147 |
| Upset scoring (scores) | 1.47±1.40 | 1.51±1.71 | 0.439 |
| Outcome | TCM group (n = 49) | Control group (n = 49) | P valuea | P valueb | P valuec | |
|---|---|---|---|---|---|---|
| BCVA (difference from baseline) | 1 week | -0.06±0.18 | -0.06±0.18 | 0.318 | <0.001 | 0.452 |
| 2 weeks | -0.13±0.21 | -0.10±0.22 | ||||
| 1 month | -0.22±0.24 | -0.16±0.29 | ||||
| 2 months | -0.25±0.31 | -0.22±0.32 | ||||
| 3 months | -0.29±0.32 | -0.23±0.31 | ||||
| 6 months | -0.30±0.32 | -0.20±0.42 | ||||
| Number of relapses within 6 months [n (%)] | 3 (6.1) | 12 (24.5) | 0.022 | NA | NA | |
| Number of IOP elevations >10 mm Hg within 6 months [n (%)] | 1 (2) | 4 (8.2) | 0.646 | NA | NA | |
Table 2 Comparison of changes in BCVA, IOP, and recurrence rate among groups
| Outcome | TCM group (n = 49) | Control group (n = 49) | P valuea | P valueb | P valuec | |
|---|---|---|---|---|---|---|
| BCVA (difference from baseline) | 1 week | -0.06±0.18 | -0.06±0.18 | 0.318 | <0.001 | 0.452 |
| 2 weeks | -0.13±0.21 | -0.10±0.22 | ||||
| 1 month | -0.22±0.24 | -0.16±0.29 | ||||
| 2 months | -0.25±0.31 | -0.22±0.32 | ||||
| 3 months | -0.29±0.32 | -0.23±0.31 | ||||
| 6 months | -0.30±0.32 | -0.20±0.42 | ||||
| Number of relapses within 6 months [n (%)] | 3 (6.1) | 12 (24.5) | 0.022 | NA | NA | |
| Number of IOP elevations >10 mm Hg within 6 months [n (%)] | 1 (2) | 4 (8.2) | 0.646 | NA | NA | |
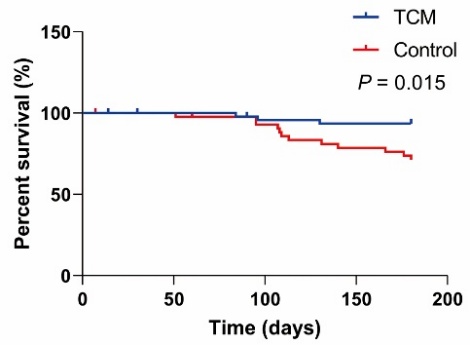
Figure 2 Effect of different treatment regimens on the recurrence rate of non-infectious anterior uveitis Control group: treated with topical glucocorticoids and pupil-dilating eye drops. TCM group: treated with Jiawei Simiao Yongan granules in addition to the control regimen. Cumulative recurrence-free survival rates were estimated using the Kaplan-Meier method. Data are presented as percentages. Cox regression analysis was performed to assess the impact of different treatment regimens on recurrence rate.
| Characteristic | HR | 95% CI | P value |
|---|---|---|---|
| Gender | 0.455 | 0.155, 1.332 | 0.151 |
| Age (years) | 0.169 | 0.913, 1.016 | 0.169 |
| TCM | 0.208 | 0.059, 0.737 | 0.015 |
| Control | - | - |
Table 3 Cox regression analysis for the impact of gender, age, and different treatment regimens on recurrence rate
| Characteristic | HR | 95% CI | P value |
|---|---|---|---|
| Gender | 0.455 | 0.155, 1.332 | 0.151 |
| Age (years) | 0.169 | 0.913, 1.016 | 0.169 |
| TCM | 0.208 | 0.059, 0.737 | 0.015 |
| Control | - | - |
| Outcome | Time | TCM group (n = 49) | Control group (n = 49) | P valuea | P valueb | P valuec |
|---|---|---|---|---|---|---|
| Anterior chamber cells | 1 week | -0.88±0.75 | -0.63±0.78 | 0.441 | <0.001 | 0.619 |
| 2 weeks | -1.18±0.78 | -1.11±0.84 | ||||
| Conjunctival hyperemia | 1 week | -0.72±0.63 | -0.59±0.66 | 0.265 | <0.001 | 0.222 |
| 2 weeks | -1.07±0.80 | -1.00±0.79 | ||||
| The number of patients with kp (+) | 1 week | 27 | 27 | >0.999 | NA | NA |
| 2 weeks | 8 | 9 | 0.79 | NA | NA | |
| Eye pain | 1 week | -1.96±1.94 | -1.02±1.64 | 0.033 | 0.002 | 0.144 |
| 2 weeks | -2.16±1.91 | -1.59±1.83 | ||||
| Photophobia | 1 week | -2.08±2.89 | -1.67±3.04 | 0.62 | 0.002 | 0.418 |
| 2 weeks | -2.57±3.27 | -2.49±3.23 | ||||
| Lacrimation | 1 week | -1.67±2.75 | -0.65±2.06 | 0.068 | 0.033 | 0.758 |
| 2 weeks | -2.04±3.35 | -1.14±2.71 |
Table 4 Comparison of changes in the scoring of ocular signs and symptoms among groups
| Outcome | Time | TCM group (n = 49) | Control group (n = 49) | P valuea | P valueb | P valuec |
|---|---|---|---|---|---|---|
| Anterior chamber cells | 1 week | -0.88±0.75 | -0.63±0.78 | 0.441 | <0.001 | 0.619 |
| 2 weeks | -1.18±0.78 | -1.11±0.84 | ||||
| Conjunctival hyperemia | 1 week | -0.72±0.63 | -0.59±0.66 | 0.265 | <0.001 | 0.222 |
| 2 weeks | -1.07±0.80 | -1.00±0.79 | ||||
| The number of patients with kp (+) | 1 week | 27 | 27 | >0.999 | NA | NA |
| 2 weeks | 8 | 9 | 0.79 | NA | NA | |
| Eye pain | 1 week | -1.96±1.94 | -1.02±1.64 | 0.033 | 0.002 | 0.144 |
| 2 weeks | -2.16±1.91 | -1.59±1.83 | ||||
| Photophobia | 1 week | -2.08±2.89 | -1.67±3.04 | 0.62 | 0.002 | 0.418 |
| 2 weeks | -2.57±3.27 | -2.49±3.23 | ||||
| Lacrimation | 1 week | -1.67±2.75 | -0.65±2.06 | 0.068 | 0.033 | 0.758 |
| 2 weeks | -2.04±3.35 | -1.14±2.71 |
| Outcomes | Time | TCM group (n = 49) | Control group (n = 49) | P valuea | P valueb | P valuec |
|---|---|---|---|---|---|---|
| Bitter taste | 1 week | -0.59±1.50 | -0.20±1.37 | 0.022 | 0.163 | 0.009 |
| 2 weeks | -1.00±1.44 | -0.09±1.61 | ||||
| Insomnia | 1 week | -0.65±1.32 | 0.00±1.29 | 0.001 | 0.218 | 0.063 |
| 2 weeks | -1.02±1.42 | 0.04±1.58 | ||||
| Headache | 1 week | -0.78±1.28 | -0.53±1.21 | 0.326 | 0.153 | 0.957 |
| 2 weeks | -0.90±1.30 | -0.68±1.34 | ||||
| Upset | 1 week | -0.45±1.31 | -0.53±1.46 | >0.999 | 0.385 | 0.384 |
| 2 weeks | -0.61±1.30 | -0.55±1.99 |
Table 5 Comparison of changes in the TCM symptom scores among groups
| Outcomes | Time | TCM group (n = 49) | Control group (n = 49) | P valuea | P valueb | P valuec |
|---|---|---|---|---|---|---|
| Bitter taste | 1 week | -0.59±1.50 | -0.20±1.37 | 0.022 | 0.163 | 0.009 |
| 2 weeks | -1.00±1.44 | -0.09±1.61 | ||||
| Insomnia | 1 week | -0.65±1.32 | 0.00±1.29 | 0.001 | 0.218 | 0.063 |
| 2 weeks | -1.02±1.42 | 0.04±1.58 | ||||
| Headache | 1 week | -0.78±1.28 | -0.53±1.21 | 0.326 | 0.153 | 0.957 |
| 2 weeks | -0.90±1.30 | -0.68±1.34 | ||||
| Upset | 1 week | -0.45±1.31 | -0.53±1.46 | >0.999 | 0.385 | 0.384 |
| 2 weeks | -0.61±1.30 | -0.55±1.99 |
| 1. |
Rosenbaum JT, Bodaghi B, Couto C, et al. New observations and emerging ideas in diagnosis and management of non-infectious uveitis: a review. Semin Arthritis Rheum 2019; 49: 438-45.
DOI URL |
| 2. |
Tsirouki T, Dastiridou A, Symeonidis C, et al. A Focus on the epidemiology of uveitis. Ocul Immunol Inflamm 2018; 26: 2-16.
DOI PMID |
| 3. | Joltikov KA, Lobo-Chan AM. Epidemiology and risk factors in non-infectious uveitis: a systematic review. Front Med (Lausanne) 2021; 8: 695904. |
| 4. |
Takeuchi M, Mizuki N, Ohno S. Pathogenesis of non-infectious uveitis elucidated by recent genetic findings. Front Immunol 2021; 12: 640473.
DOI URL |
| 5. | Espinosa G, Herreras JM, Muñoz-Fernández S, García Ruiz de Morales JM, Cordero-Coma M. Recommendations statement on the immunosuppressive treatment of non-infectious, non-neoplastic, non-anterior uveitis. Med Clin (Barc) 2020; 155: 220. e1-12. |
| 6. | Reddy A, Liu SH, Brady CJ, Sieving PC, Palestine AG. Corticosteroid implants for chronic non-infectious uveitis. Cochrane Database Syst Rev 202329; 8: CD010469. |
| 7. |
Zhao C, Zhang M. Immunosuppressive treatment of non-infectious uveitis: history and current choices. Chin Med Sci J 2017; 32: 48-61.
DOI PMID |
| 8. |
Jap A, Chee SP. Immunosuppressive therapy for ocular diseases. Curr Opin Ophthalmol 2008; 19: 535-40.
DOI PMID |
| 9. |
Pujari SS, Kempen JH, Newcomb CW, et al. Cyclophosphamide for ocular inflammatory diseases. Ophthalmology 2010; 117: 356-65.
DOI PMID |
| 10. |
Pasadhika S, Kempen JH, Newcomb CW, et al. Azathioprine for ocular inflammatory diseases. Am J Ophthalmol 2009; 148: 500-9.e2.
DOI PMID |
| 11. |
Kaçmaz RO, Kempen JH, Newcomb C, et al. Cyclosporine for ocular inflammatory diseases. Ophthalmology 2010; 117: 576-84.
DOI PMID |
| 12. |
Okhravi N, Lightman S. Cystoid macular edema in uveitis. Ocul Immunol Inflamm 2003; 11: 29-38.
PMID |
| 13. |
Agrawal H, Doan H, Pham B, et al. Systemic immunosuppressive therapies for uveitis in developing countries. Indian J Ophthalmol 2020; 68: 1852-62.
DOI PMID |
| 14. |
Murphy CC, Greiner K, Plskova J, et al. Cyclosporine vs tacrolimus therapy for posterior and intermediate uveitis. Arch Ophthalmol 2005; 123: 634-41.
DOI URL |
| 15. |
Taylor DO, Barr ML, Radovancevic B, et al. A randomized, multicenter comparison of tacrolimus and cyclosporine immunosuppressive regimens in cardiac transplantation: decreased hyperlipidemia and hypertension with tacrolimus. J Heart Lung Transplant 1999; 18: 336-45.
DOI URL |
| 16. |
Samson CM, Waheed N, Baltatzis S, Foster CS. Methotrexate therapy for chronic noninfectious uveitis: analysis of a case series of 160 patients. Ophthalmology 2001; 108: 1134-9.
PMID |
| 17. |
Gangaputra S, Newcomb CW, Liesegang TL, et al. Methotrexate for ocular inflammatory diseases. Ophthalmology 2009; 116: 2188-98.e1.
DOI PMID |
| 18. | Kremer JM, Alarcón GS, Lightfoot Jr RW, et al. Methotrexate for rheumatoid arthritis. Suggested guidelines for monitoring liver toxicity. American College of Rheumatology. Arthritis Rheum 1994; 37: 316-28. |
| 19. |
Galor A, Jabs DA, Leder HA, et al. Comparison of antimetabolite drugs as corticosteroid-sparing therapy for noninfectious ocular inflammation. Ophthalmology 2008; 115: 1826-32.
DOI PMID |
| 20. |
Wu X, Tao M, Zhu L, Zhang T, Zhang M. Pathogenesis and current therapies for non-infectious uveitis. Clin Exp Med 2023; 23: 1089-106.
DOI |
| 21. |
Agrawal R, Lee C, Phatak S, Pavesio C. Immunopharmacotherapy of non-infectious uveitis: where do we stand? Expert Opin Biol Ther 2014; 14: 1719-22.
DOI PMID |
| 22. |
Imrie FR, Dick AD. Biologics in the treatment of uveitis. Curr Opin Ophthalmol 2007; 18: 481-6.
PMID |
| 23. | Zhang DD, Yao J, Sun H, Wang JD, Cao CH. The advantages and characteristics of Chinese medicine in prevention and treatment of anterior uveitis. Zhong Guo Zhong Yi Yan Ke Za Zhi 2021; 31: 530-2. |
| 24. | Wei D, Zhang WD, Zhou WY. Clinical discussion on the treatment of uveitis with Si Miao Yong An Tang. Zhong Guo Zhong Yi Yan Ke Za Zhi 2020; 30: 741-4. |
| 25. |
Fu S, Yu F, Sun T, Hu Z. Drug-drug interaction by metabolites: Challenges and solutions during therapeutics innovation. Drug Metab Dispos 2026; 54: 100251.
DOI URL |
| 26. | Xiao GS, Tang YZ. Encyclopedia of Ophthalmology of Traditional Chinese Medicine. Beijing: People's Medical Publishing House, 2011: 192-5. |
| 27. |
Nguyen QD, Merrill PT, Jaffe GJ, et al. Adalimumab for prevention of uveitic flare in patients with inactive non-infectious uveitis controlled by corticosteroids (VISUAL II): a multicentre, double-masked, randomised, placebo-controlled phase 3 trial. Lancet 2016; 388: 1183-92.
DOI PMID |
| 28. | Jabs DA, Nussenblatt RB, Rosenbaum JT; Standardization of Uveitis Nomenclature (SUN) Working Group. Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. Am J Ophthalmol 2005; 140: 509-16. |
| 29. |
Zou J, Xu W, Li Z, et al. Network pharmacology-based approach to research the effect and mechanism of Si-Miao-Yong-An decoction against thromboangiitis obliterans. Ann Med 2023; 55: 2218105.
DOI URL |
| 30. |
Li HY, Sun H, Zhang AH, et al. Therapeutic effect and mechanism of Si-Miao-Yong-An-Tang on thromboangiitis obliterans based on the urine metabolomics approach. Front Pharmacol 2022; 13: 827733.
DOI URL |
| 31. |
Zhang S, Shao Y, Jin R, Ma B. Combining Network pharmacology, molecular docking, molecular dynamics simulation, and experimental validation to uncover the efficacy and mechanisms of Si-Miao-Yong-An decoction in diabetic wound healing. J Inflamm Res 2025; 18: 4087-101.
DOI PMID |
| 32. | Zhou WY, Fang DY. Treatment of uveitis from collateral disease. Zhong Yi Za Zhi 2009; 50: 977-8. |
| 33. | Yu LM. Clinical study of Simiaoyong’an decoction in the treatment of anterior uveitis. Henan Zhong Yi 2017; 37: 287-9. |
| 34. | Zhou WY, Wang WX, Fang DY. Fang Dingya’s experience in treating leukoaraiosis-related uveitis by staging using the theory of collateral disease. Zhong Yi Za Zhi 2021; 62: 1116-9. |
| 35. |
Takeuchi M, Usui Y, Namba K, et al. Ten-year follow-up of infliximab treatment for uveitis in Behçet disease patients: a multicenter retrospective study. Front Med 2023; 10: 1095423.
DOI URL |
| 36. | Miao M, Xiang L. Pharmacological action and potential targets of chlorogenic acid. Adv Pharmacol 2020; 87: 71-88. |
| 37. |
Naveed M, Hejazi V, Abbas M, et al. Chlorogenic acid (CGA): a pharmacological review and call for further research. Biomed Pharmacother 2018; 97: 67-74.
DOI PMID |
| 38. | Wang JZ, Zhang YF, Xu F, Shang MY, Liu GX, Cai SQ. Investigation of the in vivo metabolism of harpagoside and distribution of its metabolites in rats by HPLC-IT-TOF-MSn. Biomed Chromatogr 2018; 32: e4218. |
| 39. |
Kim JY, Cheon YH, Ahn SJ, et al. Harpagoside attenuates local bone Erosion and systemic osteoporosis in collagen-induced arthritis in mice. BMC Complement Med Ther 2022; 22: 214.
DOI |
| 40. |
Che D, Cao J, Liu R, et al. Harpagoside-induced anaphylactic reaction in an IgE-independent manner both in vitro and in vivo. Immunopharmacol Immunotoxicol 2018; 40: 173-8.
DOI URL |
| 41. |
Kowalska A, Kalinowska-Lis U. 18β-Glycyrrhetinic acid: its core biological properties and dermatological applications. Int J Cosmet Sci 2019; 41: 325-31.
DOI URL |
| 42. | Ke P, Yong L, Zhe L, Guang F. Mechanisms of Baishao (Radix Paeoniae Alba) and Gancao (Radix Glycyrrhizae) on major depressive disorder: network pharmacology and in vivo validation. J Tradit Chin Med 2025; 45: 1067-77. |
| 43. |
Li H, You J, Yang X, et al. Glycyrrhetinic acid: a potential drug for the treatment of COVID-19 cytokine storm. Phytomedicine 2022; 102: 154153.
DOI URL |
| [1] | ZHANG Mingcai, LIN Xun, DU Guoqing, LIU Congying, YUAN Weian, LI Wei, YANG Jiayu, CHEN Bo, YU Xiaoyue, XIONG Yizhe, JIANG Enyu, GAO Ningyang, JIANG Sumin, XU Zhenqiu, WANG Xiang, ZHAN Hongsheng, WU Yufeng. Efficacy and safety of Shi’s cervical manipulation versus diclofenac for patients with acute and subacute neck pain: a multicenter clinical randomized trial [J]. Journal of Traditional Chinese Medicine, 2025, 45(6): 1385-1394. |
| [2] | LI Shanyi, GAO Qi, LI Yanjie, ZHANG Yaling, CHEN Xiaoqi. Efficacy and safety of Yangxiao Fukang granule (养消复康颗粒) in the treatment of stage Ⅲ hepatitis B related liver cancer: study protocol for a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2025, 45(5): 1127-1134. |
| [3] | LU Zhenkai, TAN Chang, ZHI Yingjie, ZHANG Xuming, XIE Yanming. Efficacy and safety of Compound E’jiao Jiang (复方阿胶浆) for treating leukopenia based: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2025, 45(5): 941-953. |
| [4] | ZHANG Hongchun, LIU Jian, CHEN Sheng, ZHANG Wei, LU Xuechao, LI Ying, YU Xueqing, HUANG Yan, SU Lianhua, WEI Baolin, LI Zhuyin, PEI Shuai, LEI Xiang, YANG Daowen, GUO Jianning. Efficacy and safety of Suhuang Zhike capsule (苏黄止咳胶囊) for cough variant asthma: a multicenter, single-arm, open-label phase IV real-world clinical trial [J]. Journal of Traditional Chinese Medicine, 2025, 45(4): 817-828. |
| [5] | XU Jian, LIU Yuntao, LUO Zhihao, ZHAO Zhen, WANG Dawei, LIU Qing. Chinese patent medicine for atherosclerosis: a systematic review and Meta-analysis of randomized controlled trials [J]. Journal of Traditional Chinese Medicine, 2024, 44(6): 1082-1090. |
| [6] | ZHANG Meizhen, HAO Xiaohui, TANG Yiting, CHEN Yupeng, HE Puyu, ZHAO Liming, PANG Bing, NI Qing. Efficacy and safety of Buyang Huanwu decoction (补阳还五汤) for diabetic peripheral neuropathy: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(5): 841-850. |
| [7] | SUN Wu, ZHAO Yuwei, LIAO Liang, ZHAO Zhonghui, CHEN Shiqi, YAN Xiaoling, WANG Xueyao, CHAO Guojun, ZHOU Jian. Effectiveness and safety of Xuebijing injection for patients with coronavirus disease 2019: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(4): 631-639. |
| [8] | LUO Xin, XIE Jing, HUANG Li, GAN Wenfan, CHEN Ming. Efficacy and safety of activating blood circulation and removing blood stasis of Traditional Chinese Medicine for managing renal fibrosis in patients with chronic kidney disease: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 429-440. |
| [9] | ZHANG Yuehong, SHAO Xianzhi, ZHAO Qianlong, ZHAN Hualong, ZHANG Jianhua, DU Sisi, CHEN Jing, LIU Yingfang, ZHOU Haiwang, CHEN Xinsheng, HONG Ying, LIAN Fengmei, TONG Xiaolin, BA Yuanming. Effectiveness of Xiangsha Liujun pills (香砂六君丸) on decreased digestive function in convalescent patients of coronavirus disease 2019: a randomized, double blind, placebo controlled clinical trial [J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 552-558. |
| [10] | ZHOU Yingyan, LIANG Huasheng, YAN Jingyao, HE Xiaohong, PAN Lili, LI Xue, CHEN Xianghong, CHEN Xiumin, YANG Aicheng, HUANG Qingchun. Effectiveness and safety of tripterygium glycosides tablet (雷公藤多苷片) for lupus nephritis: a systematic review and Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2022, 42(5): 671-680. |
| [11] | LIN Yi, LI Xun, WANG Zi, ZHENG Xiaoran, HANG Haiyan, LI Lingling. Efficacy and safety of external application of Chinese herbal medicine for psoriasis vulgaris: a systematic review of randomized controlled trials [J]. Journal of Traditional Chinese Medicine, 2022, 42(4): 493-504. |
| [12] | HUANG Yusi, YANG Jiju, LI Xinyi, HAO Huifeng, LI Chong, ZHANG Fan, LIN Haiming, XIE Xianfei, HE Ke, TIAN Guihua. Effectiveness and safety of electroacupuncture for the treatment of pain after laparoscopic surgery: a systematic review [J]. Journal of Traditional Chinese Medicine, 2022, 42(4): 505-512. |
| [13] | LIU Ying, ZOU Wen, XIAN Qingfei, DENG Xin, ZHANG Fuchun, WANG Li, LI Yonghong, LUN Wenhui, WANG Jian. Efficacy and safety of Mianyi granules (+mianyi+) for reversal of immune nonresponse following antiretroviral therapy of human immunodeficiency virus-1: a randomized, double-blind, multi-center, placebo-controlled trial [J]. Journal of Traditional Chinese Medicine, 2022, 42(3): 432-438. |
| [14] | MA Tingting, WU Jie, YANG Lijie, FENG Fen, YANG Huilin, ZHANG Jinhua, ZHONG Yanjin, NING Qing, HUANG Lirong, LIN Youbing, YAN Jue, CHEN Guiquan, HOU Tianshu, WANG Li, REN Yuanfang, TAN Jing. Ginger-indirect moxibustion plus acupuncture versus acupuncture alone for chronic fatigue syndrome: a randomized controlled trial [J]. Journal of Traditional Chinese Medicine, 2022, 42(2): 242-249. |
| [15] | ZHAO Weipeng, LI Jing, ZHANG Yushuang, LI He, HUANG Jinchang, BAI Jing, LI Jianbo. Efficacy of acupuncture therapy for improving anorexia in tumor patients: a Meta-analysis [J]. Journal of Traditional Chinese Medicine, 2021, 41(4): 507-514. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||