Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (2): 294-305.DOI: 10.19852/j.cnki.jtcm.2026.02.003
• Original Articles • Previous Articles Next Articles
Xiaotan Sanjie recipe (消痰散结方) suppressed the oncogenic potential of cancer-associated fibroblasts via the stromal-derived factor 1α/C-X-C chemokine receptor type 4 pathway in gastric cancer
ZHAO Ying1, ZHU Zhenxin2, YU Zhihong3, XIU Lijuan1, LIU Yongying1, LU Ye1, WEI Pinkang1( )
)
- 1
Department of Traditional Chinese Medicine ,Second Affiliated Hospital of Naval Medical University Shanghai 200003, China
2Department of Gastrointestinal Surgery ,Second Affiliated Hospital of Naval Medical University Shanghai 200003, China
3Cancer Institute of Integrated Chinese and Western Medicine ,Zhejiang Academy of Traditional Chinese Medicine, Tongde Hospital of Zhejiang Province Hangzhou 310012, China
-
Received:2024-10-25Accepted:2025-05-29Online:2026-04-15Published:2026-04-04 -
Contact:WEI Pinkang, Department of Traditional Chinese Medicine, Second Affiliated Hospital of Naval Medical University, Shanghai 200003, China. czzyk73_W@163.com -
Supported by:National Nature Science Foundation of China: The Role and Mechanism of Xiaotan Sanjie Decoction on Cell Migration and Invasion in Gastric Cancer based on Cancer-associated Fibroblasts Proliferation and Related Factor Expression in Tumor Microenvironment(81603524)
Cite this article
ZHAO Ying, ZHU Zhenxin, YU Zhihong, XIU Lijuan, LIU Yongying, LU Ye, WEI Pinkang. Xiaotan Sanjie recipe (消痰散结方) suppressed the oncogenic potential of cancer-associated fibroblasts via the stromal-derived factor 1α/C-X-C chemokine receptor type 4 pathway in gastric cancer[J]. Journal of Traditional Chinese Medicine, 2026, 46(2): 294-305.
share this article
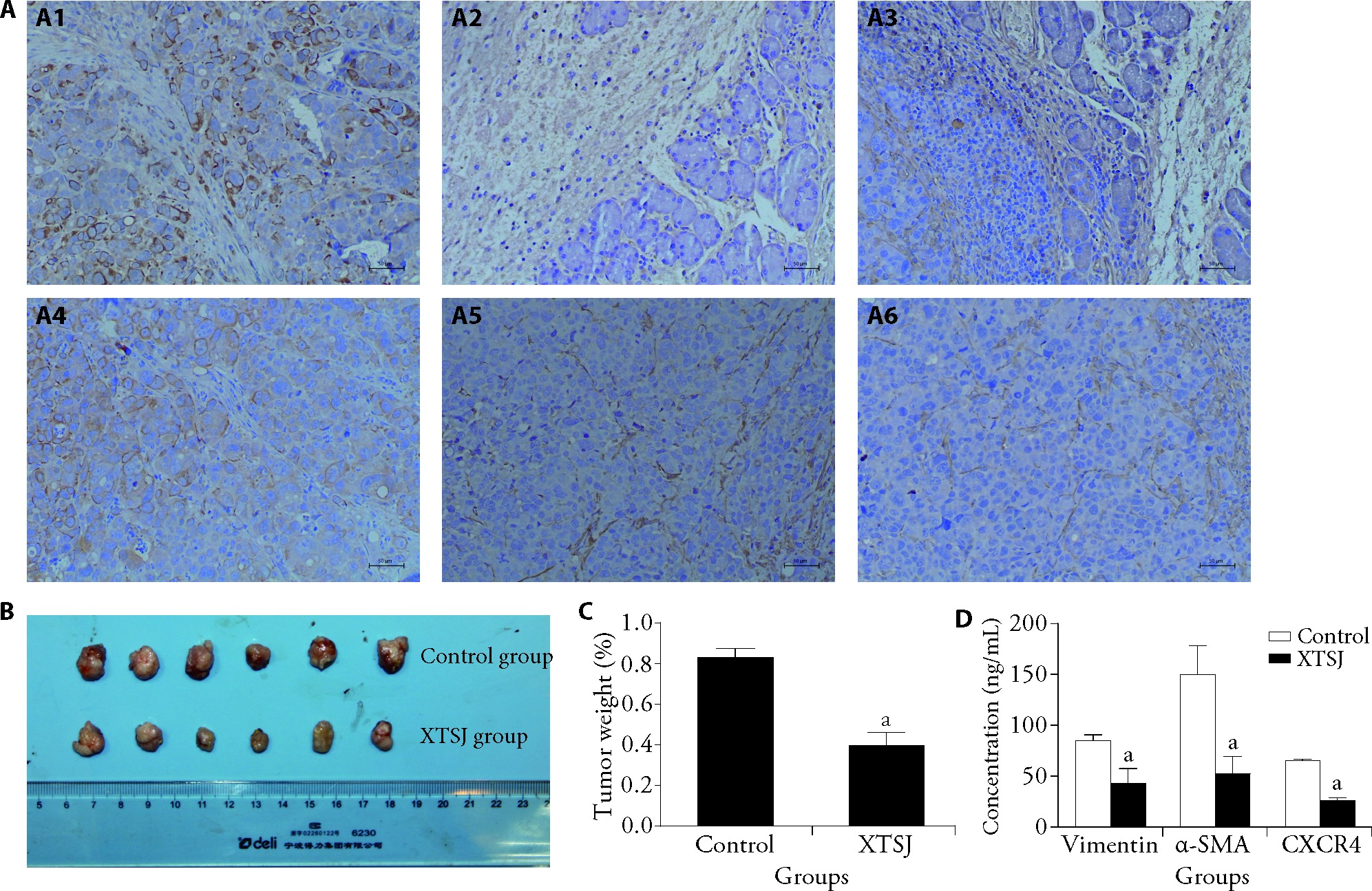
Figure 1 XTSJ inhibits the growth of GC tumors A: immunohistochemistry analysis for the expression of cytokines; A1: vimentin (control group); A2: α-SMA (control group); A3: CXCR4 (control group), A4: vimentin (XTSJ group); A5: α-SMA (XTSJ group); A6: CXCR4 (XTSJ group); B: tumor sizes; C: tumor weights; D: ELISA for vimentin, α-SMA, and CXCR4 detection. Control group: an orthotopic xenograft mouse model intragastrically administered 0.2 mL normal saline twice a day for 28 d; XTSJ group: an orthotopic xenograft mouse model intragastrically administered 0.2 mL XTSJ (1.39 g/kg) twice a day for 28 d. XTSJ: Xiaotan Sanjie; GC: gastric cancer; α-SMA: α-smooth muscle actin; CXCR4: C-X-C chemokine receptor type 4; ELISA: enzyme-linked immunosorbent assay. Statistical analyses were measured using one-way analysis of variance. Data were presented as mean ± standard deviation (n = 3). Compared with control group, aP < 0.05.
| Cytokine | Protein concentration (ng/mL) | mRNA expression | |||||
|---|---|---|---|---|---|---|---|
| NFs | CAFs | CAFs+XTSJ | NFs | CAFs | CAFs+XTSJ | ||
| IL-1β | 27845.00±1318.00 | 40919.00±621.70 | 32700.00±1711.00a | 2.82±0.61 | 5.39±0.44 | 4.72±0.15a | |
| IL-6 | 19.34±0.91 | 26.13±0.04 | 21.51±0.37a | 1.58±0.57 | 1.74±0.16 | 1.27±0.51a | |
| VEGF | 2597.00±866.40 | 3830±579.20 | 3387.00±624.50a | 3.03±1.46 | 6.33±1.84 | 4.98±0.61a | |
| SDF-1α | 26.07±1.11 | 44.57±0.21 | 28.81±0.83a | 4.54±0.57 | 8.33±0.18 | 4.90±0.10a | |
Table 1 XTSJ inhibits IL-1β, IL-6, SDF-1α, and VEGF secretion by CAFs ($ \bar{x} \pm s$)
| Cytokine | Protein concentration (ng/mL) | mRNA expression | |||||
|---|---|---|---|---|---|---|---|
| NFs | CAFs | CAFs+XTSJ | NFs | CAFs | CAFs+XTSJ | ||
| IL-1β | 27845.00±1318.00 | 40919.00±621.70 | 32700.00±1711.00a | 2.82±0.61 | 5.39±0.44 | 4.72±0.15a | |
| IL-6 | 19.34±0.91 | 26.13±0.04 | 21.51±0.37a | 1.58±0.57 | 1.74±0.16 | 1.27±0.51a | |
| VEGF | 2597.00±866.40 | 3830±579.20 | 3387.00±624.50a | 3.03±1.46 | 6.33±1.84 | 4.98±0.61a | |
| SDF-1α | 26.07±1.11 | 44.57±0.21 | 28.81±0.83a | 4.54±0.57 | 8.33±0.18 | 4.90±0.10a | |
| Cytokine | Protein concentration (ng/L) | mRNA expression | |||||
|---|---|---|---|---|---|---|---|
| Control (n = 6) | XTSJ (n = 6) | P value | Control (n = 6) | XTSJ (n = 6) | P value | ||
| IL-1β | 64.92±5.28 | 50.75±4.77 | 0.006 | 8.85±3.11 | 6.45±1.66 | 0.0067 | |
| IL-6 | 115.60±21.64 | 85.59±8.41 | 0.0101 | 2.94±0.96 | 1.86±1.08 | 0.0036 | |
| VEGF | 404.80±45.24 | 262.30±30.65 | 0.001 | 1.57±0.56 | 0.92±0.43 | 0.0004 | |
| SDF-1α | 787.10±72.38 | 448.20±35.22 | 0.001 | 1.24±0.41 | 0.71±0.24 | 0.0001 | |
Table 2 XTSJ inhibits the secretion of IL-1β, VEGF, IL-6, and SDF-1α from GC tumors ($ \bar{x} \pm s$)
| Cytokine | Protein concentration (ng/L) | mRNA expression | |||||
|---|---|---|---|---|---|---|---|
| Control (n = 6) | XTSJ (n = 6) | P value | Control (n = 6) | XTSJ (n = 6) | P value | ||
| IL-1β | 64.92±5.28 | 50.75±4.77 | 0.006 | 8.85±3.11 | 6.45±1.66 | 0.0067 | |
| IL-6 | 115.60±21.64 | 85.59±8.41 | 0.0101 | 2.94±0.96 | 1.86±1.08 | 0.0036 | |
| VEGF | 404.80±45.24 | 262.30±30.65 | 0.001 | 1.57±0.56 | 0.92±0.43 | 0.0004 | |
| SDF-1α | 787.10±72.38 | 448.20±35.22 | 0.001 | 1.24±0.41 | 0.71±0.24 | 0.0001 | |
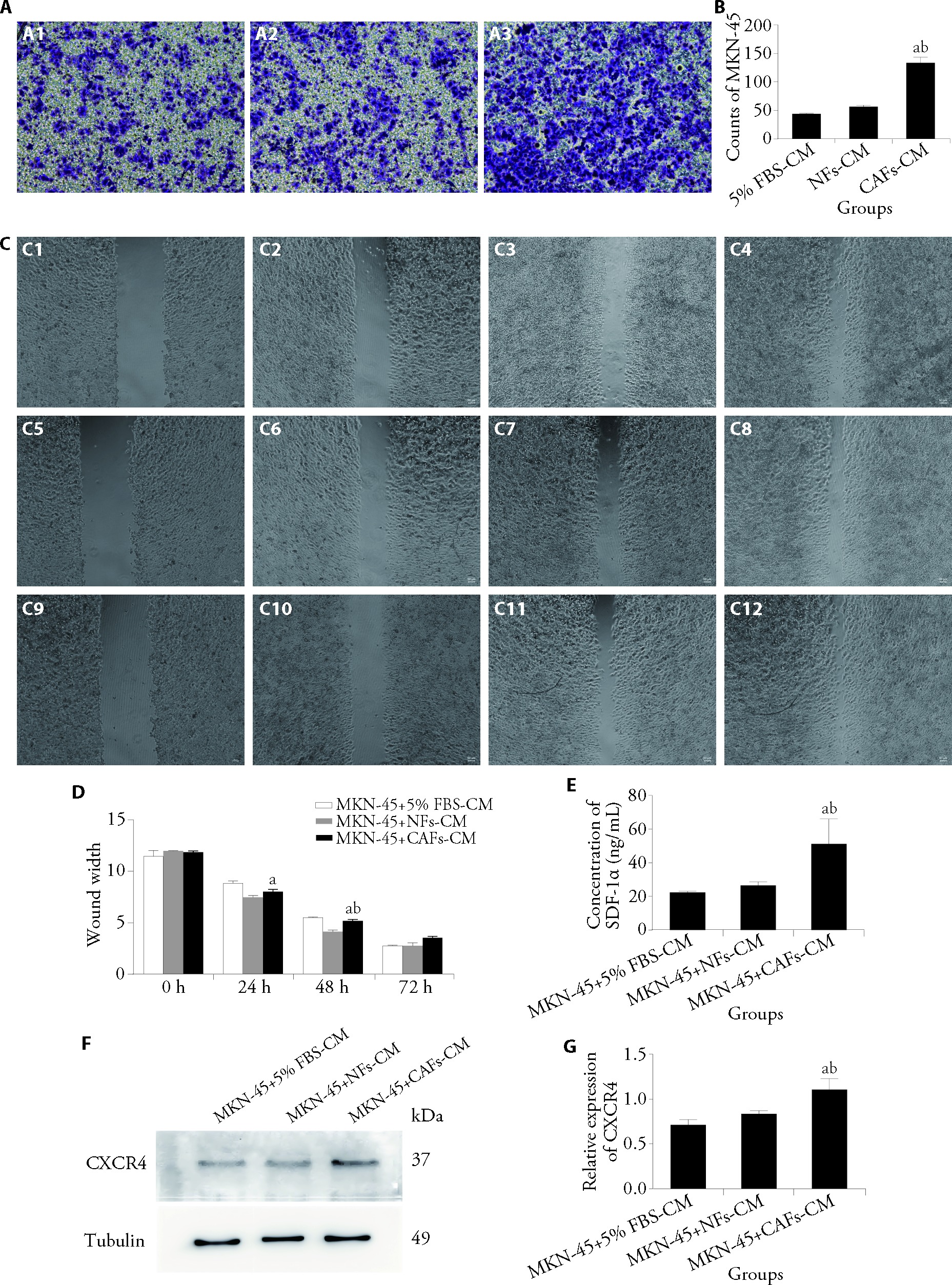
Figure 2 CAFs increase the migratory and invasive abilities of human GC MKN-45 cells A: cell invasion was assessed using the Transwell assay; A1: MKN-45 + 5% FBS-CM group; A2: MKN-45 + NFs-CM group; A3: MKN-45 + CAFs-CM group; B: quantitative analysis of cell invasion; C: cell migration was assessed using the wound healing assay; C1: MKN-45 + 5% FBS-CM group at 0 h; C2: MKN-45 + 5% FBS-CM group at 24 h; C3: MKN-45 + 5% FBS-CM group at 48 h; C4: MKN-45 + 5% FBS-CM group at 72 h; C5: MKN-45 + NFs-CM group at 0 h; C6: MKN-45 + NFs-CM group at 24 h; C7: MKN-45 + NFs-CM group at 48 h; C8: MKN-45 + NFs-CM group at 72 h; C9: MKN-45 + CAFs-CM group at 0 h; C10: MKN-45 + CAFs-CM group at 24 h; C11: MKN-45 + CAFs-CM group at 48 h; C12: MKN-45 + CAFs-CM group at 72 h; D: quantitative analysis of cell migration; E: analysis of SDF-1α levels using ELISA; F: expression of CXCR4 was examined by WB; G: relative protein expression levels of CXCR4. MKN-45 + 5% FBS-CM group: MKN-45 cells cultured with conditioned medium from 5% FBS-treated cells; MKN-45 + NFs-CM group: MKN-45 cells cultured with conditioned medium from normal fibroblasts; MKN-45 + CAFs-CM group: MKN-45 cells cultured with conditioned medium from cancer-associated fibroblasts. GC: gastric cancer; FBS: fetal bovine serum; CM: conditioned medium; NFs: normal fibroblasts; CAFs: cancer-associated fibroblasts; SDF-1α: stromal-derived factor 1α; CXCR4: C-X-C chemokine receptor type 4. Statistical analyses were measured using one-way analysis of variance. Data were presented as mean ± standard deviation (n = 3). Compared with MKN-45 + 5% FBS-CM group, aP < 0.01; compared with MKN-45 + NFs-CM group, bP < 0.05.
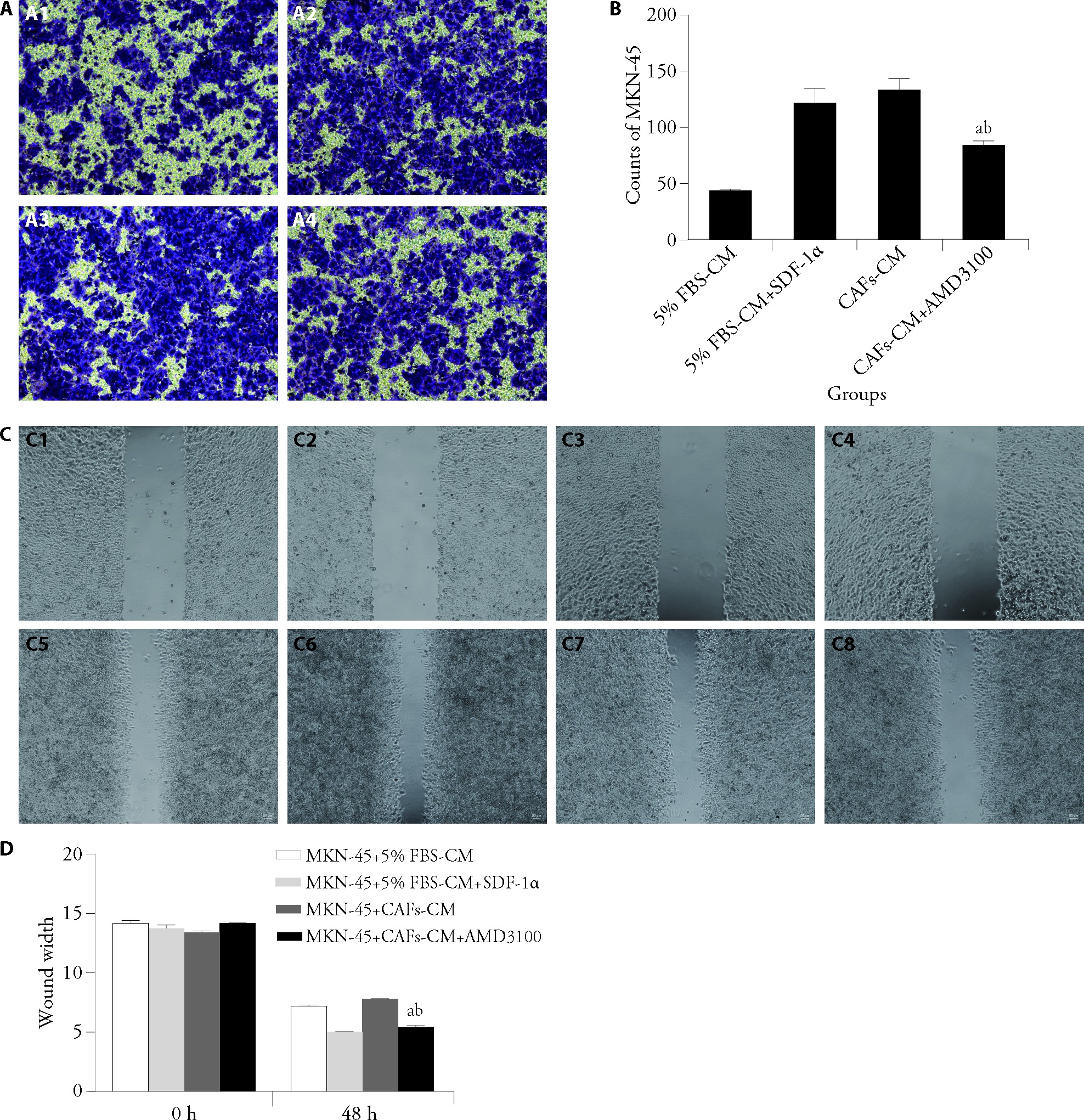
Figure 3 Role of CXCR4/SDF-1α signaling in CAF-induced invasive and migratory abilities in GC cells A: cell invasion was examined using the Transwell assay; A1: MKN-45 + 5% FBS-CM group; A2: MKN-45 + 5% FBS-CM + SDF-1α group; A3: MKN-45 + CAFs-CM group; A4: MKN-45 + CAFs-CM + AMD3100 group; B: quantitative analysis of cell invasion; C: cell migration was examined using the wound scratch assay; C1: MKN-45 + 5% FBS-CM group at 0 h; C2: MKN-45 + 5% FBS-CM + SDF-1α group at 0 h; C3: MKN-45 + CAFs-CM group at 0 h; C4: MKN-45 + CAFs-CM + AMD3100 group at 0 h; C5: MKN-45 + 5% FBS-CM group at 48 h; C6: MKN-45 + 5% FBS-CM + SDF-1α group at 48 h; C7: MKN-45 + CAFs-CM group at 48 h; C8: MKN-45 + CAFs-CM + AMD3100 group at 48 h; D: quantitative analysis of cell migration. MKN-45 + 5% FBS-CM group: MKN-45 cells cultured with 5% FBS as control; MKN-45 + 5% FBS-CM + SDF-1α group: MKN-45 cells treated with SDF-1α in 5% FBS medium; MKN-45 + CAFs-CM group: MKN-45 cells cultured with conditioned medium from cancer-associated fibroblasts; MKN-45 + CAFs-CM + AMD3100 group: MKN-45 cells cultured with CAFs-CM plus 100 nM CXCR4 inhibitor AMD3100. GC: gastric cancer; FBS: fetal bovine serum; CM: conditioned medium; SDF-1α: stromal-derived factor 1α; CAFs: cancer-associated fibroblasts. Statistical analyses were measured using one-way analysis of variance. Data were presented as mean ± standard deviation (n = 3). Compared with MKN-45 + 5% FBS-CM + SDF-1α, aP < 0.01; compared with MKN-45 + CAFs-CM, bP < 0.05.
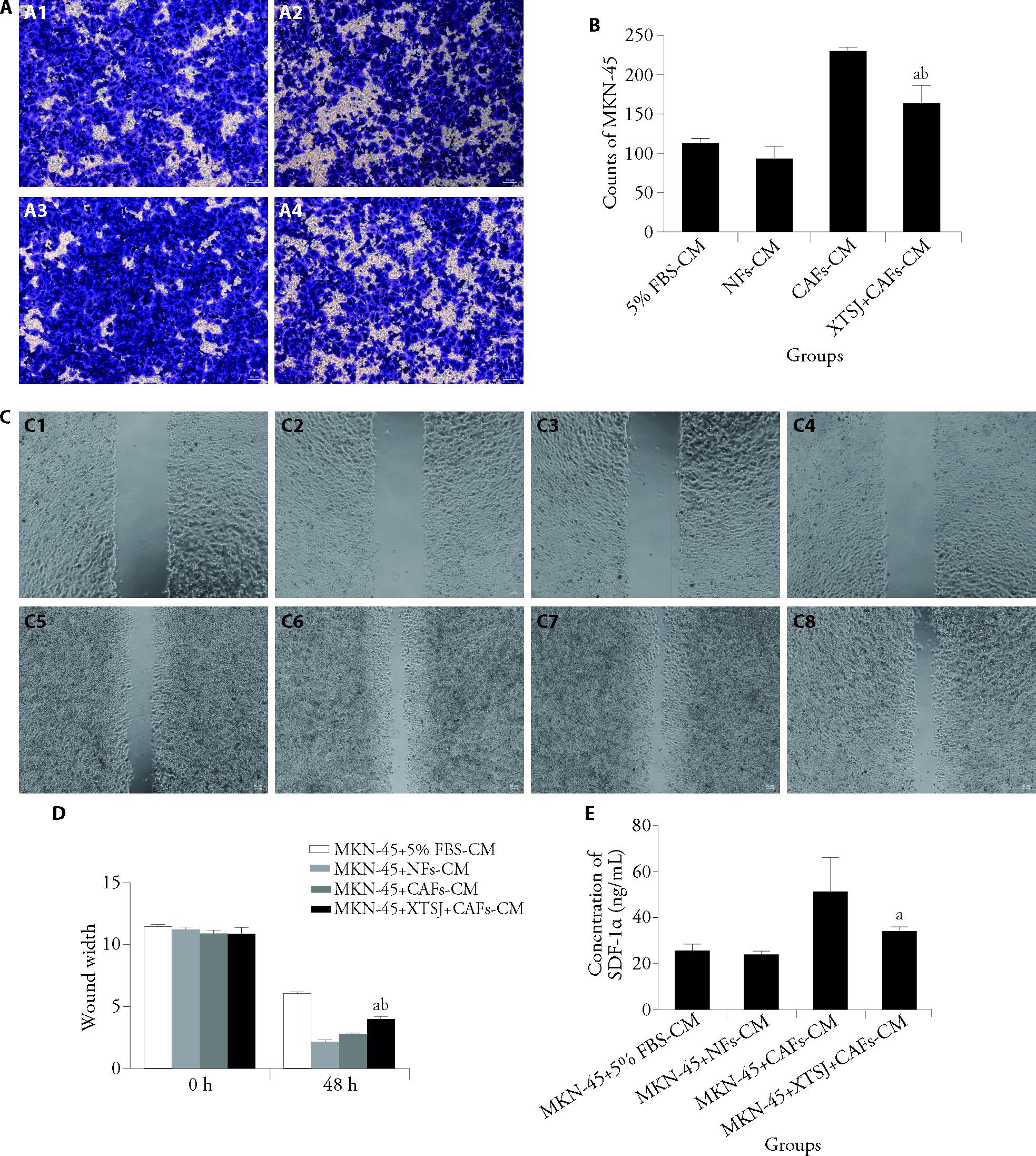
Figure 4 XTSJ suppresses CAF-mediated invasion and metastasis in GC cells A: cell invasion was determined using the Transwell assay; A1: MKN-45 + 5% FBS-CM group; A2: MKN-45 + NFs-CM group; A3: MKN-45 + CAFs-CM group; A4: MKN-45 + XTSJ + CAFs-CM group; B: quantitative analysis of cell invasion; C: cell migration was examined using the wound scratch assay; C1: MKN-45 + 5% FBS-CM group at 0 h; C2: MKN-45 + NFs-CM group at 0 h; C3: MKN-45 + CAFs-CM group at 0 h; C4: MKN-45 + XTSJ + CAFs-CM group at 0 h; C5: MKN-45 + 5% FBS-CM group at 48 h; C6: MKN-45 + NFs-CM group at 48 h; C7: MKN-45 + CAFs-CM group at 48 h; C8: MKN-45 + XTSJ + CAFs-CM group at 48 h; D: quantitative analysis of cell migration; E: SDF-1α levels were analyzed using ELISA. MKN-45 + 5% FBS-CM group: MKN-45 cells cultured with conditioned medium from 5% FBS-treated cells; MKN-45 + NFs-CM group: MKN-45 cells cultured with conditioned medium from normal fibroblasts; MKN-45 + CAFs-CM group: MKN-45 cells cultured with conditioned medium from cancer-associated fibroblasts; MKN-45 + XTSJ + CAFs-CM group: MKN-45 cells cultured with conditioned medium from cancer-associated fibroblasts treated with 10% XTSJ for 24 h. GC: gastric cancer; FBS: fetal bovine serum; CM: conditioned medium; NFs: normal fibroblasts; CAFs: cancer-associated fibroblasts; XTSJ: Xiaotan Sanjie; SDF-1α: stromal-derived factor 1α; ELISA: enzyme-linked immunosorbent assay. Statistical analyses were measured using one-way analysis of variance. Data were presented as mean ± standard deviation (n = 3). Compared with MKN-45 + NFs-CM group, aP < 0.01, compared with MKN-45 + CAFs-CM group, bP < 0.05.
| 1. |
Ajani JA, D'Amico TA, Bentrem DJ, et al. Gastric Cancer, Version 2.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 2022; 20: 167-92.
DOI URL |
| 2. |
Smyth EC, Verheij M, Allum W, et al. Gastric cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 2016; 27: v38-49.
DOI URL |
| 3. | Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021; 71: 209-49. |
| 4. | Siegel RL, Miller KD, Fuchs HE, Jemal A.Cancer statistics, 2022. CA Cancer J Clin 2022; 72: 7-33. |
| 5. |
de Visser KE, Joyce JA. The evolving tumor microenvironment: from cancer initiation to metastatic outgrowth. Cancer Cell 2023; 41: 374-403.
DOI PMID |
| 6. |
Baghban R, Roshangar L, Jahanban-Esfahlan R, et al. Tumor microenvironment complexity and therapeutic implications at a glance. Cell Commun Signal 2020; 18: 59.
DOI PMID |
| 7. |
Sahai E, Astsaturov I, Cukierman E, et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer 2020; 20: 174-86.
DOI PMID |
| 8. |
Liu T, Han C, Wang S, et al. Cancer-associated fibroblasts: an emerging target of anti-cancer immunotherapy. J Hematol Oncol 2019; 12: 86.
DOI |
| 9. |
Raskov H, Orhan A, Gaggar S, Gögenur I. Cancer-associated fibroblasts and tumor-associated macrophages in cancer and cancer immunotherapy. Front Oncol 2021; 11: 668731.
DOI URL |
| 10. |
Sun R, Sun Y, Wu C, et al. CXCR4-modified CAR-T cells suppresses MDSCs recruitment via STAT3/NF-κB/SDF-1α axis to enhance efficacy against pancreatic cancer. Mol Ther 2023; 31: 3193-209.
DOI URL |
| 11. |
Ito S, Sato T, Maeta T. Role and therapeutic targeting of SDF-1α/CXCR4 axis in multiple myeloma. Cancers (Basel) 2021; 13: 1793.
DOI URL |
| 12. | Gong J, Song Y, Xu L, et al. Upregulation of serine proteinase inhibitor clade B member 3 (SERPINB3) expression by stromal cell-derived factor (SDF-1)/CXCR4/nuclear factor kappa B (NF-κB) promotes migration and invasion of gastric cancer cells. Med Sci Monit 2020; 26: e927411. |
| 13. | Ma X, Zhang Z, Qin X, et al. Therapeutic effect of curcumol on chronic atrophic gastritis (CAG) and gastric cancer is achieved by downregulating SDF-1α/CXCR4/VEGF expression. J Oncol 2022; 2022: 3919053. |
| 14. |
Yashiro M, Kinoshita H, Tsujio G, et al. SDF1α/CXCR4 axis may be associated with the malignant progression of gastric cancer in the hypoxic tumor microenvironment. Oncol Lett 2021; 21: 38.
DOI PMID |
| 15. |
Wang X, Zhang H, He H, et al. Prognostic value of stromal cell-derived factor 1 expression in patients with gastric cancer after surgical resection. Cancer Sci 2014; 105: 1447-56.
DOI URL |
| 16. |
Sugihara H, Ishimoto T, Yasuda T, et al. Cancer-associated fibroblast-derived CXCL12 causes tumor progression in adenocarcinoma of the esophagogastric junction. Med Oncol 2015; 32: 618.
DOI PMID |
| 17. |
Gu Y, Gu W, Xie R, Chen Z, Xu T, Fei Z. Role of CXCR4 as a prognostic biomarker associated with the tumor immune microenvironment in gastric cancer. Front Cell Dev Biol 2021; 9: 654504.
DOI URL |
| 18. |
Guo F, Wang Y, Liu J, Mok SC, Xue F, Zhang W. CXCL12/CXCR4: a symbiotic bridge linking cancer cells and their stromal neighbors in oncogenic communication networks. Oncogene 2016; 35: 816-26.
DOI PMID |
| 19. |
Xu X, Chen Y, Zhang X, et al. Modular characteristics and the mechanism of Chinese medicine's treatment of gastric cancer: a data mining and pharmacology-based identification. Ann Transl Med 2021; 9: 1777.
DOI PMID |
| 20. |
Liu SH, Tang L, Zhao YN, et al. Nursing perspective of expert consensus for diagnosis and treatment of gastric cancer with integrated Traditional Chinese and Western Medicine. J Integr Nurs 2023; 5: 8-14.
DOI URL |
| 21. | Sun DZ, Ye M, Ju DW, et al. The effects of gastric cancer interstitial fluid on tumors based on Traditional Chinese Medicine 'phlegm' theory and the investigation on the mechanism through microRNA-21 regulation. J Physiol Pharmacol 2021; 72: 381-9. |
| 22. |
Liu Y, Huang T, Wang L, et al. Traditional Chinese Medicine in the treatment of chronic atrophic gastritis, precancerous lesions and gastric cancer. J Ethnopharmacol 2025; 337: 118812.
DOI URL |
| 23. |
Shi J, Wei PK. Xiaotan Sanjie decoction inhibits interleukin-8-induced metastatic potency in gastric cancer. World J Gastroenterol 2015; 21: 1479-87.
DOI URL |
| 24. |
Huang N, He HW, He YY, Gu W, Xu MJ, Liu L. Xiaotan Sanjie recipe, a compound Chinese herbal medicine, inhibits gastric cancer metastasis by regulating GnT-V-mediated E-cadherin glycosylation. J Integr Med 2023; 21: 561-74.
DOI PMID |
| 25. |
Zhou Z, Chen J, Li M, et al. Xiaotan Sanjie decoction inhibits gastric cancer cell proliferation, migration, and invasion through lncRNA-ATB and miR-200A. Biomed Res Int 2022; 2022: 7029182.
DOI URL |
| 26. |
Shi J, Lu Y, Wei P. Xiaotan Sanjie decoction inhibits angiogenesis in gastric cancer through interleukin-8-linked regulation of the vascular endothelial growth factor pathway. J Ethnopharmacol 2016; 189: 230-7.
DOI PMID |
| 27. |
Fitzgerald AA, Weiner LM. The role of fibroblast activation protein in health and malignancy. Cancer Metastasis Rev 2020; 39: 783-803.
DOI |
| 28. |
Lü Z, Li Y, Zhu D, et al. Fibroblast activation protein-alpha is a prognostic biomarker associated with ferroptosis in stomach adenocarcinoma. Front Cell Dev Biol 2022; 10: 859999.
DOI URL |
| 29. | Yasuda T, Koiwa M, Yonemura A, Akiyama T, Baba H, Ishimoto T. Protocol to establish cancer-associated fibroblasts from surgically resected tissues and generate senescent fibroblasts. STAR Protoc 2021; 2: 100553. |
| 30. | Jiu G, Dao R, Wu D, et al. Effects of herbal therapy on intestinal microbiota and serum metabolomics in different rat models of Mongolian medicine. Evid Based Complement Alternat Med 2022; 2022: 7255780. |
| 31. |
Shi J, Wei PK, Zhang S, et al. OB glue paste technique for establishing nude mouse human gastric cancer orthotopic transplantation models. World J Gastroenterol 2008; 14: 4800-4.
DOI URL |
| 32. |
Nair A, Morsy MA, Jacob S. Dose translation between laboratory animals and human in preclinical and clinical phases of drug development. Drug Dev Res 2018; 79: 373-82.
DOI URL |
| 33. | Abdel-Tawab MS, Fouad H, Othman AM, et al. Evaluation of gene expression of PLEKHS1, AADAC, and CDKN3 as novel genomic markers in gastric carcinoma. PLoS One 2022; 17: e0265184. |
| 34. | Xu X, Yu Z, Zeng S. Investigating the therapeutic mechanism of Xiaotan Sanjie formula for gastric cancer via network pharmacology and molecular docking: a review. Medicine (Baltimore) 2023; 102: e35986. |
| 35. |
Sun DZ, Wei PK, Yue XQ. Xiaotan Sanjie decoction normalizes tumor permissive microenvironment in gastric cancer (Review). Oncol Rep 2023; 49: 74.
DOI URL |
| 36. |
Rébé C, Ghiringhelli F. Interleukin-1β and cancer. Cancers (Basel) 2020; 12: 1791.
DOI URL |
| 37. |
Kumari N, Dwarakanath BS, Das A, Bhatt AN. Role of interleukin-6 in cancer progression and therapeutic resistance. Tumour Biol 2016; 37: 11553-72.
DOI URL |
| 38. |
Yang Y, Cao Y. The impact of VEGF on cancer metastasis and systemic disease. Semin Cancer Biol 2022; 86: 251-61.
DOI PMID |
| 39. |
Pasquier J, Abu-Kaoud N, Abdesselem H, et al. SDF-1alpha concentration dependent modulation of RhoA and Rac1 modifies breast cancer and stromal cells interaction. BMC Cancer 2015; 15: 569.
DOI PMID |
| 40. |
Xiang Y, Guo Z, Zhu P, Chen J, Huang Y. Traditional Chinese Medicine as a cancer treatment: modern perspectives of ancient but advanced science. Cancer Med 2019; 8: 1958-75.
DOI URL |
| 41. |
Chen F, Li J, Wang H, Ba Q. Anti-tumor effects of Chinese medicine compounds by regulating immune cells in microenvironment. Front Oncol 2021; 11: 746917.
DOI URL |
| 42. |
Hoffman RD, Li CY, He K, et al. Chinese herbal medicine and its regulatory effects on tumor related T cells. Front Pharmacol 2020; 11: 492.
DOI PMID |
| [1] | ZHANG Yang, SHI Youyang, LIU Xiaofei, YANG Rui, LI Qiong, LI Feifei, YANG Xiaojuan, WANG Yi, SUN Chenping, HAN Xianghui, LIU Sheng. Wenshen Zhuanggu Fang (温肾壮骨方) reduces breast cancer bone metastasis by regulating macrophage polarization: a bioinformatics analysis combined with experimental validation [J]. Journal of Traditional Chinese Medicine, 2025, 45(6): 1295-1306. |
| [2] | ZHANG Guangshun, XU Xiaonan, XU Chuyun, LIAO Guanghui, XU Hao, LOU Zhaohuan, ZHANG Guangji. Actinidia chinensis polysaccharide interferes with the epithelial-mesenchymal transition of gastric cancer by regulating the nuclear transcription factor-κB pathway to inhibit invasion and metastasis [J]. Journal of Traditional Chinese Medicine, 2024, 44(5): 896-905. |
| [3] | WU Ruixin, FANG Qingliang, GUAN Sisi, WEI Xianglong, SHAN Mengjun, MAO Zhujun, GONG Yabin, XU Ling, ZHOU Di, DONG Changsheng. A pilot study of precision treatment for patients with lung cancer pain by Longteng Tongluo recipe (龙藤通络方) using serum genomics [J]. Journal of Traditional Chinese Medicine, 2024, 44(5): 1006-1016. |
| [4] | GONG Hui, LI Yang, FENG Lei, XIAO Yujie, HUANG Lizhong, MAO Dan, ZHANG Hui. Yanghe decoction (阳和汤) attenuated pain hypersensitivity induced by michigan cancer foundation-7 injection in rats with bone metastases from breast cancer by inhibiting transient receptor potential ankyrin 1 [J]. Journal of Traditional Chinese Medicine, 2022, 42(6): 948-955. |
| [5] | ZHAO Shuai, TAN Jun, YU Hengmei, TIAN Ying, WU Youfang, LUO Rui, GUO Jianjun. In vivo and in vitro antiproliferative and antimetastatic effects of hemolymph of Aspongopus chinensis Dallas on breast cancer cells [J]. Journal of Traditional Chinese Medicine, 2021, 41(4): 523-529. |
| [6] | Liu Minghua, Yang Ying, Liu Dajun, Cao Ye, Li Yan. Parthenolide increases the sensitivity of gastric cancer cells to chemotherapy [J]. Journal of Traditional Chinese Medicine, 2020, 40(6): 908-916. |
| [7] | Zheng Lixiang, Zheng Qing, Yu Zhipeng, Wang Jian, Ren Xiaoying, Gong Yan, Yang Xue, Hunag Ping, Weng Meizhi, Liu Hongning, Liu Haizhou. Liuwei Dihuang pill suppresses metastasis by regulating the wnt pathway and disrupting β-catenin/T cell factor interactions in a murine model of triple-negative breast cancer [J]. Journal of Traditional Chinese Medicine, 2019, 39(06): 826-832. |
| [8] | Yin Yukun, Feng Li, Zhou Lei, Li Jie, Gao Yin, Wang Ningjun, Yu Jianhua, Jiang Zhenglong, He Shengqi, Lu Dianrong, Wang Fang, Du YanlinYin. Effects of Yishengukang decoction on expression of bone-specific alkaline phosphatase, carboxyterminal propeptide of typeⅠprocollagen, and carboxyterminal cross-linked telepeptide of typeⅠcollagen in malignant tumor patients with bone metastasis [J]. Journal of Traditional Chinese Medicine, 2017, 37(01): 30-34. |
| [9] | Wang Shuo, Zhang Ying, Cong Weihong, Liu Jie, Zhang Yuren, Fan Huiting, Xu Yonggang, Lin Hongsheng. Breast cancer stem-like cells can promote metastasis by activating platelets and down-regulating antitumor activity of natural killer cells [J]. Journal of Traditional Chinese Medicine, 2016, 36(04): 530-537. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

