Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (2): 285-293.DOI: 10.19852/j.cnki.jtcm.2026.02.002
• Original Articles • Previous Articles Next Articles
Gut microbiota-mediated short-chain fatty acids contribute to the protective effects of Xiaoxuming decoction (小续命汤) against lipopolysaccharide-induced acute lung injury
XIANG Yijin1( ), YANG Zhigang2, GONG Shaomin3, LI Xiangting1, CAI Min1
), YANG Zhigang2, GONG Shaomin3, LI Xiangting1, CAI Min1
- 1
Department of Integrative Medicine ,Zhongshan Hospital, Fudan University Shanghai 200032, China
2Department of Neurosurgery ,Zhongshan Hospital, Fudan University Shanghai 200032, China
3Department of Nephrology ,Zhongshan Hospital, Fudan University Shanghai 200032, China
-
Received:2024-09-26Accepted:2025-06-24Online:2026-04-15Published:2026-04-04 -
Contact:XIANG Yijin, Department of Integrative Medicine, Zhongshan Hospital, Fudan University, Shanghai 200032, China. qqdemi728@126.com; Telephone: +86-21-64041990
Cite this article
XIANG Yijin, YANG Zhigang, GONG Shaomin, LI Xiangting, CAI Min. Gut microbiota-mediated short-chain fatty acids contribute to the protective effects of Xiaoxuming decoction (小续命汤) against lipopolysaccharide-induced acute lung injury[J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 285-293.
share this article
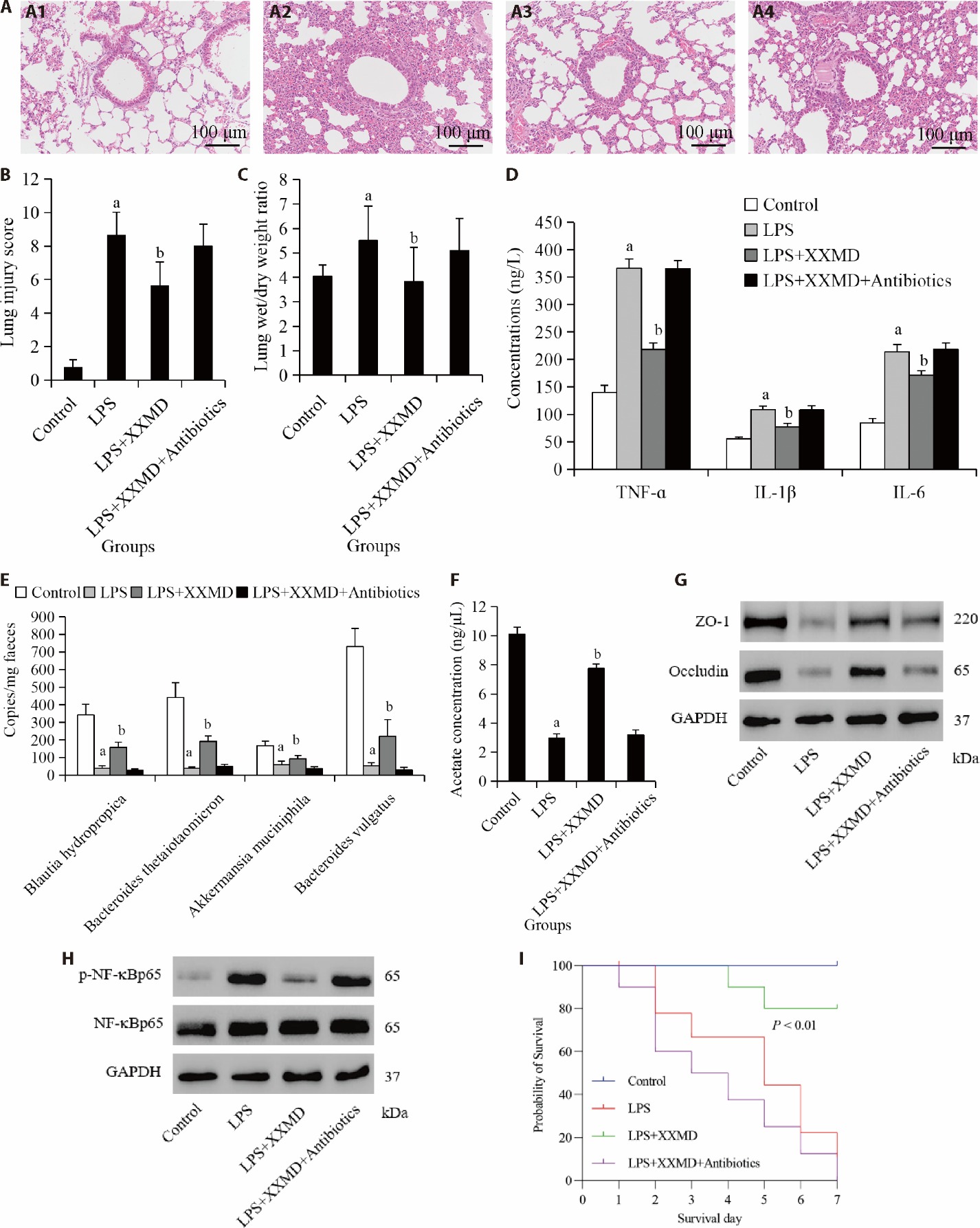
Figure 1 The gut microbiota contributes to the protective effects of XXMD against LPS-induced acute lung injury A: lung tissue HE staining was used to analyze the pathological changes of lung injury; B: histological injury score of the lungs; C: lung wet-to-dry weight ratio; D: ELISA was used to detect the content of TNF-α, IL-1β, and IL-6 in the BALF of the mouse; E: qRT-PCR was used to detect the levels of acetate-producing bacteria — Blautia hydropropica, Bacteroides thetaiotaomicron, Akkermansia muciniphila, Bacteroides vulgatus in feces; F: biochemical detection of the fecal acetate content; G: Western blot detection of lung tissue ZO-1 and occludin expression; H: Western blot detection of lung tissue p-NF-κBp65 and NF-κBp65 expression; I: seven-day survival rates; A1: control group; A2: LPS group; A3: LPS + XXMD group; A4: LPS + XXMD + Antibiotics group. Control group: give purified water 0.3 mL/d; LPS group: give 5 mg/kg LPS; LPS + XXMD group: give 5 mg/kg LPS and 100 mg/kg XXMD (twice a day). LPS + XXMD + Antibiotics group: give 5 mg/kg LPS, 100 mg/kg XXMD (twice a day) and antibiotics (120 units/mL polymyxin B and 0.6 mg/mL neomycin). XXMD: Xiaoxuming decoction; LPS: lipopolysaccharide; HE: hematoxylin and eosin; ELISA: enzyme-linked immunosorbent assay; TNF-α: tumor necrosis factor-alpha; IL-1β: interleukin-1beta; BALF: bronchoalveolar lavage fluid; qRT-PCR: quantitative real time-PCR; ZO-1: zonula occludens-1; p-NF-κB: phosphorylated nuclear factor-kappa B. Statistical analyses were measured by using one-way analysis of variance to analyze the differences between the groups. Data are presented as the mean ± standard deviation (n = 8). Compared with control group, aP < 0.01; compared with LPS group, bP < 0.01.
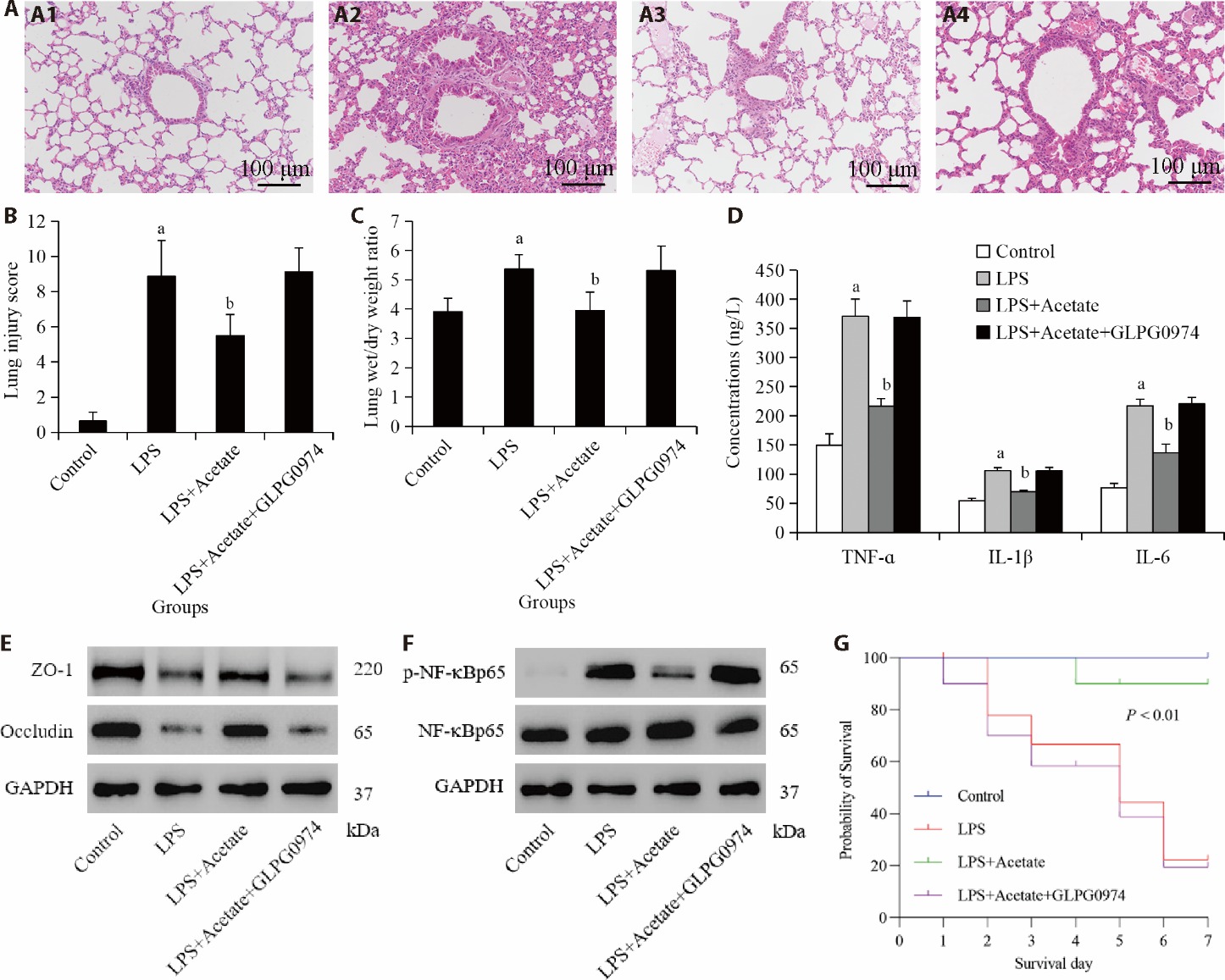
Figure 2 Acetate inhibits LPS-induced ALI via SCFA receptor GPR43 A: lung tissue HE staining was used to analyze the pathological changes of lung injury; B: histological injury score of the lungs; C: Lung wet-to-dry weight ratio; D: ELISA was used to detect the content of TNF-α, IL-1β, and IL-6 in the BALF of the mouse; E: Western blot detection of lung tissue ZO-1 and occludin expression; F: Western blot detection of lung tissue p-NF-κBp65 and NF-κBp65 expression; G: seven-day survival rates. A1: control group; A2: LPS group; A3: LPS + Acetate group; A4: LPS + Acetate + GLPG0974 group. Control group: give purified water 0.3 mL/d; LPS group: give 5 mg/kg LPS; LPS + Acetate group: give 5 mg/kg LPS and 500 mg/kg acetate (twice a day); LPS + Acetate + GLPG0974 group: give 5 mg/kg LPS, 500 mg/kg acetate (twice a day) and GLPG0974 (1 mg·kg-1·d-1). LPS: lipopolysaccharide; ALI: acute lung injury; SCFA: short-chain fatty acid; GPR43: G-protein coupled receptor 43; HE: hematoxylin and eosin; ELISA: enzyme-linked immunosorbent assay; TNF-α: tumor necrosis factor-alpha; IL-1β: interleukin-1 beta; BALF: bronchoalveolar lavage fluid; ZO-1: zonula occludens-1; p-NF-κB: phosphorylated nuclear factor-kappa B. Statistical analyses were measured by using one-way analysis of variance to analyze the differences between the groups. Data are presented as the mean ± standard deviation (n = 8). Compared with control group, aP < 0.01; compared with LPS group, bP < 0.01.
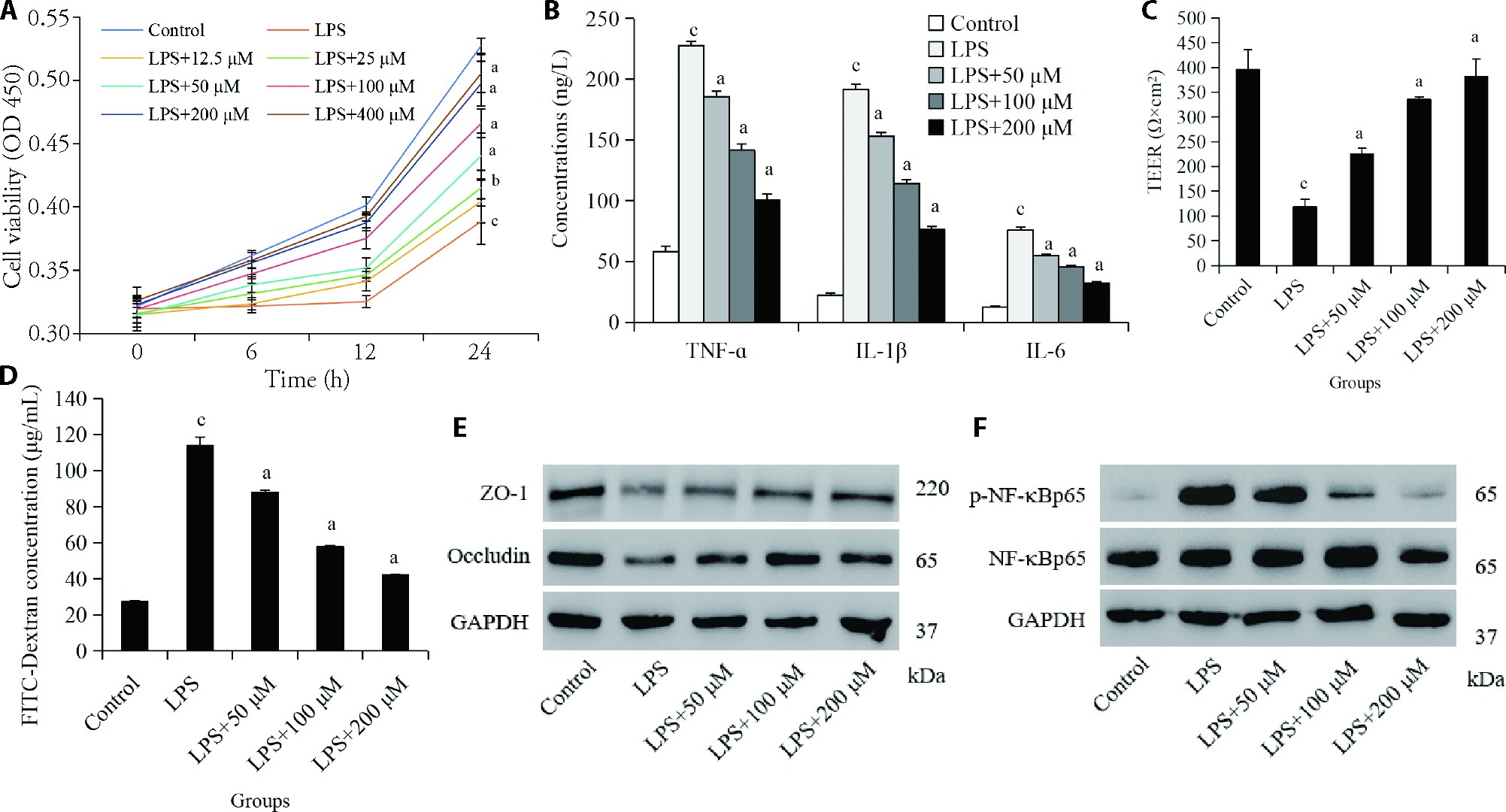
Figure 3 Acetate inhibits LPS-mediated cell viability and permeability in HPAEpiCs A: HPAEpiCs were pretreated with acetate at concentrations of 0, 12.5, 25, 50, 100, 200, and 400 μM for 24 h, followed by LPS treatment at a concentration of 10 mg/L, and CCK-8 was used to detect cell viability at 0, 6, 12, and 24 h; B: ELISA was used to detect TNF-α, IL-1β, and IL-6 expression; C: the TEER analysis; D: FITC leakage experiments were used to analyze cell permeability; E: the Western blot analysis was used to detect ZO-1 and occludin expression; F: the Western blot analysis was used to detect p-NF-κBp65 and NF-κBp65 expression. Control group: untreated HPAEpiCs; LPS group: HPAEpiCs treated with 10 mg/L LPS; LPS + 12.5 μM group: HPAEpiCs treated with 10 mg/L LPS and 12.5 μM acetate; LPS + 25 μM group: HPAEpiCs treated with 10 mg/L LPS and 25 μM acetate; LPS + 50 μM group: HPAEpiCs treated with 10 mg/L LPS and 50 μM acetate; LPS + 100 μM group: HPAEpiCs treated with 10 mg/L LPS and 100 μM acetate; LPS + 200 μM group: HPAEpiCs treated with 10 mg/L LPS and 200 μM acetate; LPS + 400 μM group: HPAEpiCs treated with 10 mg/L LPS and 400 μM acetate. LPS: lipopolysaccharide; CCK-8: cell counting kit-8; ELISA: enzyme-linked immunosorbent assay; TNF-α: tumor necrosis factor-alpha; IL-1β: interleukin-1 beta; TEER: transepithelial electrical resistance; FITC: fluorescein isothiocyanate; ZO-1: zonula occludens-1; p-NF-κB: phosphorylated nuclear factor-kappa B. Statistical analyses were measured by using one-way analysis of variance to analyze the differences between the groups. Data are presented as the mean ± standard deviation (n = 3). Compared with LPS group, aP < 0.01 and bP < 0.05; compared with control group, cP < 0.01.
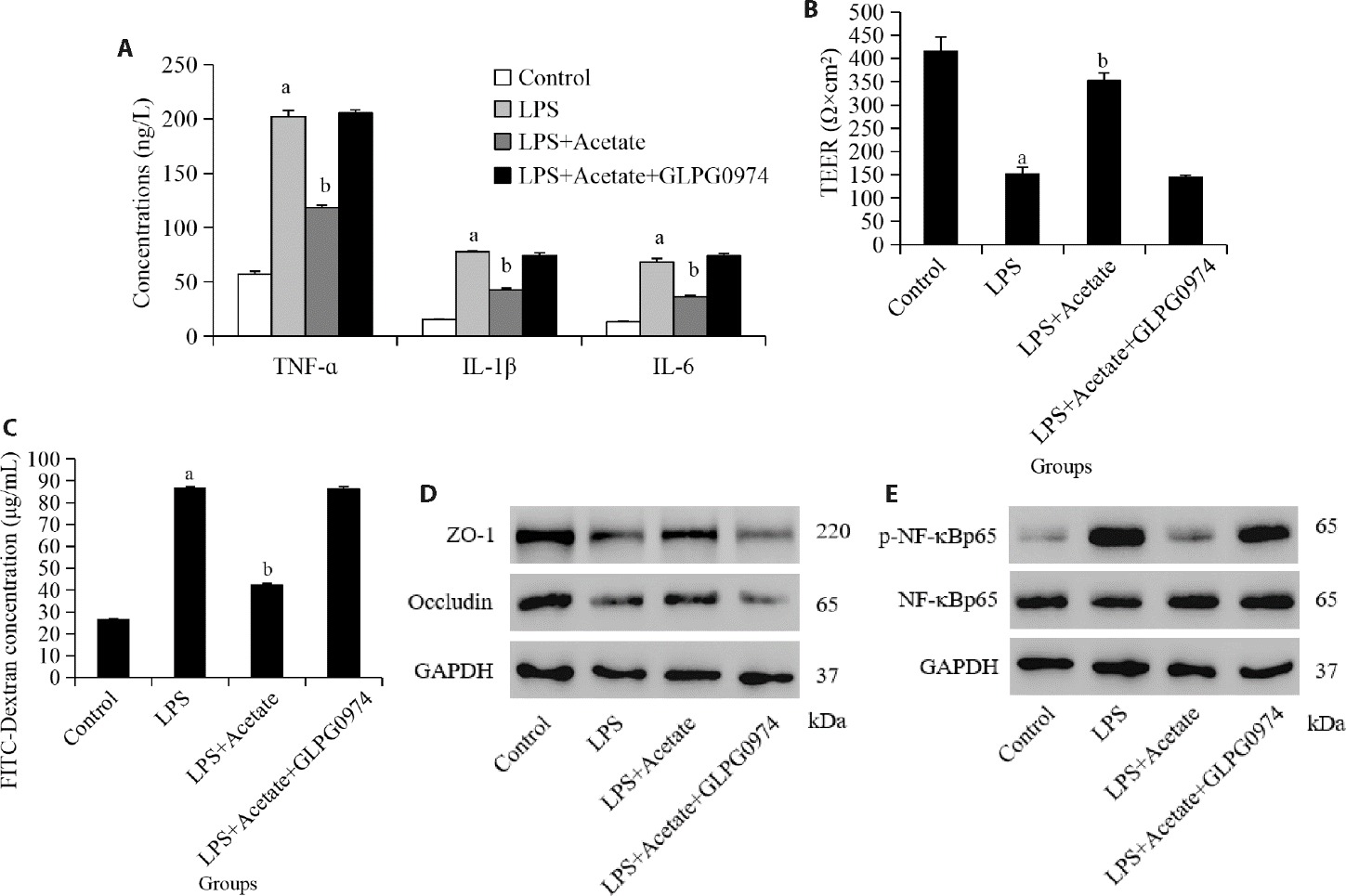
Figure 4 Acetate inhibits LPS-mediated cell viability and permeability in HPAEpiCs via GPR43 A: ELISA was used to detect TNF-α, IL-1 β, and IL-6 expression; B: The TEER analysis; C: the FITC leakage assay was used to analyze cell permeability; D: the Western blot analysis was used to detect ZO-1 and occludin expression; E: the Western blot analysis was used to detect p-NF-κBp65 and NF-κBp65 expression. Control group: untreated HPAEpiCs; LPS group: HPAEpiCs treated with 10 mg/L LPS; LPS + Acetate group: HPAEpiCs treated with 10 mg/L LPS and 200 μM acetate; LPS + Acetate + GLPG0974 group: HPAEpiCs treated with 10 mg/L LPS, 200 μM acetate and 100 nM GLPG0974. LPS: lipopolysaccharide; GPR43: G-protein coupled receptor 43; ELISA: enzyme-linked immunosorbent assay; TNF-α: tumor necrosis factor-alpha; IL-1β: interleukin-1beta; TEER: transepithelial electrical resistance; FITC: fluorescein isothiocyanate; ZO-1: zonula occludens-1; p-NF-κB: phosphorylated nuclear factor-kappa B. Statistical analyses were measured by using one-way analysis of variance to analyze the differences between the groups. Data are presented as the mean ± standard deviation (n = 3). Compared with control group, aP < 0.01; compared with LPS group, bP < 0.01.
| 1. |
Parveen S, Khan KUR, Iqbal SM, et al. Exploration of the anti-inflammatory potential of Polygonum bistorta L.: protection against LPS-induced acute lung injury in rats via NF-ĸβ pathway inhibition. Front Pharmacol 2024; 15: 1500085.
DOI URL |
| 2. |
Jian YJ, Lü Q, Du L, Lei CC, Zhi LP, Liu XH. Discovery of a novel pyrimidine derivative for treatment of acute lung injury through reducing oxidative stress and inflammatory response. RSC Med Chem 2025; 16: 1441-58.
DOI URL |
| 3. | Ren L, Hai Y, Yang X, Luo XQ. Yemazhui (Herba Eupatorii Lindleyani) ameliorates lipopolysaccharide-induced acute lung injury modulation of the toll-like receptor 4/nuclear factor kappa-B/nod-like receptor family pyrin domain-containing 3 protein signaling pathway and intestinal flora in rats. J Tradit Chin Med 2024; 44: 303-14. |
| 4. |
Guo J, Zhu J, Wang Q, Wang J, Jia Y. Comparative efficacy of seven kinds of Chinese Medicine injections in acute lung injury and acute respiratory distress syndrome: a network Meta-analysis of randomized controlled trials. Front Pharmacol 2021; 12: 627751.
DOI URL |
| 5. |
Chen YP, Wang KX, Cai JQ, et al. Detecting key functional components group and speculating the potential mechanism of Xiao-xu-ming decoction in treating stroke. Front Cell Dev Biol 2022; 10: 753425.
DOI URL |
| 6. |
Li DH, Su YF, Sun CX, Fan HF, Gao WJ. A network pharmacology-based identification study on the mechanism of Xiao-xu-ming decoction for cerebral ischemic stroke. Evid Based Complement Alternat Med 2020; 2020: 2507074.
DOI URL |
| 7. | Xiang Y, Cai M, Li X, Bao X, Cai D. Protective effect of Xiao-xu-ming decoction-mediated inhibition of ROS/NLRP3 axis on lipopolysaccharide-induced acute lung injury in vitro and in vivo. Evid Based Complement Alternat Med 2021; 2021: 8257495. |
| 8. |
Wang J, Xue X, Zhao X, et al. Forsythiaside A alleviates acute lung injury by inhibiting inflammation and epithelial barrier damages in lung and colon through PPAR-γ/RXR-α complex. J Adv Res 2024; 60: 183-200.
DOI URL |
| 9. |
Xie L, Wang L, Liao Y, et al. Therapeutic potential of short-chain fatty acids for acute lung injury: a systematic review and Meta-analysis of preclinical animal studies. Front Nutr 2024; 11: 1528200.
DOI URL |
| 10. |
Hung KY, Wu SY, Pao HP, Liao WI, Chu SJ. Acetate, a gut bacterial product, ameliorates ischemia-reperfusion induced acute lung injury in rats. Int Immunopharmacol 2022; 111: 109136.
DOI URL |
| 11. |
Shao S, Wang D, Zheng W, et al. A unique polysaccharide from Hericium erinaceus mycelium ameliorates acetic acid-induced ulcerative colitis rats by modulating the composition of the gut microbiota, short chain fatty acids levels and GPR41/43 respectors. Int Immunopharmacol 2019; 71: 411-22.
DOI PMID |
| 12. |
Wu Z, Qian S, Zhao L, et al. Metabolomics-based study of the potential interventional effects of Xiao-xu-ming decoction on cerebral ischemia/reperfusion rats. J Ethnopharmacol 2022; 295: 115379.
DOI URL |
| 13. |
Wang Z, Liu J, Li F, et al. The gut-lung axis in severe acute pancreatitis-associated lung injury: the protection by the gut microbiota through short-chain fatty acids. Pharmacol Res 2022; 182: 106321.
DOI URL |
| 14. |
Liu S, Su X, Pan P, et al. Neutrophil extracellular traps are indirectly triggered by lipopolysaccharide and contribute to acute lung injury. Sci Rep 2016; 6: 37252.
DOI PMID |
| 15. |
Yehya N, Smith L, Thomas NJ, et al. Definition, incidence, and epidemiology of pediatric acute respiratory distress syndrome: from the second pediatric acute lung injury consensus conference. Pediatr Crit Care Med 2023; 24: S87-98.
DOI URL |
| 16. |
Lee J, d'Aigle J, Atadja L, et al. Gut microbiota-derived short-chain fatty acids promote poststroke recovery in aged mice. Circ Res 2020; 127: 453-65.
DOI PMID |
| 17. | Ashley SL, Sjoding MW, Popova AP, et al. Lung and gut microbiota are altered by hyperoxia and contribute to oxygen-induced lung injury in mice. Sci Transl Med 2020; 12: eaau9959. |
| 18. |
Ye J, Guan M, Lu Y, Zhang D, Li C, Zhou C. Arbutin attenuates LPS-induced lung injury via Sirt1/ Nrf2/ NF-kappaBp65 pathway. Pulm Pharmacol Ther 2019; 54: 53-9.
DOI URL |
| 19. |
Hildebrand CB, Lichatz R, Pich A, et al. Short-chain fatty acids improve inflamm-aging and acute lung injury in old mice. Am J Physiol Lung Cell Mol Physiol 2023; 324: L480-92.
DOI URL |
| 20. |
Wu Z, Shen J, Xu Q, et al. Epigallocatechin-3-gallate improves intestinal gut microbiota homeostasis and ameliorates clostridioides difficile infection. Nutrients 2022; 14: 3756.
DOI URL |
| 21. |
Xiong RG, Zhou DD, Wu SX, et al. Health benefits and side effects of short-chain fatty acids. Foods 2022; 11: 2863.
DOI URL |
| 22. |
Kibbie JJ, Dillon SM, Thompson TA, Purba CM, McCarter MD, Wilson CC. Butyrate directly decreases human gut lamina propria CD4 T cell function through histone deacetylase (HDAC) inhibition and GPR43 signaling. Immunobiology 2021; 226: 152126.
DOI URL |
| 23. |
Wen C, Xie T, Pan K, et al. Acetate attenuates perioperative neurocognitive disorders in aged mice. Aging 2020; 12: 3862-79.
DOI URL |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

