Journal of Traditional Chinese Medicine ›› 2026, Vol. 46 ›› Issue (1): 119-126.DOI: 10.19852/j.cnki.jtcm.2026.01.011
• Original Articles • Previous Articles Next Articles
Baitouweng Tang (白头翁汤) alleviates dextran sulfate sodium-induced ulcerative colitis in mice: a network pharmacology combined with experimental study
SUN Yan1, ZHOU Haozheng2, FENG Jianhui2, LIN Zikai2, HE Jie2, WANG Zhenhua2, GUO Yuting1, WEN Shaohong2, LI Gang2( )
)
- 1 College of Traditional Chinese Medicine, Shandong University of Traditional Chinese Medicine, Jinan 250000, China
2 Center for Mitochondria and Healthy Aging, College of Life Sciences, Yantai University, Yantai 264000, China
-
Received:2024-12-12Accepted:2025-05-07Online:2026-02-15Published:2026-01-28 -
Contact:LI Gang, Center for Mitochondria and Healthy Aging, College of Life Sciences, Yantai University, Yantai 264000, China.ligang@ytu.edu.cn ;Telephone: +86-535-6902638 -
About author:SUN Yan andZHOU Haozheng are co-first authors and contributed equally to this work -
Supported by:Shandong Provincial Natural Science Foundation Project, Study on the Structure-Activity Relationship and Mechanism of Licorice Chalcone Components in Synergizing with Immune Checkpoint Inhibitors for Anticancer Therapy(ZR2020MH380);Mechanisms of the Novel Flavone C-Glycoside 6'-O-Rhamnosyllutonarin from Dianthus superbus Improves Non-alcoholic Fatty Liver Disease via Modulating the Juxtaposed with Another Zinc Finger gene 1/Adenosine Monophosphate-activated Protein Kinase/ Sterol Regulatory Element-Binding Protein Pathway(ZR2024MC209)
Cite this article
SUN Yan, ZHOU Haozheng, FENG Jianhui, LIN Zikai, HE Jie, WANG Zhenhua, GUO Yuting, WEN Shaohong, LI Gang. Baitouweng Tang (白头翁汤) alleviates dextran sulfate sodium-induced ulcerative colitis in mice: a network pharmacology combined with experimental study[J]. Journal of Traditional Chinese Medicine, 2026, 46(1): 119-126.
share this article

Figure 1 Intervention extent of PD on UC and histopathological changes in colonic tissue A: DAI scores; B: macroscopic colon appearance; C: measured colon length in the indicated groups; D: changes in body weight in the indicated groups; E: colon tissue HE staining; B1, E1, E6: control group; B2, E2, E7: model group; B3, E3, E8: mesalazine group; B4, E4, E9: PD low group; B5, E5, E10: PD high group; E1; E2; E3; E4; E5: × 100; E6; E7; E8; E9; E10: ×400. Control group: a normal control group provided with a normal diet and water; model group: a model group subjected to DSS modeling, with alternating saline and 2% DSS administration; mesalazine group: a mesalazine group mesalazine (300 mg/kg) was used to gavage mice when drinking 2% DSS; PD low group: a low-dose PD group in which mice treated with DSS were gavaged with PD (15 mL/kg); PD high group: a high-dose PD group in which in which mice treated with DSS were gavaged with PD (30 mL/kg). PD: Baitouweng Tang (Pulsatilla decoction); UC: ulcerative colitis; DAI: disease activity index; HE: hematoxylin-eosin; DSS: dextran sulfate sodium. Data were presented as mean ± standard error of the mean (n = 3). The t test was used for measurement data; one-way analysis of variance was used for multiple comparisons. Compared with the control group, aP < 0.001; compared with the model group, bP < 0.01.

Figure 2 Effect of PD on the levels of pro-inflammatory and anti-inflammatory cytokines in the serum of DSS-induced UC model mice A: IL-6. B: TNF-α. C: TGF-β. D: IL-4. E: IL-10. Control group: a normal control group provided with a normal diet and water; model group: a model group subjected to DSS modeling, with alternating saline and 2% DSS administration; mesalazine group: a mesalazine group mesalazine (300 mg/kg) was used to gavage mice when drinking 2% DSS; PD low group: a low-dose PD group in which mice treated with DSS were gavaged with PD (15 mL/kg); PD high group: a high-dose PD group in which in which mice treated with DSS were gavaged with PD (30 mL/kg). PD: Baitouweng Tang (Pulsatilla decoction); UC: ulcerative colitis; DSS: dextran sulfate sodium; IL: interleukin; TNF-α: tumor necrosis factor-alpha; TGF-β: transforming growth factor-beta. Data were presented as mean ± standard error of the mean (n = 3). The t test was used for measurement data; one-way analysis of variance was used for multiple comparisons. Compared with the control group, aP < 0.001; compared with the model group, bP < 0.001, cP < 0.01.
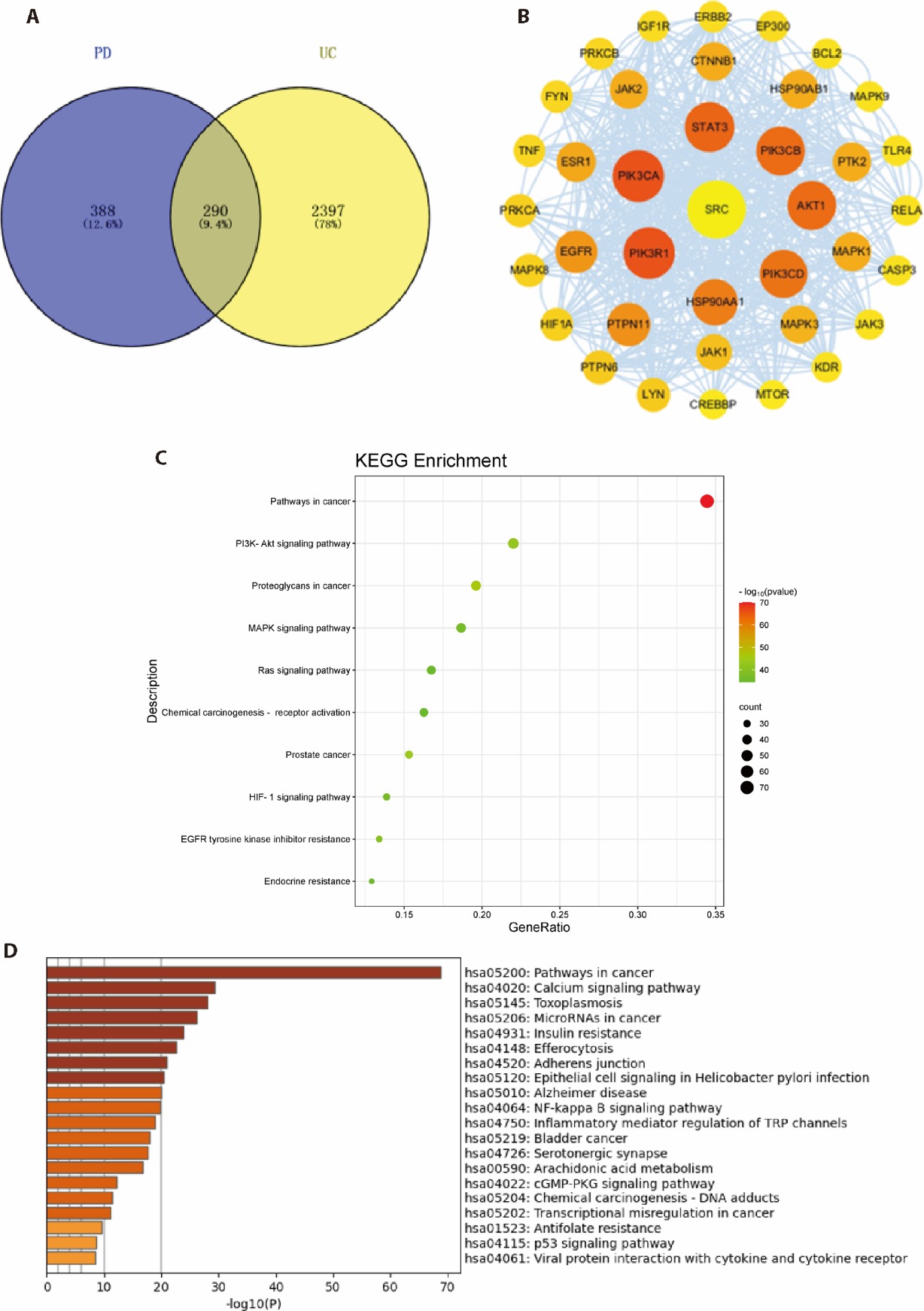
Figure 3 Network pharmacology analysis A: intersecting targets; B: core targets in the PPI network; C: KEGG analysis; D: associated pathways. PD: Baitouweng Tang (Pulsatilla decoction); UC: ulcerative colitis; PPI network: protein-protein interaction networks; GO: gene ontology; KEGG: kyoto encyclopedia of genes and genomes; TLR4/ NF-κB: toll-like receptor 4/ nuclear factor-kappa B; PI3K: phosphatidylinositol 3 kinase; Akt: protein kinase B; MAPK: mitogen-activated protein kinase. The abbreviations and their full names appeared in Figure 3B are presented in supplementary Table 2.

Figure 4 Effect of PD on mRNA and protein levels in the colon A: effect of PD on mRNA levels in the colon; A1: TLR4 mRNA; A2: NF-κb mRNA; B: effect of PD on protein levels in the colon; B1: TLR4 protein; B2: NF-κb protein; B3: Western blot images. Control group (1 and 2): a normal control group provided with a normal diet and water; model group (3 and 4): a model group subjected to DSS modeling, with alternating saline and 2% DSS administration;mesalazine group (5 and 6): a mesalazine group mesalazine (300 mg/kg) was used to gavage mice when drinking 2% DSS; PD low group (7 and 8): a low-dose PD group in which mice treated with DSS were gavaged with PD (15 mL/kg); PD high group (9and 10):a high-dose PD group in which in which mice treated with DSS were gavaged with PD (30 mL/kg). PD: Baitouweng Tang (Pulsatilla decoction); TLR4: toll-like receptor 4; NF-κb: nuclear factor-kappa B; DSS: dextran sulfate sodium. Data were presented as mean ± standard error of the mean (n = 3). The t test was used for measurement data; one-way analysis of variance was used for multiple comparisons. Compared with the model group, aP < 0.01; bP < 0.05; dP < 0.001; compared with the control group, cP < 0.01; eP < 0.05.
| 1. |
Wangchuk P, Yeshi K, Loukas A. Ulcerative colitis: clinical biomarkers, therapeutic targets, and emerging treatments. Trends Pharmacol Sci 2024; 45: 892-903.
DOI URL |
| 2. |
Le Berre C, Honap S, Peyrin-Biroulet L. Ulcerative colitis. Lancet 2023; 402: 571-84.
DOI PMID |
| 3. |
Lao J, Yan S, Yong Y, et al. Lacticaseibacillus casei IB 1 alleviates DSS-induced inflammatory bowel disease by regulating the microbiota and restoring the intestinal epithelial barrier. Microorganisms 2024; 12: 1379.
DOI URL |
| 4. |
Elkholy SE, Maher SA, Abd El-Hamid NR, et al. The immunomodulatory effects of probiotics and azithromycin in dextran sodium sulfate-induced ulcerative colitis in rats via TLR4-NF-κB and p38-MAPK pathway. Biomed Pharmacother 2023; 165: 115005.
DOI URL |
| 5. |
Shi G, Kong J, Wang Y, Xuan Z, Xu F. Glycyrrhiza uralensis Fisch. alleviates dextran sulfate sodium-induced colitis in mice through inhibiting of NF-κB signaling pathways and modulating intestinal microbiota. J Ethnopharmacol 2022; 298: 115640.
DOI URL |
| 6. |
Rossier L, Matter C. Overview and update on treatment in ulcerative colitis. Ther Umsch 2023; 80: 386-92.
PMID |
| 7. | Zhang ZJ. Differentiation of the pulse. Symptom complex, and treatment of the Jueyin (greater Yin syndrome). In: Shang Han Lun. Translated by Luo Xiwen. Beijing: New World Press, 2007: 221-2. |
| 8. |
Wei P, He Q, Liu T, et al. Baitouweng decoction alleviates dextran sulfate sodium-induced ulcerative colitis by suppressing leucine-related mTORC 1 signaling and reducing oxidative stress. J Ethnopharmacol 2023; 304: 116095.
DOI URL |
| 9. |
Fu YJ, Xu B, Huang SW, et al. Baicalin prevents LPS-induced activation of TLR4/NF-κB p65 pathway and inflammation in mice via inhibiting the expression of CD14. Acta Pharmacol Sin 2021; 42: 88-96.
DOI |
| 10. |
Xiong T, Zheng X, Zhang K, et al. Ganluyin ameliorates DSS-induced ulcerative colitis by inhibiting the enteric-origin LPS/ TLR4/NF-κB pathway. J Ethnopharmacol 2022; 289: 115001.
DOI URL |
| 11. |
Pisani LF, Tontini G, Vecchi M, Croci GA, Pastorelli L. NF-kB pathway is involved in microscopic colitis pathogenesis. J Int Med Res 2022; 50: 3000605221080104.
DOI URL |
| 12. |
Hu N, An R, Yu K, Chang Y, Gao W. PF4 induces inflammatory response through NF-kB signal pathway in rats with intracerebral haemorrhage. Folia Neuropathol 2023; 61: 379-86.
DOI PMID |
| 13. |
Wirtz S, Popp V, Kindermann M, et al. Chemically induced mouse models of acute and chronic intestinal inflammation. Nat Protoc 2017; 12: 1295-309.
DOI PMID |
| 14. |
Rentea RM, Renaud E, Ricca R, et al. Surgical management of ulcerative colitis in children and adolescents: a systematic review from the APSA outcomes and evidence-based practice committee. J Pediatr Surg 2023; 58: 1861-72.
DOI PMID |
| 15. |
Pigneur B, Ruemmele FM. A critical review of adalimumab for the treatment of moderate-to-severe active ulcerative colitis in children. Expert Rev Gastroenterol Hepatol 2022; 16: 1023-8.
DOI URL |
| 16. |
Mirsepasi-Lauridsen HC. Therapy used to promote disease remission targeting gut dysbiosis, in UC patients with active disease. J Clin Med 2022; 11: 7472.
DOI URL |
| 17. |
Jadhav A, Jagtap S, Vyavahare S, Sharbidre A, Kunchiraman B. Reviewing the potential of probiotics, prebiotics and synbiotics: advancements in treatment of ulcerative colitis. Front Cell Infect Microbiol 2023; 13: 1268041.
DOI URL |
| 18. |
Segal JP, LeBlanc JF, Hart AL. Ulcerative colitis: an update. Clin Med (Lond) 2021; 21: 135-9.
DOI PMID |
| 19. |
Nakase H, Sato N, Mizuno N, Ikawa Y. The influence of cytokines on the complex pathology of ulcerative colitis. Autoimmun Rev 2022; 21: 103017.
DOI URL |
| 20. |
Wu J, Luo Y, Shen Y, et al. Integrated metabonomics and network pharmacology to reveal the action mechanism effect of Shaoyao decoction on ulcerative colitis. Drug Des Devel Ther 2022; 16: 3739-76.
DOI URL |
| 21. |
Shi YJ, Huang C, Gong PQ, Liu C, Hu ZQ, Wang H. The protective role of TLR4 in intestinal epithelial cells through the regulation of the gut microbiota in DSS-induced colitis in mice. Front Biosci (Landmark Ed) 2023; 28: 175.
DOI URL |
| 22. |
Li M, Lan L, Zhang S, et al. IL-6 downregulates hepatic carboxylesterases via NF-κB activation in dextran sulfate sodium-induced colitis. Int Immunopharmacol 2021; 99: 107920.
DOI URL |
| 23. |
Shao XX, Xu Y, Xiao HY, Hu Y, Jiang Y. Higenamine improves DSS-induced ulcerative colitis in mice through the Galectin-3/TLR4/NF-κB pathway. Tissue Cell 2023; 82: 102111.
DOI URL |
| 24. |
Althagafy HS, Ali FEM, Hassanein EHM, et al. Canagliflozin ame-liorates ulcerative colitis via regulation of TLR4/MAPK/NF-κB and Nrf2/PPAR-γ/SIRT 1 signaling pathways. Eur J Pharmacol 2023; 960: 176166.
DOI URL |
| 25. |
Chen Y, Yan W, Chen Y, et al. SLC6A14 facilitates epithelial cell ferroptosis via the C/EBPβ-PAK6 axis in ulcerative colitis. Cell Mol Life Sci 2022; 79: 563.
DOI |
| 26. |
Saez A, Herrero-Fernandez B, Gomez-Bris R, Sánchez-Martinez H, Gonzalez-Granado JM. Pathophysiology of inflammatory bowel disease: innate immune system. Int J Mol Sci 2023; 24: 1526.
DOI URL |
| 27. |
Feng Z, Zhou P, Wu X, Zhang J, Zhang M. Hydroxysafflor yellow A protects against ulcerative colitis via suppressing TLR4/NF-κB signaling pathway. Chem Biol Drug Des 2022; 99: 897-907.
DOI URL |
| 28. |
Qu Y, Li X, Xu F, et al. Kaempferol alleviates murine experimental colitis by restoring gut microbiota and inhibiting the LPS-TLR4-NF-κB axis. Front Immunol 2021; 12: 679897.
DOI URL |
| 29. |
Zheng J, Zhang J, Zhou Y, et al. Taurine alleviates experimental colitis by enhancing intestinal barrier function and inhibiting inflammatory response through TLR4/NF-κB signaling. J Agric Food Chem 2024; 72: 12119-29.
DOI URL |
| 30. |
Yu C, Wang D, Yang Z, Wang T. Pharmacological effects of poly-phenol phytochemicals on the intestinal inflammation via targeting TLR4/NF-κB signaling pathway. Int J Mol Sci 2022; 23: 6939.
DOI URL |
| 31. |
Hu LH, Liu JY, Yin JB. Eriodictyol attenuates TNBS-induced ulcerative colitis through repressing TLR4/NF-kB signaling pathway in rats. Kaohsiung J Med Sci 2021; 37: 812-908.
DOI URL |
| 32. | Sun J, Zhao P, Ding X, et al. Cayratia japonica prevents ulcerative colitis by promoting M2 macrophage polarization through blocking the TLR4/MAPK/NF-κB pathway. Mediators Inflamm 2022; 2022: 1108569. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

