Journal of Traditional Chinese Medicine ›› 2023, Vol. 43 ›› Issue (3): 514-522.DOI: 10.19852/j.cnki.jtcm.20230328.001
Previous Articles Next Articles
Waggle needling Yanglingquan (GB34) enhances cerebral blood flow and ameliorates condition of spastic paresis in rats with middle cerebral artery occlusion induced by surgical nylon suture
ZHANG Zhou1, MU Jiedan1, MA Liangxiao1,2( ), YU Wenyan1, SUN Tianyi1, QIAN Xu1, TIAN Yuan1, ZHANG Yidan1, WANG Junxiang3
), YU Wenyan1, SUN Tianyi1, QIAN Xu1, TIAN Yuan1, ZHANG Yidan1, WANG Junxiang3
- 1 School of Acupuncture-Moxibustion and Tuina, Beijing University of Chinese Medicine, Beijing 100029, China
2 the Key Unit of State Administration of Traditional Chines Medicine, Evaluation of Characteristic Acupuncture Therapy, Beijing 100029, China
3 School of Nursing, Beijing University of Chinese Medicine, Beijing 100029, China
-
Received:2022-01-12Accepted:2022-04-10Online:2023-06-15Published:2023-03-28 -
Contact:Prof. MA Liangxiao, School of Acupuncture-Moxibustion and Tuina, Beijing University of Chinese Medicine, Beijing 100029, China and the Key Unit of State Administration of Traditional Chines Medicine, Evaluation of Characteristic Acupuncture Therapy, Beijing 100029, China. maliangxiao@vip.sina.com. Telephone: +86-10-53912199 -
Supported by:Study on the Mechanism of Waggle Needling GB34 Relieving Spasticity in Post-stroke Rats Based on KCC2-GABAA Receptor Pathway(81774417);Research and Development of Physical Therapy Technology and Equipment for Regulation of Human Functional State(2020-JYB-ZDGG-062)
Cite this article
ZHANG Zhou, MU Jiedan, MA Liangxiao, YU Wenyan, SUN Tianyi, QIAN Xu, TIAN Yuan, ZHANG Yidan, WANG Junxiang. Waggle needling Yanglingquan (GB34) enhances cerebral blood flow and ameliorates condition of spastic paresis in rats with middle cerebral artery occlusion induced by surgical nylon suture[J]. Journal of Traditional Chinese Medicine, 2023, 43(3): 514-522.
share this article
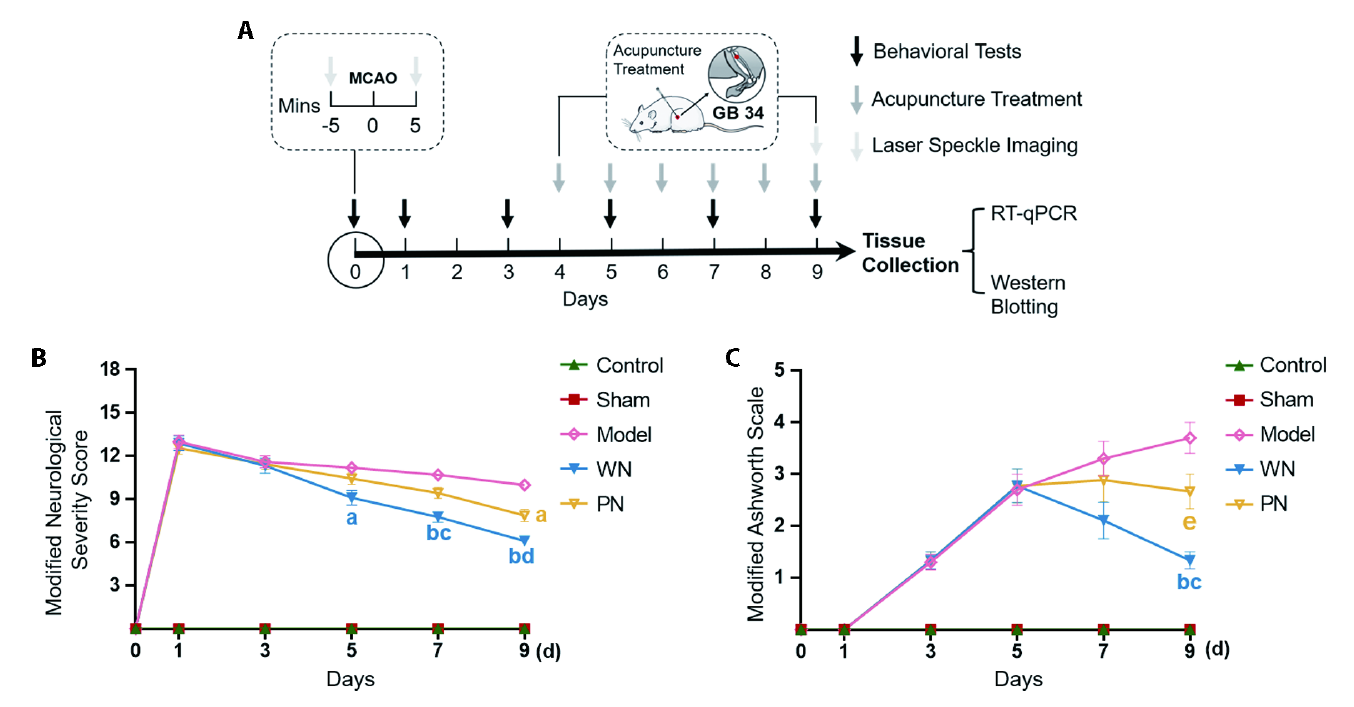
Figure 1 Experimental design of this study and the effect of acupuncture at Yanglingquan (GB34) on neurological deficit and muscle tone in MCAO-induced SP rats A: experimental design of this study. Behavioral tests included the modified neurological severity score and modified Ashworth scale; B: effect of acupuncture at Yanglingquan (GB34) on neurological deficit in MCAO-induced spastic paresis rats. The change of neurological deficit assessed by mNSS at days 0, 1, 3, 5, 7 and 9; C: effect of acupuncture at Yanglingquan (GB34) on muscle tone in MCAO-induced spastic paresis rats. The change of hyper muscle tone assessed by MAS at days 0, 1, 3, 5, 7 and 9. Control group: Rats without any intervention (n = 9). Sham group: Rats received sham operation (n = 9); Model group: MCAO rats without treatment (n = 10); Waggle needling group: MCAO rats + waggle needling Yanglingquan (GB34) for 6 d (n = 9); Perpendicular needling group: MCAO rats + perpendicular needling Yanglingquan (GB34) for 6 d (n = 9). MCAO: middle cerebral artery occlusion; GB34: Yanglingquan acupoint; RT-qPCR: real‐time quantitative polymerase chain reaction; mNSS: the modified neurological severity score; MAS: Modified Ashworth Scale; SP: spastic paresis; Control: control group; Sham: sham group; Model: model group; WN: waggle needling group; PN: perpendicular needling group. All data were expressed as mean ± standard error of mean. aP<0.01, bP<0.001, eP<0.01, versus model group; cP<0.01, dP<0.001 versus PN group.

Figure 2 Effect of acupuncture at Yanglingquan (GB34) on cerebral blood flow recovery in MCAO-induced SP rats A: representative LSI images assessing the CBF. The dynamic change of CBF was recorded in the ROI; B: summary data of cortical CBF in ROI at 5 min before MCAO, 5 min after MCAO and after 6 days’ treatment. Intervention included waggle needling and perpendicular needling Yanglingquan (GB34) in each group. C: representative LSI images of cortical CBF in different groups at 5 min before MCAO, 5 min after MCAO and after 6 days’ treatment. c1-c5: The representative LSI images of rats in Control group, Sham group, Model group, Waggle needling group and Perpendicular needling group at 5 min before MCAO. c6-c10: The representative LSI images of rats in Control group, Sham group, Model group, Waggle needling group and Perpendicular needling group at 5 min after MCAO. c11-c15: The representative LSI images of rats in Control group, Sham group, Model group, Waggle needling group and Perpendicular needling group at 5 min after 6 days’ treatment. Control group: Rats without any intervention (n = 9). Sham group: Rats received sham operation (n = 9). Model group: MCAO rats without treatment (n = 10). Waggle needling group: MCAO rats + waggle needling Yanglingquan (GB34) for 6 d (n = 9). Perpendicular needling group: MCAO rats + perpendicular needling Yanglingquan (GB34) for 6 d (n = 9). ROI: region of interest. MCAO: middle cerebral artery occlusion; SP: spastic paresis; LSI: laser speckle imaging; CBF: cortical cerebral blood flow; ROI: region of interest; Pre MCAO: 5 min pre MCAO operation; Post MCAO: 5 min post MCAO operation; After Intervention: after intervention at day 9. Control: control group; Sham: sham group; Model: model group; WN: waggle needling group; PN: perpendicular needling group. Scale bar = 3 mm. All data were expressed as mean ± standard error of mean. aP<0.001 versus model group; bP<0.001 versus PN group.
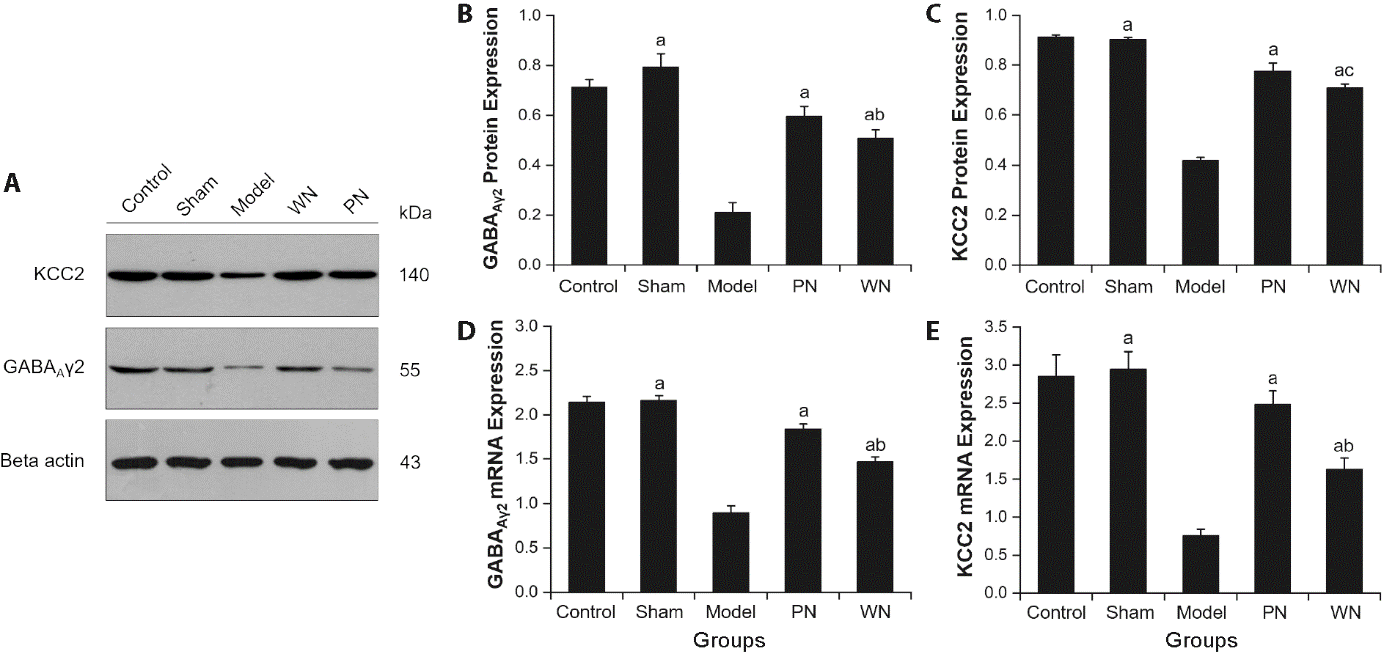
Figure 3 Effect of acupuncture at Yanglingquan (GB34) on the expressions of GABAAγ2 and KCC2 in the ischemic cortex in SP rats A: representative Western blots of GABAAγ2 and KCC2 in the cortex; B, C: protein levels of GABAAγ2 and KCC2 in the cortex; D, E: mRNA levels of GABAAγ2 and KCC2 in the cortex. Control group: rats without any intervention (n = 9). Sham group: rats received sham operation (n = 9). Model group: MCAO rats without treatment (n = 9). Waggle needling group: MCAO rats + waggle needling Yanglingquan (GB34) for 6 d (n = 9). Perpendicular needling group: MCAO rats + perpendicular needling Yanglingquan (GB34) for 6 d (n = 9). GABAAγ2: γ2 subunit of the γ-aminobutyric acid receptor A; KCC2: K+-C-cotransporter 2; SP: spastic paresis; Control: control group; Sham: sham group; Model: model group; WN: waggle needling group; PN: perpendicular needling group. All data were expressed as mean ± standard error of mean. aP<0.001 versus Model group; bP<0.01, cP<0.05 versus PN group.
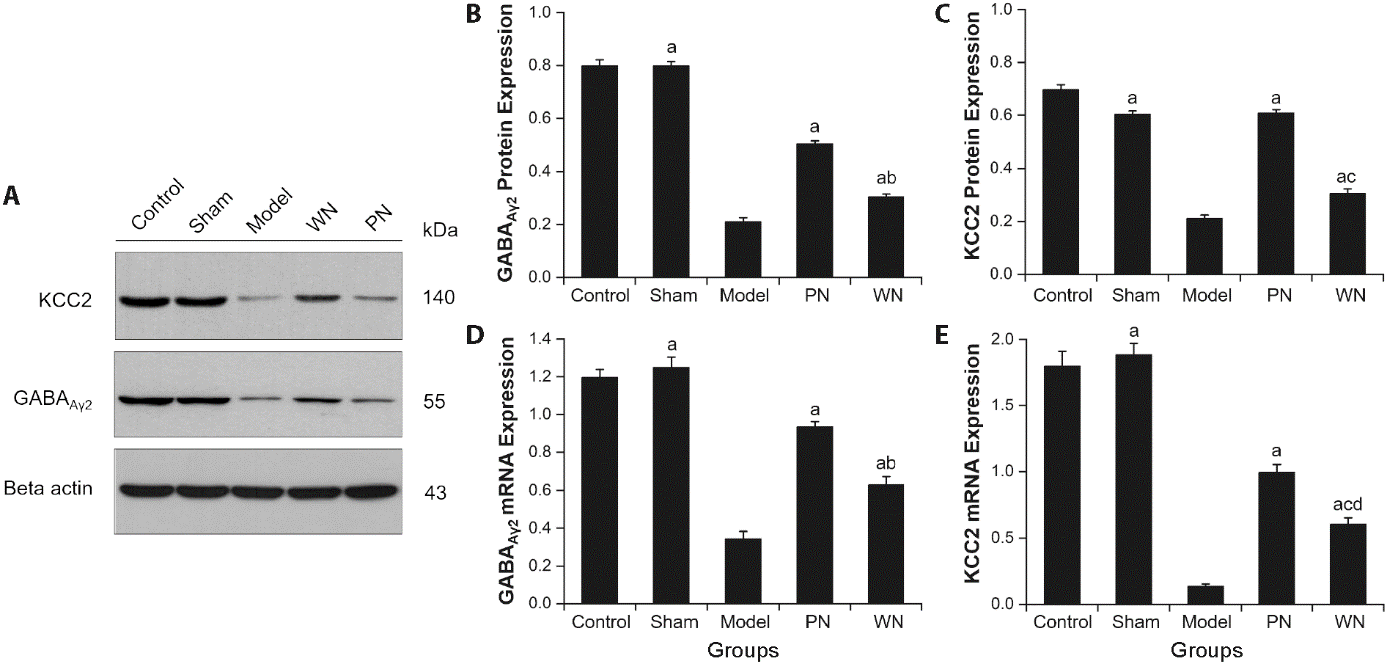
Figure 4 Effect of acupuncture at Yanglingquan (GB34) on the expressions of GABAAγ2 and KCC2 in the lumbar enlargement in SP rats A: representative Western blots of GABAAγ2 and KCC2 in the lumbar enlargement; B, C: protein levels of GABAAγ2 and KCC2 in the lumbar enlargement; D, E: mRNA levels of GABAAγ2 and KCC2 in the lumbar enlargement. Control group: Rats without any intervention (n = 9). Sham group: Rats received sham operation (n = 9). Model group: MCAO rats without treatment (n = 9). Waggle needling group: MCAO rats + waggle needling Yanglingquan (GB34) for 6 d (n = 9). Perpendicular needling group: MCAO rats + perpendicular needling Yanglingquan (GB34) for 6 d (n = 9). GABAAγ2: γ2 subunit of the γ-aminobutyric acid receptor A; KCC2: K+-Cl-cotransporter 2; SP: spastic paresis; Control: Control group; Sham: Sham group; Model: Model group; WN: Waggle needling group; PN: perpendicular needling group. All data were expressed as mean ± standard error of mean. aP<0.001, dP<0.01 versus Model group; bP<0.001, cP<0.01 versus PN group.
| [1] |
Zhou MG, Wang HD, Zeng XY, et al. Mortality, morbidity, and risk factors in China and its provinces, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019; 394: 1145-58.
DOI PMID |
| [2] |
Wissel J, Schelosky LD, Scott J, Christe W, Faiss JH, Mueller J. Early development of spasticity following stroke: a prospective, observational trial. J Neurol 2010; 257: 1067-72.
DOI PMID |
| [3] | Zorowitz RD, Gillard PJ, Brainin M. Poststroke spasticity: sequelae and burden on stroke survivors and caregivers. Neurology 2013; 80: S45-52. |
| [4] |
Zhang X, Chen XP, Lin JB, Xiong Y, Liao WJ, Wan Q. Effect of enriched environment on angiogenesis and neurological functions in rats with focal cerebral ischemia. Brain Res 2017; 1655: 176-85.
DOI PMID |
| [5] |
Zhu XQ, Hu HX, Li ZF, Lin RH, Mao JJ, Chen LD. Gua Lou Gui Zhi decoction attenuates post‑stroke spasticity via the modulation of GABAB receptors. Mol Med Rep 2015; 12: 5957-62.
DOI URL |
| [6] |
Wang JX, Mu JD, Ma LX, et al. Waggle needling wields preferable neuroprotective and anti-spastic effects on post-stroke spasticity rats by attenuating γ-aminobutyric acid transaminase and enhancing γ-aminobutyric acid. Neuroreport 2020; 31: 708-16.
DOI URL |
| [7] |
Sutherland BA, Papadakis M, Chen RL, Buchan AM. Cerebral blood flow alteration in neuroprotection following cerebral ischemia. J Physiol 2011; 589: 4105-14.
DOI URL |
| [8] | Grigoras IF, Stagg CJ. Recent advances in the role of excitation-inhibition balance in motor recovery post-stroke. Fac Rev 2021; 10: 58. |
| [9] | Chen CR, Zhou X, He JL, Xie ZX, Xia SF, Lu GL. The roles of GABA in ischemia-reperfusion injury in the central nervous system and peripheral organs. Oxid Med Cell Longev 2019; 2019: 4028394. |
| [10] |
Pozzi D, Rasile M, Corradini I, Matteoli M. Environmental regulation of the chloride transporter KCC2: switching inflammation off to switch the GABA on? Transl Psychiatry 2020; 10: 349.
DOI |
| [11] |
Wu HJ, Che XR, Tang JJ, Ma FQ, Pan K, Zhao MF. The K (+)-Cl (-) cotransporter KCC2 and chloride homeostasis: potential therapeutic target in acute central nervous system injury. Mol Neurobiol 2016; 53: 2141-51.
DOI URL |
| [12] |
Gracies JM, Brashear A, Jech R, et al. Safety and efficacy of abobotulinumtoxinA for hemiparesis in adults with upper limb spasticity after stroke or traumatic brain injury: a double-blind randomised controlled trial. Lancet Neurol 2015; 14: 992-1001.
DOI URL |
| [13] |
Gupta AD, Chu WH, Howell S, et al. A systematic review: efficacy of botulinum toxin in walking and quality of life in poststroke lower limb spasticity. Syst Rev 2018; 7: 1
DOI URL |
| [14] |
Fan WJ, Kuang X, Hu JW, et al. Acupuncture therapy for poststroke spastic hemiplegia: a systematic review and Meta-analysis of randomized controlled trials. Complement Ther Clin Pract 2020; 40: 101176.
DOI URL |
| [15] |
Yang L, Tan JY, Ma H, et al. Warm-needle moxibustion for spasticity after stroke: a systematic review of randomized controlled trials. Int J Nurs Stud 2018; 82: 129-38.
DOI PMID |
| [16] |
Cai YY, Zhang CS, Liu SN, et al. Electroacupuncture for poststroke spasticity: a systematic review and Meta-analysis. Arch Phys Med Rehabil 2017; 98: 2578-89. e4.
DOI URL |
| [17] | Ling Shu Jing. Beijing: People' s Medical Publishing House, 2005: 22-4. |
| [18] | Liu J, Chen LF, Zhou J, Wang CY, Fang JQ. Efficacy on the range of motion of the lower limbs in patients of post-stroke spasmodic paralysis regulated with multi-directional stimulation technique. Zhong Guo Zhen Jiu 2016; 36: 1013-7. |
| [19] | Sui YJ, Ma TM, Bian D, Tian H. Clinical observation of post-stroke upper limb spasmodic hemiplegia treated with relaxing needling technique combined with wheat-grain moxibustion. Zhong Guo Zhen Jiu 2015; 35: 423-7. |
| [20] |
Wang JX, Ma LX, Mu JD, et al. Anti-spastic effect induced by waggle needling correlates with KCC2-GABAA pathway in post-stroke spasticity rats. Neurosci Lett 2021; 750: 135810.
DOI URL |
| [21] |
Fang JQ, Chen LF, Ma RJ, et al. Comprehensive rehabilitation with integrative medicine for subacute stroke: a multicenter randomized controlled trial. Sci Rep 2016; 6: 25850.
DOI PMID |
| [22] |
Ratmansky M, Levy A, Messinger A, et al. The effects of acupuncture on cerebral blood flow in post-stroke patients: a randomized controlled trial. J Altern Complement Med 2016; 22: 33-7.
DOI URL |
| [23] |
Moon SK, Kwon S, Cho SY, et al. Effects of GB34 acupuncture on hyperventilation-induced carbon dioxide reactivity and cerebral blood flow velocity in the anterior and middle cerebral arteries of normal subjects. Acupunct Med 2019; 37: 277-82.
DOI URL |
| [24] |
Chen X, Zhang H, Zou Yh. A functional magnetic resonance imaging study on the effect of acupuncture at GB34 (Yanglingquan) on motor-related network in hemiplegic patients. Brain Res 2015; 1601: 64-72.
DOI PMID |
| [25] |
Ning YZ, Li KS, Fu CH, et al. Enhanced functional connectivity between the bilateral primary motor cortices after acupuncture at Yanglingquan (GB34) in right-hemispheric subcortical stroke patients: a resting-state fMRI study. Front Hum Neurosci 2017; 11: 178.
DOI PMID |
| [26] |
Longa EZ, Weinstein PR, Carlson S, Cummins R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 1989; 20: 84-91.
DOI PMID |
| [27] |
Wieters F, Weiss Lucas C, Gruhn M, Büschges A, Fink GR, Aswendt M. Introduction to spasticity and related mouse models. Exp Neurol 2021; 335: 113491.
DOI URL |
| [28] |
Sunnerhagen KS. Predictors of spasticity after stroke. Curr Phys Med Rehabil Rep 2016; 4: 182-5.
DOI URL |
| [29] |
Li S, Francisco GE. New insights into the pathophysiology of post-stroke spasticity. Front Hum Neurosci 2015; 9:192.
DOI PMID |
| [30] |
Bieber M, Gronewold J, Scharf AC, et al. Validity and reliability of neurological scores in mice exposed to middle cerebral artery occlusion. Stroke 2019; 50: 2875-82.
DOI PMID |
| [31] |
Kayaci S, Aydin MD, Ozoner B, Cakir T, Bas O, Sipal S. Relationship between subarachnoid and central canal hemorrhage and spasticity: a first experimental study. J Spinal Cord Med 2021; 44: 748-56.
DOI |
| [32] |
Hu XM, De Silva TM, Chen J, Faraci FM. Cerebral vascular disease and neurovascular injury in ischemic stroke. Circ Res 2017; 120: 449-71.
DOI PMID |
| [33] |
Cuccione E, Padovano G, Versace A, Ferrarese C, Beretta S. Cerebral collateral circulation in experimental ischemic stroke. Exp Transl Stroke Med 2016; 8: 2.
DOI PMID |
| [34] |
Miao PF, Wang CH, Li P, et al. Altered gray matter volume, cerebral blood flow and functional connectivity in chronic stroke patients. Neurosci Lett 2018; 662: 331-8.
DOI PMID |
| [35] |
Zhu W, Ye Y, Liu Y, et al. Mechanisms of acupuncture therapy for cerebral ischemia: an evidence-based review of clinical and animal studies on cerebral ischemia. J Neuroimmune Pharmacol 2017; 12: 575-92.
DOI URL |
| [36] |
Chavez LM, Huang SS, MacDonald I, Lin JG, Lee YC, Chen YH. Mechanisms of acupuncture therapy in ischemic stroke rehabilitation: a literature review of basic studies. Int J Mol Sci 2017; 18: 2270.
DOI URL |
| [37] |
Howard VJ, McDonnell MN. Physical activity in primary stroke prevention: just do it! Stroke 2015; 46: 1735-9.
DOI PMID |
| [38] |
Pianta S, Lee JY, Tuazon JP, et al. A short bout of exercise prior to stroke improves functional outcomes by enhancing angiogenesis. Neuromolecular Med 2019; 21: 517-28.
DOI |
| [39] |
Jembrek MJ, Vlainic J. GABA receptors: pharmacological potential and pitfalls. Curr Pharm Des 2015; 21: 4943-59.
DOI URL |
| [40] | Li J, Chen LT, Guo F, Han XH. The effects of GABAergic system under cerebral ischemia: spotlight on cognitive function. Neural Plast 2020; 2020: 8856722. |
| [41] |
Miller PS, Aricescu AR. Crystal structure of a human GABAA receptor. Nature 2014; 512: 270-5.
DOI |
| [42] |
Schulte JT, Wierenga CJ, Bruining H. Chloride transporters and GABA polarity in developmental, neurological and psychiatric conditions. Neurosci Biobehav Rev 2018; 90: 260-71.
DOI PMID |
| [43] |
Chen T, Zhang WW, Chu YX, Wang YQ. Acupuncture for pain management: molecular mechanisms of action. Am J Chin Med 2020; 48: 793-811.
DOI URL |
| [44] |
Li S. Spasticity, motor recovery, and neural plasticity after stroke. Front Neurol 2017; 8: 120.
DOI PMID |
| [45] |
Attwell D, Buchan AM, Charpak S, Lauritzen M, Macvicar BA, Newman EA. Glial and neuronal control of brain blood flow. Nature 2010; 468: 232-43.
DOI |
| [46] |
Hao JJ, Jiang KY, Zhang X, Wu M, Ma BT. "Glu/GABA-Gln" metabolic loop abnormalities in iminodipropionitrile (IDPN)-induced dyskinetic syndrome. Neurol Sci 2021; 42: 4697-706.
DOI |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

